Abstract
Enterotoxemia caused by Clostridium perfringens type D is an important disease of sheep and goats with a worldwide distribution. Cerebral microangiopathy is considered pathognomonic for ovine enterotoxemia and is seen in most cases of the disorder in sheep. However, these lesions are poorly described in goats. In this article, we describe the vasculocentric brain lesions in 44 cases of caprine spontaneous C. perfringens type D enterotoxemia. Only 1 goat had gross changes in the brain, which consisted of mild cerebellar coning. However, 8 of 44 (18%) cases showed microscopic brain lesions, characterized by intramural vascular proteinaceous edema, a novel and diagnostically significant finding. The precise location of the edema was better observed with periodic acid–Schiff, Gomori’s, and albumin stains. Glial fibrillary acidic protein and aquaporin 4 immunostaining revealed strong immunolabeling of astrocyte foot processes surrounding microvessels. The areas of the brain most frequently affected were the cerebral cortex, corpus striatum (basal ganglia), and cerebellar peduncles, and both arterioles and venules were involved. Most of the goats of this study showed lesions in the intestine (enteritis, colitis, and typhlitis), although pulmonary congestion and edema, hydrothorax, hydropericardium, and ascites were also described. Although the intramural edema described, for the first time, in these caprine cases is useful for the diagnosis of enterotoxemia when observed, its absence cannot exclude the disease.
Enterotoxemia caused by Clostridium perfringens type D is an important disease of sheep and goats of worldwide distribution. 19,31 C. perfringens type D can be a normal inhabitant of the intestine of several animal species, including goats. 21,32 Occasionally, when the intestine is altered by sudden changes in diet or other factors that are not yet well understood, C. perfringens type D proliferates in large numbers and produces toxins that are then absorbed to the systemic circulation and/or act locally in the intestine to produce disease. 15,19
C. perfringens has been conventionally classified into 5 types (A, B, C, D and E), according to the production of 4 so-called major toxins: α, β, ∊ (ETX), and ι. However, a new typing schedule has recently been proposed, which includes 2 additional types (ie, F and G) based on the production of enterotoxin and necrotic enteritis β-like toxin, respectively. 17 Several other toxins, such as perfringolysin and β2, may be produced by different strains of C. perfringens, although they are not used in the classification of this microorganism. 20
Strains of C. perfringens type D encode, at a minimum, α and ∊ toxin. 15,19 ETX is the main virulence factor of this toxinotype, although it is possible that 1 or more additional toxins act synergistically with ETX to produce disease. 15 Most clinical signs and pathological changes of naturally occurring enterotoxemia have been reproduced by intravenous administration of ETX in sheep and goats. 4,9 –11 Reverse genetic experiments have also demonstrated that ETX is required for enterotoxemia type D in goats. 7
Field cases of enterotoxemia in goats are considered to occur in 3 clinical forms: peracute, acute, and chronic. 2,18,25 An excess of protein-rich pericardial, pleural, and/or abdominal fluid; interstitial and/or alveolar pulmonary edema; and subepicardial and subendocardial hemorrhages are frequently observed in the peracute form of the disease, although cases in which no gross or microscopic lesions are found may also occur. In the chronic form, fibrinohemorrhagic enterocolitis is the most consistent lesion. 1,25 A combination of lesions found in the peracute and chronic forms of the disease is frequently seen in the acute cases. 25 Gross findings suggestive of enterotoxemia can be found during postmortem examination, although none are pathognomonic. 21
Descriptions of histological changes in enterotoxemia of goats are scant and, with few exceptions, are based on experimental disease. 6,9,21,23,26,28,30 Cerebral microangiopathy, characterized by perivascular and intramural leakage of proteinaceus, eosinophilic fluid in certain areas of the brain, is considered pathognomonic for ovine enterotoxemia and is seen in most ovine cases of the disease. 3,12 In goats with type D enterotoxemia, only perivascular but not intramural edema has been described. This change is, however, considered unusual in goats, with only 1 published report describing it in 2 confirmed spontaneous cases of caprine enterotoxemia. 23
This article describes the histological lesions in the brain and its microvasculature of 44 cases of naturally occurring enterotoxemia caused by C. perfringens type D, and it is proposed that the distinctive intramural vascular edema found in a proportion of these cases is useful for diagnosis of this neurologic disorder, particularly when corroborated by detection of ETX in intestinal contents.
