Abstract
Canine urothelial carcinoma (UC) has a poor prognosis and high metastatic rate. Human epidermal growth factor receptor 2 (HER2), a receptor tyrosine kinase involved in cell proliferation and differentiation regulation, has been attracting interest as a therapeutic target molecule for human breast cancer. This study investigated expression of the canine homolog of HER2 (ERBB2) in canine UC, and its association with clinical factors. Since it has been controversial whether commercial anti-human HER2 antibody (Dako A0485) correctly recognizes the canine homolog of HER2, an application of the antibody using a canine UC cell line was validated first. By Western blot, a single band at the appropriate size for canine HER2 (185 kDa) was recognized. Immunohistochemistry for HER2 was performed on 23 samples of UC, 8 samples of polypoid cystitis, and 8 samples of normal urinary bladder, and the results were scored as either 0, 1+, 2+, or 3+ with reference to the evaluation method for human UC. Intense membranous HER2 immunoreactivity was frequently observed in neoplastic cells, especially in grade 2 UC. Minor HER2 expression was found in the epithelial cells of polypoid cystitis and normal bladder. The incidence of HER2 positivity (scores of 2+ or 3+) was 14 of 23 (60.9%) in UC, 3 of 8 (37.5%) in polypoid cystitis, and 0 of 8 (0%) in normal bladder. There was no significant correlation between HER2 positivity and clinical factors. While increased HER2 expression was observed in a subset of urothelial carcinomas, further mechanistic studies are needed to determine its role in the pathogenesis and targeted therapy of this cancer.
Canine urothelial carcinoma (UC), also known as transitional cell carcinoma, is a malignant tumor derived from the transitional epithelium of the urinary tract. 21 It is the most common tumor in the urinary bladder of dogs, accounting for approximately 74% of all primary bladder tumors. 20 The tumor has a poor prognosis and frequent metastasis to the regional lymph nodes, lungs, vertebral bones, and other organs. Unfortunately, most dogs with UC ultimately die or are euthanized. 13,20 Combined with the propensity for metastasis and its resistance to treatment, it is considered as one of the most malignant tumors in dogs. 20
Human epidermal growth factor receptor 2 (HER2) is a transmembrane tyrosine kinase receptor that is involved in cellular growth, survival, and migration. 10,23,38 The increased activity of this molecule is identified in a subset of human breast cancers and is strongly associated with increased disease recurrence and poor prognosis. 33 Its overexpression facilitates excessive dimer formation, which in turn drives oncogenic cell survival, proliferation, apoptosis suppression, and angiogenesis. 2,37 It also has a significant role as a therapeutic target. Administration of trastuzumab, a HER2-targeted monoclonal antibody, given in combination with or after adjuvant chemotherapy, has been found to improve both disease-free survival (DFS) and overall survival (OS) in human patients with early HER2-positive breast cancer with moderate-to-high risk of recurrence. 26,27 Pertuzumab, another humanized monoclonal antibody against HER2, and lapatinib, a small-molecule intracellular tyrosine kinase inhibitor, have also proven to be effective in the treatment of the disease. 5,32 Thus, the expression profile of HER2, combined with those of estrogen receptor, progesterone receptor, and Ki-67, has been routinely assessed by immunohistochemistry in order to classify breast tumors into distinct subtypes. 12,36
HER2 overexpression, gene amplification, and mutation have been identified in other human cancers, including non–small-cell lung cancer, gastric adenocarcinoma, ovarian adenocarcinoma, endometrial adenocarcinoma, pancreatic ductal adenocarcinoma, cervical squamous cell carcinoma, salivary duct carcinoma, and UC. 24 The frequency of HER2 overexpression in human UC varies from 9% to 76%. 1,4,7,8,11,14 –17,24 A previous study 14 showed a correlation between worse disease-related survival and immunohistochemical HER2 overexpression in human muscle-invasive UC. The Cancer Genome Atlas project has reported the mutation or amplification of the HER2 gene in a subset of human UCs. 34 These observations suggest that HER2-targeted therapies might also be effective for the treatment of other cancers in addition to breast cancer.
Our knowledge of HER2 expression in canine cancers, in contrast to human cancers, extends only to mammary gland tumors, 3,6 osteosarcomas, 19 and, most recently, UCs. 22 Furthermore, it has been controversial whether commercially available anti-human HER2 antibody correctly recognizes the canine homolog of HER2. Burrai et al 3 evaluated the specificity of the most commonly used polyclonal anti-HER2 antibody (Dako A0485) by using normal and neoplastic canine mammary tissues. However, the immunohistochemistry results suggested a lack of specificity with a diffuse cytoplasmic staining pattern, and Western blot and subsequent mass spectrometry could not show specific evidence of its validity.
From our recent work of RNA-sequencing–based transcriptome analysis against canine UC, ERBB2, encoding HER2, was the highest differentially expressed upstream regulator related to carcinogenesis, indicating that HER2 could be involved in the pathogenesis and progression of the disease. 18 Thus, in this study, we first validated an application of HER2 antibody using a canine UC cell line. Then, we investigated HER2 expression in canine UC by immunohistochemistry and evaluated the associations of tumor malignancies and clinical parameters.
