Abstract
This study reports cytomorphological, histomorphological, and immunological characterization of 608 biopsy cases of canine malignant lymphoma, with epidemiological and clinical data, collected from 7 French veterinary pathology laboratories. It compares morphological characteristics of malignant lymphoma in canines, per the updated Kiel classification system, with those reported in humans, per the World Health Organization (WHO) classification system. Of tumors described, 24.5% and 75.5% were classified as low- and high-grade malignant lymphomas, respectively. Presenting clinical signs included generalized or localized lymphadenopathy (82.4%) and extranodal diseases (17.6%) involving the skin (12.34%) and other sites (5.26%). Immunohistochemistry confirmed 63.8% B-cell (CD3–, CD79a+), 35.4% T-cell (CD3+, CD79a–), and 0.8% null-cell (CD3–, CD79a–) lymphomas. Most B-cell cases (38.49%) were of high-grade centroblastic polymorphic subtype; most T-cell cases (8.55%), high-grade pleomorphic mixed and large T-cell lymphoma subtypes. Some B-cell tumors showed morphologic characteristics consistent with follicular lymphomas and marginal zone lymphomas per the Revised European American Classification of Lymphoid Neoplasms and WHO canine classification systems and the WHO human classification system. Unusual high-grade B-cell subtypes included an atypical high-grade small B-cell lymphoma (0.66%), Burkitt-type B-cell lymphoma (1.64%), plasmacytoid lymphoma (0.99%), and mediastinal anaplastic large B-cell lymphoma (0.16%). Unusual T-cell subtypes included a previously undescribed high-grade canine immunoblastic T-cell type (1.15%), a rare low-grade prolymphocytic T-cell lymphoma (0.16%), and a recently described high-grade canine T-cell entity—aggressive granulocytic large-cell lymphoma (0.16%). Marginal zone lymphomas were common (10.86%); follicular lymphomas were rare (0.49%). Canine primary cutaneous malignant lymphoma subtypes were present (11.84%). There was no significant difference between B- and T-cell malignant lymphoma in regard to canine age and sex. A significant overrepresentation of Boxers (24.19%) was found for T-cell lymphomas.
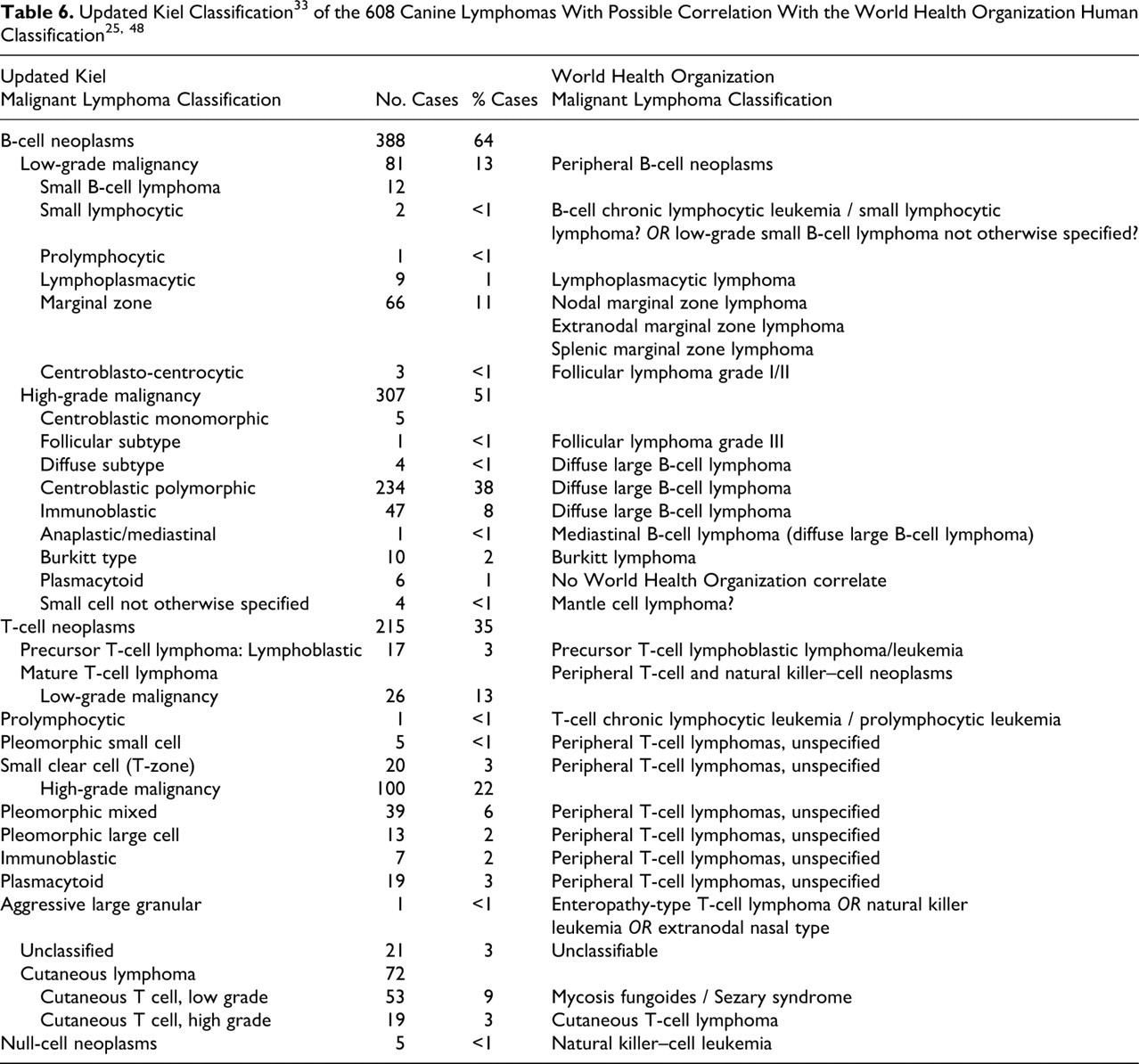
Non-Hodgkin lymphomas (NHLs) are common tumors in dogs, with an incidence previously evaluated at 33 per 100,000. 49 They are also common in humans and are on noticeable increase. 1,5,7,46 As a close companion of humans that is subjected to the same environmental influences, the dog has often been proposed as a model for studying the etiology and therapy of NHLs. The value of this model depends on the possibility of distinguishing the main lymphoma subgroups in both species with a reliable classification of canine NHLs following the schemes currently used for human NHLs. The morphologic canine classifications 3,15,18,30,50,56 have been successively based on different human classifications—namely, the updated Kiel classification, 33 Revised European American Classification of Lymphoid Neoplasms (REAL), 21,22 and more recently, the World Health Organization (WHO) classification 25,39,48 —taking account of epidemiological, clinical, phenotypic, and genetic features and identifying clinicomorphological entities.
The aims of this article are, first, to identify and describe the common B-cell, T-cell, and null-cell malignant lymphoma entities observed in canines in France using the updated Kiel classification system; second, to confirm previously obtained results concerning the high frequency of T-cell lymphomas in dogs as compared with their frequency in humans, using a large and statistically significant number of cases; and, third, to identify new, rare, or poorly described lymphoma subtypes in this large number of canine cases that earlier veterinary studies might have missed. Finally, we attempt a morphological comparison of disease entities of canine lymphoma, as defined by the updated Kiel classification system and the human WHO classifications, to view initial similarities and differences between canine and human lymphoma subtypes. This study presents a framework for further investigations, which will include developing new immunological markers and collecting additional epidemiological, clinical, immunological, and genetic data with a view to confirming the value of the dog species as a model for human NHLs.
Materials and Methods
Collection of Cases
This study was performed over 1 year, from May 2000 to May 2001. Histological paraffin-embedded specimens and case information for 702 biopsy admissions of canine lymphomas were collected from 7 private and university veterinary pathology laboratories in France. Laboratory selection was based on the number of pathologic cases diagnosed per year by each laboratory and on their geographic location in France. Fine-needle aspirates (FNAs) were available for all 93 cases submitted to the Laboratoire de Pathologie Clinique, Prof Fournel-Fleury.
From the initial 702 cases, 608 were included in the study. Ninety-four cases were excluded because of insufficient and inadequate tissue (n, 87) or misdiagnosis, including myeloid proliferation (n, 2) and benign lymphoid hyperplasia (n, 5), as confirmed by morphological and immunological tests and clinical follow-up. For all cases included in this study, the following data were collected: contributing laboratory, tumor topography, main clinical signs upon presentation, age, breed, and sex (without concern for sexual sterility of dogs). Tumor topography was assigned to one of the following categories: single lymph node involvement, generalized lymph node involvement, and extranodal involvement.
Histological Evaluation
Biopsy specimens from enlarged lymph nodes, extranodal masses, or cutaneous lesions in all 608 cases were fixed in 10% neutral buffered formalin at room temperature for 48 hours and embedded in paraffin wax by each laboratory. Tissue sections 5 μm in thickness were stained with hematoxylin and eosin (HE) and examined by one pathologist from each contributor laboratory to confirm a diagnosis of malignant lymphoma. Paraffin wax–embedded tumor tissues were subsequently sent to Laboratoire de Pathologie Clinique et Unité d’Oncologie Ecole Nationale Vétérinaire de Lyon, France, and sections were processed as described in previous studies. 4,15,16,18,40,42 Two pathologists (T.M. and associate) and one cytologist (C.F.F.) separately examined HE-stained slides before immunophenotyping. Finally, all authors examined all slides.
Cytological Evaluation
Cytological smears obtained by FNA of 93 nodal or extranodal masses were air-dried, fixed, and stained using the May–Grünwald–Giemsa technique as previously described. 4,15,16,18,40,42,54 Cytological slides were examined by one cytologist (C.F.F.). Morphological criteria based on the updated Kiel histological and cytological classification was the same as previously published. 15,16,18,40 –42
Immunophenotyping
Immunophenotyping was performed on paraffin wax–embedded sections from all 608 samples, with antibodies directed against human antigens but cross-reacting with the equivalent canine antigens (see Table 1 ). A polyclonal rabbit antibody against CD3 was used as a pan–T marker, 13 and a monoclonal antibody directed against CD79a was used as a pan–B marker (Dako, Glostrup, Denmark). 27,28 A standard avidin–biotin–immunoperoxidase procedure was implemented as described previously. 14,15,18 Immunophenotyping using pan–B markers and pan–T markers was examined by another scientist (F.P.) independently from the morphological study. Additional immunolabelings (Table 1) were performed only if initial immunophenotyping was insufficient to provide accurate tumor classification (51 cases). The same procedure was used as for paraffin-waxed sections.