Materials and Methods
Case Selection
Forty-four cases were selected from the archives of the California Animal Health and Food Safety Laboratory System (San Bernardino and Davis branches). The inclusion criteria were those previously described for the diagnosis of type D caprine enterotoxemia. They were (1) characteristic history and clinical signs, including 1 or more of sudden death, colic, and/or diarrhea; (2) suggestive pathological findings, including 1 or more of colitis, hydropericardium, hydrothorax, ascites, and/or pulmonary edema; and (3) detection of ETX toxin in the intestinal contents. 22,32 The cases occurred between January 2000 and March 2017. These goats were between 2 weeks and 6 years of age at the time of death; 24 were female and 20 were male, and several breeds were represented. In addition, 10 adult goats of various breeds (5 males and 5 females) with diagnoses other than enterotoxemia and that had negative results for ∊ toxin enzyme-linked immunoassay (ELISA) in the intestine were used as controls. The causes of death of these control goats were pneumonia (n = 6), ketosis (n = 1), mycoplasmosis (n = 1), coccidiosis (n = 1), and Clostridium difficile enteritis (n = 1). Details of signalment and clinical history are shown in Supplemental Table S1.
Gross and Microscopic Pathology
The necropsy records of all cases were reviewed, with special emphasis on signalment, clinical history, and gross findings in the central nervous system. At the time of necropsy, samples from multiple organs, including heart, lung, liver, pancreas, rumen, reticulum, omasum, abomasum, small and large intestine, kidney, spleen, lymph nodes, and brain, from all animals were collected and fixed by immersion in 10% neutral buffered formalin for 24 to 48 hours. The brain was then cut into ∼0.5-cm-thick coronal sections, and blocks were collected from all those areas where pathological findings in sheep and goats with enterotoxemia had been previously reported, 6,9,10,21,26 –28 including cerebral cortex, corpus striatum (basal ganglia and internal capsule), thalamus, hippocampus, midbrain at the level of anterior colliculi, cerebellum, and medulla oblongata. All the tissues were paraffin embedded, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). For this study, selected brain sections were also stained with periodic acid–Schiff (PAS) to evaluate the integrity of the basement membranes of blood vessels affected by edema and to further characterize the vasculocentric edema. In addition, to assess the integrity of the tunica adventitia (containing collagen type III) of the blood vessels, Gomori’s reticulin stain was performed. All brain sections were examined by 2 pathologists blinded to the identity of individual cases (J.O. and J.M.V.), with special emphasis on vascular lesions and the topographical distribution of these morphological changes.
Immunohistochemistry
For this study, selected sections of the sampled brains were examined immunohistochemically for the presence of injured axons (amyloid precursor protein [APP], the most sensitive, early marker of axonal injury), astrocytic reaction (glial fibrillary acidic protein [GFAP]), the water channel protein, aquaporin 4 (AQP4), plasma albumin, and a microglial/macrophage marker (ionized calcium binding adaptor molecule 1 [Iba1]), as previously described. 7 Briefly, the following primary antibodies were used: an anti-APP mouse monoclonal antibody (clone 22C11, Chemicon; Millipore, Billerica, MA), an anti-GFAP rabbit polyclonal antibody (Pab) (Dako, Carpinteria, CA), a rabbit Pab against AQP4 (Millipore, Burlingame, CA), a goat Pab against rat albumin (Cappel, West Chester, PA), and a rabbit Pab against Iba1 (Wako Pure Chemical Industries, Osaka, Japan). Rabbit, mouse, or goat IgGs were used as primary antibodies in negative control sections. The Dako EnVision+ horseradish peroxidase (HRP) kit (Dako) was used for APP, according to the instructions of the manufacturer and standard operating procedures of the California Animal Health and Food Safety Laboratory, UC Davis. For GFAP, an avidin-biotin-peroxidase method (Vectastain Elite Kit; Vector Laboratories, Burlingame, CA) was used according to the manufacturer’s instructions and standard operating procedures of the Veterinary Medical Teaching Hospital, UC Davis. Serum albumin, AQP4, and Iba1 were incubated with a streptavidin-conjugated peroxidase tertiary (Pierce, Pasadena, CA), according to the instructions of the manufacturer. For APP and GFAP, immunoreactivity was visualized using the chromogen 3-amino-9-ethylcarbazole (AEC K4001; Dako). Albumin, AQP4, and Iba1 were visualized with 3,3′-diaminobenzidine tetrahydrochloride (DAB). After chromogen incubation, all sections were counterstained with hematoxylin, cleared, and mounted.