Materials and Methods
Western Blot
To confirm the cross-reactivity of commercially available anti-human HER2 antibodies to the canine homolog of HER2, Western blot was performed using cell lysate of a canine transitional cell carcinoma cell line (TCCUB). 29,39 In this study, we used 2 primary antibodies, rabbit polyclonal anti-human c-erbB-2 oncoprotein (HER2/neu) antibody (A0485, Dako, Glostrup, Denmark, 1:1000 dilution) and mouse monoclonal anti-human HER2 antibody (clone 19D2, nanoTools, Teningen, Germany, 1:200 dilution). IRDye 800CW donkey anti-rabbit (1:5000 dilution) and IRDye 680RD donkey anti-mouse (1:10 000 dilution) secondary antibodies were purchased from M&S TechnoSystems Inc (Osaka, Japan). Equal sample concentrations (12 μg) were loaded in 7.5% sodium dodecyl sulfate polyacrylamide gel electrophoresis gels for electrophoresis. After separation, proteins were transferred to a nitrocellulose membrane and blocked in Tris-buffered saline (TBS) containing 5% skim milk at room temperature for 1 hour. Primary antibodies were incubated at 4°C overnight. Then, the membrane was washed using TBS with 0.2% Tween 20 and incubated with the secondary antibodies at room temperature for 1 hour. Proteins were detected using the ODYSSEY CLx (M&S TechnoSystems Inc). The cell lysate of a human ovarian cancer cell line (SKOV3) was used as a positive control according to the manufacturer’s protocol.
Study Population
From an archive of formalin-fixed, paraffin-embedded canine urinary bladder tissues, 23 UC samples were recruited (Supplemental Table S1). The samples consisted of 5 males (3 intact and 2 neutered) and 18 females (9 intact and 9 neutered). The median age of the samples was 124 months (range, 85–136 months). The different breeds included Papillon (n = 5), Miniature Dachshund (n = 4), Pembroke Welsh Corgi (n = 3), Beagle (n = 1), Chihuahua (n = 1), French Bulldog (n = 1), Maltese (n = 1), Miniature Pinscher (n = 1), mongrel (n = 1), Shiba Inu (n = 1), West Highland White Terrier (n = 1), Wire Fox Terrier (n = 1), Scottish Terrier (n = 1), and Yorkshire Terrier (n = 1). All UC samples underwent total cystectomy and subsequent urinary diversion to the prepuce or vagina. 28 Eight polypoid cystitis samples were used as disease controls (Supplemental Table S1). This group included 5 males (4 intact and 1 neutered) and 3 females (2 intact and 1 neutered). The median age of this group was 140 months (range, 83–175 months). The breeds were as follows: mongrel (n = 2), Beagle (n = 1), Cavalier King Charles Spaniel (n = 1), Miniature Pinscher (n = 1), Shetland Sheepdog (n = 1), Shih Tzu (n = 1), and Toy Poodle (n = 1). All polypoid cystitis samples underwent partial cystectomy. Medical treatments prior to and after surgery of UC/polypoid cystitis cases are listed in Supplemental Table S1. The tissue samples of UC and polypoid cystitis were obtained from the Veterinary Medical Center of the University of Tokyo and were submitted to the Laboratory of Veterinary Pathology at the University of Tokyo between November 2011 and December 2016.
Eight Beagle dogs were used as healthy controls. All dogs were intact males. The median age was 120 months (range, 120–132 months). Normal urinary bladder tissues were obtained from these dogs via necropsy. All procedures in this study were approved by the Animal Care Committee of the Graduate School of Agricultural and Life Sciences, the University of Tokyo, and conducted in accordance with the guidelines.
Tissue Processing and HER2 Immunohistochemistry
Tissues were fixed with 10% neutral buffered formalin, processed routinely, embedded in paraffin wax, sectioned, and placed on glass slides (APS- or MAS-coat type, Matsunami Glass, Osaka, Japan). Hematoxylin and eosin–stained specimens were checked and verified by a veterinary pathologist (M.T.). All UC samples were evaluated in full thickness, and histological gradings of grade 1 (low grade), grade 2 (intermediate grade), and grade 3 (high grade) were assigned according to previous reports on canine proliferative urothelial lesions. 25,31
HER2 immunohistochemistry was performed using rabbit polyclonal anti-human c-erbB-2 oncoprotein (HER2/neu) antibody (A0485, Dako), according to the manufacturer’s protocol. For each sample, 4-µm-thick sections were deparaffinized, rehydrated, and treated with 3% hydrogen peroxide–methanol at room temperature for 5 minutes and then heated in a water bath at 98°C for 40 minutes in target retrieval solution (Dako, pH 9.0). After being cooled for an hour, the sections were washed in TBS and then incubated in 8% skimmed milk-TBS at 37°C for 40 minutes. The sections were incubated with the primary antibody (1:100 in dilution) at 37°C for 40 minutes and then with Envision horseradish peroxidase–labeled anti-rabbit IgG polymer (Dako) at 37°C for an hour. The reacted products were visualized using 3.3′-diaminobenzidine (Dojindo, Kumamoto, Japan) and 0.03% H2O2 in TBS. Counterstaining was conducted with Mayer’s hematoxylin. Positive controls were prepared with canine mammary cancer tissue (Supplemental Fig. S1), which is known to show HER2 overexpression according to a previous report. 6 Negative controls were performed by omitting the primary antibody or by adding isotype control antibody instead of primary antibody (Supplemental Fig. S2).
