Abstract
A myositis syndrome has been recognized for more than a decade in California sea lions (CSLs; Zalophus californianus) but a detailed description of the lesions and potential causes of this condition is lacking. The tissues of 136 stranded CSLs with rhabdomyositis were examined. Rhabdomyositis was considered incidental in 67% (91/136) of the CSLs, and a factor contributing to the animal stranding (significant rhabdomyositis) in 33% (45/136). Of the 91 cases with incidental rhabdomyositis, lesions consisted of a few small foci of lymphohistiocytic inflammation. Of the 45 cases with significant rhabdomyositis, 28 (62%) also presented with major comorbidities such as leptospirosis (2 animals) and domoic acid toxicosis (6 animals), whereas 17 (38%) had severe polyphasic rhabdomyositis as the only major disease process associated with mortality. In these animals, most striated muscles had multiple white streaks and diffuse atrophy. Microscopically, there was myofiber necrosis surrounded by lymphocytes and histiocytes admixed with areas of myofiber regeneration, and/or moderate to severe rhabdomyocyte atrophy usually adjacent to intact Sarcocystis neurona cysts. At the interface of affected and normal muscle, occasional T lymphocytes infiltrated the sarcoplasm of intact myocytes, and occasional myofibers expressed MHCII proteins in the sarcoplasm. S. neurona antibody titers and cyst burden were higher in animals with significant polymyositis antibody titers of (26125 ± 2164, 4.5 ± 1.2 cysts per section) and active myonecrosis than animals with incidental rhabdomyositis antibody titers of (7612 ± 1042, 1.7 ± 0.82 cysts per section). The presented findings suggest that S. neurona infection and immune-mediated mechanisms could be associated with significant polyphasic rhabdomyositis in CSLs.
Myositis has been described in a wide range of wild avian and mammalian species. 23,28,35 Similar to domestic animals, many inflammatory myopathies of wildlife have a traumatic, nutritional, toxic or infectious origin with the relevance of each of those factors depending on the animal species, habitat and biology of the studied populations. 11,23,27,35 In marine mammals, myositis has been sporadically reported in cetaceans and pinnipeds, and infectious agents such as Clostridium perfringens, Toxoplasma gondii, Sarcocystis neurona, and Klebsiella pneumoniae are some of the reported causes. 1,3,6,28,30 However, in most of these cases, muscle inflammation is secondary to systemic infection, which primarily targets other organs and tissues, and pathogens that only affect the muscle of marine mammals have been rarely reported. 1,5
For more than a decade, a myopathy syndrome has been recognized in California sea lions (CSLs; Zalophus californianus) stranded along the central California coast. 4,6 These animals are usually very depressed, with variable muscle wasting and sometimes clinical pathology findings that suggest muscle inflammation and necrosis (Whoriskey et al, 2017, unpublished data). 1,6 In some of these animals, Sarcocystis neurona infection has been associated with myositis, and antiprotozoan and corticosteroid therapy have proved effective. 6 However, the cause of this condition in most cases has not been determined and the role of potential contributory factors other than protozoan infection has not been studied.
Protozoal infections are a well-known cause of morbidity and mortality among marine mammals, especially along the west coast of North America. 2 T. gondii and S. neurona have been recognized for more than 2 decades as a significant cause of disease and strandings of Southern sea otters (Enhydra lutris), and sporadically of Pacific harbor seals (Phoca vitulina). 12,19,23,25 In these host species these protozoa cause mainly meningoencephalitis although myositis has been described in cases with systemic infection. 11 Recently, a retrospective study identified the most common presentations of T. gondii in CSLs, which included meningoencephalitis and rarely myositis. 5 The role of S. neurona as agent of systemic disease or primary myopathy in CSLs, however, has not been extensively studied, although clinical cases in free-ranging and captive animals suggest that myositis and secondary renal damage due to S. neurona infection can occur in CSLs. 1,6 In addition, the seroprevalence of this protozoan in stranded CSLs populations is approximately 5.4%, 4 suggesting an important level of exposure to this pathogen. Therefore, the objective of this investigation was to characterize the polyphasic inflammatory myopathy of CSLs by retrospectively investigating cases of myositis/myopathy in animals stranded along the central California coast and to explore the potential cause(s) of this condition.
Materials and Methods
Animals
The stranding data base of The Marine Mammal Center (TMMC), Sausalito, California, was assessed for CSLs stranded between 2001 and 2016 along the central coast of California (37° 42’N, 123° 05’W to 35° 59’N, 121° 30’W), and admitted to the rehabilitation hospital and necropsied. The full clinical records, and necropsy and histopathology reports of the cases initially identified in the database search were reviewed to confirm the diagnosis of myositis and to register the reported clinical signs, postmortem findings and ancillary testing results of each case. Animals included in the study were all those that met the following inclusion criteria: full necropsy and histopathology reports were available; inflammatory infiltrate, necrosis or muscle atrophy were observed microscopically in at least 2 foci; and more than 2 striated muscles were examined histologically.
Necropsy and Histopathology
For all animals, complete postmortem examinations were performed within 12 to 24 hours after death, using a standard protocol. 9 Representative tissue samples from all major organs were collected for histopathology including in most cases brain, tongue, tonsils, pharynx, esophagus, heart and skeletal muscles (diaphragm, pectoral, masseter, temporalis, intercostal, gluteal, laryngeal muscles, longissimus and/or psoas muscles). Tissues were fixed in 10% neutral buffered formalin and sent to 4 different veterinary diagnostic laboratories. As part of the standard diagnostic work up on each case, tissues were routinely processed and stained with hematoxylin and eosin and selected sections were stained with Gram and acid fast stains (n = 21) and PAS reaction (n = 15). All cases were re-reviewed by 2 pathologists for consistency (K Colegrove, M Seguel).