Antibodies Used for Immunocytohistochemical Staining of Canine Lymphomas
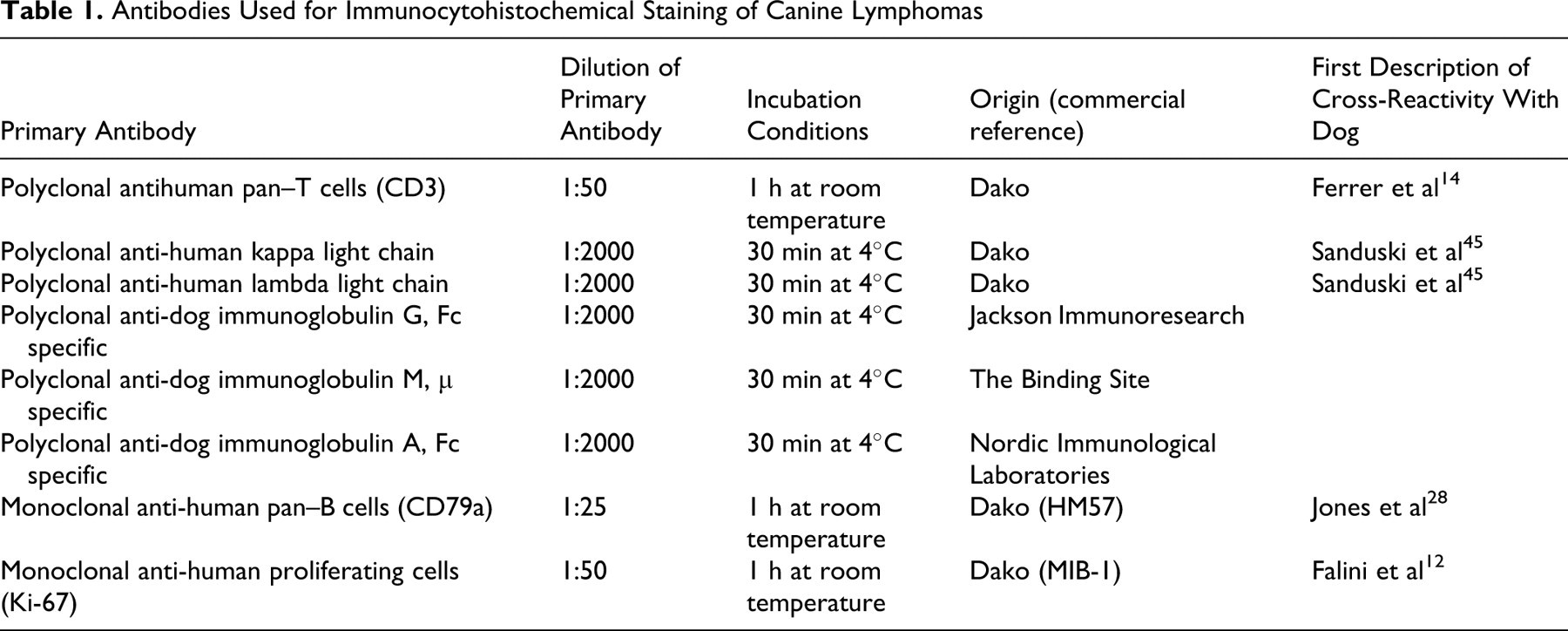
Fourteen cases were evaluated for immunoglobulin characterization with antibodies to human kappa and lambda immunoglobulin light chain antigens that cross-reacted with equivalent canine antigens. These immunoglobulin antibodies were used in 9 potentially immunosecretory cases (because of their plasmacytic morphologic appearance) and in 5 null-cell lymphomas (CD3–, CD79a–) for additional investigation. 43,45 Canine-specific polyclonal antibodies detecting immunoglobulin G, immunoglobulin M (IgM), and immunoglobulin A were used in the same 14 cases. 15 The Ki-67 proliferation index was determined for only intermediate, atypical, or doubtful cases (n, 33), based on MIB-1 immunolabeling as previously described. 12,15,18
Classification
Classification of the 608 cases was initially based on cellular morphology and immunophenotype according to the updated Kiel classification adapted to account for specific canine subtypes, as previously described. 15,18,40,42 B-cell morphologies that did not fit into the Kiel classification system—particularly, marginal zone lymphomas—were described according to the REAL and WHO human classifications and previous veterinary publications 15,18,40,42 and confirmed more recently by the REAL and WHO canine classification systems. 56
Grading was determined by the size of cells (majority of small-sized cells or majority of medium- and large-sized cells) and by mitotic index (MI). Cases showing a majority of small-sized cells and a low or medium MI were classified as low-grade lymphomas, whereas those showing a majority of medium- and large-sized cells and a high MI were classified as high-grade lymphomas. For intermediate, atypical, and/or doubtful cases, final tumor subclassification was based on the MIB-1 immunolabeling results for Ki-67 index. 16
Cutaneous T-cell lymphomas were identified as primary if there were tumor cell infiltration into the skin without evidence of visceral organ involvement. Secondary skin metastases from a primary visceral growth were not considered cutaneous tumors.
Canine Versus Human Morphologic Comparison
After clear tumor identification with the updated Kiel classification, a comparison was attempted for tumor histomorphology as reported in the human WHO classification system. B-cell morphologies that did not fit into the Kiel classification system, including marginal zone and follicular lymphomas, were described per the canine WHO classification system and then compared directly with the human WHO classification system. 48
Statistical Analysis
Means were compared with Student t test, and variances were compared with the F test. Frequencies and distributions were compared with the Pearson chi-square test. Significance was taken as P < .05.
Results
Table 2 presents clinical and epidemiological data. Tables 3, 4, and 5 summarize morphological data. Tumor subtypes indicated in bold font in the tables represent new, recently recognized, and/or rare lymphoma subtypes as presented in canines in this study. Table 6 lists the respective proportion of cases of the various updated Kiel classification tumor subtypes in the canine species with morphological correlations with the human WHO classification subtypes.
Epidemiological and Clinical Features of Canine Lymphomas According to the Morphological Subtypes
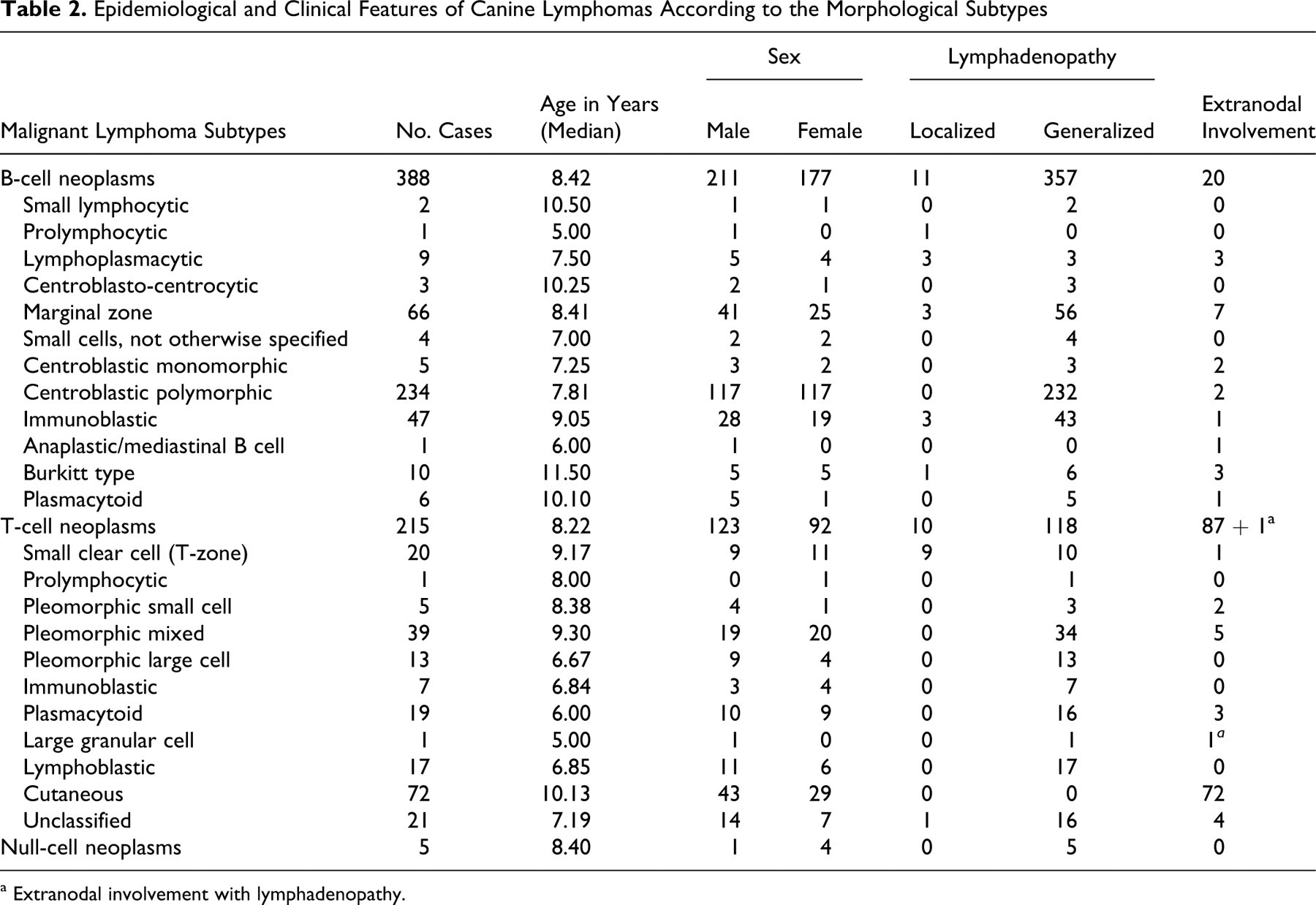
a Extranodal involvement with lymphadenopathy.