Clinical Outcomes of UC Samples
For survival analyses, the status (alive, dead, or lost) and the presence of recurrence at the end of the study (May 11, 2017) were obtained based on medical records or on fax interviews with the referring veterinarians. A diagnosis of recurrence was based on ultrasonography and urine cytology. DFS was defined as the time from surgery to the first occurrence of any of the following events: local recurrence of UC, distant metastasis, or death of an animal at the end of the study. OS was defined as the interval between surgery and the established cause of death of an animal at the end of the study.
Statistical Analyses
To compare the populations for categorical variables, Fisher exact test was performed. Survival curves were generated with the Kaplan-Meier method, and survival rates were compared using the log-rank test. These statistical analyses were performed using Prism software, version 5.0.1 (Graph Pad Software, San Diego, CA, USA). Statistical significance was defined as P < .05.
Results
Antibody Specificity
Two antibodies, Dako rabbit polyclonal and nanoTools mouse monoclonal anti-human HER2 antibodies (A0485 and clone 19D2, respectively), were investigated for their specificity against canine HER2. Western blot analysis showed a strong band at approximately 185 kDa in a human ovarian cancer cell line SKOV3 (positive control; Figs. 1 and 2). The nanoTools clone 19D2 antibody showed 2 bands, a strong band at approximately 185 kDa and a faint band at approximately 60 kDa, in the canine UC cell line TCCUB (Fig. 1). Dako A0485 antibody detected a clear single band at approximately 185 kDa in the canine UC cell line TCCUB (Fig. 2), suggesting the cross-reactivity of this A0485 antibody to canine HER2.
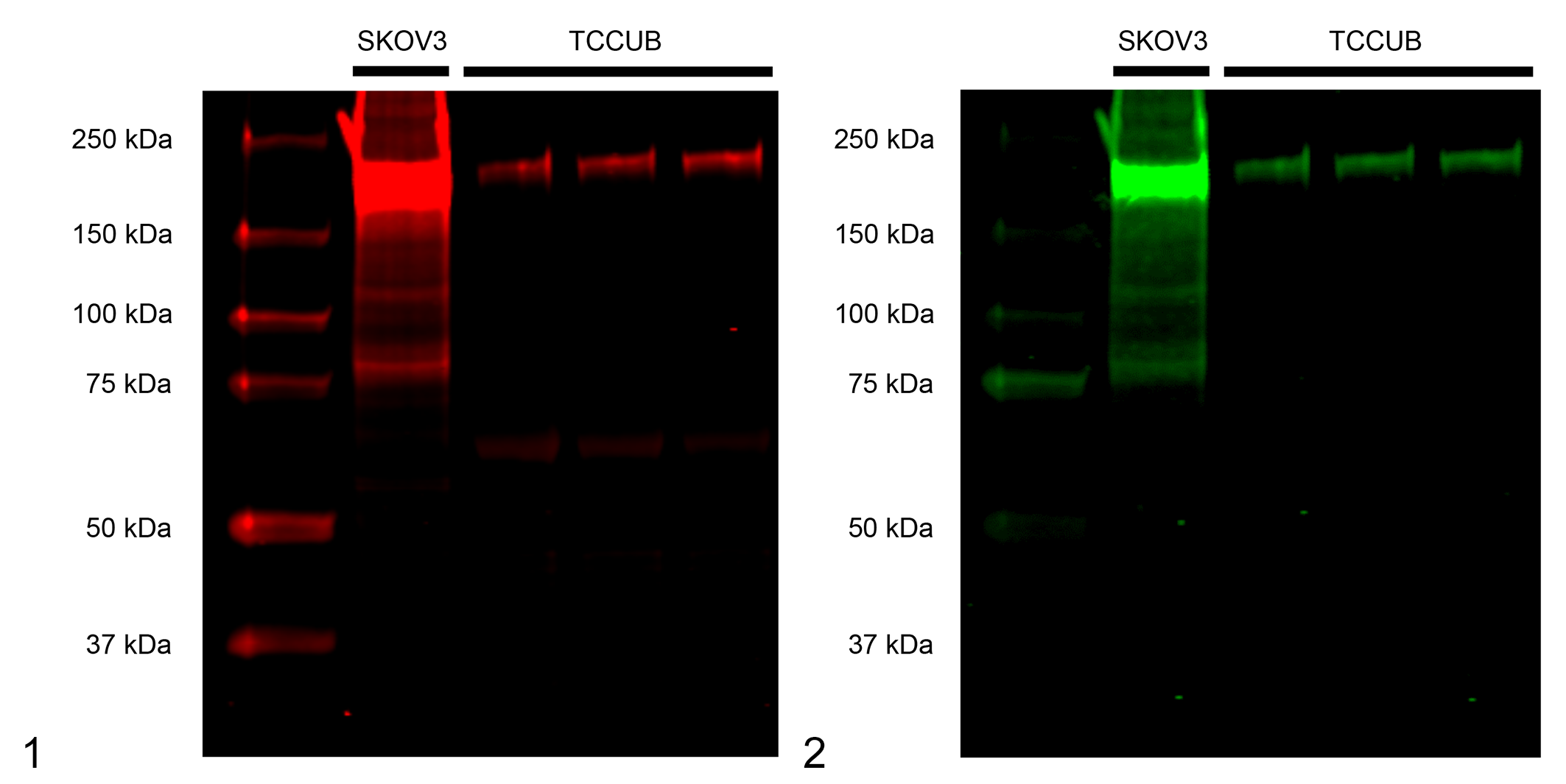
Cross-reactivity of anti-human human epidermal growth factor receptor 2 (HER2) antibodies with canine HER2 in a canine urothelial carcinoma cell line (TCCUB). A human ovarian cancer cell line (SKOV3) was used as the positive control.
