Abstract
Prior studies have failed to detect a convincing association between histologic lesions of inflammation and clinical activity in dogs with inflammatory bowel disease (IBD). We hypothesized that use of a simplified histopathologic scoring system would improve the consistency of interpretation among pathologists when describing histologic lesions of gastrointestinal inflammation. Our aim was to evaluate the correlation of histopathologic changes to clinical activity in dogs with IBD using this new system. Forty-two dogs with IBD and 19 healthy control dogs were enrolled in this retrospective study. Endoscopic biopsies from the stomach, duodenum, ileum, and colon were independently scored by 8 pathologists. Clinical disease activity was scored using the Canine Inflammatory Bowel Disease Activity Index (CIBDAI) or the Canine Chronic Enteropathy Clinical Activity Index (CCECAI), depending on the individual study center. Summative histopathological scores and clinical activity were calculated for each tissue (stomach, duodenum, ileum, and colon) and each tissue histologic score (inflammatory/morphologic feature). The correlation between CCECAI/CIBDAI and summative histopathologic score was significant (P < .05) for duodenum (r = 0.42) and colon (r = 0.33). In evaluating the relationship between histopathologic scores and clinical activity, significant (P < .05) correlations were observed for crypt dilation (r = 0.42), lamina propria (LP) lymphocytes (r = 0.40), LP neutrophils (r = 0.45), mucosal fibrosis (r = 0.47), lacteal dilation (r = 0.39), and villus stunting (r = 0.43). Compared to earlier grading schemes, the simplified scoring system shows improved utility in correlating histopathologic features (both summative histology scores and select histologic scores) to IBD clinical activity.
Diagnosis of canine idiopathic inflammatory bowel disease (IBD) requires histopathologic confirmation of inflammation in intestinal biopsies. 12,16,17,26,33,40 Previous studies have found it difficult to correlate histopathologic findings with clinical disease activity due to a lack of agreement among pathologists when describing histopathologic changes 37 and inconsistent grading schemes reported by different groups* investigating canine and feline chronic enteropathies. The World Small Animal Veterinary Association (WSAVA) gastrointestinal (GI) standardization grading scheme 5 was an attempt to rectify some of these problems, but even this was associated with poor agreement among pathologists. 39
Prior studies have failed to detect a convincing association of mucosal histopathology with clinical signs, biomarkers of inflammation, or response to therapy and outcome in dogs with IBD. † One explanation for these findings is that histopathology and clinical activity are not associated, whereas another possibility is that current WSAVA guidelines require refinement to improve diagnostic consistency. 36 Recently, use of a simplified histopathologic scoring system has shown excellent agreement among pathologists in defining duodenal inflammation in dogs with IBD. 14
We hypothesized that use of the simplified histopathologic scoring system would improve the consistency of interpretation among pathologists when evaluating gastric, duodenal, ileal, and colonic endoscopic biopsies. The aim of the present study was to evaluate the correlation of histopathologic changes to clinical disease activity in dogs with IBD using this new system.
Methods
Ethical Animal Use
The collection and analysis of intestinal biopsies obtained endoscopically from healthy dogs and dogs with IBD were previously approved by the University of Giessen and the Iowa State University (ISU) Institutional Animal Care and Use Committee (IACUC). Written informed consent was obtained from all owners of dogs enrolled in separate trials (University of Giessen: V54-19c 20 15 (1) GI 18/17 Nr. 36/2011; ISU IACUC log numbers: 1-11-7061-K, 12-11-7269-K).
Animals
Two groups of dogs were studied. Group 1 comprised a cohort of 42 dogs diagnosed with idiopathic IBD according to previously published criteria. 2,12,16,33 Dogs with IBD were enrolled from 2 study centers: the Royal Veterinary College (RVC) (K.A.A.) and Iowa State University (A.E.J.) from January 2010 to May 2012. Over this 2.5-year period, approximately 150 dogs at ISU and 300 dogs at the RVC were referred for diagnostic evaluation of chronic gastrointestinal signs. In many instances, individual dogs underwent upper, lower, or both upper and lower GI endoscopy with collection of mucosal biopsies as part of the diagnostic investigation for their chronic GI signs. The inclusion criteria included persistent (>3 weeks’ duration) GI signs, failure to respond to appropriate dietary trials (elimination diet fed exclusively for at least 3 weeks) and antimicrobial therapy (metronidazole or tylosin administered exclusively for 14 days or more), failure to document other causes for gastroenteritis by thorough diagnostic testing, and histopathologic evidence of mucosal inflammation in biopsy specimens.
The diagnostic evaluation in all dogs with IBD consisted of extensive medical histories taken over 1 or more clinical examinations, hematological and serum biochemistry analyses, urinalysis, fecal examinations for parasites, diagnostic imaging (abdominal radiographs in all and abdominal sonography performed in 31/42 dogs), and histopathologic examination of GI mucosal biopsy specimens. In some dogs, samples were also collected for a measurement of serum pancreatic-lipase immunoreactivity (cPLI), trypsin-like immunoreactivity (cTLI), cobalamin, and/or folate concentrations.
