Abstract
Feline panleukopenia virus (FPV) infections are typically associated with anorexia, vomiting, diarrhea, neutropenia, and lymphopenia. In cases of late prenatal or early neonatal infections, cerebellar hypoplasia is reported in kittens. In addition, single cases of encephalitis are described. FPV replication was recently identified in neurons, although it is mainly found in cells with high mitotic activity. A female cat, 2 months old, was submitted to necropsy after it died with neurologic deficits. Besides typical FPV intestinal tract changes, multifocal, randomly distributed intracytoplasmic vacuoles within neurons of the thoracic spinal cord were found histologically. Next-generation sequencing identified FPV-specific sequences within the central nervous system. FPV antigen was detected within central nervous system cells, including the vacuolated neurons, via immunohistochemistry. In situ hybridization confirmed the presence of FPV DNA within the vacuolated neurons. Thus, FPV should be considered a cause for neuronal vacuolization in cats presenting with ataxia.
Keywords
Feline panleukopenia virus (FPV) is a small non-enveloped single-stranded DNA virus of the family Parvoviridae and is closely related to canine parvovirus (CPV). Replication takes place within the host cell nucleus. 15 Cats are predominantly infected through the fecal-oral route, and as a non-enveloped DNA virus, FPV is very stable in the environment, with infectivity persisting for up to 1 year within infected organic material. Consequently, there is a high risk of transmission, which is especially a problem under conditions of high population densities, as seen in animal shelters. 11,15 Upon oral or intranasal infection, initial replication of the virus takes place within 18 to 24 hours in the oropharynx. After 2 days to 1 week, viremia results in distribution of virus, primarily in tissues with high mitotic rates, and is associated with clinical signs of anorexia, lymphopenia induced by direct lymphocytolysis, vomiting, and diarrhea. 15 In the bone marrow, parvoviral replication in early progenitor cells is associated with development of panleukopenia in FPV-infected cats. 15 In the intestine, clinical signs are mainly caused by infection and damage of rapidly replicating cells within the crypts of the intestinal mucosa, resulting in malabsorption and increased permeability. 15
In contrast, fetal or neonatal FPV infections of kittens are known to cause central nervous system (CNS) defects, especially cerebellar malformation. 1 FPV infection of neuroblasts of the external granular layer during cerebellar development, which takes place during the late gestation and early neonatal phases, may cause cerebellar hypoplasia due to reduced and distorted cell layers. 1,3,5,10 Additionally, DNA and antigen from FPV and CPV have been detected within neurons, especially Purkinje cells in cats and dogs. 5,12 Thus, parvoviruses are thought to be capable of using neurons for replication. 5 Recent reports show that feline parvoviruses are able to maintain their host cells in the S phase by a proposed ERK signaling pathway, as an ERK1/2 nuclear accumulation was observed, as well as the intranuclear occurrence of the key cell cycle arrest player S139 phospho-H2A1, in parvovirus-infected neurons. 9 FPV infection with mild lymphohistiocytic inflammation of the meninges and cerebellum was described in a 2-week-old cat, which exhibited parvoviral antigen in neurons of the granular layer of the cerebellum and in the gray matter of the spinal cord, as well as in most other brain regions, in macrophages, microglia, astrocytes, and ependymal cells. 13 Similarly, FPV antigen was detected in glial cells and neurons of 4 cats showing focal neuronal satellitosis and neuronophagia, especially of the interthalamic adhesion. 5 Furthermore, FPV was assumed as a cause of demyelination within the spinal cord and caudal cerebellar peduncles in cats; however, the findings are considered controversial. 4 Additionally, FPV antigen and nucleic acids were detected in neurons other than Purkinje cells, accompanied by an extracellular vacuolization. 16 So far, neuronal vacuolization has not been described in FPV CNS infections. Similar distribution patterns of parvovirus infections as described for cats, including neurotropism, can be observed in dogs infected with CPV, suffering from puppy shaker syndrome and leukoencephalopathy. 12 Furthermore, an association of CPV infection with leukoencephalopathies and vacuolization of the neuropil in dogs was discussed, although causality appeared unlikely. 12
Here, we describe for the first time neuronal vacuolization as the only pathologic alteration found in the CNS in association with parvoviral antigen and FPV-specific nucleic acids in the CNS of a 2-month-old cat suffering from ataxia.