HER2 Expression in Normal Bladder, Polypoid Cystitis, and UC
HER2 immunoreactivities were quantified based on previous reports on human UC. 8,16,17 A score of 0 denotes no reactivity at all (Fig. 3), a score of 1+ represents incomplete and weak immunoreactivity in <10% of tumor cells (Fig. 4), a score of 2+ represents incomplete but intense immunoreactivity in ≥10% of tumor cells (Fig. 5), and a score of 3+ represents intense with complete immunoreactivity (known as “chicken-wire pattern”) in ≥10% of tumor cells (Fig. 6). Samples with scores of 0 and 1+ were classified as HER2-negative, and those with scores of 2+ and 3+ as HER2-positive. Although cytoplasmic immunoreactivity was detected in some tumor cells and normal bladder epithelial cells, these were not taken into account.
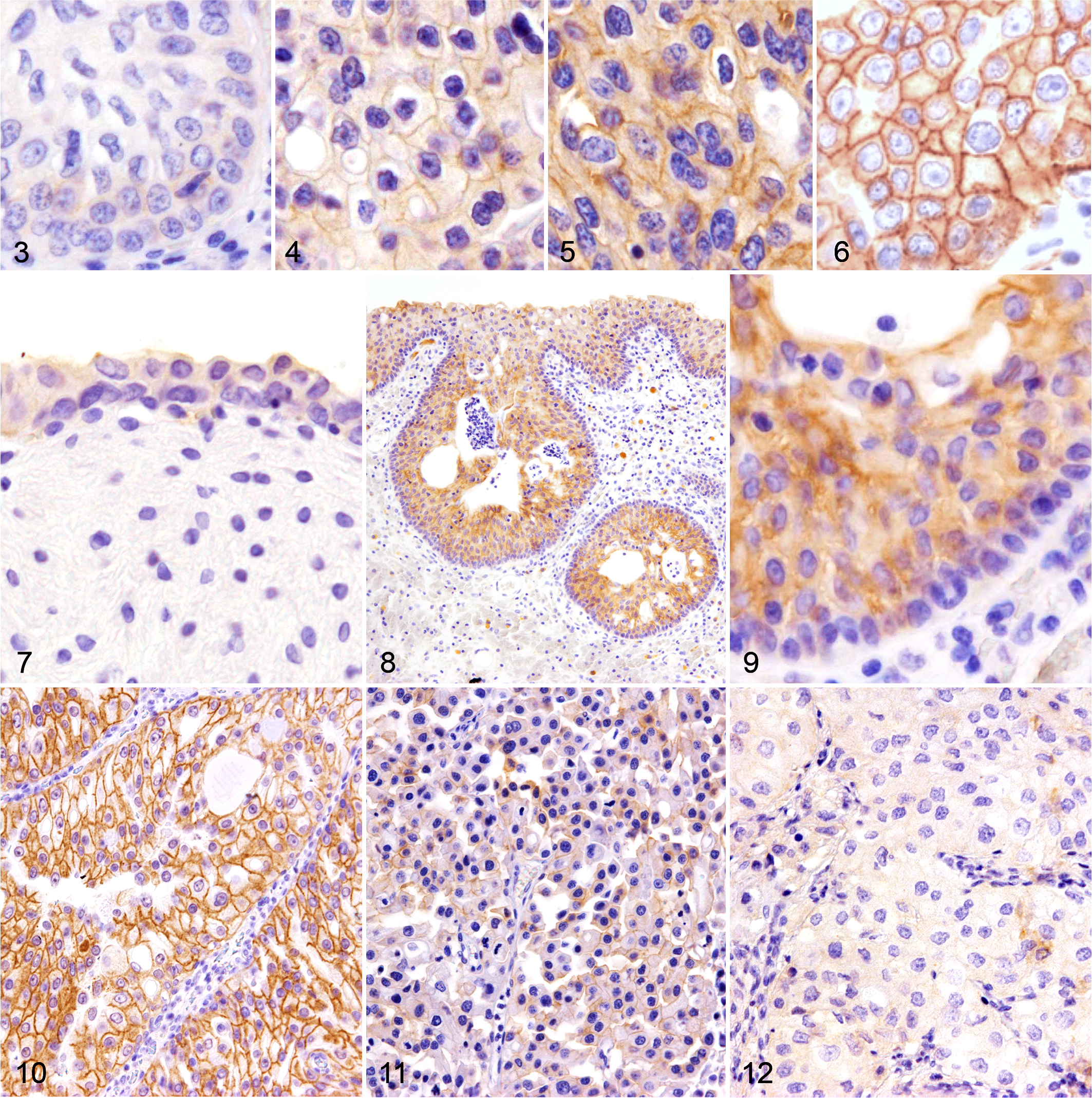
Urinary bladder, dog. Immunohistochemistry for human epidermal growth factor receptor 2 (HER2).