Group 2 dogs served as controls and comprised 19 young (<2 years old) healthy laboratory-reared beagles (n = 12) and beagle cross mongrels (n = 7). Each of these dogs was free of GI signs over several months preceding diagnostic evaluation. Moreover, control dogs were judged to be healthy on the basis of normal results on physical examination, hematological and serum biochemical analysis, urinalysis, multiple fecal examinations, and Dirofilaria antigen assay. Mucosal biopsies of the small and large intestines were obtained from each dog as previously described. 2,16,33
Simplified Histopathologic Scoring System for Gastrointestinal Inflammation 14
The simplified histopathologic scoring system for GI inflammation in dogs was based on the morphologic and inflammatory features contained in the original WSAVA GI standardization data set graded by the 4 individual pathologists who participated in that study. In brief, χ2 analysis of these data was performed and compared the extent of agreement among study pathologists for each inflammatory and morphologic feature. The resultant P values were then used to determine the interobserver agreement among study pathologists for each single inflammatory or morphologic feature. The 3 to 5 histopathologic features that showed the least interobserver variability (based on χ2 analyses) for GI inflammation involving the stomach, small intestine, and colon comprised the simplified scoring system.
The current study independently validates use of the simplified scoring system that was first tested on the original WSAVA study population. We now report use of this same system in a separate study population with tissues graded by a different group of pathologists. This allowed for quantification of parameters resulting in the derivation of summative histopathologic scores and individual histopathologic scores that could then be correlated to clinical disease activity in dogs with IBD.
Histopathologic Assessment
Hematoxylin and eosin (HE)–stained tissue sections of formalin-fixed, paraffin-embedded endoscopic biopsies from the stomach, duodenum, ileum, and/or colon of each dog were evaluated for histologic lesions using a simplified scoring system for defining GI histopathological changes. Only tissue sections of adequate diagnostic quality were used in the study. 38 Poor-quality tissues (those graded as marginal or inadequate) were excluded. Individual glass slides from the different regions of the GI tract of healthy and diseased dogs were scanned on-site using an Aperio digital pathology whole-slide scanning system housed at Texas A&M University, College Station, Texas. Digital tissue images were then sent to each pathologist separately for review. Pathologists were blinded to the dogs’ status as a control dog or IBD patient. Up to 5 morphologic/inflammatory features in randomly sorted, HE-stained digital slides from each area of the GI tract were independently scored by the 8 study pathologists (Table 1). Individual pathologists’ lesion scores for each parameter were graded using a 4-point scale (ie, 0 = normal, 1 = mild, 2 = moderate, 3 = marked histopathologic change). This resulted in the generation of both region-specific scores and summative histopathologic scores used for statistical analysis. The same histopathologic grading criteria used for duodenal biopsy specimens were used for diagnostic interpretation of ileal tissue specimens.
Parameters Included in the Simplified Histopathologic Scoring System for Canine Inflammatory Bowel Disease.
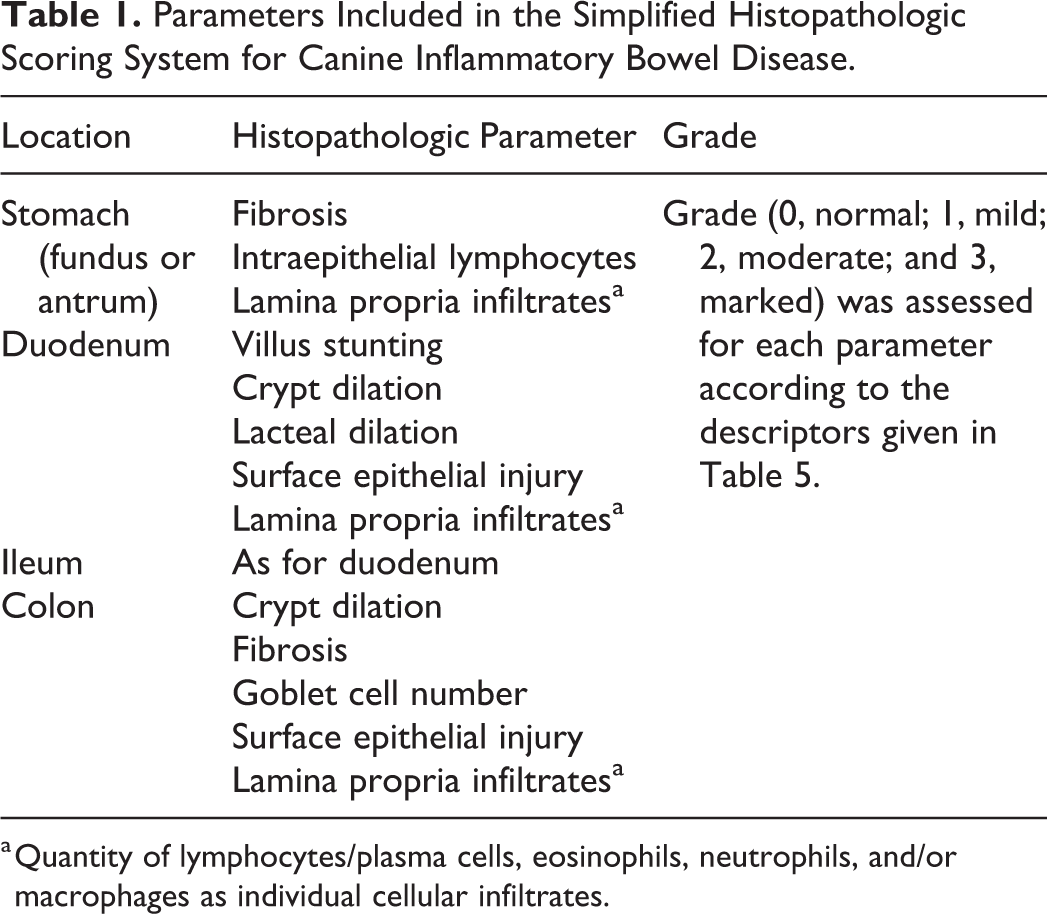
a Quantity of lymphocytes/plasma cells, eosinophils, neutrophils, and/or macrophages as individual cellular infiltrates.