A female 2-month-old cat died due to clinical signs of ataxia, anorexia, polydipsia, hypersalivation, and vomitus and was submitted for pathologic examination. Following complete necropsy, samples of various tissues were collected, fixed in 10% neutral buffered formalin, and embedded in paraffin wax or were stored at –20°C and –80°C for further investigations. For histologic examination, 3-µm-thick sections were stained with hematoxylin and eosin as previously described. 12
Macroscopic and histopathologic evaluation of the cat revealed lesions characteristic for FPV infection, including catarrhal enteritis, enlarged mesenteric lymph nodes, and atrophy of the thymus. The intestine showed severe villus atrophy and fusion with multiple dilated crypts, crypt epithelial hyperplasia, necrosis, and regeneration of enterocytes as well as moderate depletion of Peyer's patches. The spleen showed mild follicular hyperplasia. The thymus revealed a moderate depletion, and the mesenteric lymph nodes were edematous. Additionally, multifocal intracytoplasmic vacuoles were found within randomly distributed neurons of the gray matter of the thoracic spinal cord but not in other CNS regions (Fig. 1). To screen for a possible etiology of the alterations, frozen CNS and small intestine samples of the cat were used for next-generation sequencing analyses, as formerly described with minor variations. 7,8 Briefly, tissues were homogenized, and viral enrichment procedures were applied prior to the isolation of nucleic acids. Random DNA amplification was performed with a modified sequence-independent single-primer amplification protocol. Library preparation was carried out as indicated by Nextera XT DNA Sample Preparation Kit protocol (Illumina). Sequencing was performed on an Illumina Miseq system. High-throughput data were analyzed with the CLC Genomics Workbench 9.0.1. Alignments were calculated with MAFFT. Phylogenetic analyses were performed by MEGA 6.06. Tamura 3-parameter nucleotide substitution model was selected as a best-fit model according to Bayesian information criteria. Sequences obtained by next-generation sequencing showed >1.2 million reads mapping to FPV from both tissues. Complete genome sequences were constructed and compared. No amino acid changes were found between FPV in the CNS and the small intestine of the cat. Phylogenetic analyses of the VP2 gene (Fig. 2) showed that FPV from this study was closest related to Asian strains V211 and ChangC2007 (99.8% sequence identity; GenBank accession Nos. AB054227, FJ936171), but altogether, all analyzed sequences were highly identical.
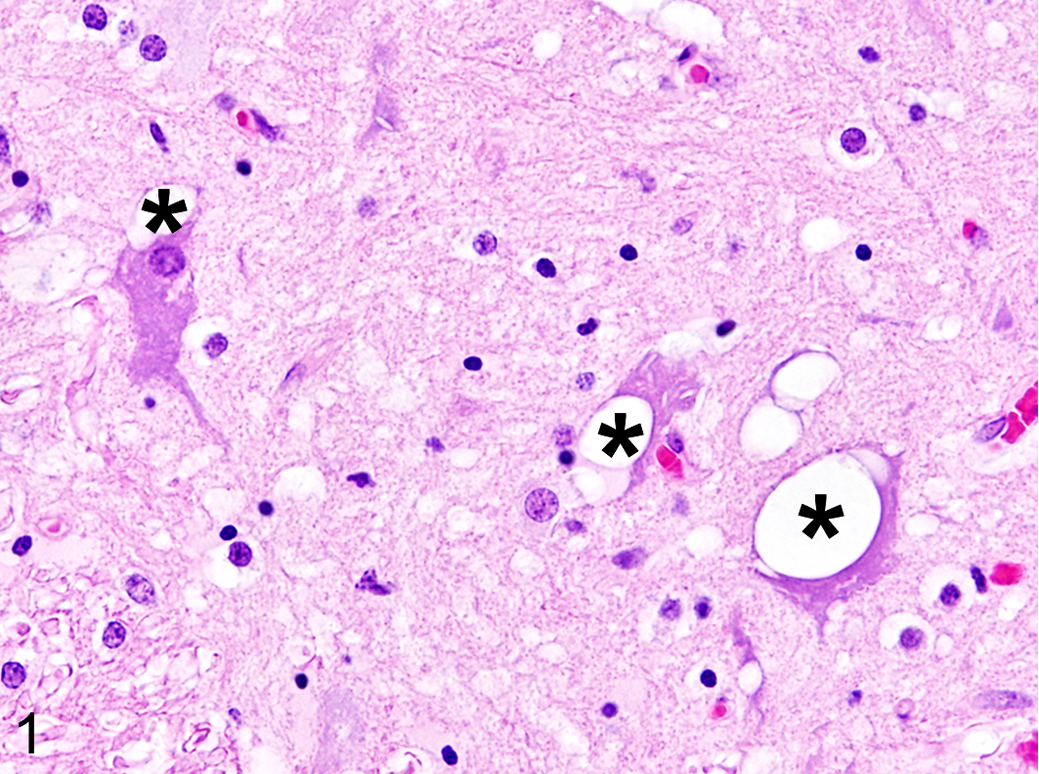
Parvovirus-associated neuronal vacuolization, thoracic spinal cord, cat. Multifocal vacuoles are present within the cytoplasm of multiple neurons (asterisks) of the gray matter. Hematoxylin and eosin.

