Abstract
Postmortem evaluation of racehorses has focused primarily on musculoskeletal injuries; however, horses also die suddenly on the track (sudden death [SD]). Although cardiac conditions are frequently suspected as a cause of death, SD racehorses are often autopsy negative; however, previous studies have been limited due to inconsistent or insufficient cardiac sampling and lack of controls. SD in New York (NY) and Maryland (MD) racehorses was evaluated in an observational case vs control study comparing clinical information, postmortem evaluation including cardiac dissection, and cardiac conduction system histopathology. In the study period, there were 40 cases of SD. In NY, SD occurred in 12% (37/316) of submissions, and 36 (11%) cases of SD were exercise associated (EASD); 3 EASD cases occurred in MD. In NY/MD EASD cases with histologic examination of the heart, 11 of 36 (31%) had significant lesions, including mesenteric artery rupture (1), axial trauma (2), systemic inflammation (2), pulmonary hemorrhage (1), and cardiac disease (5). Mild myocardial fibrosis, mild inflammation, coronary arteriosclerosis, and variation in cardiac nodal connective tissue were present in both SD cases and controls and thus were not considered to be causes of SD. While not excluding a genetic basis for SD, analysis of the genotypes (GGP Equine 70 K Array) of cases and controls did not reveal significant differences in allele frequencies at any locus. Most SD racehorses were autopsy negative; further research using standardized protocols and controls is needed to understand the underlying causes of SD, which is crucial to protecting the viability of racing.
Horseracing is one of America’s oldest sports and its history is intertwined with the American experience. Equine athletes are charismatic animals whose death or euthanasia in the public eye has a negative impact on the perception of the horseracing industry. In addition, perceptions of drug use have contributed to a beleaguered public image of horseracing and threaten this historic economic resource. While rider safety remains primary, concern over the safety and health of racehorses has led to investigation of the underlying causes of death or euthanasia, particularly in high-profile jurisdictions. 6,9,20,26 –29,36 In 2012, the New York Task Force on Racehorse Health and Safety was formed to investigate the circumstances related to 21 equine racing fatalities that occurred at the winter meet at Aqueduct Racetrack. Based on task force recommendations, all racehorses that die at New York (NY) racetracks are presented to the NY State Animal Diagnostic Center (AHDC) for postmortem examination. 33,40
Postmortem evaluation of racehorses has focused primarily on detection of preexisting conditions associated with fatal musculoskeletal injury (FMSI); however, apparently healthy horses also die suddenly or collapse on the track, often during exercise. 6,9,15,16,18,20,27,29,35 There is confusing usage of the terms sudden death and sudden cardiac death by lay and medical personnel. Sudden death (SD) is defined as nonviolent, nontraumatic, unexpected death that is instantaneous or within a few minutes of an abrupt change in clinical state and is associated with a variety of causes. 38 Sudden cardiac death applies to cases where SD is attributed to cardiac causes. In humans, frequent causes vary by age group and include atherosclerotic coronary artery disease/infarction, coronary anomaly, cardiomyopathy, valvular heart disease, and drug use; sudden cardiac death cases lacking an apparent cause of death on postmortem or histologic evaluation are “autopsy negative” and may be related to primary arrhythmogenic disorders. 1,37,38,41 Exercise-associated sudden death (EASD) has been used to describe the subset of SD occurring during exercise or within an hour thereof. 16
Cardiac conditions are frequently suspected but unproven as a cause of SD or collapse in racehorses. 15,35 SD in racehorses is often EASD and has been associated with axial skeleton fracture, exercise-induced pulmonary hemorrhage, vessel rupture and internal hemorrhage, myocardial or other cardiovascular lesions, or associated with injection. 6,9,15,16,18,20,27 –29,35
SD in racehorses shares similarities with SD in young adult humans (under age 35), which frequently occurs during physical activity. In humans, up to 10% to 30% of sudden cardiac deaths do not have identifiable abnormalities, and death may be related to rare electrophysiologic disorder and lethal arrhythmia. Specific genetic tests (“molecular autopsy” if testing is postmortem) are available for humans with compatible clinical evidence or family history; however, genetic testing does not always identify disease-causing mutations. 37,42 The occurrence, prevalence, and inheritance of similar mutations in horses and their role in SD are unknown.
There are gaps in our knowledge of the cardiac health, causes, and pathophysiology of SD in horses. Furthermore, it is unknown if there is a genetic basis for SD in the horse. Understanding the underlying causes of SD/collapse of racehorses is crucial to improving racehorse health, maintaining a positive public perception of the racing industry, and protecting its viability. We hypothesized that racehorses have occult heart or genetic arrhythmogenic abnormalities that cause SD. The first objective of this study was to systemically evaluate the cardiac pathology of NY and Maryland (MD) SD cases compared to controls guided by the epidemiology of nonatherosclerotic SD in humans. To evaluate for a genetic basis for SD, we also compared genotypes of SD racehorses and controls; given the numerous genes and exons implicated in inherited arrhythmias syndromes in humans, this genome-wide association study (GWAS) is the first stage of exploring the possible genetic basis of SD in racehorses.
Materials and Methods
Case Selection
Postmortem evaluation was performed on racehorses with racing, training, or nonexercise fatalities at NY racetracks at the AHDC at Cornell University (Ithaca, NY) as previously described. 33 In addition, tissues from a subset of horses from the Maryland Jockey Club with non-FMSI deaths were examined. SD cases were identified based on submission information indicating observation of sudden collapse/death, apparent cardiovascular collapse, lack of FMSI, axial fractures considered secondary to the trauma of collapse and/or impact with other horses (ie, due to loss of consciousness), or horses with an undetermined cause of death. Control horses euthanized due to FMSI, colic, or laminitis identified from a similar time period were included based on availability of cardiac measurement data, standardized cardiac histopathology, and/or availability of tissue for DNA isolation (see below). Horses with axial fractures were considered controls if history/clinical information indicated rearing. Not all data or tissues were available for all animals for this observational case vs control study.