Clinical Disease Activity
Severity of clinical disease (activity) at diagnosis was scored using either the Canine Chronic Enteropathy Clinical Activity Index (CCECAI) 2 or the Canine Inflammatory Bowel Disease Activity Index (CIBDAI) 18 at the different clinical trial centers.
Statistical Methods
Correlations between histopathologic variable(s) and clinical disease activity were calculated for both cohorts. For each dog, the average summative histopathologic score in each area of the GI tract (ie, averaged over the 3–5 histopathologic parameters) and individual histopathologic scores at each tissue location (stomach, duodenum, ileum, and colon) were calculated for correlation to clinical activity indices. Pearson correlation coefficients between histopathological scores and clinical disease activity were calculated for each tissue location and combination of tissue and histologic score. Significance of the correlation was determined by Fisher z-transformation tests using PROC CORR in SAS version 9.4 (SAS Institute, Cary, NC). Prior to this, differences among pathologists on those correlations were evaluated using F tests, after verifying the model assumptions of independence, homoscedasticity, and normality. For all tests, P values <.05 were considered statistically significant.
Interobserver Agreement Among Pathologists by Histopathologic Variable
When no significant differences in the (histopathological/clinical) correlations were found among pathologists, Cohen κ statistics were used to assess the extent of agreement among individual raters for each histopathologic variable. 31 This was performed using a binary or 2-point scale (ie, normal/mild and moderate/severe) as previously described. 39 The resulting κ statistic was interpreted as <0, less than chance agreement; 0.01 to 0.20, slight agreement; 0.21 to 0.40, fair agreement; 0.41 to 0.60, moderate agreement; 0.61 to 0.80, substantial agreement; and 0.81 to 0.99, almost perfect agreement. This output generated both κ statistics and P values to assess the significance of the κ scores.
Results
Of the 42 dogs with IBD assessed for clinical disease severity, 25 were enrolled at the RVC and scored using the CCECAI while 17 were enrolled at ISU and scored using the CIBDAI. For the RVC cohort, 11 dogs had mild clinical activity, 8 dogs had moderate clinical activity, and 6 dogs had severe clinical disease. For the ISU cohort, 2 dogs had mild clinical activity, 7 dogs had moderate clinical activity, and 8 dogs had severe clinical disease. Overall, 14 of 42 (33%) dogs with IBD had severe clinical disease (ie, CIBDAI/CCECAI score >9) with 7 of 42 (17%) dogs having hypoalbuminemia and/or ascites at IBD diagnosis. Thus, overall, 35 of 42 (83%) study dogs had identical CIBDAI and CCECAI scores for correlation to histopathologic indices.
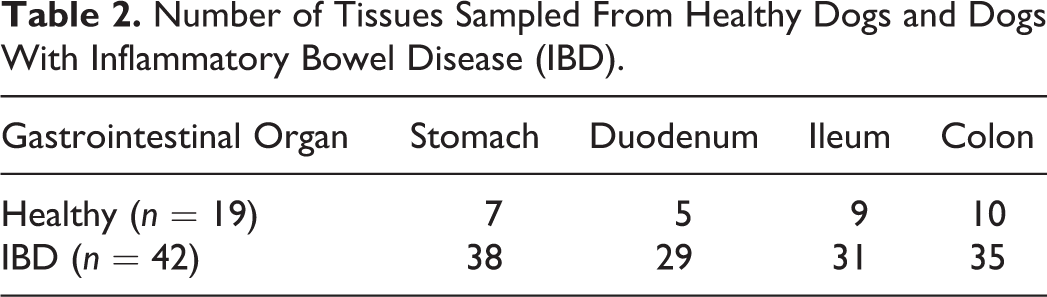
The areas of the GI tract available for diagnostic evaluation in the 2 dog groups are presented in Table 2. The collection of endoscopic biopsies from dogs with IBD was guided by localization of the predominant GI signs to the small intestine (enteritis, n = 8 dogs), large intestine (colitis, n = 4 dogs), or both small and large intestines (enterocolitis, n = 30 dogs). Digital images contained 6 to 15 individual endoscopic tissue specimens per glass slide depending on the area sampled. For some dogs, tissue samples from all 4 areas of the GI tract were not obtained due to anesthetic complications and time constraints, difficulty with endoscopic examination (retrograde ileoscopy), and/or clinical disease localization to a single area of the GI tract (eg, signs of colitis alone).