C. perfringens and C. perfringens Toxin ELISAs
Samples of small and/or large intestinal content from all the goats were analyzed for Clostridium perfringens alpha toxin (CPA), Clostridium perfringens beta toxin (CPB), and ETX by a monoclonal antibody-based capture ELISA (BioX, Brussels, Belgium), according to the manufacturer’s instructions. In addition to the 3 toxins mentioned, this ELISA also detects the presence of C. perfringens. The results were read in an ELISA reader (Pharmacia, Uppsala, Sweden) with a 450-nm filter. Positive or negative readings were obtained according to the instructions of the manufacturer. CPA, CPB, and ETX were used as positive controls. This technique was previously found to detect as little as 25 MLD50/ml of ETX in intestinal content of sheep and goats. 29
Results
CPA and ETX were detected by ELISA in the small and/or large intestine of the 44 goats with type D enterotoxemia. The gross and histological lesions in these goats are summarized in Supplemental Table S1. Although this study focused on brain pathology of caprine enterotoxemia, a brief description of the most significant gross lesions observed in other organs is included. Grossly, the most common lesion was hemorrhagic and/or necrotizing inflammation of different regions of the intestinal tract (enteritis, colitis, and/or typhlitis), affecting 24 of 44 (55%) of the goats. The presence of fluid in cavities (ie, ascites, hydrothorax, and/or hydropericardium) was the second most frequent