Abstract
Although Chlamydia causes disease of the urethra and prostate of male koalas, its impact on the testis and epididymis has not been examined. This study describes chronic-active and granulomatous orchitis and epididymitis with interstitial fibrosis associated with infection by Chlamydia pecorum in 2 of 18 adult male koalas being euthanized at a koala hospital, 8 of which also had chlamydial prostatitis. By immunohistochemistry and transmission electron microscopy, chlamydial inclusions were demonstrated within Sertoli cells directly associated with mild inflammation surrounding intact seminiferous and epididymal tubules, marked pyogranulomatous inflammation around disrupted tubules, replacement of tubules by interstitial fibrosis, and aspermia. The presence of C. pecorum but not Chlamydia pneumoniae was detected by quantitative polymerase chain reaction of formalin-fixed tissues of the left and right testes and right epididymis in 1 animal. This is the first report of orchitis and epididymitis in a koala infected with C. pecorum.
Chlamydia pneumoniae and Chlamydia pecorum are well known to cause disease of the urogenital tract and conjunctiva of male and female koalas, with infection of the female reproductive tract commonly resulting in infertility. Both species have been recovered from the prostate and urethra of the male koala, and although Chlamydia has been reported to infect the testis and epididymis of the bull, ram, human, boar, and guinea pig, there are no published reports of it infecting the koala testis or epididymis. 1,2,7 –9 In humans, chlamydial orchitis and epididymitis affects fertility adversely by directly interfering with spermatogenesis and the mature sperm cell, respectively. This report describes 2 cases of chlamydial orchitis and epididymitis in male koalas, with 1 confirmed as involving C. pecorum.
The right testis, right epididymis, and prostate were collected from 18 sexually mature male wild koalas (case Nos. 1–18) presenting with clinical signs of cystitis and fixed in 10% buffered formalin and in 3% glutaraldehyde in 0.2M sodium cacodylate buffer (pH 7.3); all animals were euthanized for welfare reasons at the Queensland Government Moggill Koala Hospital or the Australian Wildlife Hospital, both in South East Queensland, Australia. Only 2 koalas (case Nos. 1, 2) had histologic lesions in the testes; the findings of the remaining 16 koalas are provided in Supplemental Table 1. The left testis was also processed in Case No 1. Procedures were conducted under the approval of the University of Queensland Animal Ethics Committee (permit SAS/483/09/UQ).
Formalin-fixed tissues were processed by routine histologic methods and stained with hematoxylin and eosin and antichlamydial LPS antibody. 4 On demonstration of chlamydial inclusions in the testicular parenchyma of case No. 1 by immunohistochemistry (IHC), glutaraldehyde-fixed tissue samples from the same testis were washed twice in 0.1M sodium cacodylate buffer and then processed for transmission electron microscope by 4 repeats of postfixing in 2% osmium tetroxide in the microwave with vacuum for 2 minutes and then without vacuum for 2 minutes. Samples were then dehydrated through graded acetone solutions and embedded in Epon resin (ProSciTech, Thuringowa, Queensland, Australia). Thin sections were cut using a Leica Ultracut 6 ultramicrotome (Leica Microsystems, Vienna, Austria) and stained with uranyl acetate and lead citrate. Tissues were examined under a JEOL JEM 1010 transmission electron microscope (JEOL Pty Ltd, Tokyo, Japan). To confirm the species of Chlamydia in Chlamydia-infected tissues by quantitative polymerase chain reaction (qPCR), tissue sections (6 µm) were cut from paraffin blocks and extracted with 100 µl of QuickExtract FFPE DNA extraction solution (EPICENTRE Biotechnologies, Wisconsin, USA) before qPCR with C. pneumoniae– and C. pecorum–specific primers, utilizing primers for koala beta-actin as a control for DNA quality and PCR inhibition. 3