Maximum likelihood phylogenetic reconstruction with 1000 bootstrap replicates of the VP2 gene of feline panleukopenia viruses. Canine parvoviruses were used as the outgroup. Scale bar indicates nucleotide substitution per site. Taxon names are presented as GenBank accession number / virus strain. S1025/15 is the sequence identified in the present study.
To study FPV cell tropism, immunohistochemistry was performed on formalin-fixed paraffin-embedded tissue samples from the CNS, including cerebrum, cerebellum, brainstem, and spinal cord, as well as small intestine, bone marrow, thymus, spleen, tonsils, and mesenteric lymph nodes, as described using a monoclonal mouse anti-CPV1-2A1 antibody (dilution: 1:500; Custom Monoclonal Antibodies International) as the primary antibody and a biotin-conjugated goat anti-mouse antibody (dilution: 1:200; Vector Laboratories, Inc) as the secondary antibody. 12 Additionally, immunohistochemistry based on a polyclonal rabbit anti–glial fibrillary acidic protein antibody (dilution 1:1000; DakoCytomation) and a biotin-conjugated goat anti-rabbit antibody (dilution: 1:200; Vector Laboratories, Inc) as second antibody was performed on the spinal cord sections to investigate a potential astrogliosis. Briefly, after incubation with first and second antibodies, the avidin-biotin-complex method (Vector Laboratories, Inc) was used for visualization of antigen-antibody complexes, and sections were counterstained with Mayer's hemalaun (Carl Roth GmbH). 12 The proportion of FPV-immunopositive cells was scored semiquantitatively as follows, depending on the percentage of positive cells: mild (+, <25% per cell type), moderate (++, 25%–50%), and severe (+++, >50%). Neurons, glial cells (mainly cells with astrocytic morphology), and endothelial cells were positive in all examined CNS localizations, including cerebrum, cerebellum, brainstem, and spinal cord (Fig. 3), and the signal was mainly cytoplasmic. Abundant endothelial cells of the cerebrum and within all examined localizations of the spinal cord (cervical, thoracic, and lumbar) stained positively for parvovirus antigen, whereas a moderate number of parvovirus-positive endothelial cells was detected in the brainstem and a low number within the cerebellum. A moderate number of immunopositive glial cells was detected in cerebrum (coronal section including hippocampus, pineal gland, and thalamus) and spinal cord (cervical, thoracic, and lumbar), whereas a low number of parvovirus antigen–positive glial cells were observed in cerebellum and brainstem. A moderate number of neurons of the spinal cord were positive for parvovirus antigen. Only few parvovirus antigen–positive neurons were found within the cerebrum (coronal section, including hippocampus, pineal gland, and thalamus), cerebellum (Purkinje cells), and brainstem. Examination of the spinal cord areas revealed the highest number of parvovirus antigen–positive cells within the CNS (Fig. 3), including the vacuolated neurons in the thoracic spinal cord.