Number of Tissues Sampled From Healthy Dogs and Dogs With Inflammatory Bowel Disease (IBD).
Results of the F test comparing the histopathological/clinical correlations among the 8 pathologists did not show any significant differences when averaging summative histopathologic scores for the stomach, duodenum, ileum, or colon. Moreover, there was no significant difference among pathologists in the correlation of individual histopathological scores at each GI location and the clinical activity indices. In this instance, the histopathologic scores from all 8 pathologists were pooled together to calculate Pearson correlation coefficients and increase the power of the study for detection of statistically significant differences between histopathologic changes assigned by the 8 pathologists.
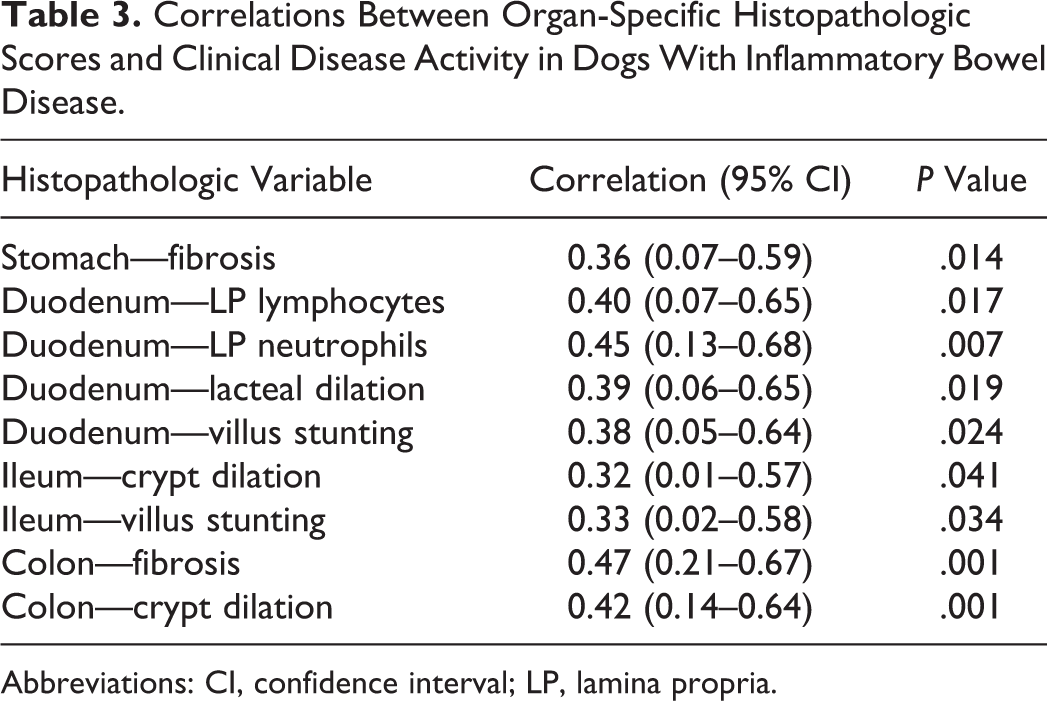
The correlation between the CCECAI/CIBDAI and the summative histology score was significant (P < .05) for duodenum (r = 0.42; 95% confidence interval [CI], 0.08–0.65) and colon (r = 0.33; 95% CI, 0.04–0.57). The correlation was nearly significant for ileum (P = .06, r = 0.29; 95% CI, –0.02 to 0.55) but nonsignificant for stomach (P = .7, r = 0.05; 95% CI, –0.24 to 0.34). Evaluation of individual histopathologic scores showed numerous significant correlations between histology and clinical disease activity for all areas of the GI tract, particularly the duodenum, ileum, and colon (Table 3). These lesions encompassed a variety of morphologic and inflammatory changes, including mucosal fibrosis, villus stunting, and crypt dilation, as well as changes in the character of the mucosal cellular infiltrate in dogs with IBD (Figs. 1-4).
Correlations Between Organ-Specific Histopathologic Scores and Clinical Disease Activity in Dogs With Inflammatory Bowel Disease.
Abbreviations: CI, confidence interval; LP, lamina propria.