Morphology of the Various Subtypes of Canine B-Cell Lymphomas: 608 Cases 15
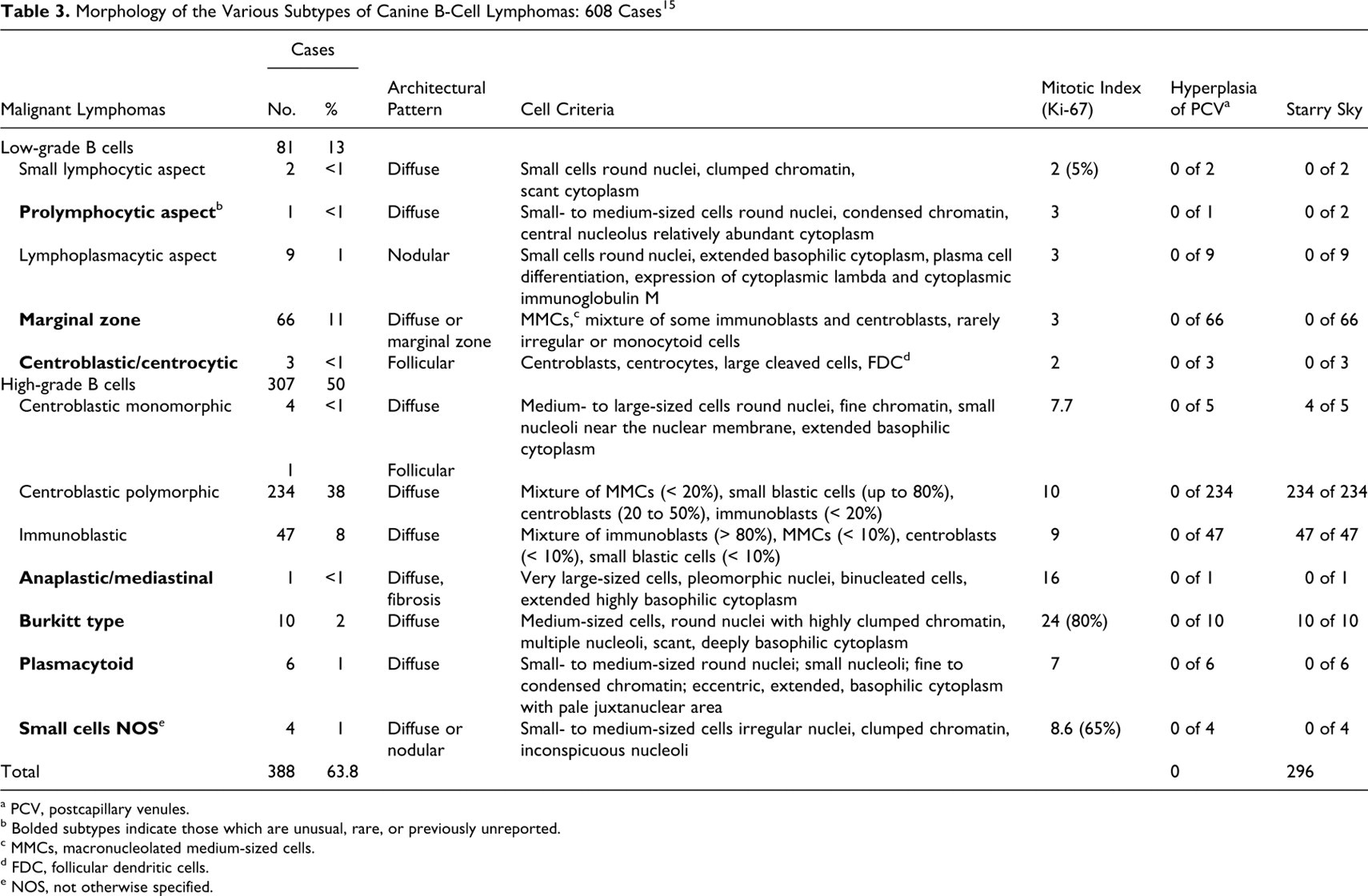
a PCV, postcapillary venules.
b Bolded subtypes indicate those which are unusual, rare, or previously unreported.
c MMCs, macronucleolated medium-sized cells.
d FDC, follicular dendritic cells.
e NOS, not otherwise specified.
a PCV, postcapillary venules.
b Bolded subtypes indicate those which are unusual, rare, or previously unreported.
c Null-cell tumor morphology subtypes.
General Findings, Epidemiology, and Clinical Data
There was no significant difference in age or sex distribution for B- and T-cell malignant lymphomas in canines (Table 2). Boxers appeared to be overrepresented (10.03%) among affected dogs, in comparison with an overall 2% of Boxers in the French canine population (P < .0001). The proportion of Boxers with T-cell lymphomas (24.19%) was significantly higher than that reported in the national canine population (P < .0001), whereas proportions of Boxers with B-cell lymphoma (2.32%) were not.
For all included dogs with malignant lymphoma, clinical signs of nodal involvement, either generalized or localized lymphadenopathy, were observed in 82.40% of cases. Extranodal disease (17.60% of cases) manifested most commonly as skin involvement (12.34%), whereas other sites (5.26%) included the digestive tract (1.48%), spleen (1.80%), tonsils (1.48%), and eyes (0.33%).
Immunohistochemistry
Immunohistochemically, 388 cases (63.4%) were identified as CD3–, CD79a+ (B-cell lymphoma), and 215 cases (35.4%) were identified as CD3+, CD79a– (T-cell lymphoma). Five cases (0.8%) were negative for CD3 and CD79a and were therefore classified as null-cell lymphomas. Additional testing was performed in 51 cases that required further immunological data for accurate classification purposes, including 37 cases for Ki-67 expression and 14 cases of immunoglobulin characterization. Of these latter 14 cases, 9 with a plasmacytoid appearance suggesting possible immunosecretion (lymphoplasmacytic lymphomas) were positive for lambda immunoglobulin light chains and for IgM, whereas 5 were negative for all the immunolabelings, confirming their null-cell type.
Histomorphology and Cytomorphology
Tumor subtypes are described in the tables. Tumor subtypes that are unusual, poorly recognized, or rare (as identified in the tables in bold font) are described in greater detail. Remaining tumor subtypes have been well described within the literature.
B-cell Malignant Lymphoma
All 388 cases of B-cell malignant lymphoma (63.8%) were determined to represent mature B-cell lymphomas (Table 3). No precursor B-cell lymphomas (ie, lymphoblastic lymphoma) were observed. Mature B-cell lymphomas, mimicking normal stages of B-cell differentiation, have been classified and tabulated on the basis of their cell size, MI, and normal postulated counterpart.
High-grade B-cell malignant lymphomas were most common, with 234 cases of centroblastic polymorphic B-cell lymphoma, 5 centroblastic monomorphic cases (with 4 diffuse and 1 follicular subtype), 47 immunoblastic cases, 1 anaplastic mediastinal case, 10 Burkitt-type cases, 6 plasmacytoid cases, and 4 unusual cases of small-cell lymphoma not otherwise specified (NOS).
The single isolated new case of anaplastic mediastinal large B-cell subtype was identified by FNA on pleural effusion from a 6-year-old male German Shepherd with a mediastinal mass. Cytological examination of the pleural fluid showed large or giant cells, frequently binucleated or multinucleated and highly nucleolated, with irregularly clumped chromatin and extended highly basophilic cytoplasm (Fig. 1 ). Histologically, the mediastinal mass showed marked fibrosis and diffuse infiltration by large cells with pleomorphic nuclei, presence of binucleated cells, and extended highly basophilic cytoplasm (Fig. 2 ).
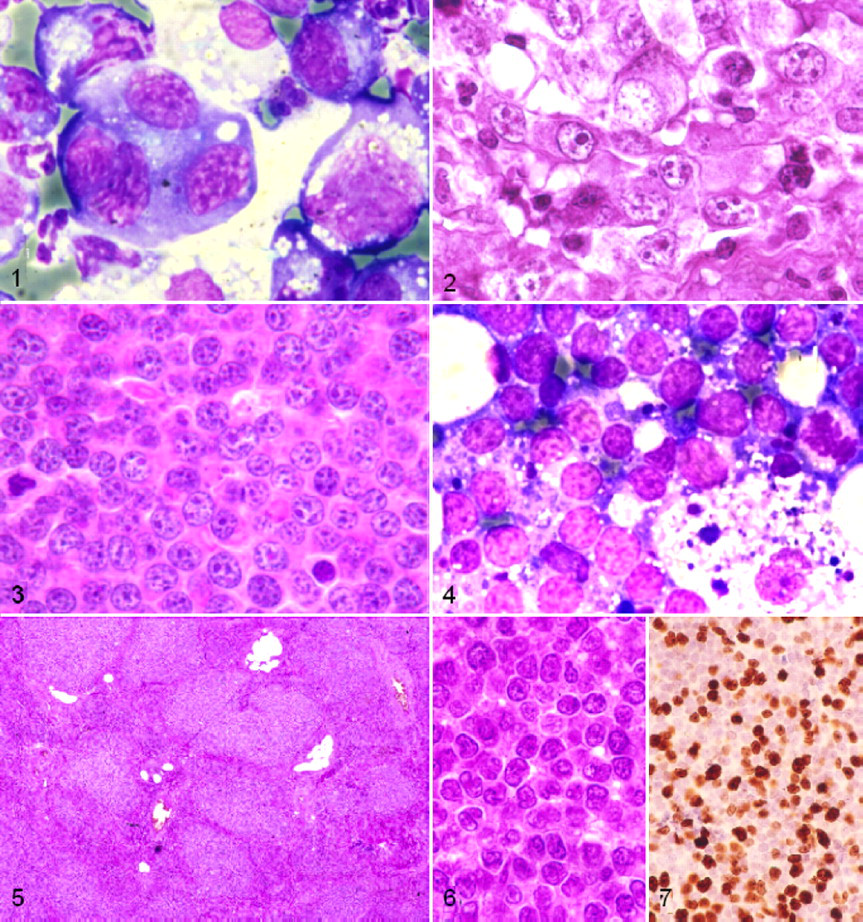
The 10 cases of Burkitt-type lymphoma showed a marked starry-sky pattern and diffuse infiltration by medium-sized cells with round nuclei, markedly clumped chromatin, several nucleoli, and a small ring of hyperbasophilic cytoplasm (Figs. 3 , 4). All exhibited a high MI (median, 24 per high-powered field [HPF], 400×) and Ki-67 index (80%). Three cases presented a slight plasmacytoid differentiation of 10% of cells with extended and eccentric hyperbasophilic cytoplasm. Three other cases showed 5% of atypical cells with slightly irregular nuclei, more prominent nucleoli, and the highest MI. Clinically, 6 Burkitt-type cases presented with generalized lymphadenopathy and 1 case with localized lymphadenopathy. The other 3 Burkitt-type cases had extranodal involvement: 1 with splenomegaly, 1 with enlarged tonsils, and 1 with uveitis.
The 6 provisional plasmacytoid B-cell lymphomas appeared slightly different from the Burkitt-type owing to a marked plasmacytoid appearance and fine to condensed chromatin in small- to medium-sized cells with no starry-sky pattern and a lower MI (median, 7 per HPF). Clinically, all presented with generalized lymphadenopathy except one, which presented mainly with skin nodules.
Small B-cell lymphomas generally have low mitotic activity and are considered a low-grade phenotype. The 4 cases of small-cell lymphoma NOS had nodular to diffuse infiltration of a typical small cell with irregular nuclei, inconspicuous nucleoli, and clumped chromatin (Figs. 5, 6) but a high mitotic rate and Ki-67 index (65%), suggesting a high-grade lymphoma phenotype (Fig. 7). These tumors also contained increased numbers of intermixed medium-sized cells.