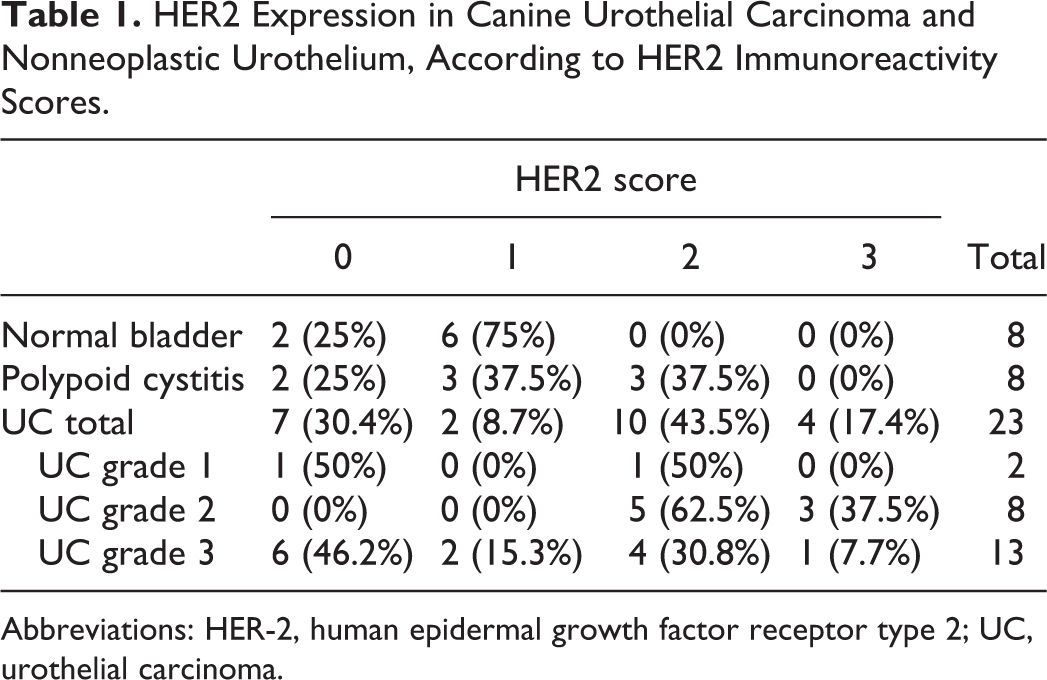
The immunoreactivity score of HER2 in each sample is listed in Table 1. All normal bladders (8/8) were HER2-negative (HER2 score: 0 or 1+), and among them, 6 of 8 samples were scored as 1+, showing faint membranous immunolabeling at the superficial layer of the urothelium (Fig. 7). In polypoid cystitis, 3 of 8 samples were HER2-positive (HER2 score: 2+). In these samples, membranous HER2 immunoreactivity was detected across the entire superficial epithelium layer of the polyps and in the subepithelial proliferative lesions of Brunn’s nest as well (Figs. 8 and 9). In UC, 14 of 23 (60.9%) samples were HER2-positive (score 2+ or 3+) at the primary site. Of the two samples of grade 1, one showed no immunoreactivity (HER2 score: 0), while the other had a score of 2+. HER2-positivity (scores 2+ and 3+) was highest in grade 2 UC (8/8). Furthermore, a score of 3+ was most frequently observed in grade 2 tumors (3/8; Fig. 10), compared with other grades. In grade 3 UC, 5 of 13 (38.5%) were HER2-positive, and HER2 immunoreactivities varied from score 0 to 3+. They also showed heterogeneity within individual tumors; some tumor cells showed an intense membranous expression, while other cells showed faint or no staining (Fig. 11). Six of 13 samples (46.2%) of grade 3 UC were completely negative for HER2, which was valued as score 0 (Fig. 12). Of 3 cases with regional lymph node metastasis, the HER2 score in metastatic lymph nodes was identical to the primary site in cases 12 and 16, both with scores of 2+ (Supplemental Figs. S3, S4, S5, and S6). The HER2 score of the metastatic lymph node in case 22 was not evaluated, as it was not excised during the surgery.
HER2 Expression in Canine Urothelial Carcinoma and Nonneoplastic Urothelium, According to HER2 Immunoreactivity Scores.
Abbreviations: HER-2, human epidermal growth factor receptor type 2; UC, urothelial carcinoma.