finding (20/44, 46%), followed by pulmonary congestion and edema (17/44, 39%). Only 1 goat in the study presented gross changes in the brain, which consisted of mild, diffuse swelling of the cerebrum, with the caudal portion of the cerebellum extending into the foramen magnum (cerebellar coning) (Fig. 1).
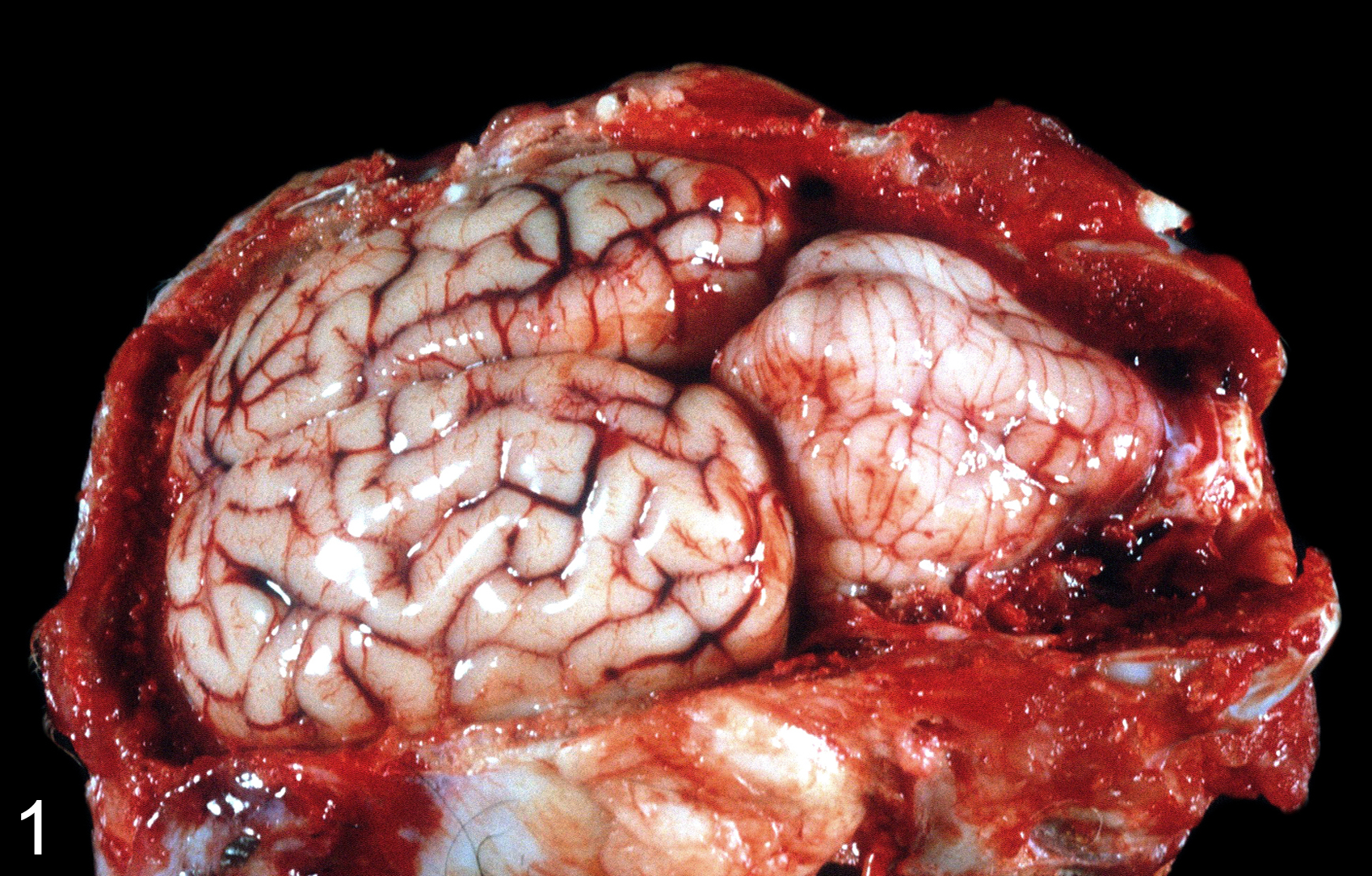
Enterotoxemia, brain, goat No. 1. The caudal portion of the cerebellum extends into the foramen magnum (cerebellar coning).
Microscopically, 8 (18%) of the 44 goats with type D enterotoxemia showed lesions in the brain. They were observed in 5 males and 3 females, and most (n = 7) were 1 year of age or younger (Table 1). The cerebral cortex (n = 6), basal ganglia of the corpus striatum (n = 6), and cerebellar peduncles (n = 5) were the most frequently affected areas, followed by the thalamic nuclei (n = 1) (Table 1). Microscopic lesions in the brain were almost exclusively vasculocentric, with some involvement of the surrounding neuropil. Vascular lesions were found in both arterioles and venules, the former predominantly in the cerebral cortex and basal ganglia and the latter in the cerebellar peduncles. In those goats in which vascular lesions were observed, only a very small number of vessels were affected, always less than 5% of the total number per section.
Distribution of Mural Edema in the Brain of 8 Goats With Clostridium perfringens Type D Enterotoxemia.

Abbreviations: A, arterioles; GM, gray matter; V, venules; WM, white matter.
