Abstract
Metal phosphides, particularly zinc and aluminum phosphide, occasionally poison horses and other equids following their use as rodenticides and insecticides. Grain-based aluminum phosphide baits are used to control rodents such as prairie dogs. The clinical course in intoxicated horses is short (<24–48 h), and animals may be found dead. Hepatic lesions caused by phosphine poisoning are not well described. Laboratory confirmation depends on detecting phosphine gas in gastric contents. Eight horses and a mule were exposed to zinc phosphide used to control prairie dogs on a Wyoming ranch. Three of 9 exposed equids developed some combination of sweating, ataxia, anxiety, and colic; 2 died acutely, and 1 recovered. A diagnosis of zinc phosphide was made by detecting phosphine in stomach contents from a horse and a mule. The liver was pale and swollen in the affected horse, which died after a clinical course of ~12 h. Other changes were generalized congestion and edema, pulmonary edema, and acute cerebrocortical edema. There was diffuse hepatocellular microvesicular steatosis. Similar histologic lesions were present in 7 equine livers from 2 previously published episodes of metallic phosphide poisoning. Older lesions (>24 h of clinical signs) had centrilobular hepatic necrosis with congestion and a mixture of microvesicular and macrovesicular steatosis. Phosphine poisoning should be considered in horses that die acutely and are found to have steatosis, either with or without hepatocellular necrosis.
Zinc phosphide, a rodenticide, and aluminum phosphide, an insecticidal fumigant, are commonly used agricultural chemicals. They are inexpensive, lack long-term residue risks, and are considered to pose only modest risks of secondary poisoning (Tietjen HP. Zinc phosphide—its development as a control agent for black-tailed prairie dogs. United States Department of Interior, Washington DC. Fish and Wildlife Service Special Science Report Wildlife 195, 1976).
7
Some non-target species, particularly carnivores, are largely protected by an effective vomit reflex that results in prompt elimination.
12
Prairie dogs (
Although used widely, zinc and aluminum phosphides have disadvantages. They are fast-acting, nonspecific, and lack an effective antidote. Intoxication of domestic animals and wildlife is well documented (Erickson W, Urban D. Potential risks of nine rodenticides to birds and nontarget mammals: a comparative approach. Office of Prevention, Pesticides and Toxic Substances. United States Environmental Protection Agency, Washington DC, 2004. Available from: https://goo.gl/C3PpsS).3,9,15 Treatment is supportive, with high case-fatality rates once sufficient phosphine gas is generated in the moist acidic environment of the stomach. 17 Some commercial products containing zinc phosphide have a restricted-use label. Their use is limited to formally trained, certified applicators employed to control small mammals in agricultural settings. Other formulations of zinc phosphide can be purchased over-the-counter or online in the United States.
Horses and other equids cannot vomit. This puts them at particular risk when given access to cereal grains coated with metallic phosphides. Two published episodes resulted in the death of 25 and 27 horses at <1–2 d after ingestion.8,21 Most reported poisonings involved fewer horses, typically 1 or 2 (Rimoldi G, et al. Phosphide poisoning in 4 equids in California. Proc Am Assoc Vet Lab Diagn Ann Mtg; Oct 2013; San Diego, CA).6,10,11
Little information exists about lesions that develop following acute metallic phosphide intoxication in horses, as is the case for other animal species. Reviews and veterinary texts state that the principal lesions are pulmonary edema, with congestion and hemorrhage in multiple organs.4,12,15,16 Gastric or enteric contents may smell of decaying fish, the result either of phosphine gas or contaminants in the pesticide formulation. 17 Phosphine gas is, however, poisonous (National Institute of Occupational Safety and Health [NIOSH]. Preventing phosphine poisoning and explosions during fumigation. Columbus, OH: NIOSH, 1999. NIOSH publication 99-126. Available from: https://goo.gl/pQsA39). Pathologists should not attempt to detect phosphine gas by inhaling odors from the gastric contents of animals exposed to metallic phosphides. Myocardial, renal, and hepatic lesions occur but are considered inconsistent and nonspecific. 15 We document hepatic steatosis in a horse poisoned by zinc phosphide. Similar lesions occurred in another 7 horses exposed to metal phosphides in 2 previously reported episodes.6,8 This suggests that detecting acute steatosis in horses that die acutely has diagnostic value given that it may prompt testing for phosphine gas, assuming that gastric contents were saved for analysis.
