Abstract
Chlamydiosis is the most documented and serious disease of koalas, characterized by ocular, urinary, and reproductive lesions. Since little attention has been paid to the pathological effects of this infection in the male reproductive system, we aimed to determine the incidence and severity of reproductive pathology associated with chlamydial infection in male koalas submitted to koala hospitals in southeast Queensland. The entire reproductive tract from 62 sexually mature male koalas not suitable for rehabilitation was evaluated and 677 tissue samples were collected for histology, immunohistochemistry (IHC), and real-time polymerase chain reaction (qPCR). Lymphoplasmacytic inflammation was observed in 178 of 677 (26.3%) tissue samples from the upper and lower reproductive tract, mainly in the prostatic, penile, and membranous urethra. IHC was positive for the chlamydial antigen in 19 of 451 normal samples (4.2%) and 46 of 178 samples with inflammation (25.8%), located within the cytoplasm of epithelial cells of the epididymis, vas deferens, prostate, bulbourethral glands, and the prostatic membranous and penile urethra. Chlamydia pecorum was detected via qPCR in 319 of 451 normal samples (70.7%) and 159 of 178 samples with inflammation (89.3%), with the highest incidence in the penile urethra, prostate, membranous urethra, and bulbourethral glands. This study suggests that Chlamydia infection in the male reproductive tract is more widespread than originally thought. Furthermore, the male reproductive tract might be a reservoir for persistent chlamydial infections in koalas, with important implications for prophylactic strategies and epidemiology.
Population numbers of koala, the most iconic Australian marsupial species, continue to decline in large parts of eastern Australia, due to a loss of habitat caused by land clearing, motor vehicle trauma, domestic dog attacks, and infectious diseases. 10,27,31 Chlamydiosis is the most documented and serious disease of koalas, with a prevalence of 72% to 100% in the northern regions, in particular Queensland and northern New South Wales. 41 The most pathogenic and prevalent Chlamydia species in wild populations is Chlamydia pecorum, which causes ocular, urinary, and reproductive diseases. 9,19 Chlamydia is a small obligate intracellular Gram-negative bacterium, alternating between 2 morphological forms, the infectious elementary bodies (EBs) and the reticulate bodies (RBs). EBs are extracellular, less metabolically active forms invading susceptible cells and differentiating into the intracellular metabolically active forms (RBs). 1 The latter undergo several cycles of binary fission reverting back to EBs, released by either lysis of infected cells 1,47 or packaged release of host membrane-bound vacuoles containing EBs, a process termed extrusion. 17 If stressful growth conditions (eg, antibiotics, nutrient deprivation, and immune response) disrupt this life cycle, aberrant RBs that neither replicate nor differentiate into EBs can result in persistent infection. 1
Another species, Chlamydia trachomatis, is the most prevalent bacterial cause of sexually transmitted infections in humans, 5 with approximately 131 million new cases of genital infection diagnosed worldwide every year. 51 In men, C. trachomatis is responsible for urethritis, epididymitis, orchitis, prostatitis, and vesiculitis. 29 The association between Chlamydia spp. and poor semen quality has been analyzed in several human clinical studies, but the detrimental effects of this infection on male fertility are still uncertain. 8,24 Experimental studies on Chlamydia-induced male genitourinary tract infection have also been performed on nonhuman primates, 32 mouse, 37 rat, 20,21 and guinea pig models. 50 Furthermore, in natural settings, Chlamydia spp. can cause seminal vesiculitis and epididymitis in bulls 44 and rams, 26 as well as chronic granulomatous orchitis in bulls. 4 Prostatitis, epididymitis, and orchitis have been reported in boars as well. 42,45
The gross and histologic lesions associated with isolation of Chlamydia spp. from the koala female reproductive tract are well documented and include acute or chronic vaginitis, salpingitis, endometritis, urogenital sinusitis, and cystic dilatation of the ovarian bursa. 3,14,16,35,36 The prevalence of reproductive lesions in sexually mature female koalas is high, affecting 31% to 57% of cases in wild populations in southeast Queensland. 25
Apart from a few descriptions of prostatitis associated with Chlamydia infection in koalas 6,15 and the first report of orchitis and epididymitis in C. pecorum–infected koalas, 22 little attention has been paid to the effects of this pathogen on male fertility, the distribution of Chlamydia in the male reproductive tract, and the associated lesions. To address this knowledge gap, we aimed to determine the incidence and severity of reproductive pathology associated with chlamydial infection in male koalas submitted to a major koala hospital in southeast Queensland.