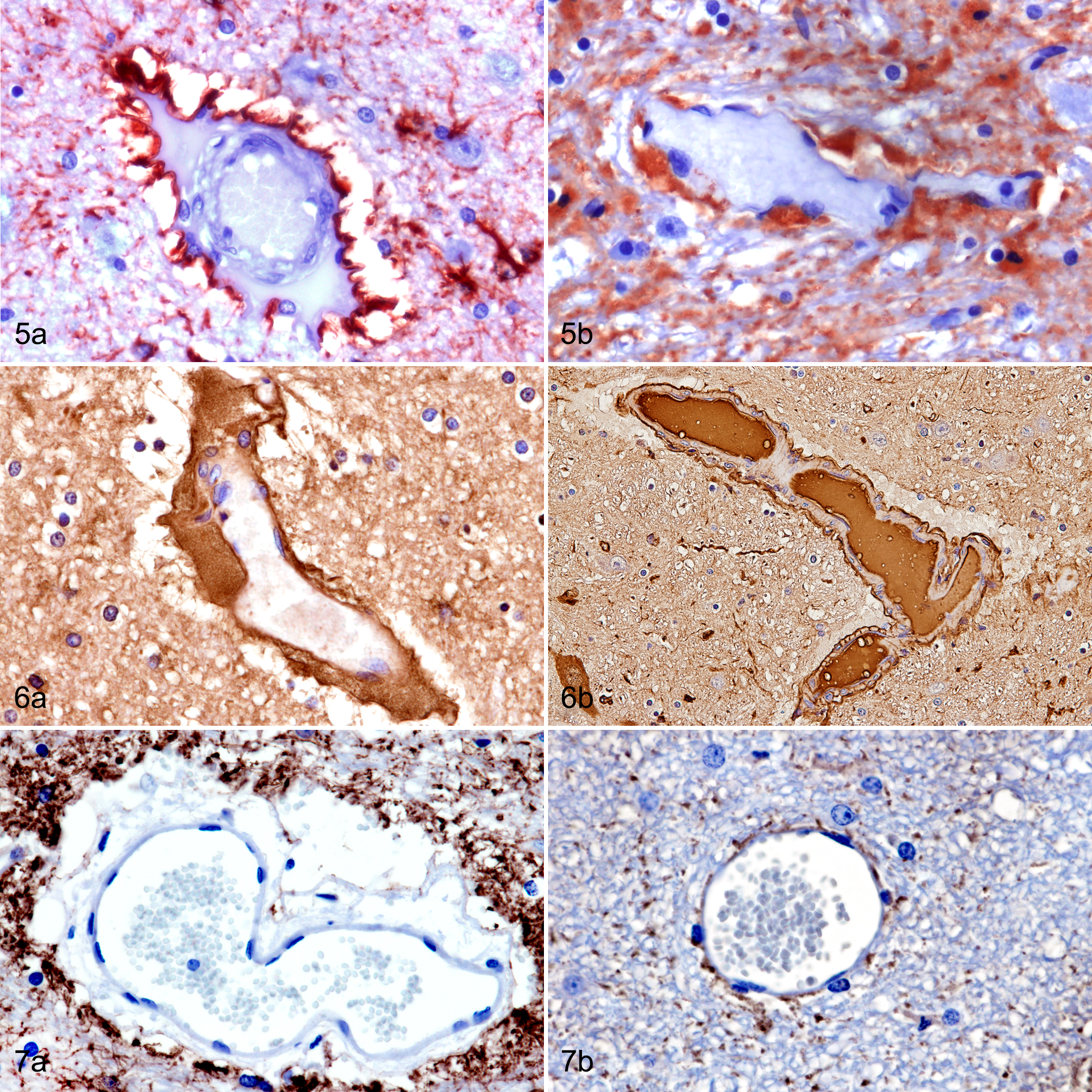
In HE-stained brain sections of these 8 goats, the vascular wall was mildly to moderately distended with lakes of extravasated eosinophilic, homogeneous, proteinaceous fluid (Fig. 2a). Special stains (PAS, Gomori reticulin) demonstrated that the high-protein edema was located between the muscularis and adventitia of the vessel wall, indicating mural involvement instead of perivascular edema (Figs. 3a, 4a). The vascular wall thickening was variable depending on the vessel size, but it ranged between 10 and 300 μm, and most of the time, it caused an elongated or oval distension of the vessel wall rather than a circumferential expansion. The extravasated fluid was PAS positive (Fig. 3a) and sometimes extended into perivascular spaces and the surrounding neuropil. GFAP immunostaining revealed, in all cases, strong immunolabeling of astrocyte foot processes around the adventitia (Fig. 5a). In blood vessels showing large mural lakes of eosinophilic proteinaceous material, this leaked fluid was strongly immunopositive to albumin (Fig. 6a). AQP4-immunoreactive granules were coarse and numerous in foot processes surrounding microvessels, especially where there was evidence of plasma protein extravasation and intramural edema, with similar robust immunolabeling of astrocytic processes in the surrounding neuropil (Fig. 7a). Iba1 immunohistochemistry identified strongly stained macrophages/microglial cells in the edematous spaces of vessels, and numerous strongly APP-immunoreactive injured axons were found in the neuropil adjacent to these vessels. Endothelial cells showed pyknosis and/or karyorrhexis.