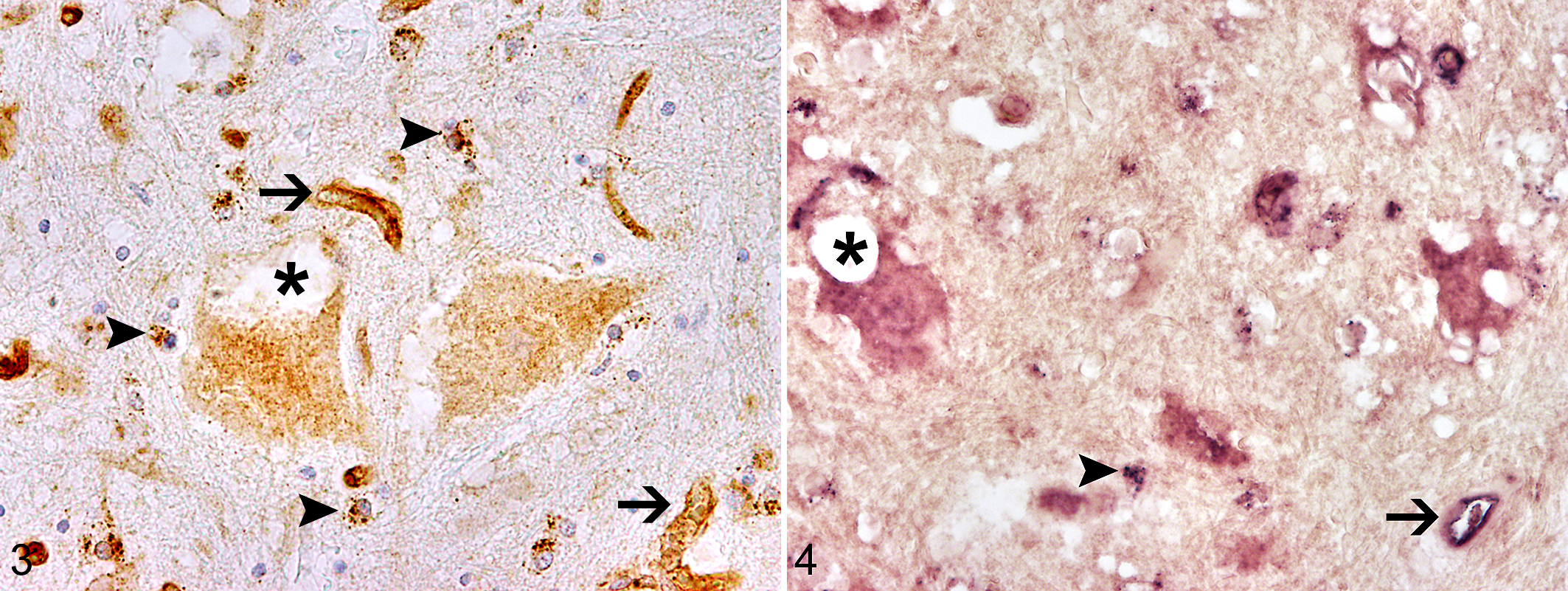
FPV infection may induce degenerative and toxic changes, as indicated by large vacuole formation in some neurons, which suggests a different pathogenesis of the observed lesions caused by the detected FPV when compared with those known to result in cerebellar hypoplasia in kittens. The observed neuronal vacuolation, probably indicating vacuolar degeneration, such as that by mitochondrial swelling, might be induced by acute injury from the FPV infection without developing an inflammatory response in the CNS, as the animal died early after disease onset. 14 Astrogliosis, detectable by glial fibrillary acidic protein immunohistochemistry, was not present in any of the spinal cord sections. Furthermore, immunohistochemistry for parvovirus antigen was positive in several affected nonneural organs. Immunohistochemical labeling of parvovirus antigen was abundant in enterocytes and lymphoid cells of the gut-associated lymphoid tissue of the small intestine, in myeloid cells of the bone marrow, and in lymphoid cells of the depleted thymus, white pulp of the spleen, and germinal centers of mesenteric lymph nodes.