The affected ranch is in Thunder Basin, eastern Wyoming. After pre-baiting with untreated oats, zinc phosphide was used in October 2016 to control a colony of black-tailed prairie dogs on a 200-hectare pasture. The user was a certified applicator employed by the U.S. Forest Service to control prairie dogs on public lands. A restricted-use formulation of zinc phosphide–coated steam-rolled oats was obtained from the county’s weed and pest office (Zinc Phosphide Prairie Dog Bait, SD Dept. of Agriculture, Rodent Control Fund, Pierre, SD). The product’s data sheet warns of toxicity to dogs, cats, birds, fish, and non-target wildlife species. Zinc phosphide–treated oats (~4 g) were placed inside active burrows. Seven horses and a mule were returned to the treated pasture 1 d after application. The owner assumed that risks to horses were minimal. His understanding was that treated oats would be inaccessible to livestock and that the risk of poisoning decreased with time. He understood that zinc phosphide degrades naturally in the environment and is effectively gone within 2 wk of coming into contact with soil.
The 8 animals were seen daily by the owner for 15 d after treated oats were set out; all appeared healthy. The following day, the owner found 1 of 7 horses (equid 1) recumbent with severe colic, whole body sweating, muscle fasciculations, depression, and apprehension. A mule in the group was dead. Another horse in the group exhibited milder signs, chiefly depression and patchy sweating. A clinical veterinarian treated equid 1 with intravenous electrolytes, and attempted unsuccessfully to pass a nasogastric tube. The horse’s condition deteriorated and it died that afternoon after a clinical course of ~12 h. The remaining 6 horses were treated orally with activated charcoal and mineral oil. The mildly affected horse recovered. None of the remaining 5 horses developed clinical signs.
The veterinarian performed a field autopsy on the mule. No distinctive gross anatomic lesions or unusual odors were recognized. The stomach contained ~1 L of oats. A sample of gastric contents, including oats, and an unfixed piece of lung were submitted for laboratory analysis. The carcass of equid 1, a 9-y-old, 595-kg Quarter Horse gelding, was submitted for laboratory autopsy. There was moderate diffuse hemorrhage and edema throughout the carcass. The mildly distended stomach contained grass and some oats (Fig. 1). The glandular mucosa of the stomach and mucosa of small and large bowel (left and right dorsal colon only) were diffusely congested. Lungs were moderately congested and edematous. No abnormal odors were recognized, including from stomach or intestines. The liver was pale and slightly swollen. Representative tissue and organ samples, including brain, were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned 5-μm thick, and stained with hematoxylin and eosin. The principal microscopic change was in the liver. There was moderate diffuse microvesicular steatosis with predominantly portal-periportal hemorrhage (Fig. 2). Hepatocytes were swollen, with centrally located nuclei surrounded by innumerable 1-μm vacuoles; necrosis was absent (Fig. 3). The lipid nature of vacuoles was confirmed in frozen sections using Oil Red O and by post-fixation in osmium tetroxide (Fig. 4). Vacuoles were negative for carbohydrates as determined by staining frozen sections using the periodic acid–Schiff method. Other changes were moderate acute perivascular hemorrhage in brain, mild multifocal intracellular edema in cerebral cortex, moderate diffuse pulmonary congestion with hemorrhage and intra-alveolar edema, and scant hemoglobin-like casts in renal tubules. No lesions were recognized in heart.