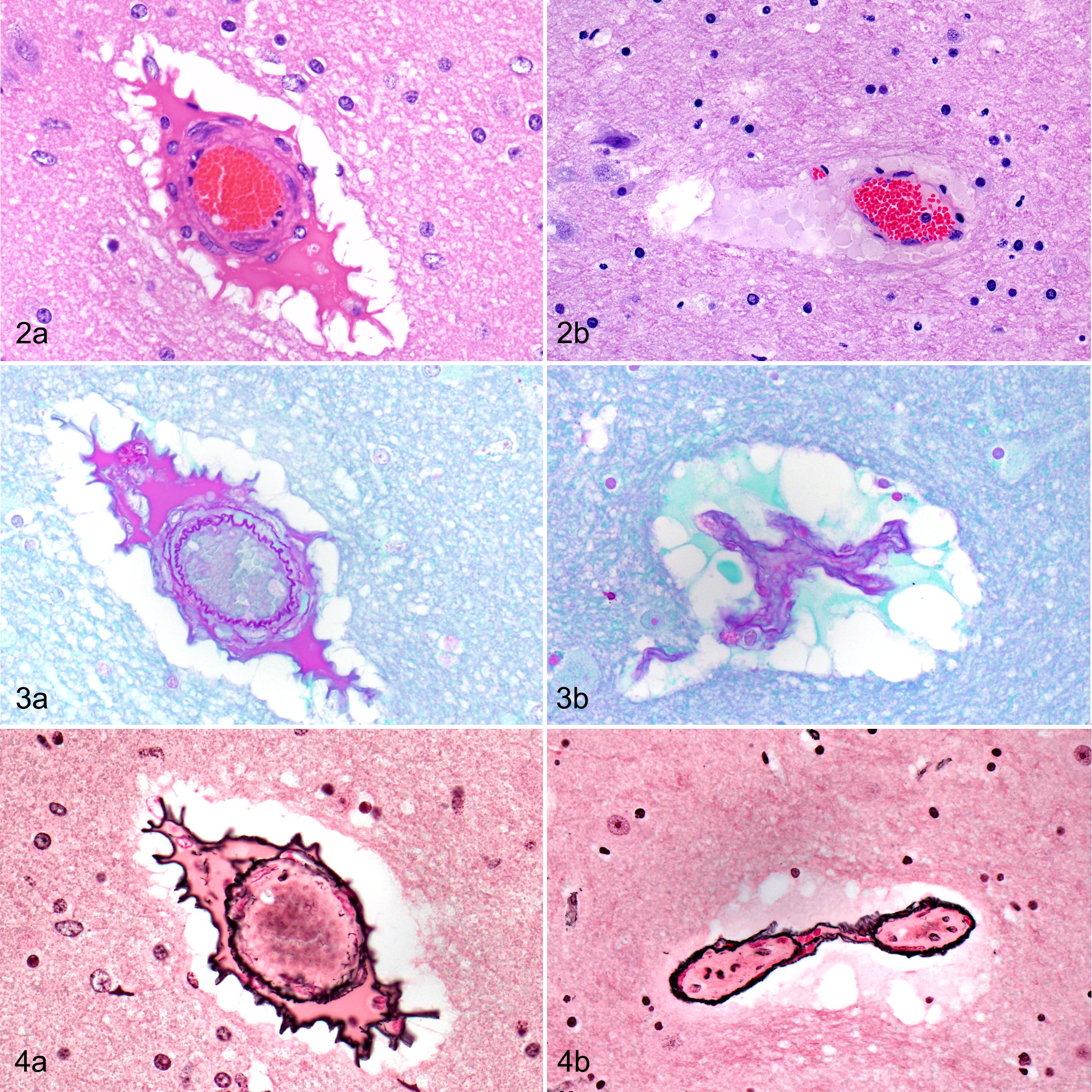
Type D enterotoxemia with intramural vascular edema (a) and controls (b), brain, goat No. 3.