Association of HER2 Expression With Clinical Parameters
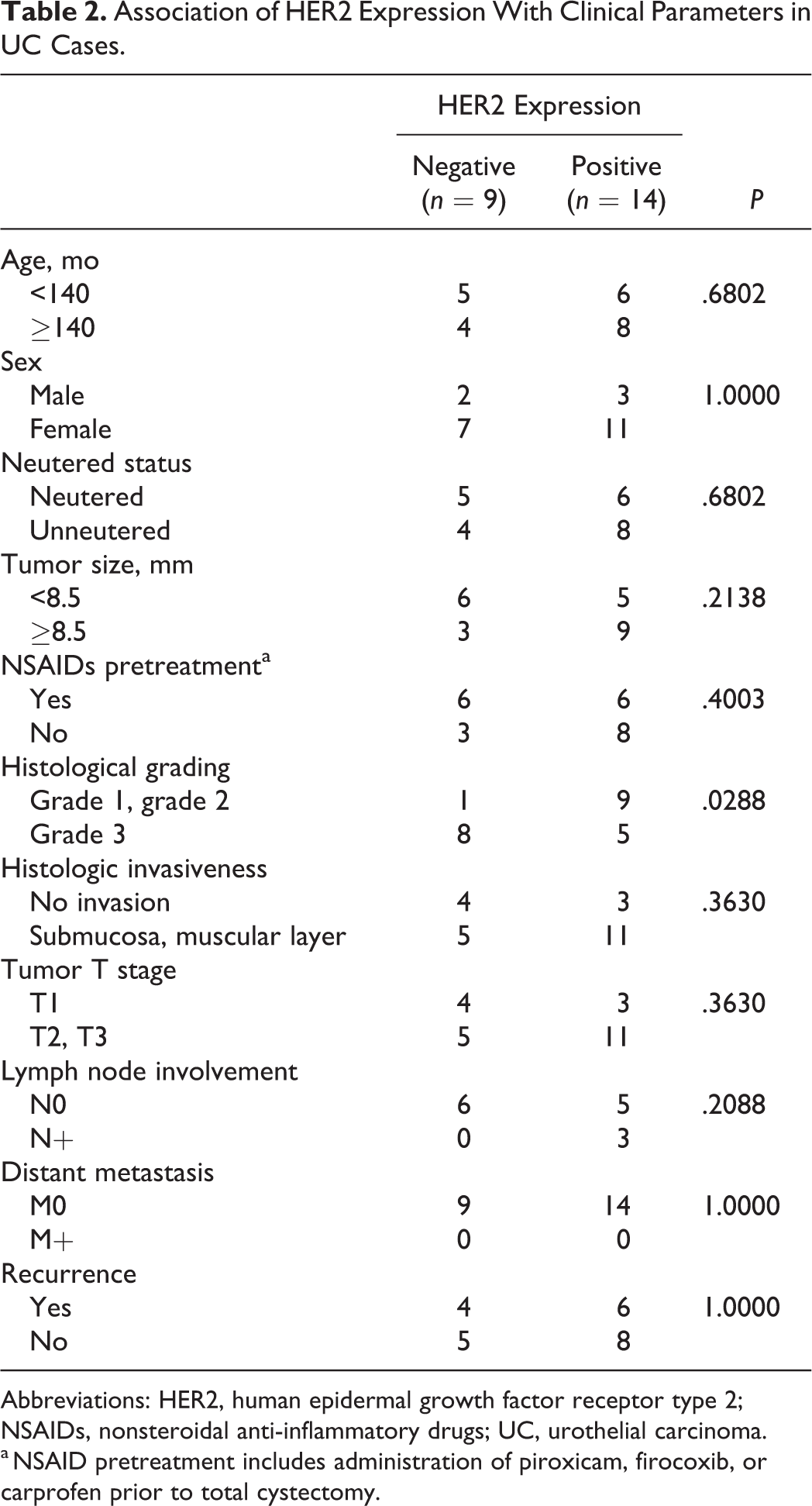
There was no significant difference between the level of HER2 expression and the age, sex, neutered status, tumor size, pretreatment status of nonsteroidal anti-inflammatory drugs, histological invasiveness, tumor T stage, lymph node involvement, distant metastasis, or recurrence in cases of UC (Table 2). However, the HER2 score tended to be higher in UC samples with a tumor diameter ≥8.5 mm. Lymph node involvement was observed only in the HER2-positive samples. HER2 positivity was significantly lower in grade 3 UC samples than in grade 1 and grade 2 UCs (P = .0288).
Association of HER2 Expression With Clinical Parameters in UC Cases.
Abbreviations: HER2, human epidermal growth factor receptor type 2; NSAIDs, nonsteroidal anti-inflammatory drugs; UC, urothelial carcinoma.
a NSAID pretreatment includes administration of piroxicam, firocoxib, or carprofen prior to total cystectomy.
Of the 23 dogs with UC, 10 (6 HER2-positive and 4 HER2-negative) relapsed during the study period. The median DFS of all UC samples was 274 days (range, 20–1271 days). The median DFS of the HER2-positive and HER2-negative samples was 192 days (range, 20–955 days) and 344 days (range, 49−1271 days), respectively. The DFS of the HER2-positive samples tended to be shorter than that of the HER2-negative samples, although there was no significant difference between them (Fig. 13; P = .2980).