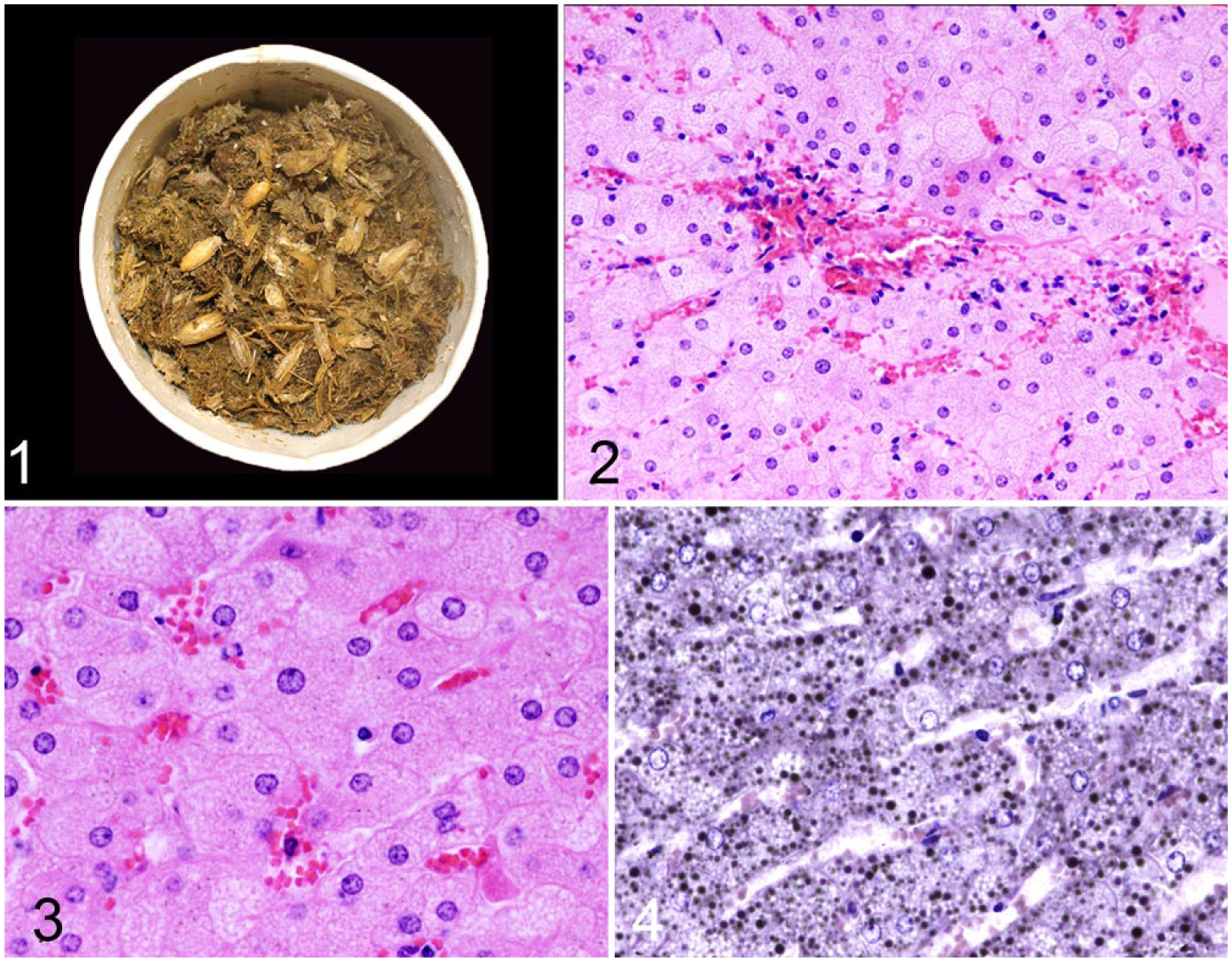
Gastric contents from equid 1. Note presence of oats.