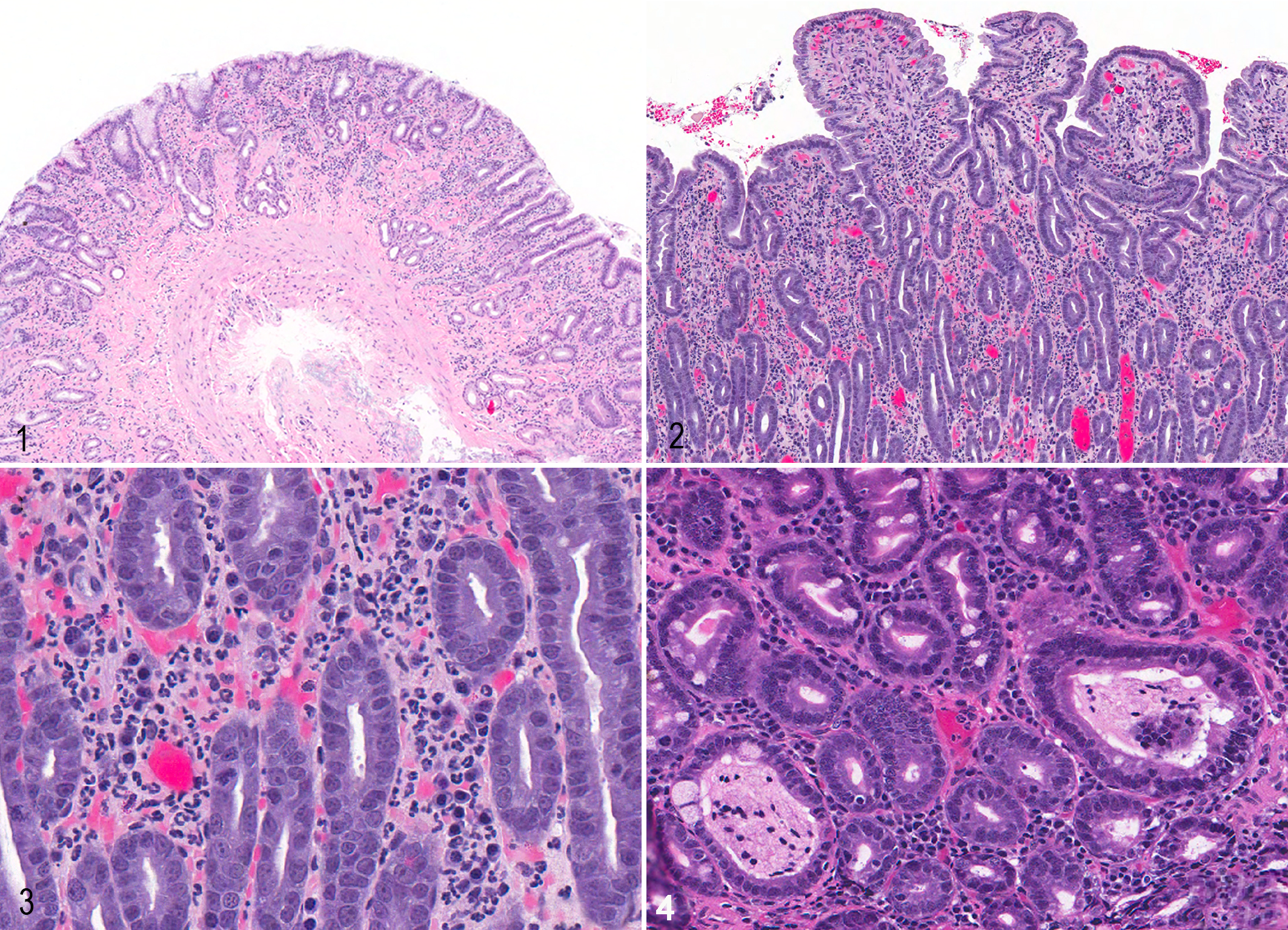
Inflammatory bowel disease, dog. Hematoxylin and eosin. Figure 1. Stomach, antrum. Within the lamina propria, there is multifocal glandular atrophy and replacement with mature fibrosis. Figure 2. Duodenum. There is diffuse moderate blunting and shortening (atrophy) of villus profiles. Figure 3. Duodenum. Within the lamina propria, there are markedly increased numbers of neutrophils. Figure 4. Ileum. There is multifocal dilation of crypts.
The κ agreement among pathologists for histopathologic scores varied across regions of the GI tract (Table 4). The best agreement for lesions was seen for colonic scores and lamina propria (LP) eosinophils across 3 of 4 areas of the GI tract. Those histopathologic lesions having at least fair to moderate agreement (>0.21–0.40) based on κ statistics included stomach (fibrosis, LP lymphocytes/plasma cells, LP eosinophils), duodenum (villus stunting, crypt dilation, lacteal dilation, LP lymphocytes/plasma cells, LP eosinophils), and colon (crypt dilation, LP eosinophils, macrophages).