Grading and Case Definitions
Muscle necrosis was defined as loss of rhabdomyocyte cellular detail and cross striations with hypereosinophilia and/or cytoplasmic fragmentation, cupping and/or lysis. Muscle inflammation was defined as presence of inflammatory cells associated with changes in cellular or tissue architectural details in the area assessed. Muscle necrosis and inflammation were graded as mild if foci involved a few muscle fibers, moderate if foci extended through several myofibers occupying on average an area of less than 30% of multiple muscle fascicles, or severe if large areas (>30%) of several muscle fascicles were affected. The protozoan cyst burden in the striated muscle sections was estimated by counting the number of cysts in the section most severely affected based on preliminary histopathological assessment of muscles. During preliminary histologic assessment of tissues, a significant degree of lymphoid hyperplasia was noted in spleen and lymph nodes (which was considered unusual based on our experience with tissues from stranded CSLs). In order to determine if lymphoid hyperplasia was correlated with myositis severity this histologic change was graded in the spleen and lymph nodes by scanning the complete tissue sections at 100X and 200X magnification. Based on this assessment, lymphoid hyperplasia was graded in the spleen according to the proportion of white pulp vs red pulp (mild = 40–50%, moderate 50–60%, severe >60% white pulp), and in the lymph nodes based on the proportion of follicles compared to rest of the parenchyma (mild = 30–40%, moderate 40–50%, severe >50% follicles).
Animals with moderate or severe myositis were included in the group of “significant rhabdomyositis” while animals with mild necrosis and inflammation were included in the group of “incidental rhabdomyositis”. If an animal had significant rhabdomyositis and no other comorbidities were identified through assessment of the clinical history, necropsy and histopathology, the animal was considered to have died most likely due to myositis (primary cause of death). Animals with significant rhabdomyositis and comorbidities that were likely to cause systemic compromise of the animal’s health (domoic acid toxicosis, leptospirosis and metastatic urogenital carcinoma) were considered to have died most likely due to a combination of these co-morbidities and myositis (contributory cause of death).
Immunohistochemistry
In some animals, immunohistochemistry (IHC) for S. neurona, T. gondii, and N. caninum was performed in formalin-fixed paraffin-embedded sections of skeletal muscle (n = 26) (masseter, diaphragm, intercostal and/or pectoral) and brain (n = 16). To characterize the inflammatory response in some of these cases with severe rhabdomyositis, IHC for CD3 (T- ymphocytes), CD79a (B lymphocytes), CD20 (B lymphocytes), major histocompatibility complex (MHC) II protein and Iba1 (macrophages) was performed in selected sections of skeletal muscle of 6 animals. MHCII staining was also performed in lymphoid tissue and skeletal muscle of 2 CSLs without rhabdomyositis and 2 animals with incidental rhabdomyositis to compare with the staining pattern observed in the animals with severe rhabdomyositis. The type of antibodies used, and other critical steps of immunohistochemical protocols are detailed in Supplemental Table S1. For all leukocyte antigens and MHCII, control tissues included lymph node from CSLs. Staining patterns of different leukocyte populations in the lymph node were always similar to those described in most mammals. 40 In addition, immunohistochemical staining for MHC I proteins was attempted on muscle, liver, kidney, lymph node and spleen using a commercial mouse monoclonal antibody targeted against the HLA-A protein and known to cross react with canine, feline, equine and bovine MHC I proteins (Novus Biologicals®, Littleton, CO, USA), however cross reactivity with sea lion MHC I was not detected in any of the tissues examined, despite the attempt of several protocols. Canine tissues used as positive controls (lymphoid tissue, liver, kidney) always yielded mild to marked cytoplasmic and membranous staining.
Serology
In 27 animals, archived serum was available and tested for the presence of IgG against S. neurona, T. gondii, and N. caninum at the Marine Ecosystem Laboratory, UC Davis, using an indirect fluorescent antibody test (IFAT) as described by Miller et al, 24 and using a fluorescein isothiocyanate (FITC)-conjugated rabbit anti-canine IgG (Bethyl Laboratories, Montgomery, TX, USA) as the secondary antibody.
Molecular Testing
In 10 animals, frozen (–80°C) muscle samples were available for molecular testing. DNA was extracted from the samples using the DNeasy Tissue Kit (Qiagen, Valencia, CA, USA) and genomic DNA preparations were screened for the presence of T. gondii, S. neurona, and N. caninum using 18 S rDNA pan- specific primers followed by sequencing. 22 ITS1 primers that amplify a 500 nucleotide fragment from S. neurona and S. falcatula were used. 22 These primers do not amplify the ITS1 section from other Sarcocystis spp, 22 and DNA sequencing of the ITS1 amplicons differentiate between S. neurona and S. falcatula. Positive controls consisted of genomic DNA preparations from well- characterized T. gondii isolates RH (Type I), 76 K (Type II) and CEP (Type III); S. neurona isolates SN1 and SN3 and N. caninum isolate NC-1 (ATCC No. 50843). Negative controls consisted of deionized water and genomic DNA from noninfected otters. Amplification products were visualized using ethidium bromide staining in 1% agarose gels. Sequencing was carried out by the Division of Biological Sciences DNA sequencing facility (University of California, Davis).
Other Testing
In 5 randomly selected animals with severe rhabdomyositis, tissue concentrations of vitamin E and selenium were determined using standard high-performance liquid chromatography methods (California Animal Health and Food Safety Laboratory, Davis, CA, USA), 21,37 and compared to 2 age matched controls (CSLs without myositis).