Low-grade B-cell lymphoma subtypes included 2 small lymphocytic subtypes, 1 prolymphocytic subtype, 9 lymphoplasmacytic subtypes, 66 marginal zone lymphomas, and 3 centroblastic/centrocytic lymphomas with follicular architecture. The small lymphocytic lymphoma showed a diffuse infiltration by small cells (which are slightly larger than normal lymphocytes), scant cytoplasm, clumped chromatin, usually a round nucleus and occasionally a small nucleolus, and an interspersed prolymphocytic cell component (small- to medium-sized cells with relatively abundant cytoplasm, clumped chromatin, central nucleolus). The MI was medium (median, 2 per HPF), and the median Ki-67 index was 5%. There was no bone marrow or blood involvement.
The sole case of prolymphocytic lymphoma had a diffuse architectural pattern with small- to medium-sized cells identical to the prolymphocytes described above. The 9 lymphoplasmacytic lymphomas had a pseudofollicular organization of small cells with round nuclei, abundant basophilic cytoplasm, and perinuclear clearing, consistent with plasmacytoid differentiation. Additional immunohistochemical testing of these tumors showed positive cytoplasmic IgM and lambda immunoexpression, confirming plasma cell differentiation.
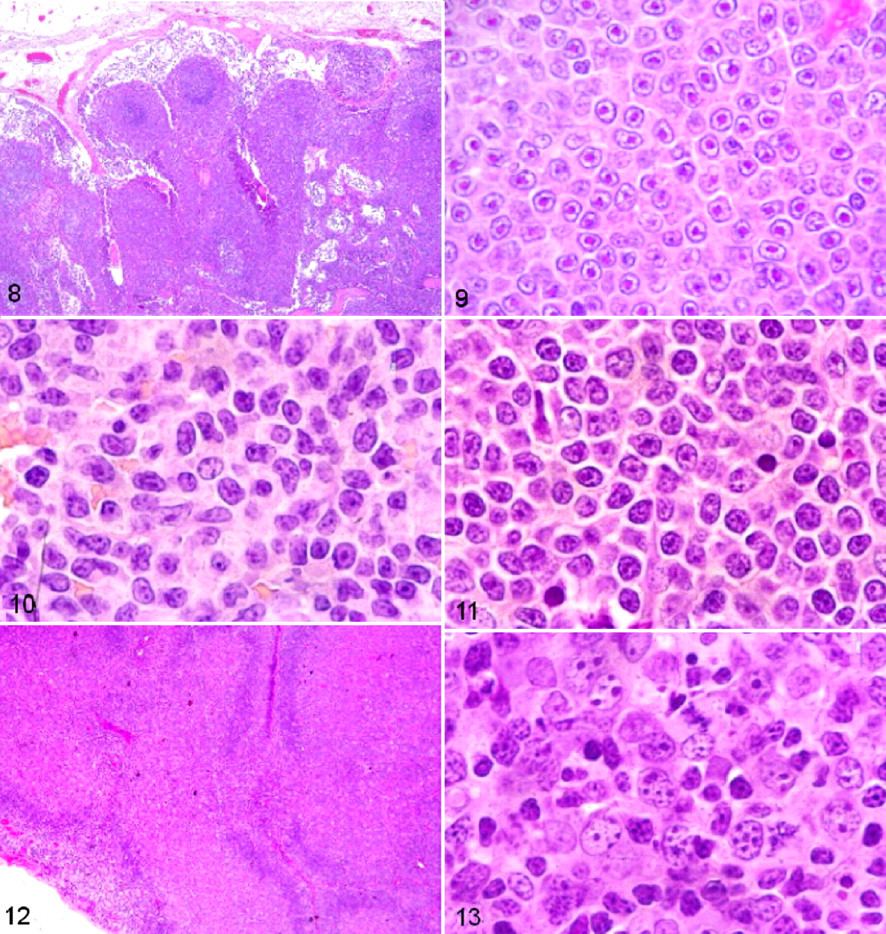
The group of marginal zone lymphomas (66 cases) was recognized as previously described in the REAL and WHO canine classification systems, both on architectural and cytological criteria. In most cases, the infiltrate was diffuse with some pseudonodular areas (58 cases), whereas in others (8 cases), the areas were highly pseudonodular with a clear marginal zone pattern (Fig. 8 ). Tumors often consisted of small- to medium-sized cells with a large central nucleolus, a low MI, and CD79a immunohistochemical positivity confirming a B-cell origin.
The 66 canine marginal zone cell lymphomas were divided into 3 subtype groups according to cytological criteria. The first group consisted of 59 cases (56 nodal and 3 extranodal lymphomas affecting spleen, tonsils, and nictitating membrane) that presented with macronucleolated medium-sized cells (MMCs). MMCs were characterized by a particularly prominent nucleolus in small- to medium-sized cells, with fine chromatin and a moderate amount of weakly basophilic cytoplasm (Fig. 9 ), as previously described. 17,35 A few large cells suggestive of centroblasts and immunoblasts were present in 29 of these 59 cases. The second group consisted of 5 cases (1 nodal and 4 digestive) characterized by monocytoid cells with paler and more abundant cytoplasm, which were interspersed with transformed blasts (Fig. 10 ). The third group concerned 2 cases with nodal involvement that were characterized by small- to medium-sized cells with slightly dispersed chromatin, inconspicuous nucleoli, irregular nuclei, and abundant cytoplasm (Fig. 11 ). Follicular lymphomas were uncommon (0.66%) and were recognized on the basis of a predominantly follicular pattern with a mix of centrocytes, centroblasts, and follicular dendritic cells (Figs. 12, 13). 25,48
T-Cell and Null-Cell Lymphomas
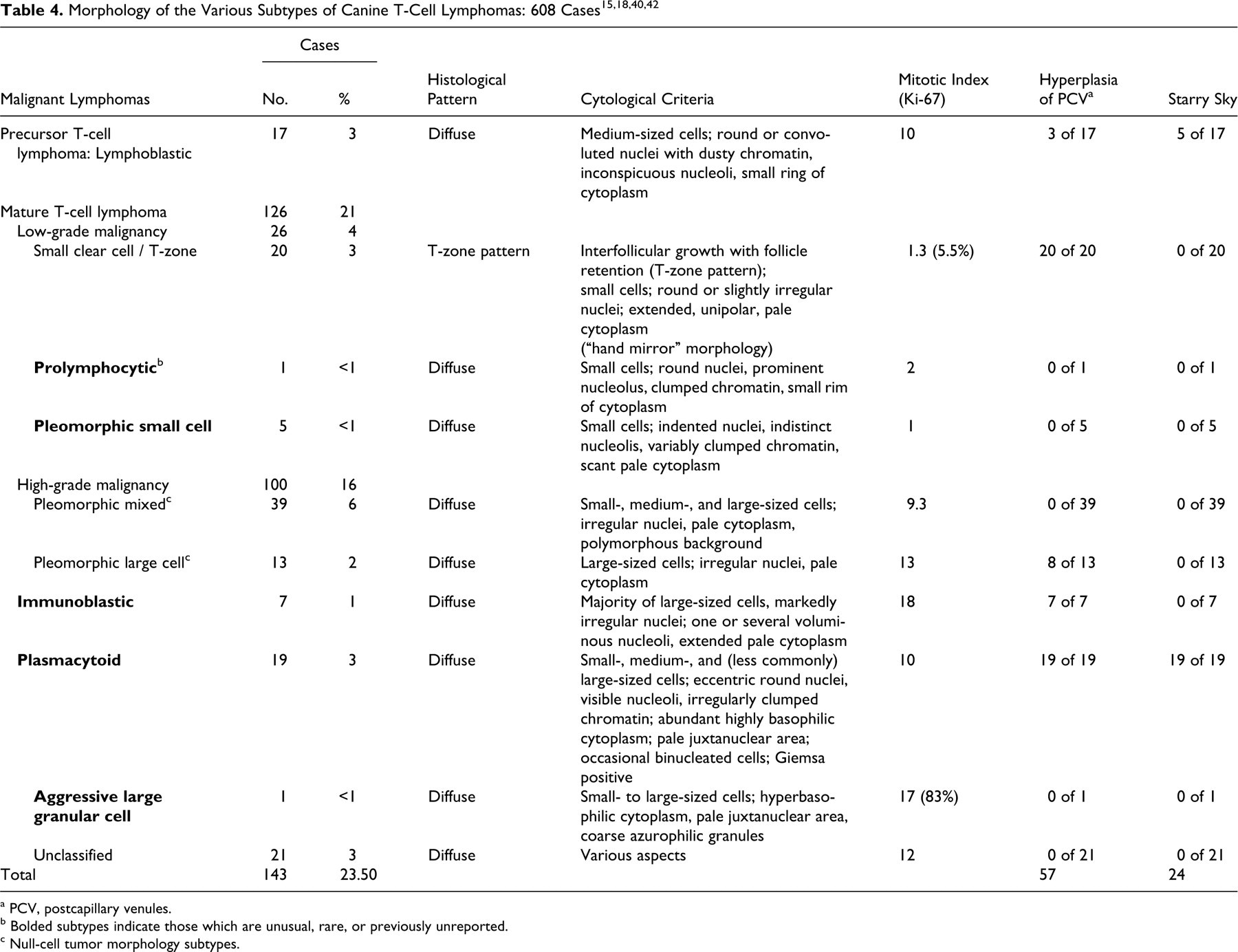
T-cell lymphoma was identified in 215 cases (34.5%), including systemic and cutaneous manifestations (Table 4). The classification of T-cell lymphomas was first based on the fundamental distinction between precursor and mature T cells (17 cases and 177 cases, respectively). Unclassifiable T-cell tumors were present in 21 cases. Among mature T-cell lymphomas, the 72 primary cutaneous lymphomas were separated from the others, as is done in humans for morphologic classification. 25,48
Precursor T-cell lymphoma
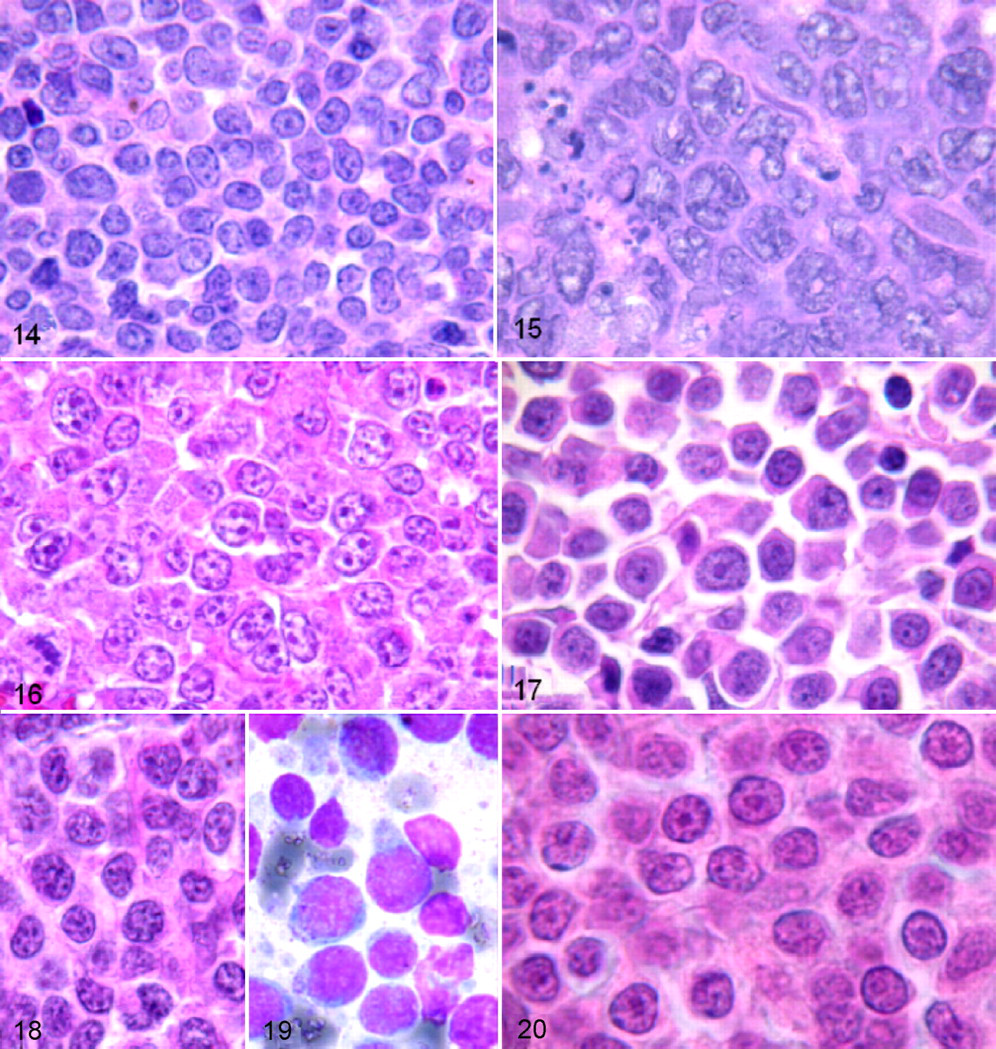
Lymphoblastic lymphoma appeared similar to previously reported cases, with a diffuse infiltration of a homogeneous proliferation of medium-sized cells with a small ring of moderately basophilic cytoplasm, a round to slightly convoluted nucleus, finely dispersed dusty chromatin, inconspicuous nucleoli, and a high MI (median, 10 per HPF) (Fig. 14 ). 40 The 17 dogs affected ranged from 3 to 11 years old (median age, 6.85 years), and all presented with polylymphadenopathy.