Materials and Methods
Animals and Sampling
This study was performed on 62 sexually mature wild male koalas (Phascolarctos cinereus) presented to Moggill Koala Hospital–Queensland (Australia) (E152.52′26.4′′; S27°34′46.56′′) (Queensland Department of Environment Heritage and Protection). Animals were selected based on the following inclusion criteria: age (sexually mature), sex (males), and welfare issues (animals not suitable for rehabilitation). Selected animals had history of conjunctivitis (29/62), cystitis (21/62), chronic weight loss (2/62), and trauma (8/62). Signalment and history were not available for 8 animals. All animals were euthanized by intracardiac pentobarbitone sodium overdose under gaseous anaesthesia. 48 At necropsy, gross lesions were recorded and tissue samples collected for histopathology, immunohistochemistry, and quantitative polymerase chain reaction (qPCR). A total of 677 tissue samples were examined, including right testis (n = 57), left testis (n = 56), right epididymis cauda (n = 34), right epididymis head (n = 57), left epididymis cauda (n = 38), right vas deferens (n = 31), left vas deferens (n = 30), prostate (n = 52), prostatic urethra (n = 52), membranous urethra (n = 35), right bulbourethral (BU) gland I (n = 32), left BU gland I (n = 58), left BU gland II (n = 27), right BU gland II (n = 33), and penile urethra (n = 31). All the procedures were conducted under the approval of the University of Queensland Animal Ethics Committee (permit number AE02305).
Histology and Immunohistochemistry
All samples were immediately fixed in 10% neutral-buffered formalin, dehydrated, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). The intensity of the inflammatory reaction was graded based on the number of inflammatory cells per five 400× fields: absent (0–10 cells), mild (11–50 cells), moderate (51–150 cells), and severe (>150 cells).
Immunohistochemistry was performed on 4% paraformaldehyde-fixed samples from the HistoTechnology Facility–Queensland Institute of Medical Research (QIMR) (Brisbane, QLD, Australia) using a Chlamydiaceae family–specific sheep polyclonal antibody directed against the chlamydial major outer membrane protein (MOMP) at 1:500 dilution. 38 Endogenous peroxidase activity was blocked using 2% H2O2 in Tris-buffered saline (TBS). Enzymatic antigen retrieval was performed by incubating the sections in pepsin for 10 minutes at 37°C. Donkey serum and fetal calf serum were used for blocking nonspecific antibody binding. Tissues were incubated with the primary antibody for 1 hour at room temperature and then with a donkey anti-sheep secondary antibody (Jackson ImmunoResearch, West Grove, PA, USA). 3,3′-Diaminobenzidine (DAB) was used to develop the signal and the sections were finally counterstained in Mayer’s hematoxylin. A Chlamydia-positive koala lung 30 was used as positive control. As a negative control, the primary antibody was substituted with phosphate-buffered saline (PBS). Positive labeling was defined as cytoplasmic staining of densely packed round structures, typical of chlamydial elementary and reticular bodies as previously described. 16
qPCR Detection for Chlamydia DNA
C. pecorum was detected using a multiplex real-time PCR previously validated 18 on 677 tissue samples and the final concentration expressed as infectious-forming units per milliliter (IFU/ml). Based on the concentration, 4 different scores of C. pecorum load were generated: (1) ≤100 IFU/ml, (2) 101 to 1000 IFU/ml, (3) 1001 to 10 000 IFU/ml, and (4) >10 000 IFU/ml. 2
Statistical Analysis
C. pecorum PCR load in immunohistochemistry (IHC)–positive and IHC-negative samples was statistically analyzed through the Mann-Whitney test. The relationship between each IFU/ml score and severity of inflammation (mild, moderate, severe) was verified by a χ2 test followed by the Cramer’s V post hoc test using GraphPad Prism (GraphPad Software, La Jolla, CA). The accepted level of significance was P < .05. For the statistical analysis, 2 Chlamydia load scores were evaluated (<1000 IFU/ml and >1000 IFU/ml) to obtain a representative number of samples for comparison.
Results
Gross Pathology
During the postmortem examination, the following gross lesions suggestive of chlamydial infection were observed: hemorrhagic cystitis (8/62), chronic cystitis (13/62), chronic interstitial nephritis (4/62), conjunctivitis (29/62), and prostatitis (1/62) (Fig. 1). No lesions were detected in 18 of 62 animals (29%).