Zinc and aluminum phosphides react rapidly with sulfuric acid and produce phosphine gas, which can be used as a rapid qualitative test. Weighed portions of gastric contents from equid 1 and the dead mule were submerged in dilute sulfuric acid and heated. Chemically specific detector tubes were used to determine the presence or absence of phosphine gas (HazMat gas detection system and phosphine 0.1/a detection tubes, National Draeger, Pittsburgh, PA). A phosphine gas detector tube was connected to each reaction vessel. A color change from yellow to pink signaled presence of the gas. Positive results were corroborated by gas chromatography–mass spectrometry performed (Dr. J Buchweitz, Toxicology section, Diagnostic Center for Population & Animal Health, Lansing, MI). 19
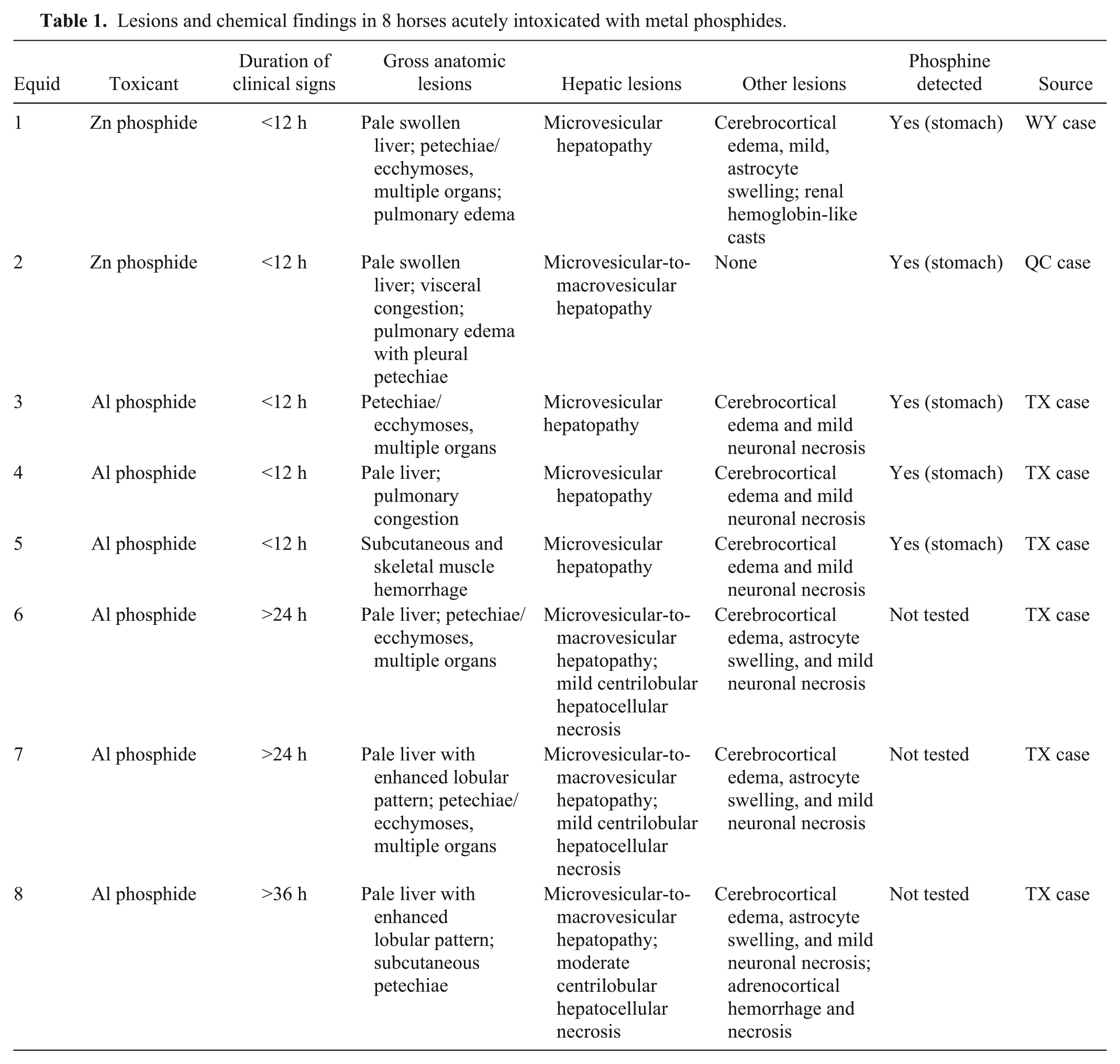
To determine whether hepatic steatosis was a feature in other horses intoxicated with metallic phosphides, archived sections of liver were reexamined from 2 previously reported episodes of phosphide intoxication. One involved a 2.5-y-old Standardbred filly (equid 2) euthanized after developing clinical signs suggestive of phosphide intoxication (profuse sweating, muscle tremors, anxiety, hyperesthesia, convulsions, and cardiac arrhythmia). 6 The other episode involved 66 horses fed a pelleted ration contaminated with aluminum phosphide (equids 3–8). 8 Metallic phosphide poisoning was confirmed in 4 of the 7 horses by detecting phosphine gas in gastric contents; the remaining 3 horses were not tested, given that they were part of the same episode (Table 1). Histologic findings in equids 1–8 are summarized. All 8 had microvesicular steatosis, with macrovesicular steatosis in those with clinical signs of longer duration. Horses with clinical signs for >24 h also had diffuse centrilobular hepatocellular necrosis of variable severity (Fig. 5). Additional microscopic features were scant intracytoplasmic eosinophilic bodies (cytosegrosomes) and swollen individualized hepatocytes (Figs. 6, 7).