Data Analyses
To determine differences in the number of myositis cases among years, the number of cases (1) vs the total number of necropsies (0) were compared between years using a generalized linear model (GLM). To detect potential seasonality in the presentation of cases the total number of cases per month was obtained and contrasted with the total number of CSL admissions during the same period. The differences in the length of stay at TMMC between groups was tested through Mann-Whitney-Wilcoxon test. In order to detect potential differences in body mass between animals with myositis as the main cause of death, secondary cause of death and incidental myositis an ANOVA test was performed. For this test a scaled body mass index was used (body length (cm) / weight (Kg)). The differences in sex and age class (adult vs juvenile) between groups (incidental, secondary cause of death and primary cause of death) were assessed through contingency tables and using a chi-square test with sex or age as risk factors and groups as outcomes. For comparisons of average number of cysts between groups a Kruskal-Wallis tests was used. The difference on antibody titers between animals with myositis as primary or contributory cause of death was assessed using a Mann-Whitney U test. Histologic diagnoses or grades were treated as frequencies and through contingency tables the differences in frequency of diagnoses or grades between different groups was assessed using Fisher’s exact test.
Results
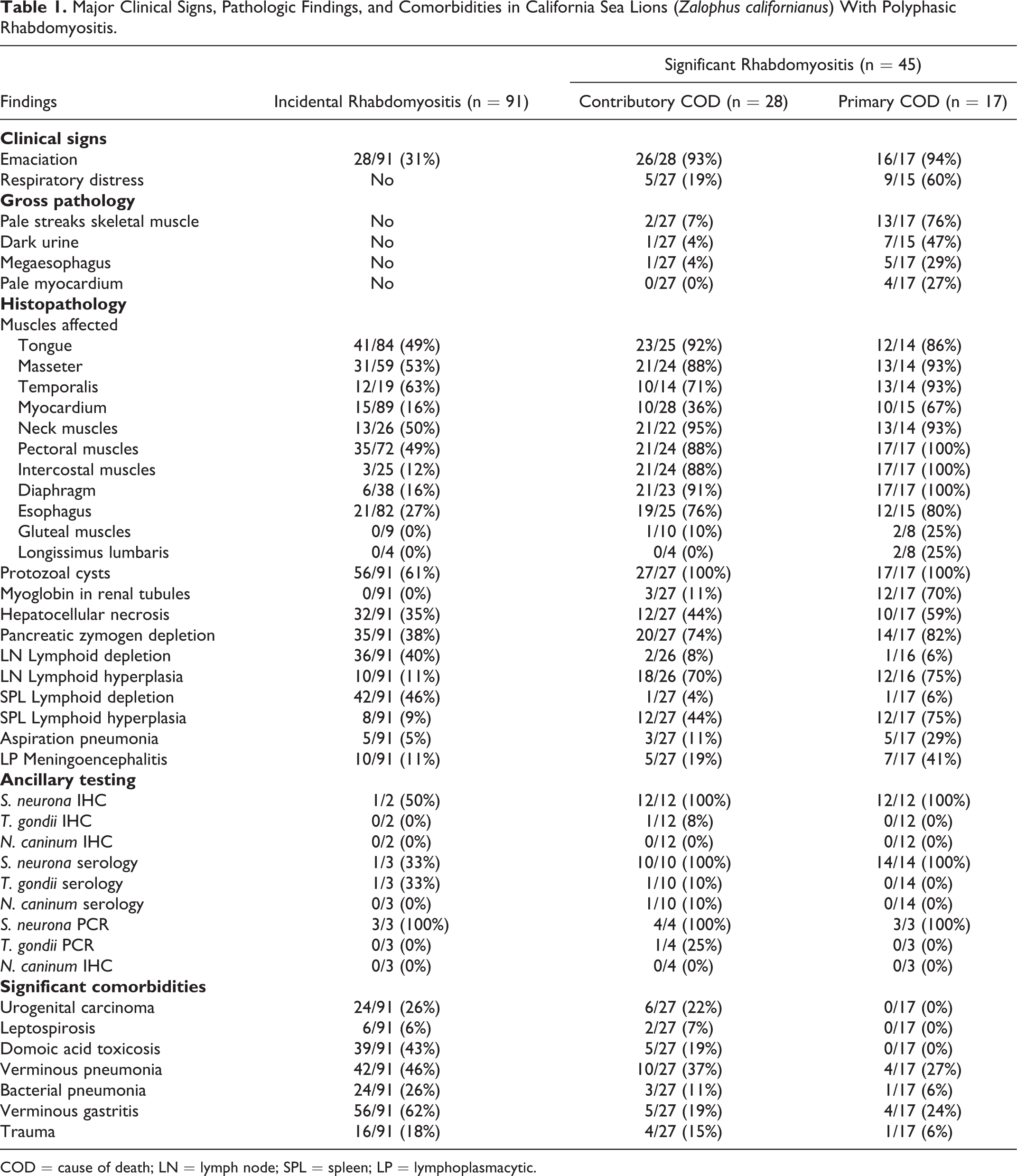
There were 136 cases of rhabdomyositis in CSLs admitted to TMMC between 2001 and 2016. Myositis was considered incidental in 91 CSLs, and a factor contributing to the stranding/death in 45 animals (significant rhabdomyositis). In 28 of the animals with significant rhabdomyositis other major disease processes included leptospirosis (n = 2) and/or domoic acid toxicosis (n = 5), trauma (n = 4) and/or metastatic urogenital carcinoma (n = 6) and verminous and bacterial bronchopneumonia (n = 13) (Table 1). Therefore, in these animals (28/45), rhabdomyositis was considered a major comorbidity but not the main cause of stranding/death. However, in 17 out of the 45 CSLs with significant polymyositis, no other major disease processes were identified and these animals were considered to have died as consequence of severe polyphasic rhabdomyositis (Table 1). Animals with incidental rhabdomyositis died or were euthanized due to domoic acid toxicosis (n = 36), metastatic urogenital carcinoma (n = 24), verminous and bacterial bronchopneumonia (n = 21), trauma (n = 16), and leptospirosis (n = 4). In this group, the proportion of animals with and without Sarcocystis sp. cysts was similar in animals with and without urogenital carcinoma (χ2 = 0.1, df = 1, P = .72).