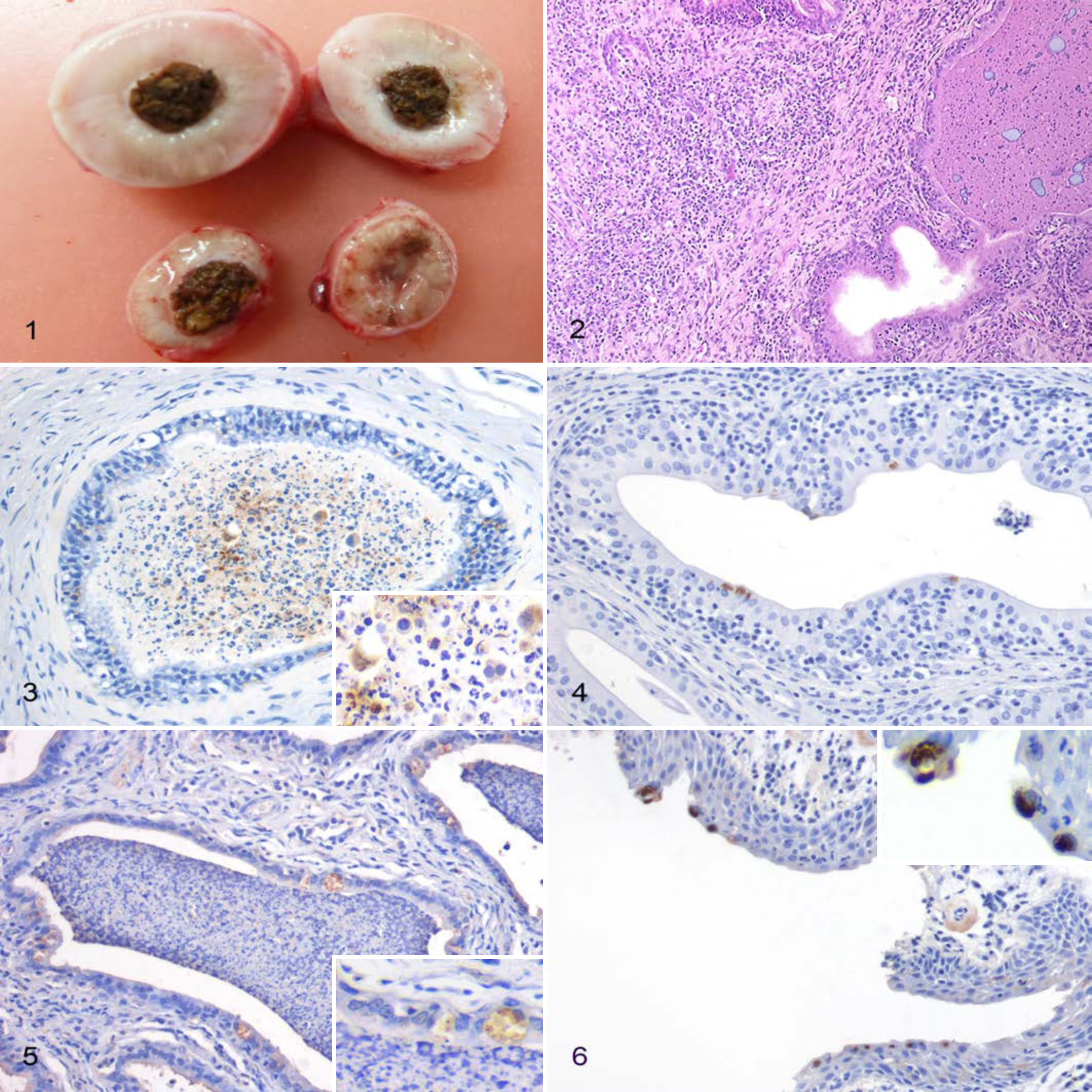
Histopathology
An inflammatory reaction was detected in 178 of 677 tissue samples (26.3%) from the 62 animals, which most frequently consisted of lymphocytes and plasma cells (171/178; 96.1%) and less frequently neutrophils (4/178; 2.3%); lymphocytes, histiocytes, and neutrophils (1/178; 0.6%); or neutrophils and lymphocytes (1/178; 0.6%) (Table 1, Suppl. Table S1). The most commonly affected organ was the prostatic urethra (46/52; 88.5%) (Supplemental Fig. S1), followed by penile urethra (26/31; 84%), membranous urethra (25/35; 71%), prostate (33/52; 63%), bulbourethral glands (25/150; 16.7%) (Fig. 2), testis (7/113; 6.2%) (Supplemental Fig. S2), vas deferens (3/61; 5%), and epididymis (7/183; 3.8%) (Supplemental Fig. S3).