Lesions and chemical findings in 8 horses acutely intoxicated with metal phosphides.
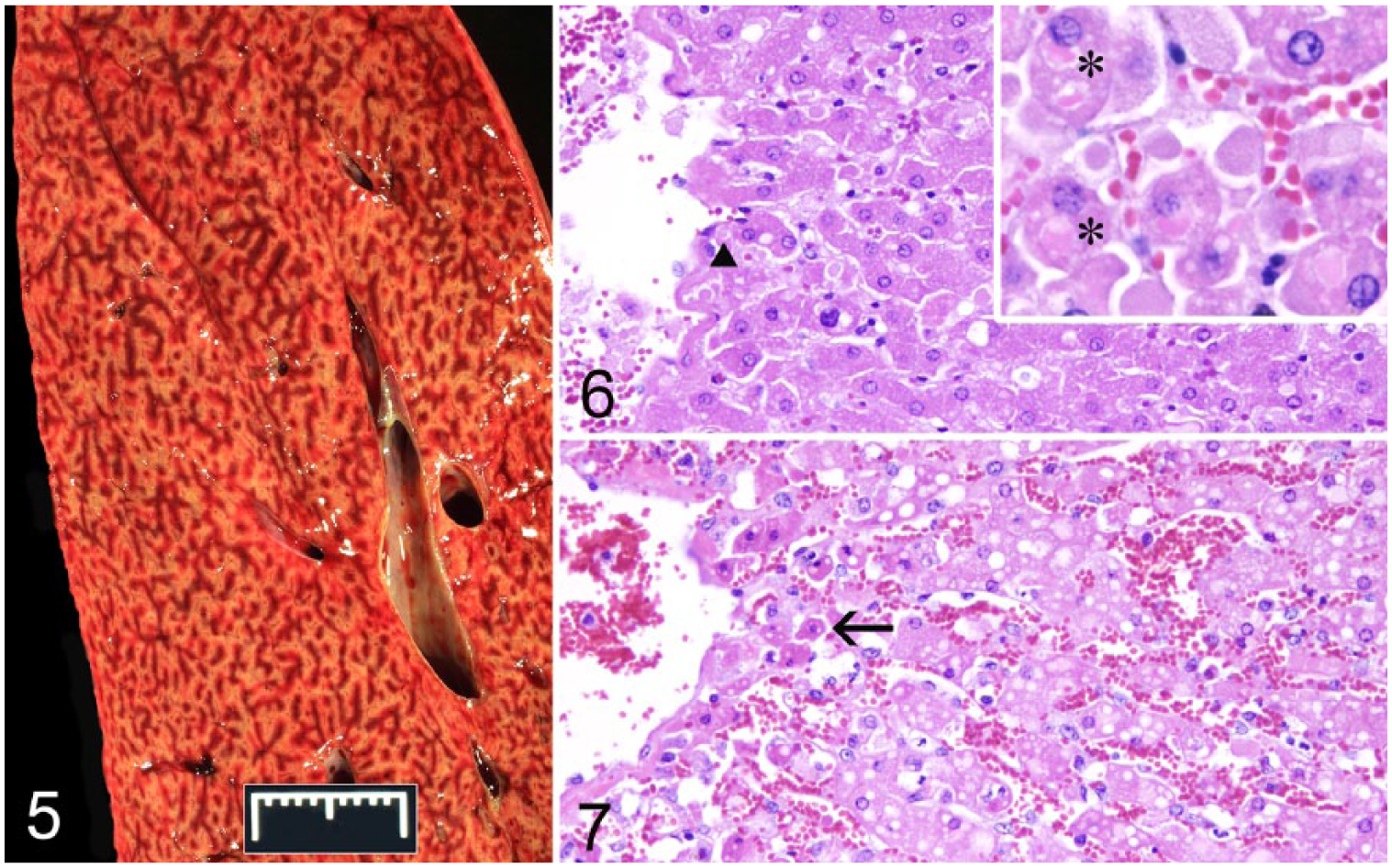
Moderate diffuse centrilobular hepatocellular necrosis manifested by enhanced lobular pattern in equid 8.