Mature or peripheral T-cell lymphomas
The majority of mature T-cell lymphomas identified in this study belonged to well-described subtypes as reported in previous studies. 15,18,42 High-grade T-cell lymphomas were most common with pleomorphic mixed-cell tumors present in 39 cases and pleomorphic large-cell tumors in 13 cases (Fig. 15 ). Remaining high-grade T-cell subtypes included 7 immunoblastic cases, 19 plasmacytoid cases, 40 and 1 recently described aggressive large granular lymphoma subtype.
A new, previously undescribed immunoblastic T-cell type occurred in 7 dogs ranging from 3 to 11 years old (median, 6.84 years). These 7 cases presented with generalized lymphadenopathy with diffuse nodal infiltration resulting in a moderate to pronounced starry-sky pattern. The lymphoma-tous population consisted of large cells with markedly irregular nuclei, fine chromatin, 1 voluminous nucleolus or several central nucleoli of variable size, and an extended pale cytoplasm (Fig. 16 ). The MI was high (median, 18 per HPF).
The plasmacytoid T-cell subtype was similar to that of previously reported cases, 42 with a population of small- to medium-sized cells or, less frequently, large cells and relatively abundant highly acidophilic cytoplasm. The nuclei were eccentrically located, round, or slightly notched (Fig. 17 ). This abundant, highly acidophilic cytoplasm—with eccentric nuclei, occasional binucleation of some cells, and juxtanuclear pale cytoplasmic area—provided a plasmacytoid appearance, which is obvious with Giemsa staining.
A rare subtype of aggressive granular large-cell lymphoma, only recently described in veterinary medicine, 8,19,53 was observed in 1 animal: A 5-year-old male Boxer presented with a primary intestinal mass, abdominal polylymphadenopathy, hepatosplenomegaly, and leukemia with 80% bone marrow infiltration. On tissue sections (Fig. 18 ), there was diffuse infiltration by cells of variable size with round or indented nuclei, fine or slightly clumped chromatin, inconspicuous or distinct nucleoli, and sometimes cytoplasmic azurophilic granules, without any angiocentric or angiodestructive growth pattern. Because samples of digestive tissue adjacent to the intestinal mass were not available, villous atrophy and/or enteropathy could not be evaluated.
On FNA examination (Fig. 19 ), the lymphomatous population was characterized by variable cell size with a majority of large cells, round and sometimes indented nuclei, fine or slightly clumped chromatin, inconspicuous or distinct nucleoli, and hyperbasophilic cytoplasm with a pale juxtanuclear area and coarse azurophilic granules. These neoplastic cells were intermingled with numerous reactive histiocytes displaying hemophagocytosis, and they had a high MI (median, 17 per HPF) and high Ki-67 proliferation index (83%). Low-grade T-cell lymphoma subtypes included 1 prolymphocytic case, 5 pleomorphic small-cell cases, and 20 cases of low-grade small clear cell–type (T-zone) lymphoma, as previously well described. 15,18
The single case of prolymphocytic lymphoma had diffuse lymph node effacement by small cells with round or slightly irregular nuclei, a visible nucleolus, irregularly clumped chromatin, and a small rim of cytoplasm (Fig. 20 ). Leukemia was not observed in this case but was observed in the only 2 cases previously reported. 15
All 5 cases of pleomorphic small-cell lymphoma had small cells with irregularly indented or deeply cleaved nuclei, and they exhibited the same morphological criteria as the 5 canine cases previously described. 15,18 The remaining 21 cases of T-cell lymphoma remained unclassifiable. All had an apparent high-grade morphology on the basis of their MI (median, 12 per HPF) but did not have enough characteristics to enter in a definite subtype.
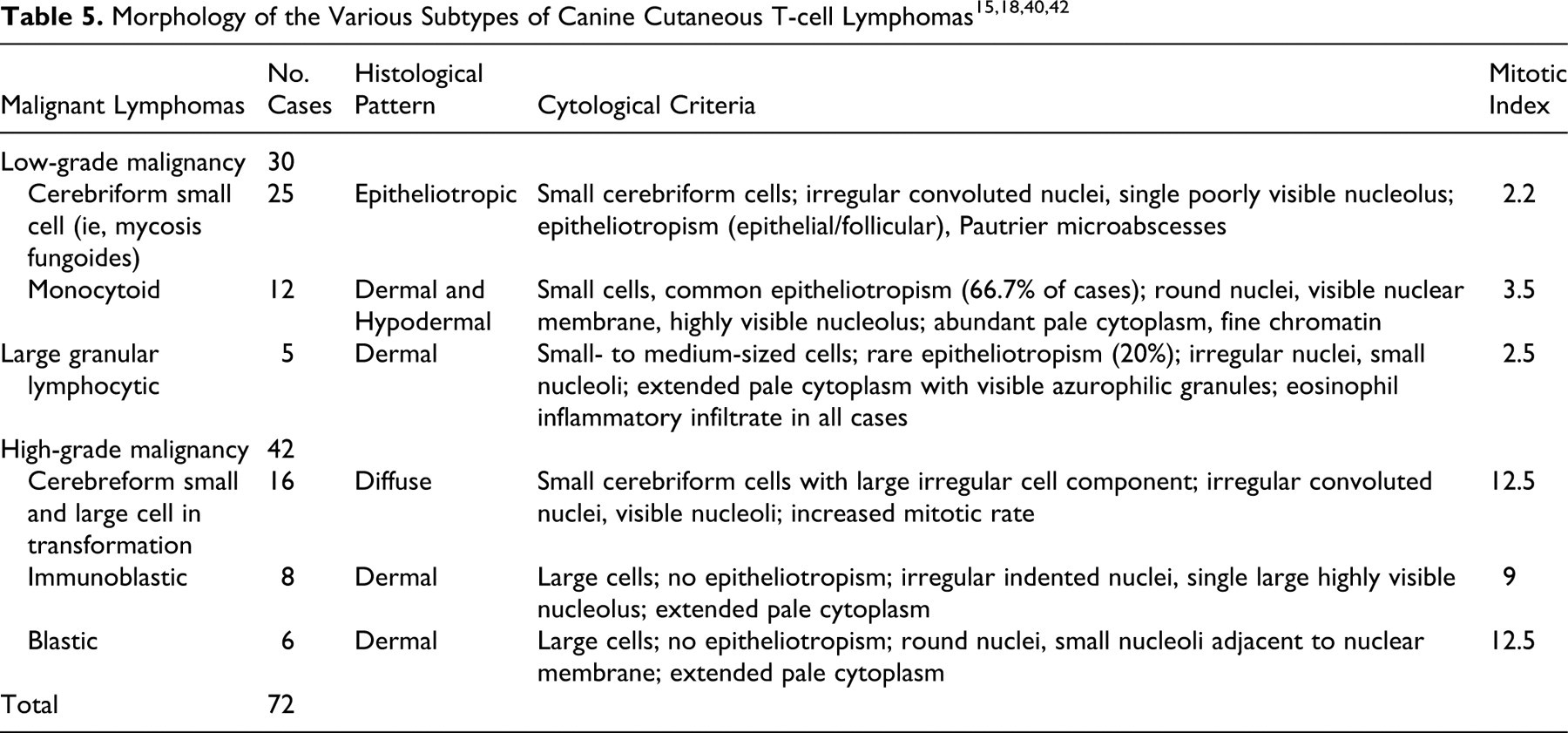
Cutaneous T-cell lymphomas
Cutaneous tumors were all immunohistochemically consistent with T-cell lymphoma (72 cases) (Table 5). This study observed 6 provisional lymphoma subtypes. The 42 cases of low-grade cutaneous lymphoma included 25 cerebriform cell types with low MI (median, 2.2 per HPF), 12 monocytoid types, and 5 large granular lymphocytic types. The 30 cases of high-grade cutaneous lymphomas included 16 cerebriform cell types with high MI (median, 12.5 per HPF), 8 immunoblastic types, and 6 blastic types. The cerebriform cell type with low MI was consistent with the classically termed mycosis fungoides (MF). 15,18 However, the transitional to high-grade cerebriform subtype appeared to have some transformation with larger cells, visible nucleoli, and increased mitotic rate.