Prevalence of Histologic Lesions and Immunohistochemical and Quantitative PCR Detection of Chlamydia Antigen in the Upper and Lower Reproductive Tract of 62 Male Koalas.
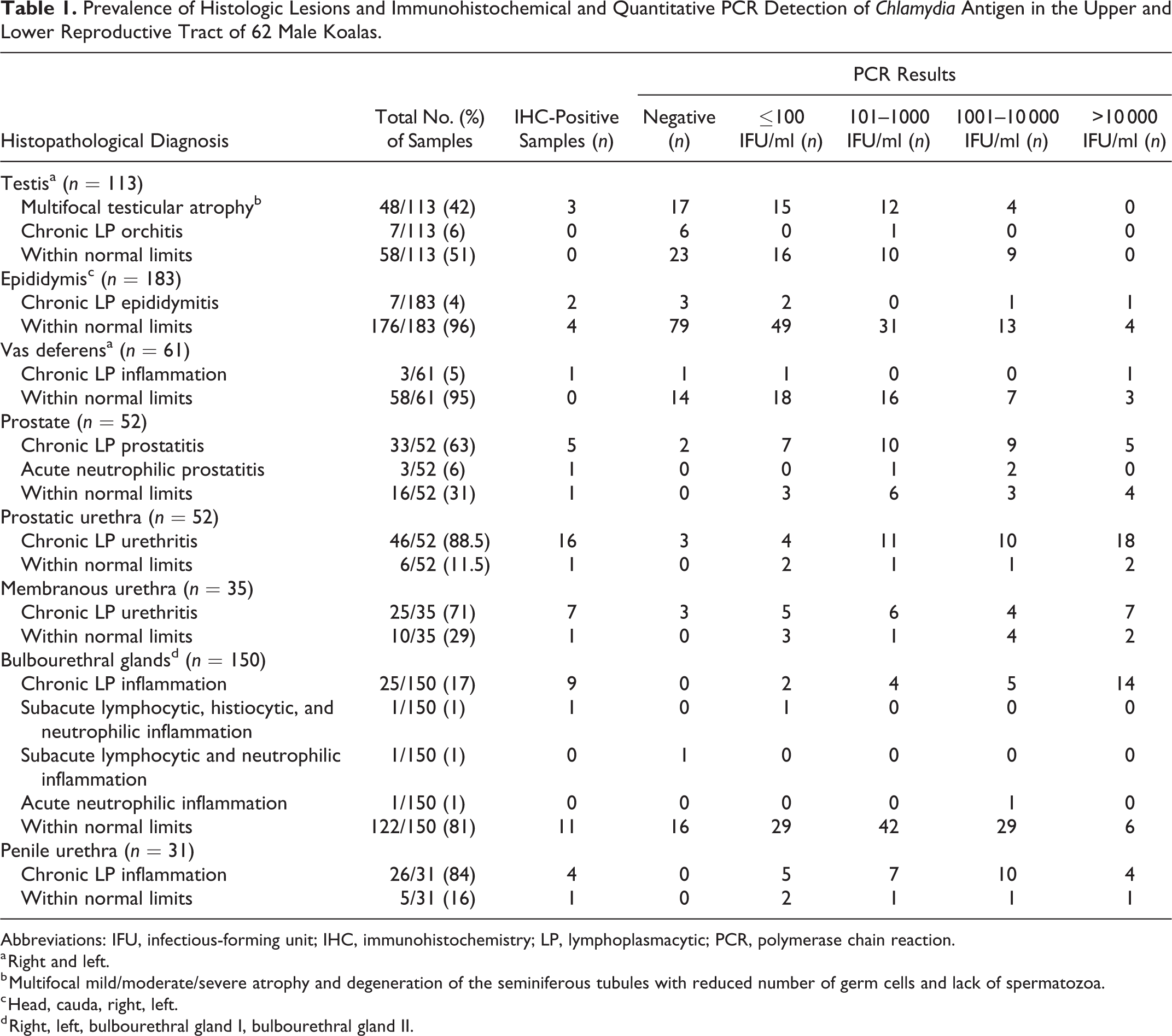
Abbreviations: IFU, infectious-forming unit; IHC, immunohistochemistry; LP, lymphoplasmacytic; PCR, polymerase chain reaction.
a Right and left.
b Multifocal mild/moderate/severe atrophy and degeneration of the seminiferous tubules with reduced number of germ cells and lack of spermatozoa.
c Head, cauda, right, left.
d Right, left, bulbourethral gland I, bulbourethral gland II.