Retrospective evaluation
A retrospective analysis was performed using the NY State AHDC Universal Veterinary Information System and VetView (Athens, GA) to identify NY Thoroughbred and Standardbred racehorse submissions. The provided submission information, clinical history, diagnostic findings (including Henneke body condition score), and available cardiac tissue were reviewed. 22
Prospective evaluation
In addition to evaluating clinical and autopsy information, a detailed cardiac dissection protocol was designed to systematically assess for causes of SD and to standardize sample collection from specific regions of the heart modeled after the “Cardiac Necropsy Guidelines for Cases of Sudden Death in Equines” of the California Animal Health and Food Safety Laboratory System (CAHFS) and the approach to SD in humans. 16,41 Prospectively, this protocol was performed on SD horses and control horses. The heart, with approximately 2 cm of aorta attached, was weighed with clots removed. The coronary artery system was traced for thrombi. To evaluate the myocardium, a series of 1.2- to 2-cm-thick short-axis “breadloaf” cross sections were made through the ventricles from the apex toward the base to a point approximately 4 cm apical to the coronary sulcus, leaving the atrioventricular apparatus intact (Suppl. Fig. S1). 12 Ventricular wall (right ventricle, interventricular septum, left ventricle) thickness measurements were taken at the midpoint between the coronary sulcus and the heart apex, avoiding trabeculae and papillary muscles. Myocardial thickness is subject to variation due to the stage of the cardiac cycle at the time of death and rigor mortis; we opted for wall measurements as dissection of the heart for ventricular weights can obfuscate the landmarks for histologic evaluation of the cardiac conduction system, which was a major study goal. 46 The heart base/atrioventricular apparatus was then dissected according to the flow of blood. Sections of sinoatrial node, atrioventricular node, right ventricular free wall, left ventricular free wall including papillary muscles, mid-chamber interventricular septum, and aorta were fixed with 10% neutral buffered formalin. The sinoatrial nodal region was identified along the cranial (superior) junction of the cranial vena cava and the right atrium by increased epicardial connective tissue with cardiac nerve branches; the sinoatrial node was trimmed by slicing the corresponding region of the sulcus terminalis on the sagittal pane (Suppl. Fig. S2 and Diab et al 16 ). The atrioventricular nodal area was identified in the atrial septum within the boundaries of the coronary sinus (lateral boundary), the septal leaflet of the right atrioventricular valve (caudal/apical boundary), and the cardiac cartilage (medial boundary); the atrioventricular node lies approximately 0.5 cm from the atrioventricular valve attachment and extends to just below the cardiac cartilage (ossified in older animals). 16 Within these boundaries, instead of a series of cross sections, a longitudinal section of the atrioventricular node was taken via a subendocardial butterfly incision in the atrial septum parallel to the right atrial endocardium to create an approximately 0.5-cm slab of endocardium and myocardium (Figs. 1, 2). Both the subendocardial section and medial section were placed cut-side down in cassettes. The histologic features of sinoatrial and atrioventricular nodal tissue have been described by others. 4,16 Histology sections were evaluated by a veterinary pathologist (K.K.) blinded to disease status.
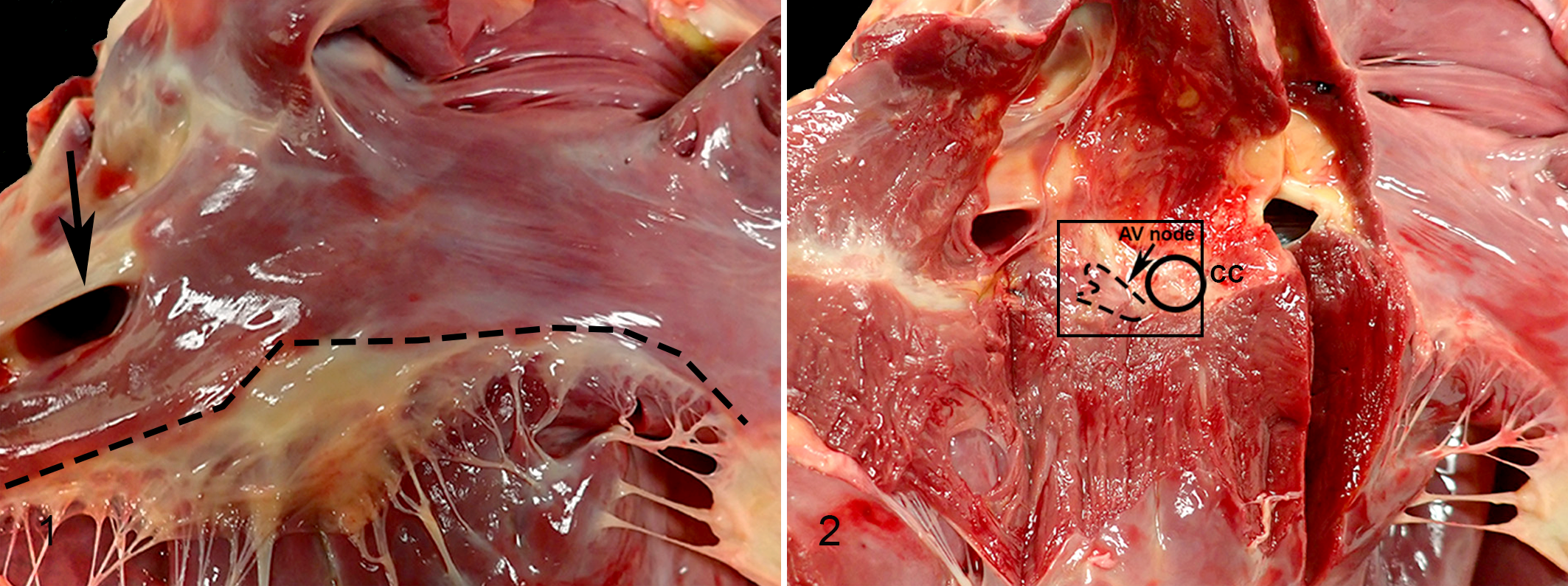
Cardiac landmarks for sampling the atrioventricular node.
SD horses were categorized as EASD if death occurred during exercise or within the subsequent hour; if SD was not associated with exercise, it was considered non-exercise-associated sudden death (NEASD). Horses with noncardiac lesions associated with death were classified as vascular, systemic, or traumatic (axial trauma). Horses with appropriate clinical history of SD after injection were considered injection associated. Horses were considered NEASD/EASD-cardiac if gross or histologic cardiac lesions were considered associated with death based on extrapolations from investigation of human SD, the experience of the single observer (K.K.), and relative to cardiac findings in controls. 3 Horses were considered autopsy negative if gross and histologic evaluation did not reveal significant lesions, autopsy insufficient if there was limited cardiac tissue for histologic evaluation, and open for cause of death if there was no cardiac tissue for histologic evaluation.
DNA was extracted from frozen spleen samples using the Omega bio-tek E.Z.N.A. Tissue DNA Kit (Norcross, GA) according to the manufacturer’s instructions. For fixed samples, two 10-μm-thick scrolls from formalin-fixed, paraffin-embedded (FFPE) spleen were sectioned into sterile Eppendorf tubes and incubated at 60°C overnight with lysis buffer containing proteinase K (Beckman Coulter, Indianapolis, IN). Samples were briefly spun in a fixed-speed mini-centrifuge and cooled to room temperature. Lysate (200 μl) was transferred to a 96-well plate for extraction using a Biomek 4000 Automated Workstation (Beckman Coulter) and Agencourt Formapure Kit reagents (Beckman Coulter). The preset program “Total Nucleic Acid” was used according to the manufacturer’s instructions. Quantity and quality/purity of extracted nucleic acid were measured with a Nanodrop ND-1000 spectrophotometer with absorbance set to 260/280 (Thermo Fisher, Waltham, Massachusetts); the average ratio of absorbance at 260/280 was 1.72; a ratio of ∼1.8 is considered the ratio for “pure” DNA. DNA samples having a concentration less than 20 ng/μl were excluded from genotyping.