Monocytoid types comprised small cells with abundant pale cytoplasm, round nuclei containing fine chromatin, and a highly visible nucleolus, and they commonly showed epitheliotropism. Large granular lymphocytic types comprised medium-sized cells with moderate cytoplasm containing azurophilic granules, slightly irregular nuclei with clumped chromatin, and small nucleoli, and they rarely had evidence of epitheliotropism.
High-grade immunoblastic and blastic types were similar in the presence of large cells, with pale cytoplasm and an absence of epitheliotropism, but they differed in nuclear morphology, with immunoblastic cells having an irregular indented nucleus with a single large prominent nucleolus and with blastic cells having a round nucleus with multiple small nucleoli adjacent to the nuclear membrane.
Null-cell lymphomas
The 5 cases of null-cell lymphomas did not express CD3 and CD79a or light or heavy chains of immunoglobulin. Infiltration was diffuse; 4 cases were morphologically classified as pleomorphic mixed lymphomas and 1 as pleomorphic large-cell lymphoma. Descriptions are the same as those given for these high-grade T-cell lymphoma subtypes in Table 4.
Canine Versus Human Lymphoma: Morphologic Comparisons
B-cell Tumors
The single case of anaplastic mediastinal lymphoma in this case study was considered atypical because of the presence of fibrosis and binucleated neoplastic cells with pleomorphic nuclei and abundant cytoplasm; otherwise, it was consistent with that reported clinically and architecturally for the mediastinal variant of diffuse large B-cell lymphomas (DLBCLs) in humans (Table 6). In contrast to humans, this variant was observed in a middle-aged to older male dog, as opposed to the common human epidemiology of a younger female. 25,48
Four canine cases were classified by the Kiel system as small-cell lymphoma NOS, which had characteristic small-cell morphology but a high mitotic rate and Ki-67 index, suggesting a high-grade lymphoma phenotype. The increased mitotic rate in conjunction with the small-cell morphology and the B-cell phenotype are suggestive of the reported human WHO classification of mantle cell lymphoma. 25,47,48,51 However, additional immunophenotyping to confirm this diagnosis was not performed, and further classification beyond the Kiel classification of small cell NOS was not possible.
Correspondence with human WHO classification for the low-grade canine B-cell Kiel classifications of 2 small lymphocytic cases and 1 small prolymphocytic case was difficult to establish because of an absence of certain morphological, immunophenotypic and genotypic features normally observed in humans. In humans, small lymphocytic lymphomas of the B-cell chronic lymphocytic leukemia/small lymphocytic lymphoma subclass of the WHO classification are a neoplasm characterized by a pseudofollicular pattern with regularly distributed pale areas containing small lymphocytes admixed with prolymphocytes and paraimmunoblasts (larger cells with dispersed chromatin and more prominent central nucleolus). Owing to the lack of these architectural features and paraimmunoblasts and in the absence of more information on immunophenotype and cytogenetic abnormalities (ie, a scoring system that helps to differentiate chronic lymphocytic leukemia/small lymphocytic lymphoma from other B-cell entities in humans), these 2 canine small lymphocytic lymphomas were classified as unspecified low-grade small B-cell lymphomas.
Regarding the sole prolymphocytic case, the lack of blood and/or bone marrow involvement is consistent with the 2 canine cases previously published. 15 Despite the same morphological aspects as human B-cell prolymphocytic leukemia, the lack of leukemia in dogs could be more suggestive of a variant of small lymphocytic lymphoma with an increased number of prolymphocytes. 25,48
Cases of canine low-grade B-cell lymphoplasmacytic lymphomas had cellular morphology and immunological findings that were suggestive of the human WHO classification of lymphoplasmacytic lymphoma, except for the growth pattern, which is diffuse for this tumor subtype in humans 21,25,48 rather than pseudofollicular as in canines. Based on this nodular orientation, plasmacytic differentiation of the cells appears more consistent with a human variant of marginal zone lymphoma with follicular colonization and plasma cell differentiation 43 than with the human lymphoplasmacytic lymphoma, which displays a diffuse pattern in the lymph node.
Marginal zone lymphomas in human medicine describe proliferation of morphologically heterogeneous small B cells, including marginal zone (centrocyte like) cells, monocytoid cells, small lymphocytes, and scattered transformed B cells 25,48,51 arranged as an encircling cuff that arises outside the mantle cell layer surrounding germinal centers. In humans, marginal zone lymphomas are separated into 3 subtypes: the nodal lymphoma and 2 subtypes that arise from extranodal sites, including MALT (mucosa-associated lymphoid tissue) lymphoma and splenic lymphoma. Canines in this study appeared to have similar subtypes as described above, with the last 2 canine subtype groups consistent with those described in cases of human marginal zone lymphoma. 25,48,51 The canine nodal version of marginal zone lymphoma, with the characteristic canine MMCs as previously described, 17,35 appeared most frequently (56 of 66). The extranodal sites in affected canines had a distribution similar to that reported in humans, with 6 apparent MALT sites affected (4 enteric, 1 tonsil, and 1 nictitating membrane) and 1 apparent splenic site affected. Whereas the majority of cases had characteristic canine MMCs, 5 cases (1 nodal, 4 digestive) showed a monocytoid appearance, and 2 other cases with nodal involvement showed a centrocyte-like appearance more consistent with the cell morphology described in cases of human marginal zone lymphoma. 21,23,25,48
The 3 low-grade B-cell centroblasto-centrocytic lymphomas in this case study displayed a clear follicular pattern with a mix of centrocytes, centroblasts, and follicular dendritic cells as observed in human follicular lymphomas. 25,48 Their grading, as described in the human WHO classification, 25,48 is based on the proportion of large centroblasts to smaller centrocytes (grade 1 = 0–5 centroblasts per HPF, grade 2 = 6–15 centroblasts per HPF, grade 3 = 15+ centroblasts per HPF). In this study, the 3 canine cases could be classified as grade 2. In addition, the sole case of high-grade lymphoma described as centroblastic monomorphic with a follicular pattern could be classified as grade 3 follicular lymphoma according to the abundance of centroblasts.
T-cell Tumors
As in humans, precursor T-cell lymphoma (ie, lymphoblastic lymphomas) in dogs is rare (2.00% and 2.82% of all NHLs, respectively) and primarily of T-cell origin (85 to 90% and 100% in our study, respectively). 25,31,40,48 As reported in people, male dogs in this study appeared more commonly affected than females (11 vs 6), and dogs affected by this subtype are younger than those for the other T-cell subtypes (6.82 vs 8.22 years). 40 Cytologically, precursor T-cell lymphomas in dogs and humans had a similar cellular morphology and a high mitotic rate, as previously described. 40
Most of the mature T-cell lymphoma subtypes observed in the dogs of this study were similar to the human WHO classification of peripheral T-cell lymphoma unspecified, including the low-grade pleomorphic small-cell and high-grade pleomorphic mixed, pleomorphic large-cell, and immunoblastic T-cell subtypes. 25,48
Canine low-grade, small, clear-cell (T-zone) lymphoma, a well-established entity in dogs, 15,18 could be compared with the human T-zone lymphoma of the Kiel classification 33 —particularly, the clear-cell-rich subtype—on the basis of its histological pattern, cytological characteristics, and a more indolent course than that of other T-cell peripheral lymphomas. In our study, CD4 immunolabeling was not performed, but both cases of canine T-zone lymphoma previously published 18 were CD4+, as reported in humans. 33 This human Kiel subtype is described in the human WHO classification as a T-zone variant of the peripheral T-cell lymphoma unspecified. 25,48
Nineteen cases of canine high-grade plasmacytoid T-cell lymphomas were observed in our study. Lennert 33 described a plasmacytoid T-cell lymphoma in the human Kiel classification; however, this category in humans has become obsolete in the WHO classification because further examination with specific antibodies and in situ probes has resulted in regrouping of entities. Furthermore, this human subtype had an indolent course (Ki-67 index, 2%), whereas our 19 cases appeared aggressive, with an MI of 10 per HPF. 25,48 Even though canine plasmacytoid T-cell lymphomas appeared clearly different from Lennert’s human plasmacytoid T-cell lymphomas, the name plasmacytoid was conserved owing to the specific cell morphology (abundant highly acidophilic cytoplasm, pale juxtanuclear area, and eccentric round nuclei).
The single case of low-grade prolymphocytic lymphoma had morphological characteristics similar to those reported in humans with T-cell chronic lymphocytic leukemia/prolymphocytic leukemia, but the absence of leukemia in this canine case makes comparisons questionable and does not allow a definite classification. 25,48
Cutaneous T-Cell
Human cutaneous T-cell lymphomas occur more frequently than do cutaneous B-cell lymphomas, and among them, MF and its variants are the most frequent (50% of cases), 6,10,11,36,48 similar to the cases observed in canines in this study (56%). Canine cases of cutaneous lymphoma with epitheliotropism and cerebriform cells are similar to those described with MF and Sezary syndrome in the human WHO classification. 25,48 In humans, it is well established that MF may transform into large T-cell lymphoma defined by the presence of more than 25% blast cells. 44 This form is more aggressive, with organ involvement and loss of epitheliotropism. 10,25,46,58 In this study, there were 16 cases that probably represent transformed MF, characterized by a mixture of large blastic cells and small cells with cerebriform nuclei and by a high MI (median 12.5 per HPF) as suggested by previously published studies. 18
Discussion
The predominance of high-grade lymphomas (75.49%) over low-grade lymphomas (24.51%), the high frequency of large B-cell lymphomas (47.38%) and pleomorphic T-cell lymphomas (9.37%), and the rarity of follicular lymphomas (0.66%) in dogs are consistent with the findings of previous veterinary studies. 15,18,20,50 From an epidemiological viewpoint, this study did not show any difference in the mean age (8 years) of dogs suffering from lymphomas when compared with previous veterinary studies. 20,26,49,50 The correlation between the age of dogs and morphological lymphoma subtypes (eg, lymphoblastic T-cell and plasmacytoid T-cell lymphomas in younger dogs and cutaneous lymphomas in older dogs) 18,40,42 and the overrepresentation of Boxers in the whole of lymphomas, particularly in T-cell lymphomas, are consistent with preestablished data. 18,26,34,36,38,40,42,50 No sex predilection was observed in this study of canine lymphomas, a highly debated point in previous studies. 18,26,29,49
Regarding frequency of T-cell lymphoma in dogs in comparison with humans, examination of a statistically significant proportion of canine cases of lymphoma in France showed that 35.4% of affected animals have T-cell lymphoma, confirming findings reported in previous studies. 16,18,49 In this study, the proportion of canine T-cell lymphoma cases, which proved much higher than that in humans, was clearly related to the frequency of cutaneous T-cell lymphoma (33.49% of T-cell NHLs). The frequency of cutaneous T-cell lymphoma observed here appears much higher than that reflected by the percentages reported in previous veterinary morphological studies. 18,20,50 The increased number of cases observed in this study could be explained by the sampling of study materials from pathology laboratories, which are more likely to receive skin masses from veterinary practices (ie, masses that are easily observed and surgically removed). An increase in the number of skin samples compared with lymph node or other organ samples would result in an apparent increase in the proportion of cutaneous lymphoma cases.