Agreement Among 8 Pathologists (κ Statistics) for Histopathologic Scoring of Lesions in Dogs With Inflammatory Bowel Disease.a
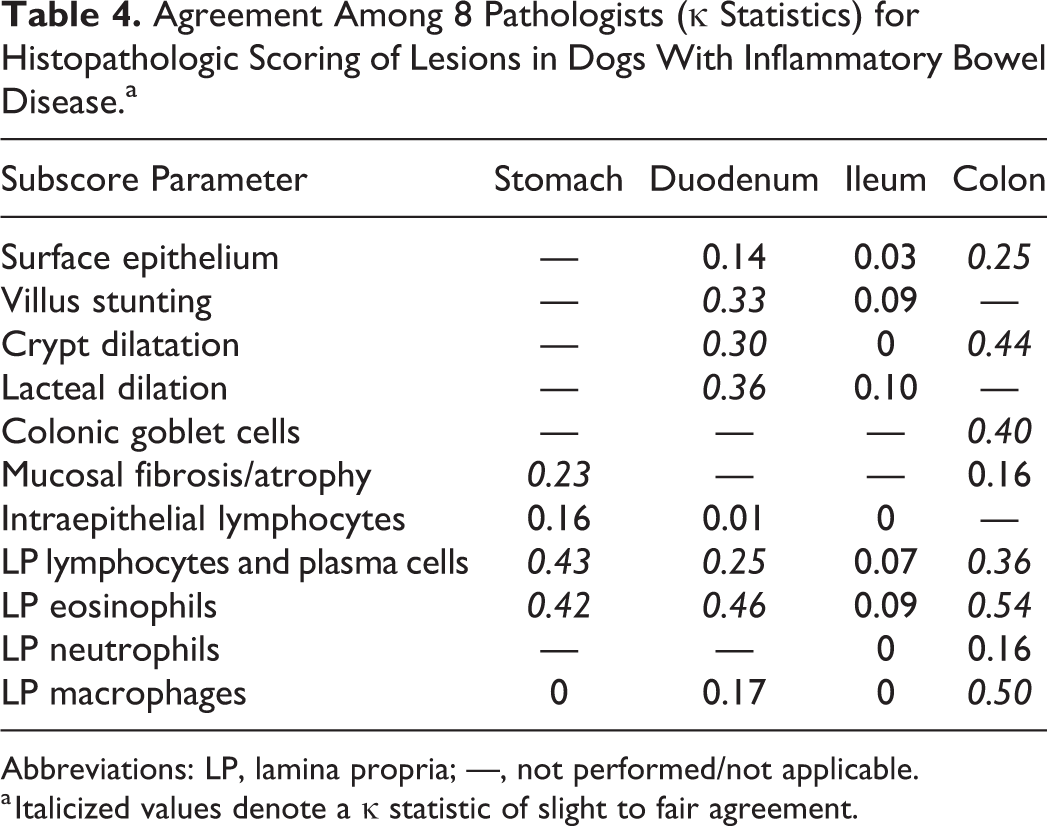
Abbreviations: LP, lamina propria; —, not performed/not applicable.
a Italicized values denote a κ statistic of slight to fair agreement.
Discussion
Characterizing the extent and severity of GI inflammation in endoscopic biopsy specimens from dogs and cats is difficult. Several studies from different institutions have shown that a variety of factors influence histopathologic interpretation of intestinal biopsies, including (1) the correct area of the GI tract to be sampled, 36 (2) the number and quality of tissue samples submitted to the laboratory, 38 (3) the quality of sample processing by the laboratory, 39 and (4) the lack of consistency in interpretation of histopathologic changes among pathologists. 5,37 Complicating these potential limitations are various grading schemes for interpretation of intestinal histopathology that have failed to gain uniform acceptance in different laboratories. In response to these concerns, the WSAVA GI Standardization Group designed a histopathologic template for characterizing the nature and severity of mucosal inflammatory and morphologic changes. It was hoped that this might reduce variation among different pathologists and different institutions. 5
Unfortunately, significant interobserver variability in the diagnostic interpretation of endoscopic mucosal specimens exists even with the use of the original WSAVA standardization criteria. 36,39 Moreover, the original WSAVA criteria do not consider some parameters, such as changes in the number of colonic goblet cells and their content of mucus, although this parameter may be decreased in granulomatous colitis (GC) and other forms of colitis 28,30 and may be significantly increased posttreatment. 20 A recent study evaluated a simplified histopathologic scoring system using select WSAVA template criteria (ie, those indices having the greatest agreement among WSAVA pathologists) to reduce variability in the diagnostic interpretation of GI inflammation among pathologists. 14 However, this investigation only graded inflammation in canine duodenal endoscopic biopsies and, like the original WSAVA study, did not include evaluation of ileal biopsy specimens.
The present study aimed to build on earlier observations using the simplified histopathologic scoring system, now extending its use to other areas of the GI tract (stomach, ileum, and colon) and correlating quantifiable histopathologic changes to clinical disease activity in dogs with IBD. Numerous studies from different groups have failed to show a consistent association between mucosal histopathologic changes and clinical disease severity either at diagnosis or in response to different treatments. Jergens et al 18 showed that clinical scores (CIBDAI) correlated best to a combination of histopathologic inflammation and serum C-reactive protein at IBD diagnosis; posttreatment histopathologic assessment was not performed in these dogs. In another study, clinical signs (CIBDAI scores) improved with medical treatment in dogs with IBD, but this did not result in significant changes in the severity of gastric or duodenal histologic inflammatory lesions. 9 In separate studies, Allenspach et al 1 showed that total lymphocyte numbers in the duodenal mucosa of dogs with IBD did not change in response to successful cyclosporine treatment and that histopathologic scores were not correlated with CCECAI scores, endoscopy scoring, or long-term outcome in dogs with IBD over 3 years. 2 In this latter study, 2 the presence of severe mucosal lesions in the duodenum (observed during duodenoscopy), hypoalbuminemia, and low serum cobalamin were significantly associated with negative outcome in dogs with chronic enteropathy. Munster et al 22 failed to demonstrate a strong correlation between treatment response (CIBDAI) and severity of histopathologic lesions of IBD. Finally, Rossi et al 25 showed reduced histopathologic lesion scores, accompanied by a positive clinical response (CIBDAI), following an 8-week course of combination probiotic therapy, while White et al 34 did not despite using the same probiotic protocol. Difficulties in showing associations in these earlier studies may relate to the use of nonstandardized histopathologic scoring systems and/or differences in study design.