Immunohistochemistry
Immunohistochemical results are summarized in Table 1 and specific details given in Supplemental Table S1. Overall, immunohistochemistry for Chlamydia spp. was positive in 64 of 677 samples (9.4%). Positive immunolabeling was detected in 3 of 113 testes (2.6%), within the cytoplasm of germ cells or interstitial cells. Six of 183 epididymides (3.3%) had positive immunolabeling in the cytoplasm of epithelial cells lining the ducts and sloughed within the lumen (Fig. 3), as well as in lymphocytes and macrophages infiltrating the stroma. Only 1 vas deferens, which was effaced by lymphoplasmacytic inflammation, contained positive immunolabeling. Chlamydial antigen was also demonstrated in the epithelial cells of the prostate (Fig. 4), prostatic urethra, membranous urethra, and penile urethra in 7 of 52 (13%), 16 of 52 (31%), 8 of 35 (23%), and 5 of 31 (16%) samples, respectively. Bulbourethral glands were positive in 21 of 150 cases (14%) (Figs. 5, 6).
Immunohistochemistry was positive in 19 of 451 normal samples (4.2%) and 46 of 178 samples with inflammation (25.8%) (Suppl. Fig. S4).
qPCR
qPCR results are summarized in Table 1 and specific details given in Supplemental Table S1. Overall, 509 of 677 samples (75.2%) were positive for C. pecorum by qPCR, with the highest frequency in the penile urethra (31/31; 100%), followed by prostate (50/52; 96%), membranous urethra (32/35; 91%), and bulbourethral glands (133/150; 88.7%). PCR detection of Chlamydia pneumoniae in all tissue types was 0/677 (0%).
The highest C. pecorum concentration (>10 000 IFU/ml as tested by qPCR) was most commonly observed in the prostatic urethra (20 of 52 samples, 38%), membranous urethra (9 of 35 samples, 26%), and prostate (9 of 52 samples, 17%). qPCR was positive in 319 of 451 histologically normal samples (70.7%) and 159 of 178 samples with inflammation (89.3%) (Suppl. Fig. S4).
Statistical Analysis
Moderate to severe inflammation was most commonly observed in tissue samples with a C. pecorum concentration >1000 IFU/ml (Suppl. Table S2), although the association between severity of the inflammatory reaction and Chlamydia concentration was statistically significant only in the bulbourethral glands (P < .05) with a Cramer’s V of 0.051.
C. pecorum PCR load was significantly higher in IHC-positive cases (mean = 495 594 IFU/ml) compared to IHC-negative samples (mean = 3103 IFU/ml) with P < .0001.
Discussion
In our study, we demonstrated colonization of epithelial cells of the penile urethra, bulbourethral glands, membranous and prostatic urethra, prostate, and occasionally the vas deferens and epididymis by Chlamydia spp., thus suggesting that the infection of the male reproductive tract may be more widespread than originally thought, despite being not clinically obvious.
In male koalas, we speculate that the primary site of infection might be the penile urethra, as already demonstrated in C. trachomatis–infected men. 8 Therefore, Chlamydia spp. might first infect epithelial cells of the penile urethra in the form of EBs and then undergo the developmental cycle to RBs, finally converting again into EBs, which infect nearby epithelial cells leading to an ascending infection. Ascending Chlamydia infection has been observed experimentally in mice, 39 monkeys, 32 and rats, 20 as well as in spontaneous infections in men 29 and bulls. 46
In addition to the penile urethra, major sites of infection in male koalas were the prostate and bulbourethral glands, where histologic lesions and DNA detection were frequently observed and where Chlamydia spp. may potentially establish a chronic persistent infection. In mouse models, the prostate represents a reservoir of continuous transmission of the infection. 29 Colonization of the male accessory glands may also result in impaired male fertility. The main function of the prostate gland is the production of a large amount of soluble proteins that provide an adequate medium for sperm survival and motility in the female reproductive tract. 11 Likewise, in the male koala, prostate secretions represent the bulk of seminal plasma, while bulbourethral glands produce secretions that have an important role in forming the seminal plug. 23 We speculate that the chronic inflammatory reaction induced by Chlamydia spp. infection might impair normal function of the glands, resulting in a potential negative impact on sperm function.