Samples were genotyped using GGP Equine 70 K Array (Neogen, Lincoln, NE), which contains >65 000 single-nucleotide polymorphisms (SNPs) and has been used to identify genetic associations in horses. 7,8,30
Data Analysis
Comparison of Thoroughbred SD and control body weight and cardiac measurements was performed using Mann-Whitney test with statistical significance set at P < .05. GraphPad Prism version 6.00 for Windows (GraphPad Software, La Jolla, CA) was used for statistical analysis.
Standardbred cardiac measurements were not compared by statistical analysis due to the low number of cases and controls.
Missing genotypes were imputed by the LD KNNi algorithm implement in TASSEL (5.2.40; Buckler Lab Cornell University, Ithaca, NY) using the default setting. After removing the individuals with missing rate >20% and SNPs with minor allele frequency <0.05, the remaining high-quality SNPs were kept for association analysis. GWAS was performed by software package TASSEL, considering population structure (G + Q; general linear model [GLM]). Population structure was represented by the first 3 principal components calculated using all markers. The observed P values from marker-trait associations were used to plot quantile-quantile (QQ) and Manhattan plots. The significant associations were considered only if the P value was lower than cutoff value P* = .05/N, where N was the total number of SNP markers. The QQ plot, Manhattan plot, and unrooted tree diagram were created by TASSEL.
Results
From November 2013 through August 2017, 316 NY racehorses submitted to the AHDC for autopsy were identified from the NY State AHDC pathology database. In NY, there were 37 SD horses (37/316, 12%), which included male Thoroughbred (n = 18; 6 stallions, 12 geldings), female Thoroughbred (n = 12), male Standardbred (n = 4, all geldings), and female Standardbred (n = 3) horses. In the study period, the majority (36) of SD horses died during exercise or immediately thereafter; thus, 11% (36/316) of NY submissions were EASD. Three EASD cases occurred on MD racetracks during 2016, which included 2 geldings and 1 female Thoroughbred. One Thoroughbred (case 18) died in the stall within 10 minutes of an intravenous injection (NEASD, injection associated) of 25 ml “Liver 7” Sterile Injectable Solution and 10 ml vitamin B12 100. Injection of B12 vitamin products containing iron is rarely associated with SD in horses. 24
The median age (median; minimum, maximum) of Thoroughbreds with SD (4 years; 2, 10) was similar to control Thoroughbred (4 years; 2, 12). While there were few Standardbreds in our cohort, those with SD had a similar median age (5 years; 2, 12) as Thoroughbreds with SD but were younger than Standardbreds with FMSI (8 years; 2, 10).
The circumstances and demographics of racehorse SDs (n = 40) in NY and MD are presented in Table 1. Of the 39 EASD (36 NYRA, 3 MD) cases, 9 died during and 12 immediately after training (21/39, 54%), and 18 horses (46%) died either during (8) or immediately after (10) a race. Over half of the racehorses (22/39, 56%) experienced EASD immediately following exercise during a period of cardiac deceleration.
Demographics and Circumstances of Sudden Death in New York and Maryland Racehorses (November 2013 Through August 2017).
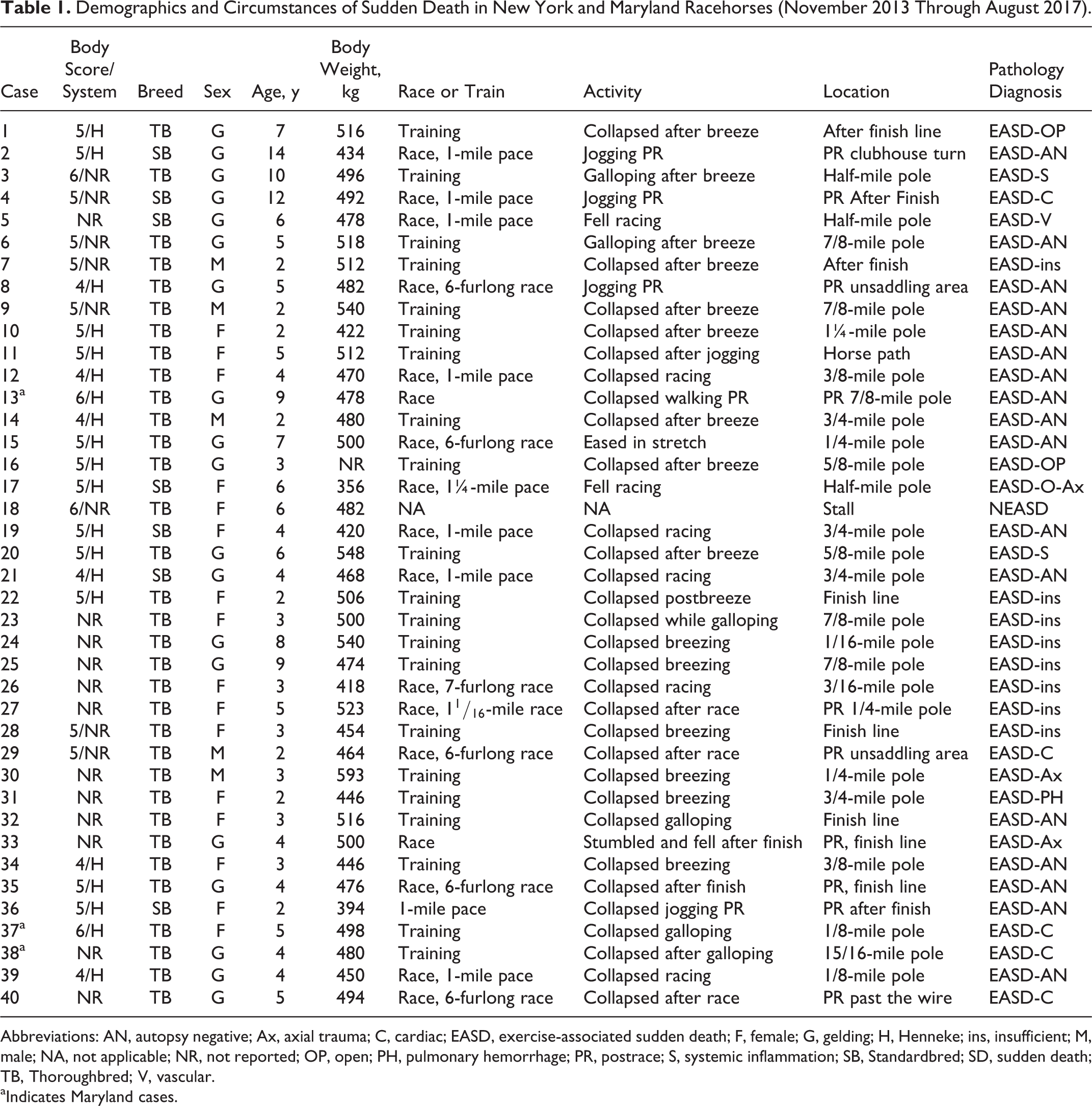
Abbreviations: AN, autopsy negative; Ax, axial trauma; C, cardiac; EASD, exercise-associated sudden death; F, female; G, gelding; H, Henneke; ins, insufficient; M, male; NA, not applicable; NR, not reported; OP, open; PH, pulmonary hemorrhage; PR, postrace; S, systemic inflammation; SB, Standardbred; SD, sudden death; TB, Thoroughbred; V, vascular.
aIndicates Maryland cases.