Type D enterotoxemia (a) and controls (b), brain, goats.
In the control group of goats, most of the brain vessels did not show significant histological changes. However, in some areas (mostly in the brainstem), the presence of extravasated erythrocytes surrounding the vessel was noted. This finding has been previously described as a common postmortem artifact. 5 In addition, a small amount of faintly eosinophilic granular or globular material surrounding the vessels was occasionally observed (Fig. 2b). This perivascular material did not react positively to PAS or albumin immunostaining (Figs. 3b, 6b) or cause any disruption to the vessel wall in Gomori-stained sections (Fig. 4b). Accordingly, these findings were interpreted as artifacts or perimortem changes and not antemortem vascular lesions. In control brains, GFAP and AQP4 immunopositivity was expressed as fine granularity of astrocytic processes (Figs. 5b, 7b).
Discussion
The main finding in the brains of goats diagnosed with C. perfringens type D enterotoxemia in the present study was microvascular damage characterized by intramural edema. This vascular lesion was found in 8 of the 44 caprine brains examined (18%), was a distinctive and therefore diagnostically useful morphologic change in this neurologic disorder, and was not found in control brains from goats diagnosed with a range of other diseases. However, in those goats in which these vascular lesions were found, a small number of blood vessels (<5%) were affected, thus mandating a thorough examination of all susceptible brain regions to detect this microangiopathy in cases of suspected type D enterotoxemia. Although many of the clinical signs and postmortem lesions of this disease were previously reproduced in goats by intravenous administration of ETX 24 or by intraduodenal inoculation of whole cultures of C. perfringens type D, 13 brain lesions in goats with naturally occurring type D enterotoxemia have only been reported in 2 animals with confirmed disease. 23 In the brains of sheep exposed to ETX, by contrast, perivascular high-protein edema has been regarded as the most reliable neuropathologic change for diagnostic purposes. 22
With routine HE staining of these goat brains, the vascular lesions were mainly manifested by eosinophilic, homogeneous, proteinaceous fluid that dissected and expanded the vessel wall (intramural edema). Much less commonly, there was perivascular deposition of extravasated protein-rich fluid, which sometimes extended further into the neuropil. It was also evident that the intramural location of the edema was better appreciated by use of PAS and Gomori’s reticulin stains, and it is therefore recommended that these stains be used in suspected cases of type D enterotoxemia in goats. Intramural vascular edema has also recently been reported in the brains of sheep experimentally inoculated with C. perfringens type D using these special stains. 10
The microangiopathy in these goat brains was also investigated using immunohistochemical techniques. GFAP immunostaining demonstrated that the astrocytic foot processes were close to the adventitia, which was separated from the muscularis due to intramural edema. Moreover, there was a marked increase in the expression of AQP4, the major water channel protein in the brain. 33,35 AQP4 regulates the movement of water in the brain. In vasogenic edema, it appears that excess water enters the extracellular space independently of AQP4, but this water is cleared from the brain via AQP4 channels in astrocytic foot processes. By contrast, AQP4 water channels in astrocytic foot processes are active in the formation of cytotoxic edema. 14 An antibody directed against endogenous albumin was used as a surrogate immunohistochemical marker of increased vascular permeability in the brains of these goats. The intramural and, less commonly, the perivascular albumin leakage was strongly immunopositive for albumin and corresponded to the distribution of PAS-positive proteinaceous material. The robustly albumin-immunostained, high-protein, blood vessel–associated edema could easily be differentiated from the much weaker, artifactual immunoreactivity of surrounding neural elements in these immersion-fixed brains, with this artifactual pattern of mild immunoreactivity only being prevented by perfusion fixation of the brain in our experience.