The role of C. trachomatis infection in chronic bacterial prostatitis is well known in men, with infiltration of mononuclear cells in the interstitium and focal to multifocal intraglandular accumulation of variable numbers of neutrophils. 49 A similar pattern has been observed in the 33 cases of chronic lymphoplasmacytic prostatitis in our study. Although the severity of the histologic lesions was not statistically associated with the qPCR load, 31 of 33 cases contained detectable C. pecorum. In the 2 qPCR-negative cases, the inflammation may have been caused by other pathogens or alternatively may be secondary to an autoimmune reaction in the absence of detectable infectious agents. 33 Mackern-Oberti et al 28 have demonstrated that genital C. trachomatis infection in male Wistar rats favored the breakdown of immune tolerance to prostate antigens in a significant number of animals leading to autoimmunity.
In addition to the organs of the lower reproductive tract, chlamydial antigen was occasionally detected in the germ cells of the seminiferous tubules and within the lumen of ductules and ducts of the epididymis, supporting the hypothesis that the infection may damage spermatozoa, spread through the semen, and affect male fertility. In the upper reproductive tract, Chlamydia-induced pathology may exert a direct negative effect on the sperm, especially when infecting seminiferous tubules and germ cells, or an indirect effect associated with the inflammatory obstruction of the tubules and ducts and the epithelial damage that results in impaired spermatogenesis and sperm transport. 8
In our study, immunohistochemistry was positive only in 9.4% of cases compared to the 75.2% of positive samples detected by qPCR. This difference in the percentage of positive samples may be explained by different sample collection and tissue sample size. Tissues for IHC and qPCR were collected from adjacent but different sites of each organ, each potentially with a different antigen distribution. In addition, Chlamydia inclusions tend to cluster, especially in folds within the epithelium, so that some of them may be partially or completely missed. Therefore, it is difficult to ensure that a tissue sample actually contains Chlamydiae when an infection is present. 34 Finally, a piece of tissue collected for extraction may have more surface epithelial cells, as well as free individual elementary bodies, with each inclusion having up to 700 gene copies, compared to the number of epithelial cells examined on a single histological section. The lack of antigen detection despite qPCR-positive results and histological lesions may be therefore related to inadequate sensitivity of IHC or the absence of the organism in the section under examination.
Finally, Chlamydia was frequently detected in male koalas without gross and histologic lesions, with immunohistochemistry and qPCR positive in 4.2% and 70.7% of normal samples, respectively. This finding raises a serious concern in terms of subclinical transmission of the infection from affected or recovered males, paralleling the typical feature of chlamydial genital tract infection in men, which is asymptomatic in up to 50% of cases. 13,40,43 That the infection was not readily detectable by routine clinical and pathological examination may mean that a high number of Chlamydia-infected koalas will go undetected unless targeted screening programs or sensitive diagnostic tests are in place. Regarding the latter, one major issue may potentially be false-negative results of urethral swabs when the infection is mainly localized in the upper reproductive tract. Seminal fluid or prostatic secretions may be Chlamydia positive in men with negative urethral swabs. 7,12 Therefore, even in male koalas, urethral swabs may not be sensitive in all cases.
Conclusions
This study confirms that (a) Chlamydia can invade cells of the upper and lower reproductive tract, (b) C. pecorum may be associated with inflammation of the bulbourethral glands and the upper genital tract, and (c) inapparent reproductive infection may be possible. The male reproductive tract might be a reservoir for persistent Chlamydia infections in koalas, and this finding will have important implications for prophylactic strategies and epidemiology. Whether or not Chlamydia leads to significant detrimental effects on male koala fertility requires further investigation.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818806963 - Chlamydia pecorum Infection in the Male Reproductive System of Koalas (Phascolarctos cinereus)
Supplemental Material, DS1_VET_10.1177_0300985818806963 for Chlamydia pecorum Infection in the Male Reproductive System of Koalas (Phascolarctos cinereus) by Chiara Palmieri, Lyndal Hulse, Sara Pagliarani, Rebecca Larkin, Damien P. Higgins, Kenneth Beagley, and Stephen Johnston in Veterinary Pathology
Footnotes
Acknowledgements
Sincere thanks to the staff of the Moggill Koala Hospital (Queensland, Australia) for the technical assistance during sample collection and to the staff of the HistoTechnology facility (QIMR, Queensland, Australia) for assisting with immunostaining.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Department of Environment and Heritage Protection, Queensland Government, project no. KRG005.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