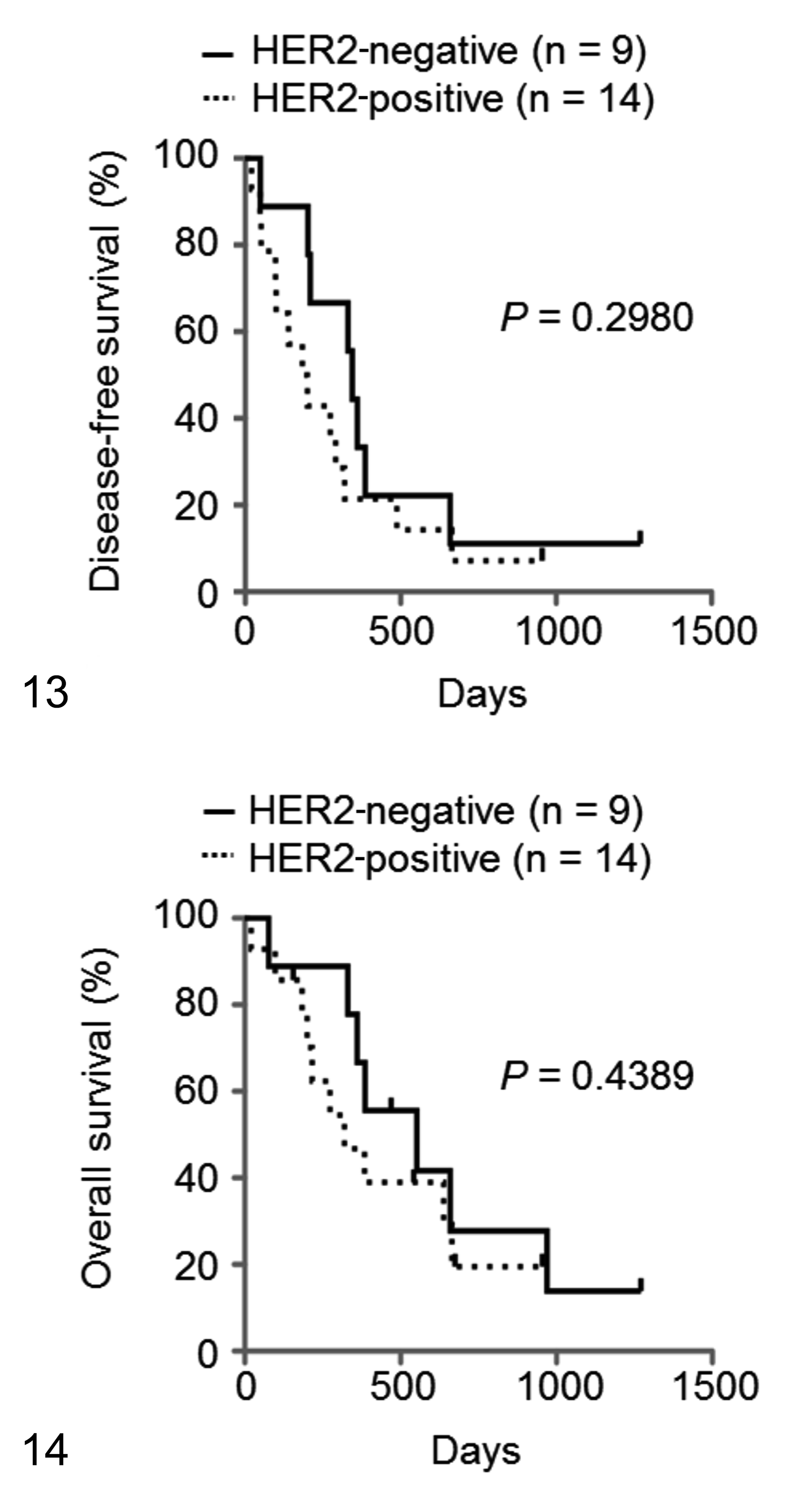
Kaplan-Meier survival curves of disease-free survival (DFS; Fig. 13) and overall survival (OS; Fig. 14) for dogs with urothelial carcinoma according to human epidermal growth factor receptor 2 (HER2) expression. There are no significant differences in DFS or OS between the HER2-positive (n = 14; dashed lines) and HER2-negative (n = 9; black lines) cases.
Of the 23 dogs with UC, 17 (10 HER2-positive and 7 HER2-negative) died during the study period. The median OS of all the UC samples was 384 days (range, 20−1271 days). The median OS of the HER2-positive and HER2-negative samples was 297 days (range, 20−955 days) and 470 days (range, 77−1271 days), respectively. The OS of the HER2-positive samples tended to be shorter than that of the HER2-negative samples, although there was no significant difference between them (Fig. 14; P = .4389).
Discussion
The clinical prognosis of canine UC is extremely poor, in part because of the lack of an effective treatment. Although surgical resection (cystectomy) is the first choice of treatment, it is rarely effective, and patients often experience recurrence and/or metastasis after surgery. 13 Conservative treatments, such as chemotherapy, cyclooxygenase-2 inhibitor treatments, or a combination of the two, are also ineffective. 13 The median survival time is reported to be about a year, regardless of the TNM stage. 13,28 Thus, the clinical field is in need of novel therapies for the treatment of this tumor.
One major reason we investigated HER2 expression in canine UC is due to the result of our previous RNA-sequence–based transcriptome analysis study. 18 As the HER2-related pathway was the highest expressed upstream regulator related to carcinogenesis, we suspected that the molecule could play a critical role in its pathogenesis. However, a previous report 3 pointed out that commercial anti-human HER2 antibody does not correctly recognize canine HER2 molecules in mammary gland carcinoma. Thus, we first evaluated its validity in canine UC cell lines. Western blot analysis revealed a single band at the appropriate molecular weight for canine HER2 (185 kDa), indicating that the two antibodies we used in this study (A0485 and 19D2) seem to be correctly reacting with canine HER2. Further investigation of immunohistochemistry using A0485 antibody showed specific membranous reactivity in a subset of cases. Thus, we concluded that the antibodies used in our study are sufficiently applicable for detecting canine HER2 molecules, at least in UC.