Major Clinical Signs, Pathologic Findings, and Comorbidities in California Sea Lions (Zalophus californianus) With Polyphasic Rhabdomyositis.
COD = cause of death; LN = lymph node; SPL = spleen; LP = lymphoplasmacytic.
Temporal, Age, and Sex Distribution of Cases
Rhabdomyositis cases represented 2.8% to 11.5% of the total animals necropsied at TMMC each year (Supplemental Table S2). The number of rhabdomyositis cases in 2006 and 2008 was significantly higher than other years (GLM, P values = .02 and .03, respectively), however these differences were not correlated with an increase in the total number of animals stranded and admitted at TMMC (Supplemental Table S2). The monthly incidence of myositis cases was the lowest in February (0.6%) and the highest in April and October (2.5%) (Supplemental Table S3) however, differences were not significant (χ2 = 17.81, df = 11, P = .08). Animals with rhabdomyositis as primary cause of death were usually males (15/17, χ2 = 7.94, df = 2, P = .018) and subadults/juveniles (14/17, χ2 = 10.47, df = 2, P = .005). Animals with incidental rhabdomyositis stayed between 1 and 75 days (11.14 ± 14.06) at TMMC and animals with significant rhabdomyositis between 1 and 48 days (8.15 ± 11.63), however differences between these 2 groups were not significant (Mann-Whitney test, P = .138)
Gross and Histologic Findings
In the 45 animals with significant rhabdomyositis, clinical signs included emaciation/muscle wasting (n = 42), respiratory distress (n = 14) and dark urine (n = 8). The scaled body mass of animals with rhabdomyositis as primary cause of death (0.35 ± 0.08) was lower than the body mass of animals with rhabdomyositis as contributory cause of death (0.37 ± 0.08) and CSLs with incidental rhabdomyositis (0.41 ± 0.07) (ANOVA, F = 4.87, df = 94, P = .009).
Gross abnormalities in the striated muscles were observed mostly in CSLs that died primarily due to rhabdomyositis (13/17) rather than those with incidental rhabdomyositis. These lesions consisted of marked atrophy of temporalis (Fig. 1), masseter and pectoral muscles. In addition, in the diaphragm (Fig. 2), longissimus cervicis and pectoral muscles, and to a lesser extent in the longissimus dorsi, intercostal, masseter, temporalis, rectus abdominis, psoas and tongue muscles there were multiple white streaks that were 5-35 mm long. In 6 animals there was marked megaesophagus, and 2 of these had esophageal rhabdomyositis with associated food impaction (Fig. 3). One of these animals died due to trauma and the other 5 due to rhabdomyositis (Table 1). None of these animals had clinical or postmortem evidence of domoic acid toxicosis. Gross abnormalities in the heart were observed in 4 individuals and consisted of pallor in the interventricular septum. In 7 animals considered to have died of rhabdomyositis and 1 animal that died of verminous pneumonia, the urinary bladder contained a moderate amount of dark-red urine and the kidneys were diffusely pale.
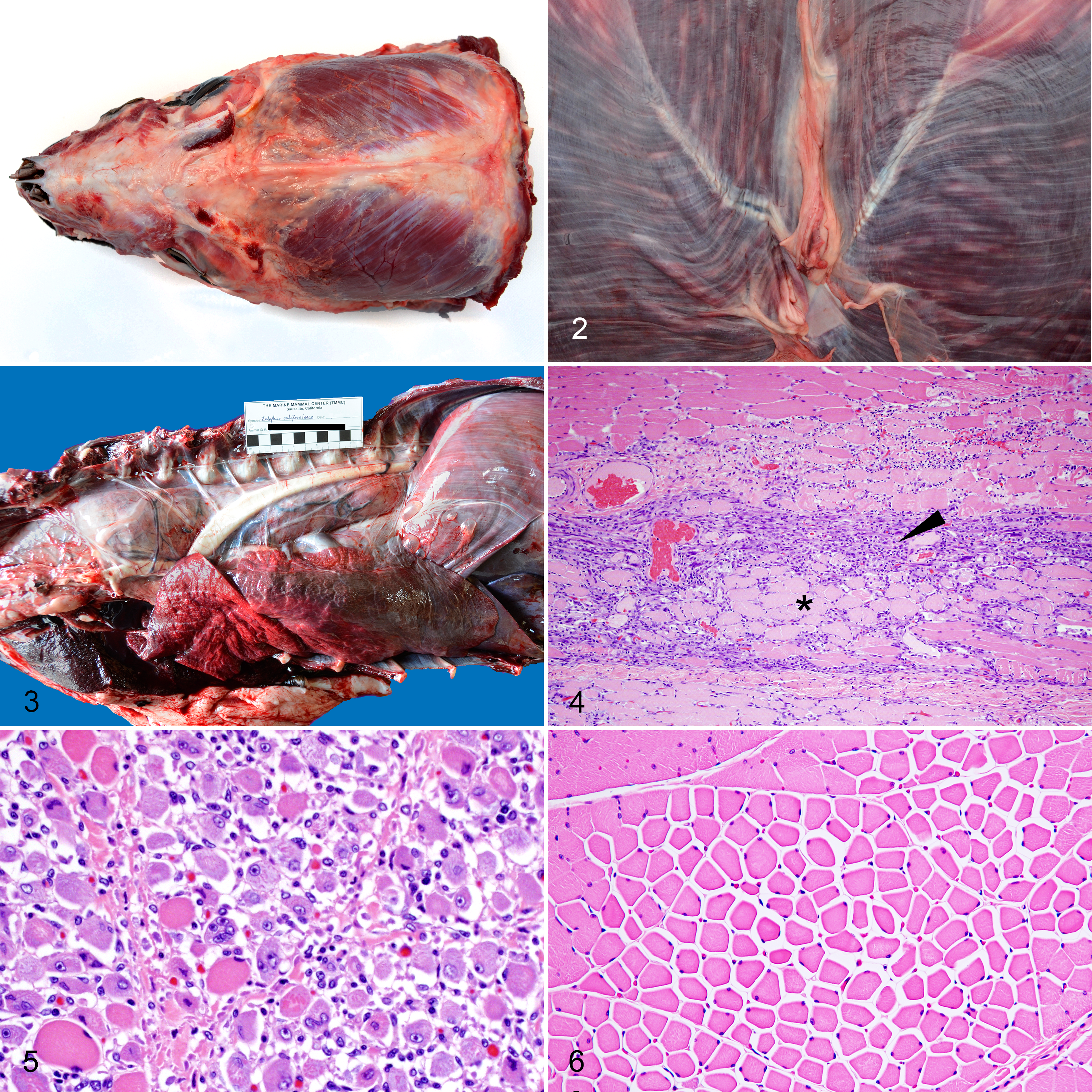
Polyphasic rhabdomyositis, California sea lions.