A median of 5913 (range, 5899–6124) Thoroughbred horses raced at NY racetracks each year between 2013 and 2017. The distribution of SD by month is shown in Fig. 3; SD cases were most numerous during August, which corresponds with peak racing season.
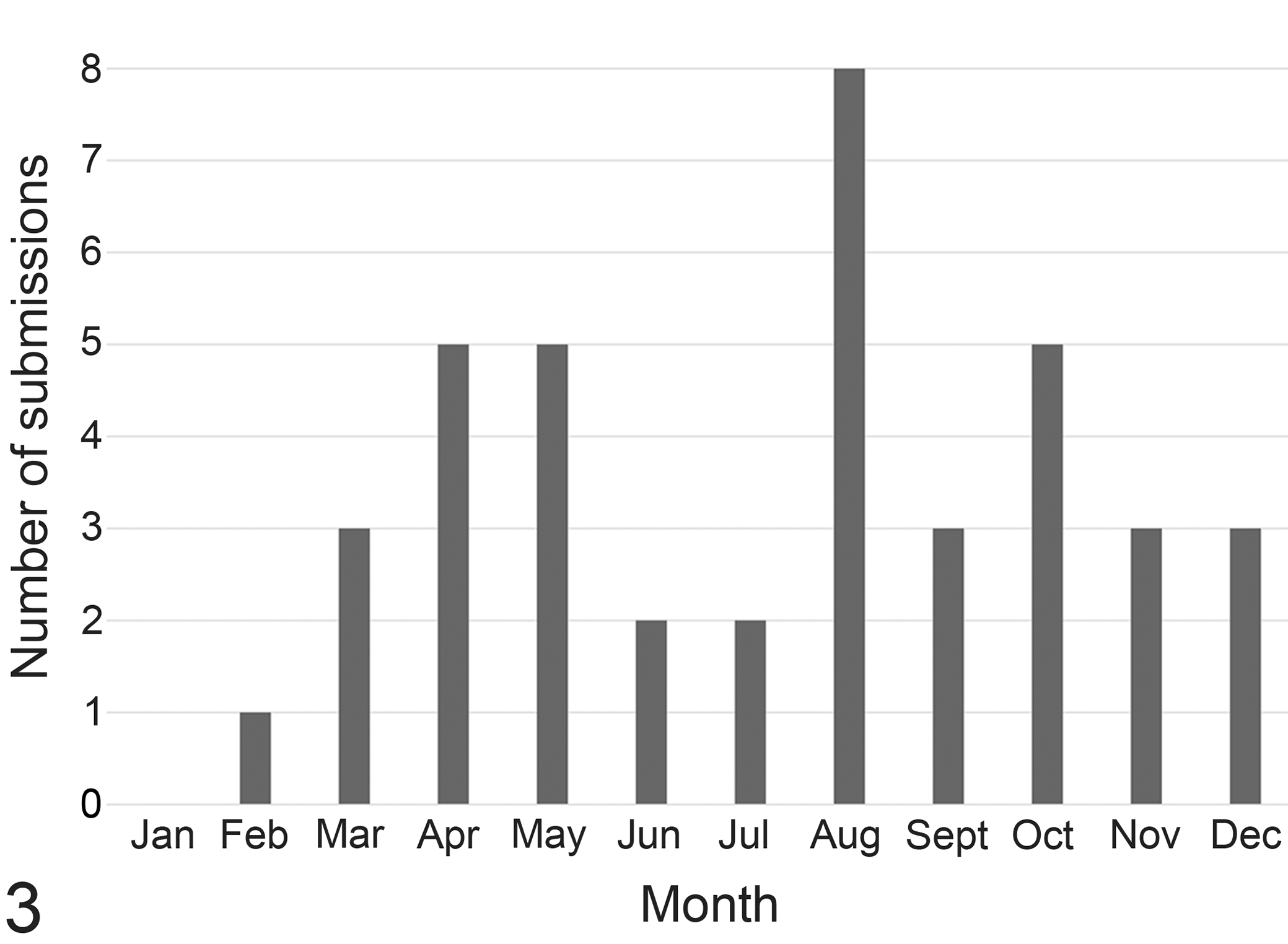
The number of New York/Maryland sudden death submissions for necropsy November 2013 through August 2017 sorted by month.
Demographic data for controls are presented in Supplemental Table S1.
Pathology
Autopsy was performed on SD cases and controls. Thoroughbred horses with SD had a nonsignificant trend toward increased body weight (n = 33; median, 494 kg; range, 368–593 kg) compared to control Thoroughbred horses (n = 65; median, 476 kg; range, 360–556 kg). The cause of death of controls was euthanasia due to FMSI (58/65), colic/enteritis (3/65), postoperative complications (1), axial trauma after rearing (1), laminitis (1), and other trauma (1).
Not all tissues were available for histologic evaluation for all SD cases. Significant lesions or circumstances associated with death were observed in 12 of 37 (32%) SD racehorses and 11 of 36 (31%) EASD cases with cardiac histology. Seven racehorses had lesions indicating noncardiac EASD. One EASD (case 5) had cranial mesenteric artery rupture with hemoabdomen (EASD-vascular). Three EASD horses sustained axial injuries (EASD-axial trauma): 2 Thoroughbred (cases 30 and 33) had cervical vertebral and spinal cord injuries while exercising and a Standardbred horse (case 17) had a skull fracture.
Histology of major organs revealed 2 EASD Thoroughbred horses (cases 3 and 20) with systemic inflammation indicative of infection (EASD-systemic). A NEASD Thoroughbred (case 18) that died immediately following an injection in the stall had a fibrinous jugular vein phlebitis with perivascular hemorrhage (injection-associated death). 15
Exercise-induced pulmonary hemorrhage has been reported as a cause of SD in racehorses. 20 Acute pulmonary hemorrhage was more frequent in horses with SD (53%, 19/36) compared to horses with FMSI (12%, 8/68). Pulmonary hemorrhage was considered the cause of death in only 1 case of EASD (case 31) based on the lack of another identifiable cause of death and estimation that pulmonary involvement (20%–75% of the pulmonary parenchyma) impaired alveolar gas exchange. 33
Beginning March 2015, a detailed cardiac dissection protocol was performed on SD racehorses and controls. Heart measurements (weight, chamber thickness, etc) recorded for EASD horses (n = 21) and controls (n = 53) are presented in Table 2 by breed. The absolute heart weight, heart to body weight ratio, right ventricular wall thickness, and left ventricular wall thickness did not differ between EASD and control horses. There was substantial heart/body weight variation (0.97–2.45 in Thoroughbreds, 1.03–2.49 in Standardbreds) regardless of outcome. The interventricular septum was thicker in Thoroughbreds with EASD compared to control horses (P = .045, Mann-Whitney test). Gross cardiac abnormalities were infrequent but identified in 4 EASD horses. One Thoroughbred (case 40) had an accessory high take-off coronary artery arising from the aorta just distal to the sinotubular junction; the origin did not indicate coronary vascular compression. 2 Three Thoroughbred horses had areas of myocardial pallor that corresponded with fibrosis or myocarditis described below: in 1 horse (case 34), there was a focal 1.5-cm-diameter area in the interventricular septum while the other 2 horses (cases 37 and 38) had multifocal to coalescing pale areas up to 2 × 1 cm in the interventricular septum and/or left ventricular free wall. Otherwise, SD and control horses did not demonstrate gross structural, valvular, or coronary defects or disease.