Case No. 1 entered the Moggill koala hospital with moderate urinary staining of rump fur (suggestive of cystitis) and poor body condition. On postmortem examination, the right testis was hemorrhagic and small, the tunica albuginea markedly thickened, and the right epididymis appeared to be fibrosed; the contralateral tissues were grossly normal. Histopathologic examination of the right testis showed chronic-active orchitis with fibrosis. The lumen of some seminiferous tubules contained exudate, consisting predominantly of neutrophils and macrophages, and a mixture of debris from seminiferous epithelial cells and cellular protein-rich debris. The interstitial tissue had mild to moderate plasma cell and lymphocyte infiltration with mild fibrosis; intact seminiferous tubules were surrounded by mild infiltrates, whereas those in which the epithelium was discontinuous were surrounded by more intense infiltrates. In the fibrotic right epididymis, granulomatous aggregates of macrophages and lymphocytes replaced some tubules, while in other areas, intact epididymal tubules were surrounded by fibrotic interstitial tissue. Epididymal tubules contained no spermatozoa within the lumen, consistent with the absence of visible sperm in the right testis. Histopathologic examination of the left testis showed no evidence of inflammation, but this testis also lacked spermatozoa, with only Sertoli cells and a few primary spermatogonia seen within the tubules.
While the right testis of koala case No. 2 showed no overt evidence of gross pathology, histopathology revealed granulomatous orchitis with interstitial fibrosis. The seminiferous tubules were reduced in number and replaced by infiltrates consisting mostly of macrophages and a few giant cells and lymphocytes; the surrounding interstitium was markedly fibrosed and contained numerous plasma cells. Some of the seminiferous tubules had lost any resemblance to normal histologic architecture, except for remnant Sertoli cells. While there was evidence of interstitial fibrosis in the epididymis, the tubules were histologically normal, other than the absence of spermatozoa.
Positive labeling of chlamydial LPS antigen was demonstrated by IHC in the right testis of case Nos. 1 and 2 and in the right epididymis of case No. 1, in direct association with pathologic changes (Fig. 1). Labeling was absent when the primary antibody was omitted. Where the epithelium of the seminiferous tubule was intact, well-defined chlamydial inclusions could be seen within the cytoplasm of some Sertoli cells. Presence of C. pecorum but not C. pneumoniae was detected by qPCR of formalin fixed tissues of the left and right testes and right epididymis of case No. 1; a biopsy of the left epididymis was not available for qPCR. Chlamydial DNA was not amplified from formalin-fixed tissues from case No. 2, but IHC findings were supported by detection of chlamydial antigen by the Clearview test on a swab from these samples. Chlamydial antigen or DNA was not detected in the testis or epididymis of any other koala in the study, consistent with absence of histologic lesions in those tissues, but chlamydial antigen was detected by using the Clearview test in the prostatic tissue of 7 of these koalas and in case No. 2. Other bacteria were not detected in tissues of case Nos. 1 and 2 by tissue Gram stain.