Microscopic lesions were similar in type and distribution between sea lions with rhabdomyositis as contributory or the main cause of death, but were more severe in those with rhabdomyositis as the primary cause of death. In these animals, the examined skeletal muscles had large areas of coagulative myofiber necrosis admixed with zones of mononuclear inflammatory cell infiltrate and myocyte regeneration (Fig. 4). The areas of regeneration had small myocytes with pale basophilic cytoplasm and multiple central rowed nuclei (Fig. 5). In muscles with grossly evident atrophy, myofibers varied markedly in size and perimysium was expanded with increased clear space (Fig. 6), whereas in other cases there was moderate replacement of myofibers by fibrous connective tissue. In the heart of these animals (rhabdomyositis as main cause of death), there were several small to medium size areas of coagulative necrosis admixed with cellular debris and surrounded by numerous macrophages, lymphocytes and fewer neutrophils.
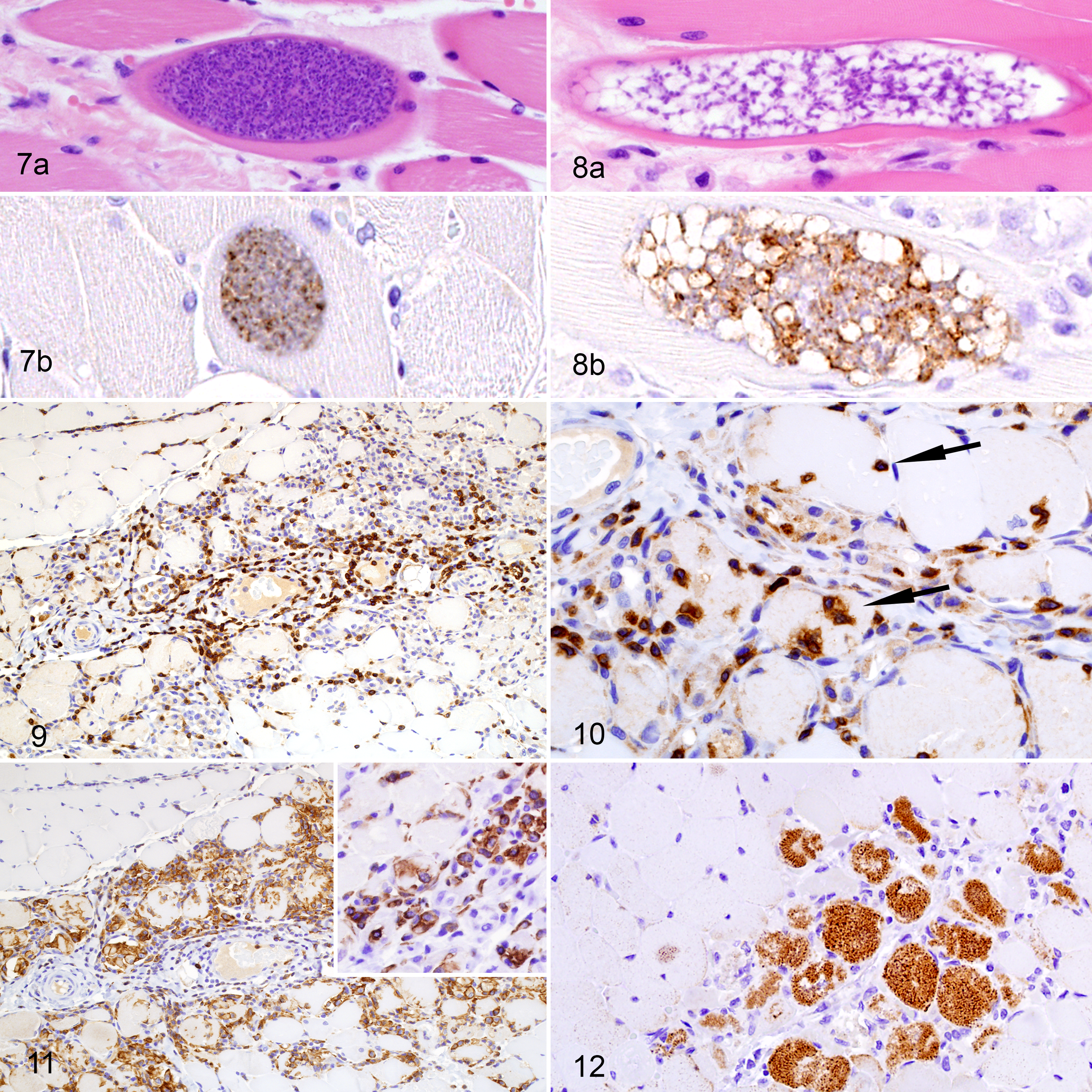
Between 2 and 8 protozoal cysts per muscle section were noted in all cases of significant rhabdomyositis adjacent to affected areas but not necessarily within areas of inflammation, necrosis, regeneration, or atrophy. Protozoal cysts had a thick (2.0–3.0 µm) striated eosinophilic wall and contained numerous 3–5 μm bradyzoites (mature cysts) (Fig. 7). Sometimes the septa of the cysts were evident on HE-stained sections and small numbers of 2–3 μm merozoites and/or bradyzoites populated the cyst (immature cysts) (Fig. 8). In nearly all animals assessed via IHC (25/26), both types of cysts had positive immunolabeling with polyclonal or monoclonal anti–S. neurona antibodies, however labeling was more intense in the zoites within immature cysts (Fig. 8b). There was no immunolabeling using antibodies against T. gondii and N. caninum in any of these samples. The average number of cysts per section was slightly larger in CSLs with rhabdomyositis as primary cause of death (4.5 ± 1.2, mean ± SD, n = 17) compared to animals with rhabdomyositis as contributory cause of death (3.37 ± 1.01, n = 22), however differences were not significant (Kruskal-Wallis test with Dunn’s multiple comparison test, P = .16).
Polyphasic rhabdomyositis, California sea lions.
The incidental microscopic lesions of rhabdomyositis corresponded to mild myonecrosis and inflammation. These lesions were restricted to a few muscle fascicles and consisted of disruption of rare myofibrils surrounded by a few lymphocytes, macrophages and rare neutrophils, sometimes (in 56 of 91 animals) adjacent to 1 to 3 protozoal cysts (35–50 μm). Animals in this group had on average (1.78 ± 0.82, n = 35) fewer cysts than in the significant rhabdomyositis groups (Kruskal-Wallis statistic = 55.76, P < .0001, Dunn’s multiple comparison tests, mean rank diff = -34.7, -54.4, P values < .0001).