Cardiac Dissection Heart Measurement Data in Exercise-Associated Sudden Death and Control Horses by Breed.
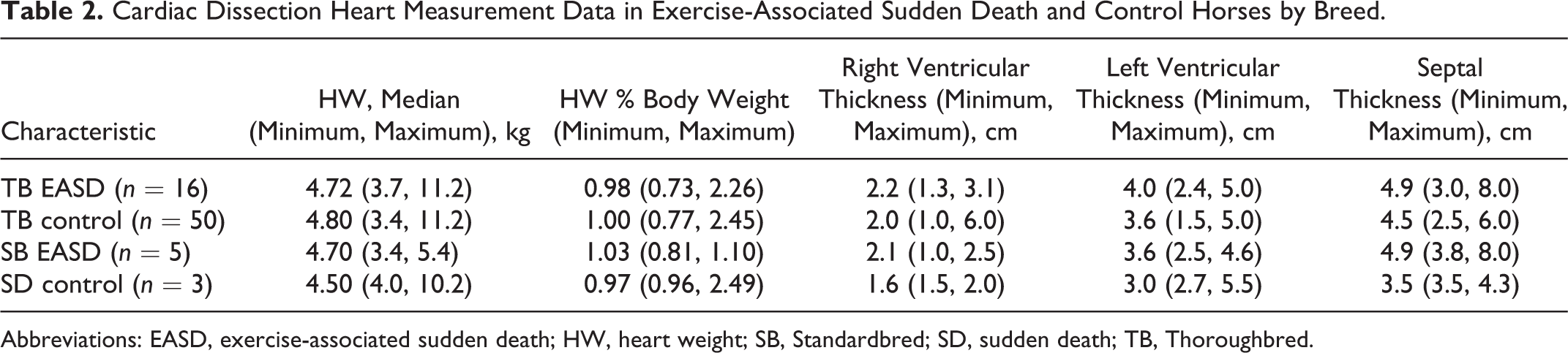
Abbreviations: EASD, exercise-associated sudden death; HW, heart weight; SB, Standardbred; SD, sudden death; TB, Thoroughbred.
Cardiac histology (prospective cardiac pathology and retrospective random sampling) was reviewed for 37 of 40 horses with SD (36/39 with EASD). Standardized cardiac assessment was available for 52 horses, including EASD horses (n = 19) and control horses (n = 33). Heart tissue from 5 additional horses (EASD = 3, control = 2) evaluated during protocol development had cardiac nodal histology and were included in the study; thus, 22 EASD horses had detailed pathologic examination of the heart. Fifteen SD horses (EASD, n = 14) had random cardiac samples available for histologic evaluation. Cardiac tissue was not available for 3 EASD horses (including 1 EASD with axial trauma). Five EASD horses had cardiac lesions not observed in controls (EASD-cardiac). In 3 of these horses, lesions were considered a probable cause or contributory to death, which included regionally extensive (1 × 1.2 cm) papillary cardiomyocyte loss with replacement fibrosis (case 4, Fig. 4); extensive mid-myocardial subacute to chronic left ventricular fibrosis and lymphohistiocytic inflammation, which involved 30% to 50% of left ventricular sections (case 37); and severe chronic myocarditis, which involved 40% to 60% of left ventricular sections (case 38, Fig. 5). In the other 2 horses, lesions were considered a possible cause or contributory to death, which included regional subepicardial coalescing areas up to 0.8 cm of left ventricular cardiomyocyte degeneration and disarray with mild interstitial fibrosis and mural coronary vessel intimal proliferation (case 40, Fig. 6) and multifocal areas of left ventricular subepicardial and subendocardial narrow cardiomyocytes in disarray within mild variable interstitial fibrosis up to 0.5 cm (case 29, Fig. 7). Seventeen EASD cases were considered autopsy negative based on evaluation of sufficient cardiac tissue and lack of significant lesions. Of these, 3 horses had no cardiac findings, 8 had nonsignificant lesions, and 6 had cardiac lesions of likely minimal but undetermined significance, including mild focal (0.3 × 0.1 cm) replacement fibrosis in the left ventricular papillary muscle (case 13); focal neutrophilic and histiocytic interstitial inflammation and hemorrhage of the atrioventricular bundle (case 12, Fig. 8); a loose nonadherent acute thrombus in a vessel adjacent to the atrioventricular node (case 14); mild focal (1 × 0.1 cm) left ventricular epicardial fibrosis and edema (case 21); mild right atrial epicardial, interstitial nodal, and epineurial neutrophilic inflammation and coalescing patchy interventricular septal fibrosis up to 0.3 × 0.1 cm (case 34); and mild subacute atrioventicular nodal phlebitis/periphlebitis (case 36, Fig. 9). Eight EASD horses that lacked significant lesions in other organs did not have sufficient cardiac tissue available for confident exclusion of cardiac disease (EASD-insufficient). Most control horses with cardiac histology were euthanized due to FMSI (33/35). Nine control horses had no cardiac lesions via detailed cardiac pathology, 23 had nonsignificant lesions, and 1 had a cardiac lesion of undetermined significance (atrial mural thrombus, 38C). There were 2 non-FMSI control horses with standardized cardiac pathology: a Thoroughbred euthanized with chronic laminitis had chronic infarction with lymphohistiocytic myocarditis and fibroplasia (22C), and the other Thoroughbred euthanized due to postoperative complications had no cardiac lesions.