The occurrence of FPV nucleic acids within the vacuolated spinal cord neurons was investigated through in situ hybridization with a parvoviral VP2 gene–specific sense and antisense probe on formalin-fixed paraffin-embedded tissue as described. 12 Briefly, slides were deparaffinized, hydrated in graded alcohol, and rinsed in diethyl pyrocarbonate–treated water. Proteolytic digestion was performed with 1 µg/ml of proteinase K (Roche Diagnostics). Following postfixation, acetylation, and prehybridization, hybridization was performed overnight at 52°C in a moist chamber. Posthybridization washing was performed for 15 minutes, 2 times, in 6× standard saline citrate (SSC) and 45% formamide at 42°C, followed by 2 times washing in 2×SSC at room temperature. After RNAse treatment, slides were rinsed 2 times in 2×SSC at room temperature and 2 times for 15 minutes in 0.2×SSC at 50°C. Digoxygenin-labeled probes were identified with an anti-digoxygenin antibody conjugated with alkaline phosphatase (Roche Diagnostics). As substrates for the enzyme reaction served nitroblue tetrazoliumchloride (Sigma-Aldrich) and 5-bromo-4-chloro-3-indolyl phosphate (X-Phosphate; Sigma-Aldrich).
Spinal cord sections were scored semiquantitatively as described for immunohistochemical analysis. With the T3-transcribed RNA probe (sense probe) that detects parvovirus genomic DNA, strong intracytoplasmic positivity was observed in endothelial cells, glial cells, and neurons (Fig. 4). The lack of a positive signal in neurons and glial cells using the T7-transcribed probe (antisense probe)—which detects cytoplasmic viral mRNA or intranuclear monomer replicative intermediate strands during FPV infection—showed that FPV transcription was missing and replication unlikely, whereas a positive signal was observed within scattered endothelial cells.
The fact that virus protein and viral nucleic acids were found primarily within the cytoplasm of infected cells may suggest a latent phase of viral infection without active replication and transcription. Nevertheless, it cannot be fully excluded that the lower signal based on the antisense probe, which detects mRNA, is due to the formalin fixation of tested tissues, although studies have shown that several genes are still detectable by real-time quantitative reverse transcription polymerase chain reaction following formalin fixation. 17 Bone marrow of the cat revealed strong positivity using both sense and antisense probes.
In summary, the presence of FPV antigen and genomic DNA within vacuolated neurons of the spinal cord of a cat suffering from ataxia, in combination with a lack of sequences of other viral agents according to next-generation sequencing, indicates that FPV may induce neuronal vacuolization in young cats. Furthermore, neuropil vacuolation, typical for feline spongiform encephalopathy, was absent in the present case, and cats with feline spongiform encephalopathy are usually >2 years old. 2,6 Nevertheless, unknown causes, such as intoxications or genetic disorders, cannot be entirely excluded as underlying causes for the observed neuronal vacuolation. Altogether, FPV infection should be taken into consideration as a cause of neurologic disorders in cats when neuronal vacuolization is the only histopathologic alteration within the CNS.
Footnotes
Acknowledgments
We thank Bettina Buck, Kerstin Rohn, Kerstin Schöne, Caroline Schütz, and Danuta Waschke for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was in part supported by the Niedersachsen Research Network on Neuroinfectiology of the Ministry of Science and Culture of Lower Saxony and by the COMPARE project and has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No. 643476.