Use of the simplified scoring system was repeatable among the different pathologists in the current study. The results comparing histopathological/clinical correlations among pathologists did not show significant differences when averaging summative histopathologic scores for the stomach, duodenum, ileum, or colon. Moreover, there was no significant difference among pathologists in the correlation of individual histopathological scores within separate areas of the GI tract and clinical disease activity. These findings suggest that histologic scores of mucosal inflammation and morphologic change can be consistently applied to the scoring system.
We also observed significant correlations between some histologic scores and clinical indices. Clinical activity in dogs with IBD was positively correlated to the summative histopathologic scores for the duodenum and colon. While the correlation between clinical disease and the summative histopathologic score approached significance (P = .06) for ileal biopsies, it was clearly not significantly correlated for the stomach. The association between histologic scores and clinical severity scored by internists was also significantly positive for several mucosal inflammatory/morphologic changes, including increased numbers of LP lymphocytes and neutrophils (duodenum), mucosal fibrosis (stomach and colon), crypt dilation (ileum and colon), and villus stunting (duodenum and ileum).
The histologic score of gastric fibrosis was correlated with clinical disease activity in our dogs with IBD. This was an interesting observation considering that the total gastric histopathology score was not correlated to disease activity in affected dogs. In the original WSAVA study, gastric fibrosis was combined with other morphologic changes indicative of mucosal injury (including glandular nesting and mucosal atrophy) to define the presence and extent of gastric inflammation. 5 The system used to grade gastritis in the present study (the simplified scoring system) was adapted from that proposed by the WSAVA standardization group and also included gastric fibrosis/mucosal atrophy as 1 parameter of morphologic injury. One standardized photographic grading scheme for evaluating gastric atrophy, fibrosis, and cellular infiltrates has also been proposed for characterizing gastritis in dogs. 11 In this previous study, expression of interleukin (IL)–1β and the presence of mast cells and atrophy were related to gastric fibrosis. While the association of cytokines and cellular infiltrates with architectural changes was not investigated in the present study, it is possible that IL-1β and other inflammatory mediators in the lesions of IBD were related to gastric fibrosis. 8,19,23
There was no correlation between the numbers of colonic goblet cells, intraepithelial lymphocytes, LP eosinophils, and macrophages with IBD clinical disease activity. Changes in the numbers of goblet cells in dogs with colitis have been previously described in separate clinical studies. Roth et al, 26 using a colitis score comprising LP cellular infiltrates, epithelial architecture, intraepithelial lymphocytes, and number of goblet cells, showed that dogs with moderate to severe IBD colitis were more likely to have reduced/absent numbers of goblet cells vs heathy dogs. Mansfield et al 20 showed that decreased numbers of goblet cells were present in a cohort of Boxers with GC and observed an increase in goblet cells and improved histopathological abnormalities following eradication of invasive intramucosal Escherichia coli with enrofloxacin administration. Colonic goblet cell numbers in dogs with IBD of the present study were not reduced compared to the numbers of goblet cells in colonic biopsies obtained from healthy dogs. As noted by Roth et al, 26 however, changes in goblet cell numbers are not always accompanied by clinical signs since 28 of 48 samples from dogs with a clinical diagnosis of chronic diarrhea and/or colitis had histopathologic normal tissues (eg, colitis grade of ≤2.0). 26
There are some potential limitations to this study. We used a retrospective study design of archived gut specimens for diagnostic investigation from only 2 study centers. The healthy dog cohort was difficult to find at any one institution; hence, this cohort was realized by pooling samples from laboratory-reared animals from different (ISU and RVC) institutions. These dogs were younger than most dogs with IBD, but the availability of age- and breed-matched control tissues for comparison to dogs with IBD was difficult due to the retrospective study design with limited availability of tissue blocks. It would have been ideal to study posttreatment biopsy samples from these same dogs with IBD to evaluate changes in disease activity and histopathology vs baseline values, but this was not possible in all cases. Last, endoscopic biopsy specimens from the stomach, duodenum, ileum, and colon were not available from all dogs for histopathologic interpretation and may have reduced the overall power of our study.
In conclusion, the simplified histopathologic scoring system provides objective and descriptive information on the extent of mucosal inflammation in the GI tract of dogs. This scoring system incorporates key WSAVA morphologic and inflammatory features that can now be applied to diagnostic interpretation of endoscopic specimens obtained from the stomach, duodenum, ileum, and colon. Compared to earlier grading schemes, the simplified scoring system shows improved utility in correlating histopathologic features (both summative histology scores and select histologic scores) to IBD clinical activity, at diagnosis, as defined by CIBDAI/CCECAI.
We now provide a quantitative simplified scoring system for use by pathologists and clinicians alike in future studies (Table 5). Finally, gastric biopsies would appear to be less clinically useful vs duodenal and colonic biopsies for defining intestinal inflammation in dogs with IBD.