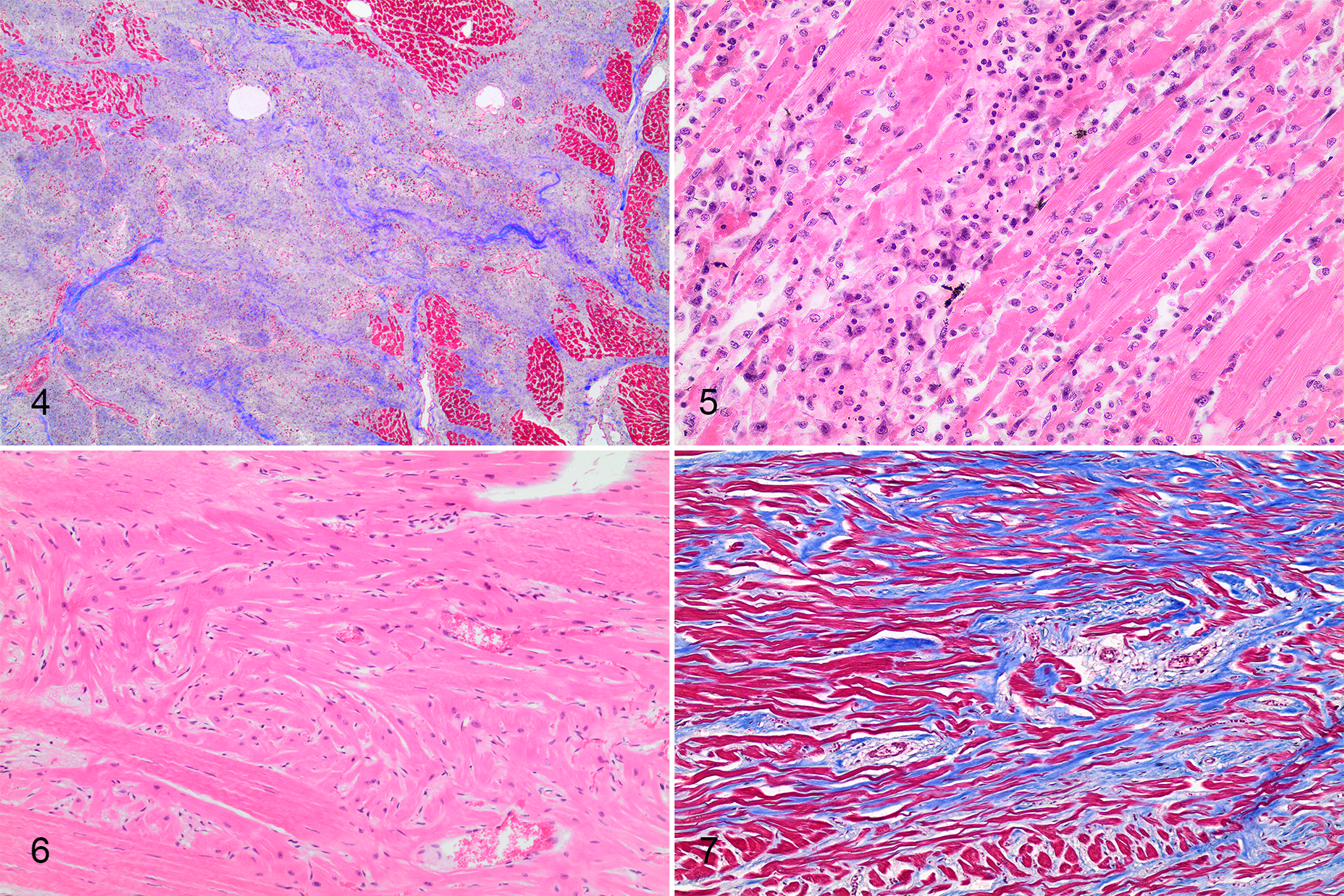
Heart, sudden death in racehorses.
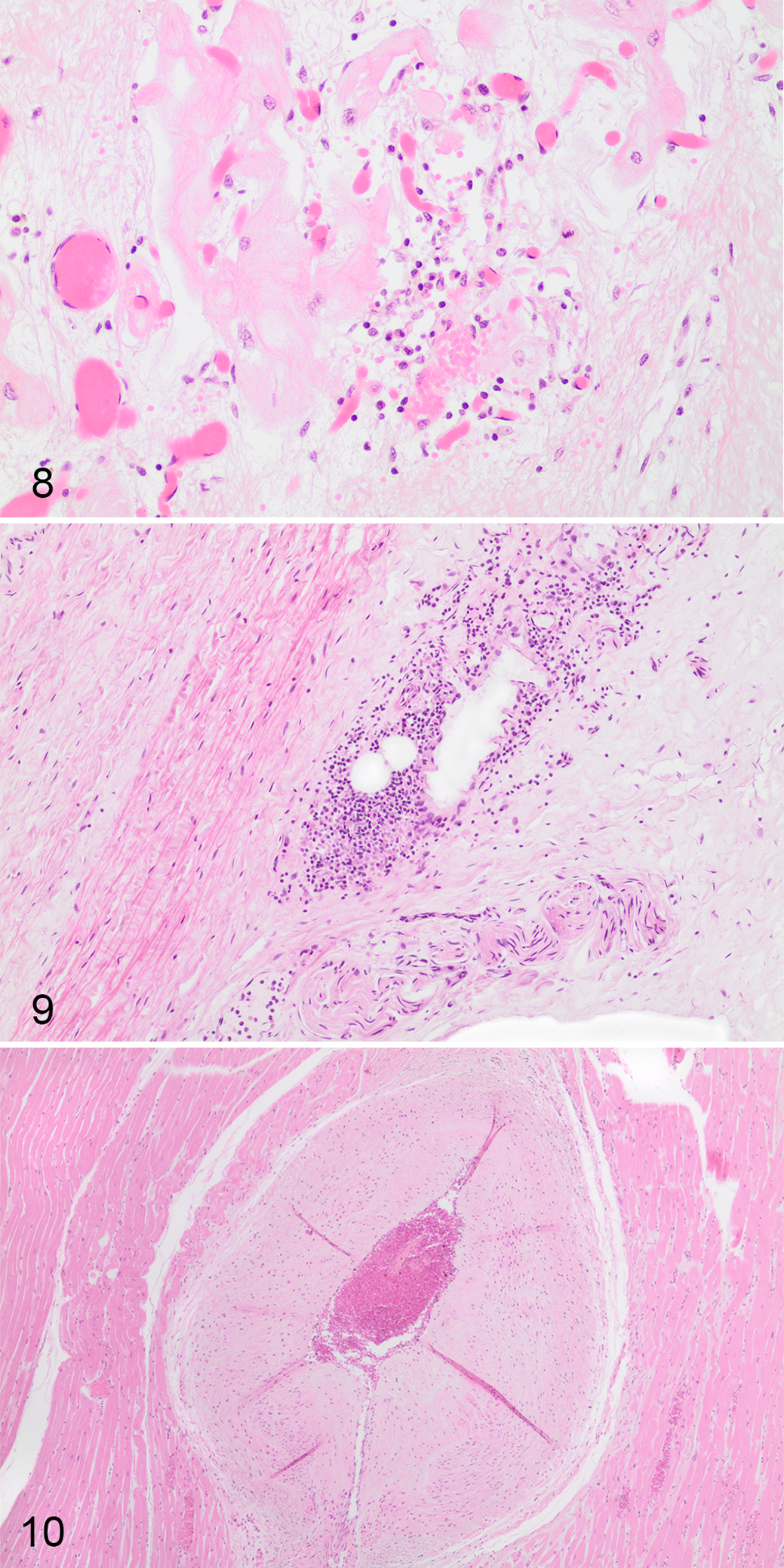
Heart, horses with sudden death and control horses.