Other common findings in animals with significant rhabdomyositis included the presence of moderate amounts of myoglobin in degenerate renal tubules (myoglobinuric nephrosis) (15/44) particularly in animals with severe myonecrosis and myositis as primary cause of death (Table 1), moderate, random individualization and necrosis of hepatocytes (22/45), depletion of pancreatic zymogen granules (34/44), moderate to marked aspiration pneumonia (8/44) and moderate to marked hyperplasia of lymph nodes and splenic lymphoid centers (26/42; Table 1). The level of splenic and lymph node lymphoid hyperplasia was higher in animals with higher scores of myonecrosis (Fisher’s exact test, P = .003). In addition, 12 CSLs presented with mild to moderate lymphoplasmacytic meningoencephalitis, and S. neurona zoites were identified in 1 out of these 12 cases when examined through IHC. In 1 case, T. gondii schizonts and meronts were identified in the brain and heart through IHC. In at least 5 cases with meningoencephalitis there were hippocampal lesions characteristic of domoic acid toxicosis. Immunohistochemistry for N. caninum was negative in all cases with meningoencephalitis.
Inflammatory Infiltrate and MHC II Expression
The inflamed muscle of animals with significant rhabdomyositis had an inflammatory cell population composed mainly by CD3-positive T- ymphocytes and Iba1-positive macrophages. T lymphocytes were in greater numbers in perivascular areas (Fig. 9), and occasional T lymphocytes were located in the endomysium or in the sarcoplasm of intact myocytes at the border of necrotic and intact muscle (Fig. 10). Macrophages were more common in areas with necrotic and fragmented myocytes, and in areas of active muscle regeneration (Fig. 11). In these areas there were also rare neutrophils and a few eosinophils. No CD79a- or CD20-positive B lymphocytes were detected in areas of inflammation, and rare CD79a- and CD20-positive B lymphocytes were observed in the perimysium of intact muscle tissue. In animals with severe rhabdomyositis occasional myofibers had moderate to marked cytoplasmic and membranous immunolabeling with anti-MHC II antibodies (Fig. 12). In inflamed areas rare leukocytes had marked cytoplasmic and membranous MHC-II positive staining. Animals used as controls (without myositis and with incidental myositis) had moderate cytoplasmic and membranous labeling of MHC-II in occasional mononuclear cells in lymphoid tissues but no labeling of striated muscles.
Ancillary Testing
Sera from 26 of 27 animals tested for antibodies against S. neurona, T. gondii and N. caninum, contained significant antibody titers against S. neurona (range 160–81920). In 2 animals, there were low and medium titers of anti–T. gondii antibodies (1:40 and 1:320) and in other CSL there were low antibody titers against N. caninum (1:160). Anti–S. neurona antibody titers were greater in CSLs with severe myositis (26175 ± 2164.87) when compared to animals with mild or moderate myositis (7612 ± 1042.74) (Mann-Whitney U test, P = .011). In 7 animals with significant rhabdomyositis and 3 CSLs with incidental rhabdomyositis tested through PCR, 18 S sequencing revealed 97–99% identity with S. neurona. In 1 animal with significant rhabdomyositis, no S. neurona was detected by PCR but T. gondii DNA was identified. No N. caninum DNA was identified the 10 examined cases. Selenium and vitamin E levels were similar in CSLs with (range 59–69 µg/mg) and without (55 and 73 µg/mg) rhabdomyositis.