Predilection sites for the development of microvascular damage, particularly intramural edema, in the 8 brains in which this lesion was found were the cerebral cortex (6/8), basal ganglia (6/8), cerebellar peduncles (5/8), and, less frequently, the thalamus (1/8). The response of the brain, especially its microvasculature, to ETX, as well as the attendant neuroanatomical distribution of lesions, is complex. ETX may cause selective or more widely distributed neurotoxicity, depending upon the dose of toxin and the time interval between toxin exposure and death. In general, higher doses of ETX tend to widen the distribution of brain lesions, although still favoring certain vulnerable areas, while lesions in subacutely intoxicated (or partially immune) animals tend to be smaller, more circumscribed, and confined to fewer selectively vulnerable regions. 7 Accordingly, the lesion distribution in brains exposed to ETX can vary from case to case within a given species, as well as between species.
ETX is a member of the aerolysin family of pore-forming toxins. 16 In mice and rats (and presumably also in sheep and goats), ETX binds to specific receptors on the microvascular endothelium in the brain, causing vascular endothelial degeneration. One domain of ETX binds to endothelial receptors, where it uses lipid rafts and caveolin to oligomerize into a prepore on the surface of the cell membrane. Another ETX domain then inserts into the membrane lipid bilayer and forms an active pore. ETX can be transiently prevented from exerting its deleterious effect by prior administration of the prototoxin, presumably by competitive inhibition of endothelial receptors. The ETX-induced endothelial cytotoxicity appears to be due, in part at least, to formation of pores that cause a rapid decline in cytoplasmic potassium levels, with a concomitant influx of sodium and chloride ions. This process, which seems to involve ATP depletion, leads to cell necrosis. 20 While the microvasculature appears to be the prime target of ETX, this potent neurotoxin could also have a direct cytotoxic effect on neural elements after it has gained access to the brain parenchyma. 8 Moreover, it is now recognized that the cerebral endothelium is heterogeneous, this being evident not only between different neuroanatomical regions as a reflection of their different functional diversity but also among capillaries and venules and even at the single cell level within individual microvessels. 34
In contrast to sheep, in which a neurologic syndrome is found in cases of type D enterotoxemia, the main clinical presentation in goats is enteric. It has been postulated that enterocolitis is more prominent in goats because of slower absorption of ETX produced in the intestine into the systemic circulation. 22 The lower circulating levels of ETX in goats could restrict the resulting cerebral microvascular injury and ensuing vasogenic edema. It could be speculated that microvascular damage in sheep brains exposed to higher levels of ETX would be more severe and that they would develop, in addition to the predominantly intramural edema we found in goats, the more commonly observed perivascular pools of extravasated proteinaceous fluid.
In conclusion, the results of this study showed that intramural microvascular edema is a distinctive and diagnostically useful neuropathologic finding in goats with C. perfringens type D enterotoxemia. However, microvascular damage in C. perfringens type D enterotoxemia seems to be much less common in goats than in sheep and, accordingly, a thorough histologic examination of the brain needs to be conducted in suspected cases of this neurological disorder. Moreover, the absence of intramural edema in cerebral blood vessels of ETX-exposed goats cannot exclude the disease.
Supplemental Material
Supplemental Material, DS1_VET_10.11770300985818817071 - Intramural Vascular Edema in the Brain of Goats With Clostridium perfringens Type D Enterotoxemia
Supplemental Material, DS1_VET_10.11770300985818817071 for Intramural Vascular Edema in the Brain of Goats With Clostridium perfringens Type D Enterotoxemia by Joaquín Ortega, José Manuel Verdes, Eleonora L. Morrell, John W. Finnie, Jim Manavis and Francisco A. Uzal in Veterinary Pathology
Footnotes
Acknowledgements
We thank Mark Anderson for sharing his gross pictures and all the pathologists from California Animal Health & Food Safety (CAHFS) Laboratory who performed the necropsies of the goats. Also, we thank S. J. Uzal for reviewing this manuscript and Julianne Beingeser for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Joaquín Ortega was supported by “Ayudas a la movilidad investigadora CEU–Banco Santander.” José Manuel Verdes was supported by CSIC-UdelaR, PEDECIBA, and ANII (Uruguay).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