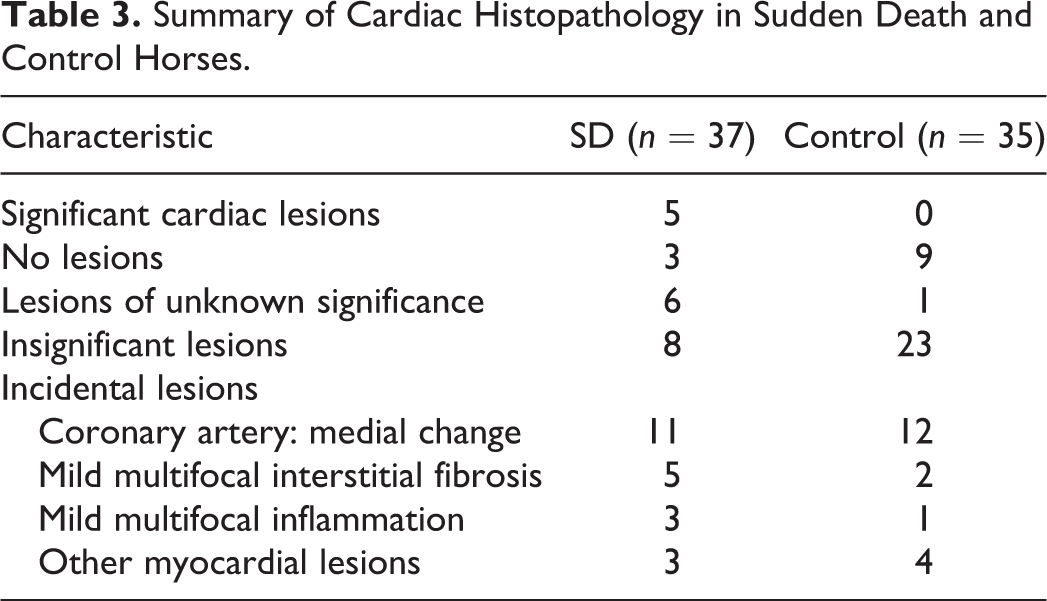
Similar proportions of SD (11/37, 30%) and FMSI (12/35 34%) control horses had variable arteriosclerosis of medium-sized intramyocardial/mural coronary arteries most frequently involving the left ventricular papillary muscles (Table 3). These coronary vascular changes primarily consisted of medial hypertrophy (Fig. 10) with less frequent intimal proliferation. We observed mild multifocal patchy isolated microscopic (foci less than <1 mm) myocardial interstitial fibrosis and inflammation in both cases and controls (Table 3).
Summary of Cardiac Histopathology in Sudden Death and Control Horses.
Full or partial sinoatrial node was available for evaluation for 34 horses, including 15 horses with EASD and 19 controls. Full or partial atrioventricular node was available for evaluation for 39 horses, including 18 EASD horses and 21 controls. Significant cardiac nodal lesions were not observed; lesions of unknown or little clinical significance included arteriosclerosis of sinoatrial (n = 3; 1 EASD, 2 controls) or atrioventricular (n = 1, control) nodal vessels, focal mild nodal interstitial and perineurial inflammation in the sinoatrial (n = 3; 1 EASD, 2 controls) or atrioventricular (n = 5; 3 EASD, 1 control) nodal regions, and single EASD cases with focal mild atrioventricular nodal phlebitis (case 36, Fig. 9) or focal venule thrombosis (case 14). One control horse had multifocal sinoatrial interstitial hemorrhage. The nodal tissue of both EASD (n = 5) and control (n = 10) horses had variation in the amount and proportion of fatty and fibrous tissue. Increased connective tissue was observed in the atrioventricular junction/ventricular crest of both SD and controls.
Although horses with SD were frequently autopsy negative, detailed histologic cardiac assessment revealed cardiac lesions in 18% (4/22) horses with available tissues while random cardiac histologic sampling detected lesions in 7% (1/15) horses.
DNA was isolated from fresh-frozen or FFPE spleen from 26 EASD-autopsy negative, 4 EASD-cardiac, 3 EASD-axial trauma, and EASD horses with exercise-induced pulmonary hemorrhage (1) and systemic inflammation (1). GWAS was performed with DNA from 35 cases (30 Thoroughbreds, 5 Standardbreds) and 34 controls (30 Thoroughbreds, 4 Standardbreds; Suppl. Table S1) using the 65 157 SNP array. FMSI was the cause of death of 32 of 34 genotyped controls. A total of 12 726 SNPs were excluded based on high missingness, monomorphism, or rarity. GWAS did not reveal significant genome-wide results (see supplemental data for QQ plot [Suppl. Fig. S3], Manhattan plots [Suppl. Fig. S4], and unrooted cladogram [Suppl. Fig. S5]). Allele frequencies at the array equine genetic markers were not different between SD racehorses and controls, although additional genotype data are required for an adequately powered study to confirm these data. Case and control Standardbreds clustered into a clade on the unrooted cladogram (Suppl. Fig. S5). There was good concordance observed for the sample with DNA isolated from both FFPE and frozen spleen (23C, Suppl. Fig. S5).