Discussion
Polyphasic rhabdomyositis has been a consistent but uncommon cause of stranding and mortality in CSLs in central California over the last 15 years. These animals present with muscle wasting and clinical signs related to abnormal function of the muscle group(s) most severely affected. On gross examination, white streaking and atrophy of skeletal muscles were common in the most severe cases; however, milder lesions were not easy to identify grossly, especially in animals with chronic emaciation or anemia, which make striated muscles smaller and paler. Microscopic features and ancillary testing suggest that protozoal infection, and particularly S. neurona exposure, is one of the factors associated with the presentation of severe polyphasic rhabdomyositis in CSLs. In addition, leukocyte-induced muscle damage may sustain sarcolemmal injury, myocyte loss and muscle atrophy.
The reasons for an increase in the number of rhabdomyositis cases in some years is not clear. In protozoal encephalitis in marine otters, environmental conditions such as increase in rainfall and runoff can be associated with more exposure to the parasites in the marine environment and higher disease prevalence. 7,23 However, in the case of CSLs, which are widely distributed along the west coast of the US, the relationship between rhabdomyositis and environmental, oceanographic, or prey-related factors is unknown and more data are necessary to explore potential relationships. Animals dying primarily from rhabdomyositis were usually young males; this might suggest that differences in diet among different pinniped age categories and sexes 33 affect the number of ingested S. neurona oocysts and thus the infection burden. On the other hand, this difference between males and females could reflect different distribution patterns of sex and age classes in otariids, a pattern that has been already described for leptospirosis in CSL. 7 Animals with more severe disease, high S. neurona titers and without comorbidities tended to be young (less than 6 years old). This could indicate some predisposition to more severe disease in this age class, similar to horses with S. neurona meningoencephalitis. 26 However, the mechanisms underlying this potential susceptibility are not clear.
Animals with severe rhabdomyositis were usually infected with S. neurona and tended to have more Sarcocystis sp. cysts in muscle sections when compared to animals with incidental rhabdomyositis. These differences suggest a role of S. neurona infection in severe polyphasic rhabdomyositis, but it is likely that additional host (eg, immune response) and environmental (eg, pollutants, diet, energy balance) factors play a role in the development of this condition. For instance, animals with severe rhabdomyositis were usually in an emaciated state and had on average lower body mass compared to animals with incidental rhabdomyositis. Therefore, a negative energy balance could have contributed to muscle catabolism and degeneration in the studied CSLs. On the other hand, some of these animals had severe megaesophagous that compromised swallowing, suggesting that rhabdomyositis could be the cause and not the consequence of emaciation. However, given the design of our study, is difficult to confirm the directionality of the relationship between low body mass/emaciation and rhabdomyositis.
S. neurona, the causative agent of equine protozoal meningoencephalitis, infects intermediate hosts when they ingest sporulated oocysts released by opossums (Didelphis virginiana). 12 This protozoan has been recognized for decades as a marine pathogen. 2,25 Oocysts probably reach marine estuarine ecosystems through runoff from agricultural and semiurban landscapes, and are later concentrated by marine organisms which are consumed by marine mammals, especially sea otters. 12,23,25
In horses and most marine mammal species, S. neurona affects primarily the central nervous system, causing meningoencephalitis and/or myelitis. 2,11,26 In CSLs, although lymphoplasmacytic meningocencephalitis was common in cases with significant rhabdomyositis, S. neurona organisms were identified in only 1 case through IHC. However, IHC is not highly sensitive for detecting protozoan infection, and although protozoa may cause meningoencephalitis, other causes of inflammation in the brain such as domoic acid are also possible. Other than 2 previously reported cases of CSLs successfully treated for S. neurona myositis, prior to this study, myositis caused by S. neurona has been reported in a few cases of systemic infection in sea otters and a captive lynx. 1,6,11,23 Although infection has been described in multiple tissues from a wide range of marine mammals, 12 its relation with primary muscle inflammatory disease is apparently unique to CSLs.
In wild and domestic animals, other protozoan species are more commonly associated with myositis and/or myocarditis. Some of these associations include N. caninum and non–S. neurona Sarcocystis sp. in canids, 10,36 T. gondii in felids and humans, 14 S. cruzi in cattle and S. fayeri in horses. 16,38 However, in these cases inflammatory reaction and myositis is usually directed against muscle cysts or numerous free merozoites and tachyzoites in the tissue. Inflammatory infiltrate in these cases is composed primarily of eosinophils, neutrophils and macrophages. 10,16,38 However, in the CSLs of this study, free zoites were not observed, and inflammation was lymphohistiocytic and associated with necrotic and regenerating myofibers adjacent to intact S. neurona cysts. These findings suggest that muscle damage might not be the result of active destruction of myofibers by protozoan or other bacterial and/or parasitic agent, but that injury may be initiated by other external factors and sustained by immune cells. In this case, the presence of intact S. neurona cysts in the presented cases could represent a background infection since S. neurona is rather common in CSLs skeletal and cardiac muscle. 15 However, the presence of higher S. neurona burden and antibody titers in animals with more severe myonecrosis and inflammation and the lack of significant cyst burden and antibody response in animals with incidental myositis suggests that S. neurona exposure may be necessary for development of the severe form of polyphasic rhabdomyositis in CSLs. An alternative explanation for the lack of evidence of a cellular inflammatory response targeted against S. neurona cysts is that exposure to this parasite and muscle infection alters mechanisms of immune tolerance in skeletal muscles. 13 Elucidating the pathogenesis of rhabdomyositis in this species will require controled studies including animals that survive rhabdomyositis, and a more detailed understanding of the immune response of CSLs to protozoan infection.