In this study, 14 of 23 (60.9%) of the canine UC samples were positive for HER2; 43.5% had a score of 2+ and 17.4% had a score of 3+. The result was congruent with a previous report that HER2 was overexpressed in 13 of 23 (56%) samples of canine urinary bladder transitional cell carcinomas. 22 Although the HER2 scoring methods were somewhat different between the present and previous reports, the HER2-positivity rate in canine UC seems to be higher than that of canine mammary tumors (17.6%–22%). 3,6 Moreover, HER2 scores in UC tended to be higher than those of healthy bladders and polypoid cystitis. Thus, these findings suggest a possible role for HER2 in the development of canine UC.
As 3 of 8 (37.5%) polypoid cystitis cases were judged as HER2-positive, HER2 overexpression does not seem to be specific for UC. A previous study, which assessed epithelial growth factor receptor (EGFR) expression in canine bladder, showed a similar result as the present study, with the highest EGFR staining scores in transitional cell carcinoma, followed by lower scores in polypoid cystitis and no significant immunohistochemical staining in normal bladder epithelium. 9 As urothelial epithelium usually proliferates in polypoid cystitis, HER2 and EGFR molecules might play an important role in both neoplastic and nonneoplastic proliferative diseases.
In the respective UC samples, HER2 immunoreactivity was highest in the grade 2 UCs. On the other hand, the HER2-positive rate of grade 3 UCs was significantly lower than that of grade 1 and 2 UCs. Moreover, HER2-negative foci were also frequently observed in some areas of high-grade UC samples with scores of 2+ and 3+, indicating severe heterogeneity in grade 3 tumors. This intriguing loss of HER2 expression may be caused by the dedifferentiation of neoplastic cells, as was reported in a previous study that reported loss of uroplakin III expression in 42% of grade 3 UCs. 31 This finding may be relevant to therapeutic targeting of HER2, as it suggests the possibility that HER2-negative neoplastic cells might confer resistance to anti-HER2 therapy.
In human UC, patients with the HER2-positive disease typically have a poor prognosis with characteristics including aggressive tumor progress and shorter patient survival times. 14 To evaluate the association between HER2-positivity and its prognosis in canine UC, we assessed the clinical parameters of the present cases. However, we did not find any significant differences in terms of age, sex, neutered status, tumor size, tumor T stage, lymph node involvement, distant metastasis, or recurrence. Moreover, although both DFS and OS appeared to be shorter in HER2-positive samples compared with HER2-negative ones, there were no significant differences. There are 2 possibilities that could account for the discrepancies between the human and canine results: either the sample size was too small for statistical analysis or there is a unique HER2 expression transition depending on the tumor grade, as mentioned above. Grade 3 UCs, which generally have a poor clinical outcome, showed a huge variety of HER2 expression patterns, suggesting the need to evaluate different subsets of the disease based on HER2 expression patterns and other factors. As such, more extensive studies will be necessary to determine their correlations.
In addition to sample size, there are several limitations in this study. First, not all the information was available for each sample, because of the retrospective design of this study. Medications after cystectomy varied among the samples as listed in Supplemental Table S1, and this might have influenced the results of DFS and OS. Second, it is beyond the scope of this study to investigate the amplification status of the HER2 gene, which is routinely assessed in human breast cancer. 35 Normally, the HER2-positivity of human breast cancer was determined by both the expression patterns of HER2 and the copy numbers of the HER2 gene by in situ hybridization. Administration of trastuzumab is generally applicable in score 3+ cancers or in score 2+ cancers with a high HER2/CEN17 ratio (>2.0) and a high average HER2 gene copy number per cell (≥6.0). 35 As we are not sure whether there is gene amplification in canine UC, its status must be further investigated to determine a discriminant criterion for HER2-positivity.
In the field of clinical medicine, molecule-targeted cancer therapies have been of strong interest for the treatment of human cancers. More recently, these therapeutic targets including HER2-targeted therapy have also been investigated for small animal medicine. The protein sequence identity between human and canine HER2 is significantly high (92%), indicating that the canine homologue also expresses the cetuximab and trastuzumab binding sites and may facilitate antibody-based immunotherapy in dogs. 30 Furthermore, HER2-directed cancer immunotherapy using recombinant Listeria monocytogenes, which expresses a chimeric human HER2/neu fusion protein, has shown encouraging results in reducing the metastasis rates of canine osteosarcoma. 19 As a large percentage of UC samples were found to express HER2, HER2-targeted medications could be a potential treatment for canine UC.
In conclusion, we found that strong HER2 immunoreactivity is frequently observed in canine UC neoplastic cells, especially in grade 2 UCs. This is the first report that investigated HER2 expression in association with the histological grades of malignancy and the correlation between the expression and clinical features. While increased HER2 expression is observed in a subset of UCs, further mechanistic studies are needed to determine its role in the pathogenesis and targeted therapy of this cancer.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818817024 - Assessment of HER2 Expression in Canine Urothelial Carcinoma of the Urinary Bladder
Supplemental Material, DS1_VET_10.1177_0300985818817024 for Assessment of HER2 Expression in Canine Urothelial Carcinoma of the Urinary Bladder by Masaya Tsuboi, Kosei Sakai, Shingo Maeda, James K. Chambers, Tomohiro Yonezawa, Naoaki Matsuki, Kazuyuki Uchida and Hiroyuki Nakayama in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS Fellows of Japan Society for the Promotion of Science, a Grant-in-Aid for Science Research (KAKENHI grant 16H06208), and Anicom Capital Research Grant (EVOLVE).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