Macrovesicular steatosis of the liver is a common physiologic and pathologic finding that provides limited diagnostic leverage. 13 By contrast, hepatic microvesicular change (microsteatosis) is less common and considered more ominous. 13 It is characterized by multiple lipid vacuoles that are smaller than hepatocyte nuclei, which are not displaced by their presence. 5 Microvesicular change in people is associated with inhibition of mitochondrial β-oxidation of fatty acids.2,5,13 It occurs following intoxication with various drugs and toxins, including valproic acid, parenteral tetracycline, salicylates, hypoglycin A, and metallic phosphides.5,13,20
Hepatic steatosis may be a useful diagnostic clue when horses die acutely or are found dead in situations in which exposure to metallic phosphides is a possibility. Reports and reviews of zinc phosphide intoxication in horses describe hepatic lesions such as a “pale yellow liver,” “liver damage,” “fatty change,” “hepatic lipidosis,” “cloudy swelling and fatty degeneration,” hepatocellular lipidosis, and centrilobular necrosis.4,6,8,12,15,16 A high proportion of human patients dying of metallic phosphides develop “fine isomorphic cytoplasmic vacuoles” in hepatocytes. 20 An early study reported changes consistent with microvesicular lipidosis (“minute vacuoles”) in hepatocytes of experimentally poisoned poultry. 18 A 2015 case study of an intoxicated dog reported microvesicular hepatic steatosis and hepatocellular necrosis. 14 The toxicity of metallic phosphides is mediated by the formation of phosphine gas in the digestive tract, resulting in inhibition of mitochondrial cytochrome C oxidase complex IV, the last enzyme of the mitochondrial respiratory chain. This blocks oxidation of NADH to NAD+, impaired fatty acid beta-oxidation of acyl-CoA to acetyl CoA, and rapid energy depletion. 17 Microvesicular lipidosis comports well with this mechanism for intoxication. Free radical formation is also thought to occur. Additional mechanisms may contribute to the short clinical course when intoxication is lethal. 17 A pathologist’s suspicion of metallic phosphide intoxication may be strengthened by concurrent findings of cerebrocortical edema, pulmonary edema, and myocardial necrosis.14,15,20 This can be confirmed by analyzing gastric contents for phosphine gas, assumed they were saved following postmortem examination.
An advantage of metallic phosphides is that they degrade quickly in the environment (within weeks; https://goo.gl/C3PpsS). Rapid degradation depends on several factors, particularly moisture. In the Wyoming case, October 2016 was unusually dry. Zinc phosphide remains stable for months to years when kept dry (Tietjen HP. Zinc phosphide). The rancher’s presumption that it degrades in a fast predictable way was therefore in error, as was his assumption that placing toxic oats in burrows guaranteed inaccessibility to horses.
Phosphine gas is hazardous to people (https://goo.gl/pQsA39). Medical personnel have been poisoned while treating canine and equine patients.1,6 There appears to be no report of pathologists or other laboratory staff being injured while performing autopsies on animals poisoned by metallic phosphides. Nevertheless, human risk is a consideration when horses are examined postmortem and metallic phosphides may be involved.
Footnotes
Acknowledgements
We thank the Wyoming rancher for background information. We thank Rebecca Ashley for histologic sections, and Dr. Drolet, author of reference 6, for permission to reexamine tissue sections from equid 2.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Institute of Food and Agriculture, U.S. Department of Agriculture, under awards WYO-568-16 and WYO-564-16.