The type of inflammatory response found in most cases with severe rhabdomyositis resemble some aspects of immune-mediated inflammatory myopathies of dogs and humans. 8,34 In human and canine polymyositis, inflammatory infiltrates are composed mainly of T lymphocytes, which can sometimes infiltrate the endomysium and sarcoplasm of intact myofibers, a finding that is considered a key diagnostic feature of immune-mediated myositis in humans. 8,34 T-lymphocyte activation and attack of muscle fibers can be elicited by production of specific antibodies against sarcolemma proteins and/or expression of MHCI and MHCII proteins in the sarcoplasm. 14 In mammals, normal mature myocytes usually do not express MHC molecules, but these are upregulated in human and equine immune-mediated myopathies. 8,13,14 Similarly, in our study we found that CSLs with severe myositis expressed sarcoplasmic MHC II proteins, therefore, it is possible that S. neurona infection in CSLs elicited immune-mediated muscle damage by producing upregulation of MHC molecules once the cell is infected. If this process is dependent on the infective dose, it could explain why animals with higher S. neurona titers have more severe myonecrosis. On the other hand, this could also be due to a more reactive immune system with consequent more severe immune-mediated damage. In a similar manner, the presence of marked lymphoid hyperplasia in emaciated CSLs is unusual and probably related to a hyperactive immune system, stressing the potential role of immune activation in the development of polyphasic rhabdomyositis in CSLs. Our findings and proposed pathogenesis are also in line what has been observed clinically in CSLs with rhabdomyositis, because besides Ponozuril therapy, corticosteroids have been effective at increasing the recovery rate in animals affected this condition (Whoriskey et al, 2017, unpublished data). 6
Further evidence of the role of the immune response in the pathogenesis of S. neurona tissue damage is the fact that in horses lacking T and B lymphocytes (foals with severe combined immunodeficiency), S. neurona experimental inoculation does not cause clinical disease. 20,32 In addition, in equine protozoal meningoencephalitis, other factors such as stress and young age have been indicated as risk factors for the presentation of disease. 26,29 In the case of CSLs, the effect of stress is not clear but likely to play a role given the fact that many CSLs with significant rhabdomyositis and moderate S. neurona titers had significant comorbidities, such as urogenital carcinoma and domoic acid toxicosis, conditions that are likely to cause significant stress.
Although myositis and Sarcocystis sp. cysts were observed in many muscle groups, these tended to be more common in diaphragm, intercostal, pectoral, neck and masticatory muscles. The reason for this distribution pattern of myositis and S. neurona cysts is unclear but it could be related to the metabolic activity of these muscle groups. 18 For instance, T. gondii conversion of tachyzoites into tissue cysts is inhibited by lactate production in myocytes. 39 Therefore, if similar processes act on S. neurona life cycle, transition from zoites to cysts could be favored in muscle groups that contain more mitochondria, because in these fibers the glycolytic pathway and lactate production are not frequent under normal physiological demand. 17,39 In otariids, these muscle groups are concentrated in the cranial portion of the body, including pectoral muscles, whereas caudal (eg, hind limbs) muscles have less mitochondrial density and are more likely to undergo anaerobic metabolism and produce lactate. 18 These host and parasite metabolic adaptations, therefore, could play a role in the observed distribution pattern of S. neurona cysts in CSLs.
In addition to infectious agents, a common cause of myopathy and myositis in wild animals, including marine mammals, is capture myopathy. 17,31 The CSLs included in this study were all restrained and transported to the rehabilitation facility where they stayed and were subjected to additional restraining for different periods of time. Therefore, it could be hypothesized that muscle use and contraction due to restraint played a role in the development of the described muscle lesions. If this was the case, similar lesions should be seen in most of the animals admitted to TMMC since all of them undergo similar capture and restraint procedures. In addition, no differences were detected in the time of stay at TMMC between animals with different severity of myositis. Furthermore, capture stress in cetaceans and otariids usually involve muscle groups related to locomotion, and not masticatory and respiratory muscles as in the condition described in this study. In addition, capture myopathy lesions are in most cases monophasic, acute or subacute and involve a mild inflammatory component compared to the cases examined in this study. 17,31
Polyphasic rhabdomyositis in CSLs can range from mild (incidental) to severe, and the latter appear to be a cause of stranding and mortality especially in young male CSLs. S. neurona is a plausible inciting agent of severe myositis in CSLs; however, immune-mediated mechanisms may be a significant component of muscle damage and clinical disease. Many aspects on the epidemiology and pathogenesis of rhabdomyositis in CSLs remain to be investigated as well as the potential role of infectious agents other than S. neurona.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819829526 - Polyphasic Rhabdomyositis in California Sea Lions (Zalophus Californianus): Pathology and Potential Causes
Supplemental Material, DS1_VET_10.1177_0300985819829526 for Polyphasic Rhabdomyositis in California Sea Lions (Zalophus Californianus): Pathology and Potential Causes by Mauricio Seguel, Kathleen M. Colegrove, Cara Field, Sophie Whoriskey, Tenaya Norris and Padraig Duignan in Veterinary Pathology
Footnotes
Acknowledgements
The authors acknowledge the contributions and diagnostic work performed on early cases by Drs Linda Lowenstine, Terry Spraker, and Tanja Zabka. We would like to thank Dr Daphne Carlson-Bremer for facilitating information on archived cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Mauricio Seguel was supported by a Morris Animal Foundation fellowship (Grant N D16ZO-413)
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