Clinically, the low frequency of extranodal involvement in canine lymphomas (18%) in comparison with human lymphomas (30%) 24,25,46,48 should be cautiously interpreted because of the retrospective nature of this study and the primary use of histological materials. Available clinical data mainly consisted of predominating presenting signs and profound lesions, which could have underestimated the degree of extranodal involvement in favor of superficial involvement (eg, superficial lymphadenopathy and cutaneous lymphomas). Additional clinical data relating to the different canine lymphoma subtypes are necessary to determine the clinical profile of each entity.
This study highlights the proportion of cases and morphological aspects of common canine lymphoma subtypes, as well as those rarer subtypes that could have been underestimated in more restricted, previously published studies. In addition to morphologic descriptions, some apparent morphologic similarities were observed among canine malignant lymphoma subtypes (as identified with the Kiel classification system in this large population from 7 different veterinary laboratories throughout France) and among the human malignant lymphoma subtypes (as reported in the WHO classification).
Canine Lymphoma Morphology
B-cell Lymphomas
B-cell lymphoma subtypes, including the high-grade large B-cell lymphomas of the centroblastic polymorphic subtype and the immunoblastic subtype, were the most common in this canine population, and they have been well documented in previous veterinary studies. 15,20 However, unusual or rare B-cell lymphoma subtypes were observed, including high-grade types such as anaplastic mediastinal, Burkitt type, and small-cell lymphoma NOS, as well as low-grade marginal zone and follicular lymphomas. In addition, a provisional subtype of high-grade Burkitt-like lymphoma with plasmacytoid morphology was identified (discussed in the comparative section below).
In this study, canine Burkitt-like tumor morphology was similar to the 3 previously published cases in veterinary medicine. 4,15,56 All cases showed a high growth fraction, with 80% of cells positive for Ki-67. This specific morphology is an important subtype to be recognized by pathologists because of the propensity for a poor prognosis as previously published. 41
Small B-cell lymphomas generally have low mitotic activity and are considered a low-grade phenotype. The 4 cases of small B-cell lymphoma NOS observed here had a classic small-cell morphology but a high mitotic rate, averaging 8.6 per HPF, compared with the usual small B-cell mitotic rate, ranging from 2 to 3 per HPF. The diffuse or nodular pattern observed in the 4 unusual cases in this study of canine small cell NOS, along with the small-cell morphology, the high MI, and the B-cell immunophenotype, may be compared to that of the 3 canine cases of mantle cell lymphoma described in a recent study by Valli et al. 57 They described 3 cases of canine splenic mantle cell lymphoma on the basis of histological and cytological criteria—namely, proliferation of small cells with internal nuclear detail, without nucleoli, and centered on end arterioles. However, in this study, the lack of cyclin D1 positivity 9,25,48 and absence of monoclonal antibodies specific to or cross-reacting with canine CD5 did not allow definite identification of a mantle cell subtype.
Among the low-grade lymphomas, the typical canine nodal marginal zone cell subtype (marginal zone lymphoma) is well defined in the literature and is clearly recognizable because of its marginal zone pattern of growth and characteristic cytological presentation, including specific MMCs. However, this study pointed out a rare cytological presentation not previously described in veterinary studies—including small- to medium-sized cells, slightly dispersed chromatin, inconspicuous nucleoli, irregular nuclei, and abundant cytoplasm—observed in 2 of the 66 canine nodal marginal zone lymphomas.
Regarding extranodal marginal zone lymphomas in this canine population, MALT and splenic marginal zone lymphomas appeared rare and slightly different from those of a recent canine study, which reported 8 tumors with exclusive splenic involvement among 46 marginal zone lymphomas. 57 Cases may be underestimated in our study given that digestive and splenic biopsies are not systematically performed in animals with evidence of lymphoma in the lymph node and/or skin.
Follicular lymphomas are well defined in the veterinary and human WHO classification systems but were not specifically described in the Kiel classification system. Follicular lymphomas in canines in this study were rarely observed, as in previous veterinary studies. 4,15,20,49,51,56,57
In addition, there was an absence of lymphoblastic B-cell lymphoma, also known as precursor B-cell lymphoma, in this significant European series, which is consistent with previous reports in French canine populations. 15,40 This result based on the Kiel classification system is slightly different from a recent veterinary book that described some cases of lymphoblastic B-cell lymphoma. 56
T-cell Lymphomas
The morphological presentation of most T-cell lymphomas is highly specific, hence enabling pathologists to strongly suspect T-cell phenotypes. 15,18 In regard to morphological presentation and proportion of cases, the findings relating to T-cell lymphomas in this study are similar to those reported in recent veterinary publications. 18,49,50,55 The low-grade pleomorphic small-cell lymphomas and high-grade pleomorphic mixed and large-cell lymphomas constituted the majority of nodal T-cell lymphomas identified.
Two previously provisionally classified tumors, considered specific T-cell subtypes on the basis of their clinical behavior, were observed in greater numbers than those previously reported. 41 These included the low-grade small clear-cell (T-zone) lymphoma and high-grade plasmacytoid lymphoma. 15,18,31,37,40,41,46,49,50 Most important, 7 cases of high-grade immunoblastic T-cell lymphoma were observed and so represent the first identified cases of an immunoblastic T-cell subtype in canines.
Two questionable types of canine T-cell lymphoma were named as provisional entities: one case of low-grade T-prolymphocytic lymphoma and one case of aggressive high-grade large granular cell lymphoma. The single case of T-prolymphocytic lymphoma appeared to be similar to the 2 previously published canine cases. 15 This may constitute a rare category of low-grade canine T-cell lymphoma/leukemia described in the updated canine Kiel classification, but further clinical evaluation is necessary for confirmation.
The single case of aggressive large granular lymphocytic T-cell lymphoma appeared to have a similar morphology to that of the 6 cases described in recent veterinary studies. 8,19,53 The low number of cases observed in these studies may be due in part to the sampling of study materials from tissue biopsies easily removed from superficial lesions in veterinary practice, whereas the clinical presentation of this type of lymphoma consists of profound infiltration (spleen, liver, abdominal lymph nodes) without superficial, visible lesions.
Finally, 21 unclassifiable T-cell lymphomas were defined as those that did not fit a definitive or provisional category of the updated Kiel classification system. Morphological, immunological, and clinical data were insufficient to venture an assumption concerning the cells of origin. Further immunophenotypic and genetic studies should be carried out on these tumors for classification purposes.
Cutaneous T-cell Lymphoma
In this study, cutaneous T-cell lymphomas occurred more frequently than B-cell lymphomas. Small-cell cerebriform epitheliotropic tumors (25 cases), which likely represent MF and its variants, were the most common (56%). 36,56 For a long time, canine cutaneous small-cell lymphomas were most commonly classified as MF based on the presence of epitheliotropism and CD3 positivity. However, 2 distinct morphologic subtypes have epitheliotropism and CD3 positivity but may be different from MF—namely, monocytoid-type lymphoma (12 cases in this study) and large granular lymphocytic lymphoma (5 cases).
Monocytoid-type lymphoma differs from MF by the presence of homogeneous cells with small round nuclei containing one highly visible nucleolus and abundant pale cytoplasm and by the absence of small cells with cerebriform nuclei as observed in cases of MF. We are unsure of the clinical course of this subtype of lymphoma and whether it exhibits the same clinical features as MF (patches, plaques, and nodules) before treatment. Prospective studies are absolutely necessary to point out clinical and therapeutic differences between monocytoid-type lymphoma and MF, if any.
Large granular lymphocytic lymphoma differs from MF primarily by the presence of cytoplasmic acidophilic granules but shows a more regular nucleus with more condensed chromatin and small nucleoli. Similar cases have shown a high proliferation rate with a poor response to chemotherapy and usually a TiA-1+ immunophenotype (personal observation).
Null-Cell Lymphoma
Publications on null-cell lymphomas are rare. 43,53 One recently reported a null-cell phenotype for aggressive large granular lymphocyte lymphomas, diagnosed owing to the presence of cytoplasmic granules. 53 In our study, the null-cell cases were classified as pleomorphic subtypes on the basis of cell morphology in the absence of cytoplasmic granules. TiA-1 or other cytotoxic markers were not available for further classification. Null-cell types remain immunologically poorly characterized because of the lack of canine CD56 immunomarkers to identify natural killer cell lymphomas, as tested by Turinelli et al. 53
Canine Versus Human Lymphoma: Morphologic Comparison
B-cell Lymphoma
High-grade large B-cell lymphomas of the centroblastic polymorphic subtype and the immunoblastic subtype, which were the most common tumors observed in this study, appear morphologically similar to the DLBCLs described in the REAL and WHO human classifications. 24,25 These findings are well documented and consistent with previous veterinary and human studies. 13,15,20,24,25,46,49
The only case of anaplastic mediastinal B-cell lymphoma observed during our study appears similar to that described in a recent veterinary study, 56 and it probably also falls within the category of DLBCL. Along with other high-grade B-cell neoplasms, including centroblastic polymorphic, diffuse centroblastic monomorphic, and immunoblastic B-cell lymphoma, mediastinal lymphomas are considered part of DLBCL as described in the human WHO classification system based on cell size, irregular nuclear outlines, abundant cytoplasm, and association with nodular sclerosis. 25,48
As in dogs, this DLBCL group of B-cell tumors is reported to be the most common lymphoid neoplasm subtype in human adults, as well as almost all other species. In humans, DLBCL tumors consist of sheets of large transformed B lymphocytes that architecturally obliterate the normal tissue morphology and occur at nodal and extranodal sites—particularly, enteric and mucosal (MALT) sites—as was observed in dogs with these tumor subtypes. In canines but not humans (as yet), MMCs are commonly observed in centroblastic polymorphic and immunoblastic lymphoma cases, as intermixed with large blastic cells, suggesting transformation of these tumor types from marginal zone lymphoma. 41
Human Burkitt-type lymphomas, classified as a high-grade B-cell NHL, are characterized by a more pronounced starry-sky pattern than are other fast-growing lymphomas, a high mitotic and apoptotic rate, medium-sized cells, round nuclei, clumped chromatin, and relatively clear parachromatin. 25,48 In this study, canine Burkitt-like tumor morphology was similar to that of the human subtype and the 3 previously published cases in veterinary medicine. 4,15 Moreover, a high growth fraction was observed in canine tumors, with 80% of cells positive for Ki-67, which is close to the almost 100% Ki-67 positivity reported in human Burkitt-type lymphoma. 13,25,48
Diagnosis of the provisional highly aggressive plasmacytoid B-cell lymphoma raises the question of a possible variant of a Burkitt-type lymphoma (plasmacytoid variant) as it exists in the human WHO classification but with some morphological differences, including smaller cells, finer chromatin, and the absence of a single central nucleolus and a starry-sky pattern. Alternatively, consideration must be given to transformation of a generally low-grade small B-cell lymphoma toward a higher grade owing to the presence of small- to medium-sized cells with higher MI. 25,47,48
The 4 canine cases of high-grade small B-cell lymphoma NOS present in this case study had morphologic similarities to the 3 canine cases of mantle cell lymphoma described in a recent study by Valli et al. 57 The mantle cell lymphoma is one of the worst malignant lymphomas in humans, with a poor median survival time (less than 3 to 5 years). 25,47,48 This high-grade small-cell lymphoma does not undergo transformation to a large-cell lymphoma, but a high MI (> 20 per HPF, 10×), peripheral blood involvement, and/or increase in nuclear size and chromatin dispersal are associated with a poor survival time. Morphologic variants are reported for human mantle cell lymphomas, such as blastoid, pleomorphic, marginal zone–like, and particularly small lymphocytic types. This last type may be difficult to differentiate from low-grade small B-cell lymphomas such as small lymphocytic lymphomas. 48 In humans, immunophenotyping (CD5 and cyclin D1 positive, CD10 and CD23 negative) and genotypic/cytogenetic features—translocation t(11;14)(q13;q32)—are usually sufficient to make this differential diagnosis. 9,25,47,48 Immunophenotyping for immunoglobulin heavy chain, cyclin D1, or gene translocations as reported in humans was not performed in either canine study owing to insufficient development of canine cytogenetic analyses. 57 In some cases, a definitive diagnosis among small B-cell lymphoma subtypes is difficult because of overlapping morphologic and immunophenotypic features and/or lack of specific genotypic anomalies. In these cases, only an early diagnosis with a preserved architecture allowing the observation of a particular mantle cell pattern, a precise morphological description, and the clinical outcome are useful to distinguish the highly aggressive mantle cell lymphomas from the other indolent small B-cell lymphomas.