Quantitative Simplified Scoring System for Defining Gastrointestinal Inflammation.
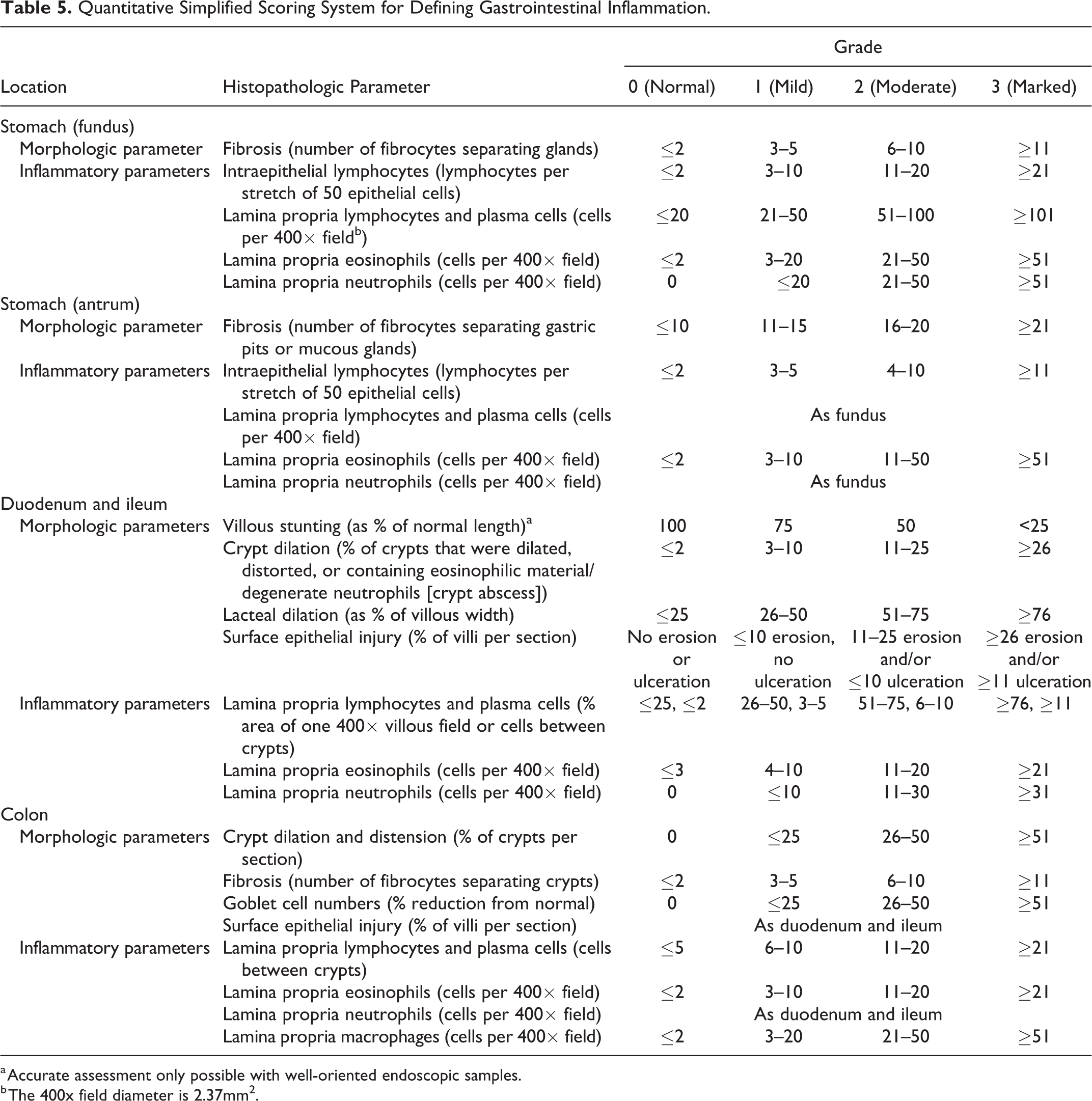
a Accurate assessment only possible with well-oriented endoscopic samples.
b The 400x field diameter is 2.37mm2.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818813090 - Correlating Gastrointestinal Histopathologic Changes to Clinical Disease Activity in Dogs With Idiopathic Inflammatory Bowel Disease
Supplemental Material, DS1_VET_10.1177_0300985818813090 for Correlating Gastrointestinal Histopathologic Changes to Clinical Disease Activity in Dogs With Idiopathic Inflammatory Bowel Disease by Karin A. Allenspach, Jonathan P. Mochel, Yingzhou Du, Simon L. Priestnall, Frances Moore, Michael Slayter, Aline Rodrigues, Mark Ackermann, Mark Krockenberger, Joanne Mansell, WSAVA GI Standardization Working Group, Nicole Luckschander, Chong Wang, Jan Suchodolski, Nora Berghoff and Albert E. Jergens in Veterinary Pathology
Footnotes
Authors’ Note
This work was performed at Iowa State University, the Royal Veterinary College, and Texas A&M University.
Acknowledgement
We thank Dr. Alejandro Suarez-Bonnet for providing excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