Discussion
The reported proportion of SD in racehorses varies substantially by jurisdiction from 9% to 68%. In NY, SD was 12% and EASD was 11% of submissions, which is similar to previous reports in other areas, including California (9%) 25,29 and the United Kingdom (12%), 29 but less than that reported in Victoria (Australia; 19%–26%), 5,6 Illinois (68%), 18 or Ontario (31%). 15 Although our sample size was relatively small, EASD occurred with equal proportion during racing and training, with more than half of EASD occurring immediately following the exercise period when heart rate transitions from maximal effort to resting levels. Cardiac arrhythmias are reported frequently in Standardbred racehorses during cardiac deceleration in association with sudden rapid increase in vagal tone. 34
This observational study builds upon the work of others by including standardized sampling and the assessment of cardiac tissues in both cases and controls. Our autopsy data, collected using a protocol that assesses recognized causes of sudden death in humans, is similar to other reports that found that racehorses dying suddenly are often autopsy negative. 15,29 In 11 of 36 (31%) NY/MD EASD cases, significant lesions associated with death were observed, which included mesenteric artery rupture (1), axial trauma (2), systemic inflammation (2), exercise-induced pulmonary hemorrhage (1), and cardiac lesions considered nonincidental (5). For the cases with axial trauma and exercise-induced pulmonary hemorrhage, it is difficult to determine the proximate cause of death and exclude whether changes are secondary to another cause of death (ie, arrhythmia, syncope, etc). In this study, the 1 case of NEASD was injection associated. 15
Our cohort had a higher proportion of SD cases that were considered autopsy negative compared to previous reports that identified a definitive cause of death in 53% of SD cases. 29 Lyle et al 29 reported frequent pulmonary hemorrhage, cardiac lesions, and combined cardiopulmonary failure in their multicenter review of postmortem findings. Gunson et al 20 reported exercise-induced pulmonary hemorrhage as a frequent cause of SD in horses on Pennsylvania racetracks (1 in 1500 starts). We attribute differences in determination of cause of death to interpretation of the significance of pulmonary and cardiac lesions. In this cohort, while acute pulmonary hemorrhage was frequent in horses with SD, it was rarely considered the cause of death, consistent with the American College of Veterinary Internal Medicine consensus statement that there is a lack of evidence to indicate that there is increased risk of SD in horses with exercise-induced pulmonary hemorrhage. 23
We observed mild multifocal isolated interstitial fibrosis and inflammation in the myocardium and cardiac nodal tissue of both cases and controls, which we considered incidental findings, consistent with background lesions observed in the normal equine myocardium. 4,16 Consideration of cardiac lesions as the cause of SD is complex and must be considered on a case-by-case basis; however, similar minor patchy fibrosis is not considered a likely cause of SD in human investigations. 41 Ischemic and fibrotic lesions near the sinoatrial node and in the atrioventricular junction have been speculated to be arrhythmogenic and involved in SD in racehorses. 27,28 Although our cardiac pathology findings do not exclude arrhythmic disorder as a cause of racehorse SD, we observed variation in the amount and proportion of connective tissue in the nodal and perinodal tissue of both SD and control horses. Our data suggest that caution be used in the interpretation of nodal fibrosis unless there are supporting clinical findings (auscultation or electrocardiography) to indicate arrhythmia or heart block as a cause of death. We agree with Diab et al 16 that microscopic lesions in the heart of SD horses do not necessarily indicate that these lesions are the cause of death. Regardless, the conduction system must be evaluated when investigating SD to exclude structural abnormalities (ie, any noncoronary cardiac defects or abnormalities). Consistent evaluation of these tissues, particularly in the context of electrocardiographic and clinicopathologic data, will progress our understanding of the role of these lesions in equine SD.
SD in humans is frequently associated with coronary atherosclerosis and its consequences. In our cohort of racehorses, SD was not associated with coronary artery pathology. Medial hypertrophy or arteriosclerosis (nonspecific thickening of arterial walls) of intramyocardial coronary arteries, most frequently involving the left ventricular papillary muscles, was observed in a similar proportion of SD and control horses and thus was not considered a significant lesion in racehorses or a cause of SD. Arteriosclerosis is considered a senile change but can be associated with myocardial infarction or hypertension in the dog and has been considered a contributing factor to myocardial fibrosis and SD in horses. 10,28,39,47 SD is associated with coronary artery anomalies in children and young adults. 41 Thus, for the exclusion of coronary artery disease (eg, structural abnormality or acute thrombosis), the coronary arteries should be evaluated when investigating SD in horses.
Drug use is an important cause of SD in humans; drug/chemical administration may also have an important role in the SD of racehorses. 31 Chemical screening was not performed in this study. Diab et al 16 have described the possible roles of intoxicants in equine SD and the challenges of toxicology testing, particularly limited by unavailability of a single analytical screening technique or combination of techniques.
As cardiac examination must be guided by history and clinical information, there is no single perfect method for cardiac assessment. The cardiac protocol used in this study includes myocardial breadloafing (multiple parallel sections) and unique longitudinal sectioning of the conduction system designed to preserve cardiac nodal morphology and allow efficient histologic examination of cardiac tissues. Although EASD horses were frequently autopsy negative, this detailed histologic cardiac assessment more frequently revealed nonincidental cardiac lesions compared to random sampling. Furthermore, it is difficult to interpret findings and confidently exclude cardiac disease from limited, random sampling. Breadloafing of the myocardium allows for thorough evaluation for infarcts and infiltrative disease otherwise overlooked when assessment is limited to the evaluation of the cardiac chambers. In this study, lesions, when present, were observed upon cross-sectional breadloafing of the interventricular septum and left ventricular free wall.
Heart weights for this cohort of racehorses were consistent with previous reports indicating physiologic ventricular hypertrophy and increased heart/body weight with training and exercise. 11,17,19,21 Heart/body weight records for our cohort of racehorses indicate substantial cardiac size variation (>2-fold variation) regardless of outcome (SD vs control), which is likely related to the stage of training or other variables and not a pathologic change in most cases. 48 Measurement and weight guidelines for autopsy evaluation of equine cardiac disease are not available; we opted for wall thickness measurements to preserve conduction system landmarks, acknowledging that myocardial thickness may vary due to the stage of the cardiac cycle at time of death and rigor mortis. 13,32,46
One of the study goals was to identify genetic loci associated with SD in racehorses to guide identification of associated genes, inform test development, and/or educate breeding decisions and ultimately reduce SD prevalence. While this GWAS was underpowered, a global assessment of the genetic architecture did not identify a location or architecture of traits linked to SD phenotype in our cohort. Patterns may not have been apparent due to multiple, unshared mutations or limited array coverage at disease loci. These data do not exclude a role for genetics in racehorse SD but suggest that there are likely other factors involved. The common lethal inherited channelopathies of long QT syndrome and catecholamine polymorphic ventricular tachycardia (CPVT) have striking similarities to racehorse SD: genetic testing is available for at-risk or affected humans, but these syndromes are associated with mutations in several large genes. CPVT in humans manifests as exercise-induced fainting/syncope or SD, often in juveniles, accounting for about 15% of autopsy-negative SD; half of the CPVT cases are associated with mutations in cardiac ryanodine receptor 2 (RYR2). 43,44 Interestingly, a GWAS of French performance horses identified a quantitative trait locus related to the performance of families of horses, 0.55 Mb from the RYR2 gene. 8 More genotype data are needed to determine the contributions of genetics to SD.
In this study, genotyping was performed on DNA isolated from FFPE samples similar to recent studies that successfully used FFPE samples for GWAS. 14,45 The deployment of technology that allows superior yield and purity of total nucleic acid from archived FFPE samples can advance genetic studies of relatively rare traits.
Our studies did not reveal a cause of death in most SD cases but support the ongoing industry effort to ensure the safety and health of racehorses and advance our understanding of the pathology and genetics of SD.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819829529 - Cardiac Pathology and Genomics of Sudden Death in Racehorses From New York and Maryland Racetracks
Supplemental Material, DS1_VET_10.1177_0300985819829529 for Cardiac Pathology and Genomics of Sudden Death in Racehorses From New York and Maryland Racetracks by Alex Molesan, Minghui Wang, Qi Sun, Virginia Pierce, Rhiannon Desideri, Scott Palmer, Rory Todhunter and Kathleen Kelly in Veterinary Pathology
Footnotes
Acknowledgements
We are grateful to the pathologists, residents, and Necropsy and Histology Laboratory staff of the New York State Animal Health Diagnostic Center with special thanks to John Hatfield. We thank Brian Caserto, Samantha Brooks, and Adam Boyko for valuable discussions. We thank the NY and MD track veterinarians.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support included research seed funding sponsored by the Cornell University Center for Vertebrate Genomics.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