The different Kiel subtypes of canine low-grade small B-cell lymphoma identified in this study, including small lymphocytic, prolymphocytic, and lymphoplasmacytic subtypes, were difficult to classify and compare with the various well-known entities of the human WHO classification. In the human WHO classification, the group of small B-cell lymphomas includes B-cell chronic lymphocytic leukemia/small lymphocytic lymphoma; mantle cell lymphoma; splenic, nodal, or MALT marginal zone lymphomas; and lymphoplasmacytic and follicular lymphomas. 9,25,47,48
Small B-cell tumors are generally indolent, but a variable proportion may undergo transformation to large-cell lymphomas. 47 The small size of the B cells is not enough to predict clinical behavior, so it is important for the pathologist to make a precise morphological analysis of each case and to perform a proliferation index, such as Ki-67, in addition to the classical MI to differentiate mantle cell lymphomas that, at least in humans, have a worse prognosis. Additional studies are important for these tumors in canines as well because they often require intensive evaluation, including early diagnosis and further diagnostics.
In regard to marginal zone lymphoma, the presence of specific MMCs has been described in canines 15,17,35 but appears to constitute an original difference from its human equivalent. 23 –25,48 This study pointed out a rare cytological presentation described earlier in 2 of the 66 canine nodal marginal zone lymphomas, which had not been described in veterinary studies. However, the cytological morphology is close to that described for the classical form of human marginal zone cells. 48,51,52 In regard to extranodal marginal zone lymphomas, the specific cytological presentation of the infiltrates of monocytoid cells interspersed with transformed blasts in the 4 extranodal digestive mucosal-associated canine cases described in this study resembled their human equivalents. 23,48
Follicular lymphomas are well defined in the veterinary and human WHO classification systems. 15,25,48,56,57 In this study, the 4 cases of follicular lymphoma could be divided into different morphological grades, as described in the human WHO classification. 25,48 Three cases showed a mixture of centrocytes and centroblasts (centroblastic–centrocytic type) and could be classified as grade 2. The fourth case, showing a majority of centroblasts (centroblastic monomorphic type) and high MI, could be classified as grade 3. Follicular lymphoma appears generally uncommon in dogs, but this trend is in opposition to reports in humans, which are more common. 25,48 Differences regarding incidence of this subtype among species may be explained either by a late diagnosis in dogs or, more likely, by the rapid and diffuse invasion of highly proliferating blastic forms of follicular origin. 15,20,49,51 For more questionable cases, the bcl-2 protein, expressed in the majority of human cases, should be further investigated in dogs. 25,48
T-cell Lymphoma
The human WHO classification of unspecified peripheral T-cell lymphoma is a general catchall that is becoming obsolete in people because of the increased use of additional genetic and diagnostic tests. However, the absence of similar testing available for canine species results in the classification of most canine T-cell tumors identified by the Kiel classification system into this human WHO subtype—including low-grade pleomorphic small-cell lymphoma, low-grade small clear-cell (T-zone) lymphoma, high-grade pleomorphic mixed and large-cell lymphomas, high-grade immunoblastic T-cell lymphomas, and high-grade plasmacytoid lymphomas.
The single case of aggressive large granular lymphocytic T-cell lymphoma observed in this study was more difficult to classify. Aggressive to fulminant clinical course, multiorgan involvement, cytoplasmic azurophilic granules within neoplastic cells, and intermingled reactive histiocytes with hemophagocytic syndrome, as reported in this case, are close to clinical and morphological features described in human aggressive natural killer cell leukemia/lymphoma or extranodal natural killer cell/T-cell lymphoma, nasal type. Because of the presence of a primary intestinal mass, this case may also be compared with human enteropathy-associated T-cell lymphoma. However, characteristic epitheliotropism of neoplastic cells found in human cases of enteropathy-associated T-cell type, along with celiac disease, was not found. 24,25,37,48 Further investigations, mainly immunophenotypical (CD56, granzyme M) and genetic (presence of T-cell receptor gene rearrangement or not), should be carried out to distinguish which of the tumors from this larger group of canine aggressive large granularlymphocytic T-cell lymphoma arise from the innate immune system (aggressive natural killer cell leukemia/lym phoma or extranodal natural killer cell/T-cell lymphoma, nasal type) or from the adaptive immune system (enteropathy-associated T-cell lymphoma). 25,48
The single case of provisionally named T-prolymphocytic lymphoma observed in this study, which is similar to the 2 previously published canine cases, 14 may constitute a rare updated Kiel classification category of low-grade canine T-cell lymphoma/leukemia. However, further clinical and immunophenotypical investigations (especially, presence of leukemia, aggressive clinical course, and TCL1 oncogene overexpression) are required to compare it with the T-prolymphocytic lymphoma/leukemia of the human WHO classification. 25,48
Finally, unclassifiable T-cell lymphomas were defined as those that did not fit either a definitive or provisional category of the updated Kiel classification system and therefore could not be further evaluated. Morphological, immunological, and clinical data were insufficient to venture an assumption concerning the cells of origin. Further immunophenotypic and genetic studies should be carried out on these tumors for classification purposes.
Cutaneous T-cell Lymphoma
In humans, primary cutaneous lymphomas may present a specifically skin-oriented morphology, such as MF, or resemble their nodal counterparts but with a different phenotype, genotype, or clinical behavior, such as cutaneous follicular lymphomas. 25,48 Canine cases of cutaneous lymphoma in this study with epitheliotropism and cerebriform cells are most consistent with MF and/or Sezary syndrome in the human WHO classification. 25,48 Several other primary cutaneous human lymphomas—including cutaneous gamma–delta T-cell lymphoma, primary cutaneous CD8+ aggressive epidermotropic cytotoxic T-cell lymphoma, or extranodal T-cell lymphoma of nasal type—share common histomorphological and immunohistochemical features with MF, such as epitheliotropism, a small pleomorphic cell component, and either surface or cytoplasmic CD3 positivity. Only aggressive or highly aggressive behavior and usual positivity for cytotoxic proteins (granzyme, perforin, TiA-1) distinguish them from MF. 32
It is more difficult to classify the other 14 cases of cutaneous lymphoma, which are composed only of large blastic cells, including 8 immunoblastic and 6 blastic types. Among these high-grade lymphomas, some may result from the complete transformation of small-cell lymphomas (such as MF), but the others are probably real primary large-cell lymphomas, as in humans. 25,32 In humans, primary cutaneous large T-cell lymphomas expressing CD30 antigen bear a more favorable prognosis. Unfortunately, no canine CD30 antibodies were available, and human monoclonal antibodies usually used to recognize CD30 antigen do not cross-react with canine lymphocytes (personal observation). 2,6,32
This large and significant population of dogs with malignant lymphoma has confirmed some previously published information as well as shown evidence of new cytohistological entities present in the French population. Moreover, many clinico-morphological entities initially characterized with the updated Kiel classification for canine NHLs appear to have similarities to those reported in the human WHO classification system, including Burkitt-type lymphoma, immunoblastic T-cell lymphoma, aggressive large granular T-cell lymphoma, various subtypes of cutaneous T-cell lymphoma, and others. To truly determine the significance of canine malignant lymphoma in relation to human malignant lymphoma, consistent classification of lymphoma subtypes between the 2 species would be useful. Although direct comparison between Kiel lymphoma subtypes and WHO classification are difficult because of the requirement of clinical data and cellular response to treatment in the Kiel classification system, this initial morphologic comparison between the 2 species will serve as a springboard for further studies.
Footnotes
Acknowledgements
We wish to thank the following 7 veterinary pathology laboratories for their valuable contribution to our study by providing the histological material: Laboratoire d’Histo-Cytopathologie Vétérinaire, Drs Mialot and Lagadic, Maisons-Alfort; Laboratoire de Pathologie Clinique, Ecole Vétérinaire de Lyon, Prof Fournel-Fleury, Prof Magnol; Laboratoire Vetopath, Drs Pitou, Breton, and Sales, Sophia Antipolis; Laboratoire d’Anatomie Pathologique Vétérinaire, Metz; Laboratoire d’Anatomie Pathologique Vétérinaire du Sud Ouest, Drs Poujade and Degorce, Toulouse; Laboratoire d’Anatomie Pathologique Vétérinaire, Drs George, Lenet, Longeart, and Amboise; Laboratoire d’Anatomie Pathologique, Ecole Vétérinaire de Nantes, Prof Wyers. We also wish to thank S. Balleydier and C. Bonnefont-Rebeix for their technical support and Dr Christy McKnight for her editorial input. This study was funded by the Association pour la Recherche contre le Cancer, Fondation pour la Recherche Médicale, Ligue contre le Cancer Région Rhône-Alpes, Fondation de France, Académie de Médecine, and Ecole Nationale Vétérinaire de Lyon.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded by the Association pour la Recherche contre le Cancer (ARC), Fondation pour la Recherche Médicale (FRM), Ligue contre le Cancer Région Rhòne-Alpes, Fondation de France, Académie de Médecine, Ecole Nationale Vétérinaire de Lyon.