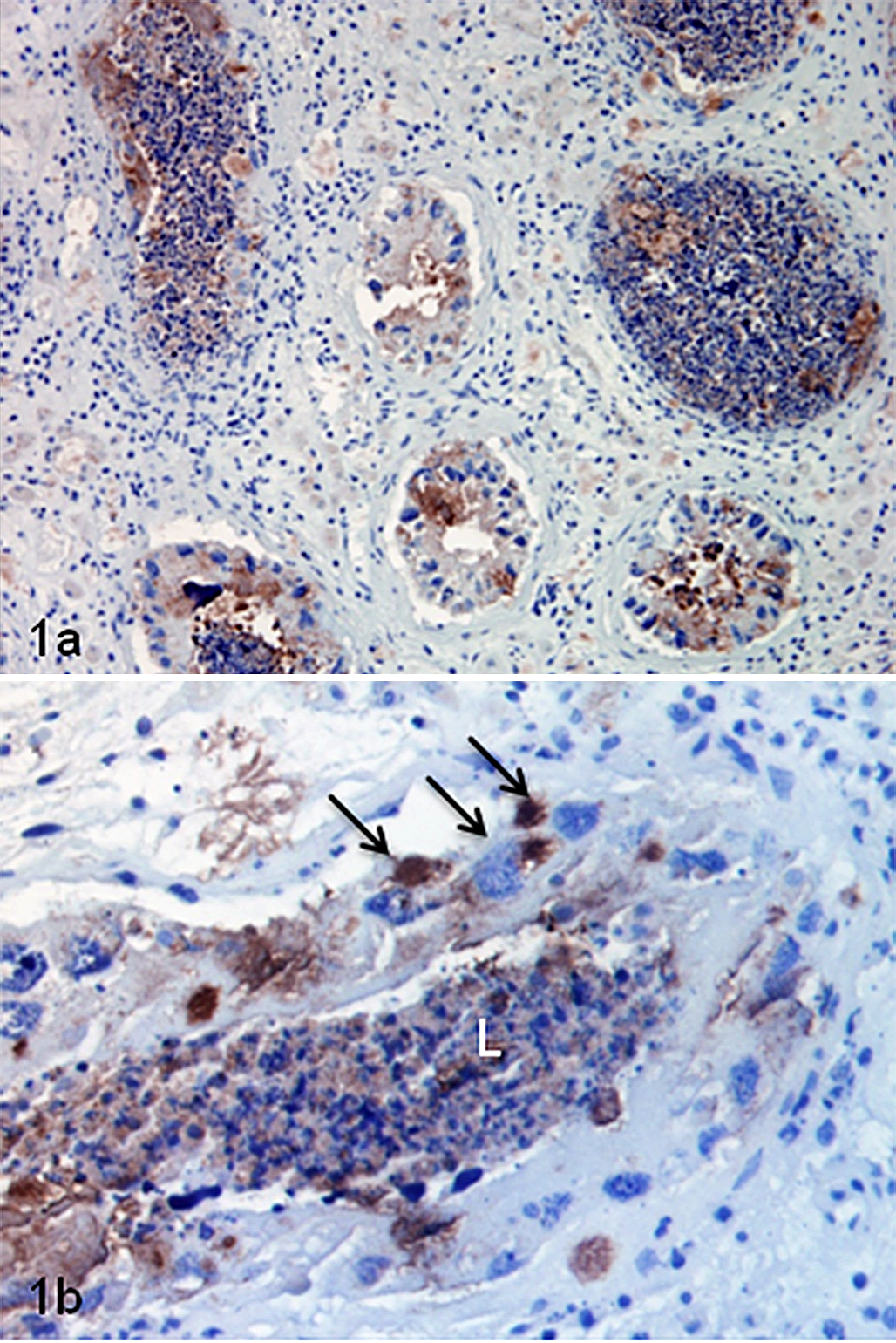
Chlamydial infection, testis; koala, case No. 1. (a) Multifocal labeling of cells lining seminiferous tubules. (b) Labeling of debris in the lumen (L) of a seminiferous tubule and well-defined chlamydial inclusions within the cytoplasm of Sertoli cells (arrows). Immunohistochemistry for chlamydial lipopolysaccharide.
Electron microscopy revealed bodies consistent in size and morphology with chlamydial elementary bodies (EBs) and reticulate bodies (RBs; not shown), aggregated between the crystalloids and the basement membrane of the Sertoli cells of the right testis of case No. 1 (Fig. 2). EBs (200–400 nm) were numerous and had highly dense eccentric nuclei, while RBs (1000 nm) were fewer and had a dispersed nuclear core. The characteristic double membrane of Chlamydia-like bodies was detected in a few EBs and RBs. Interestingly, transmission electron microscope also revealed that all spermatogenic cells were degenerated in all sections of the seminiferous tubules of the right testis of case No. 1.
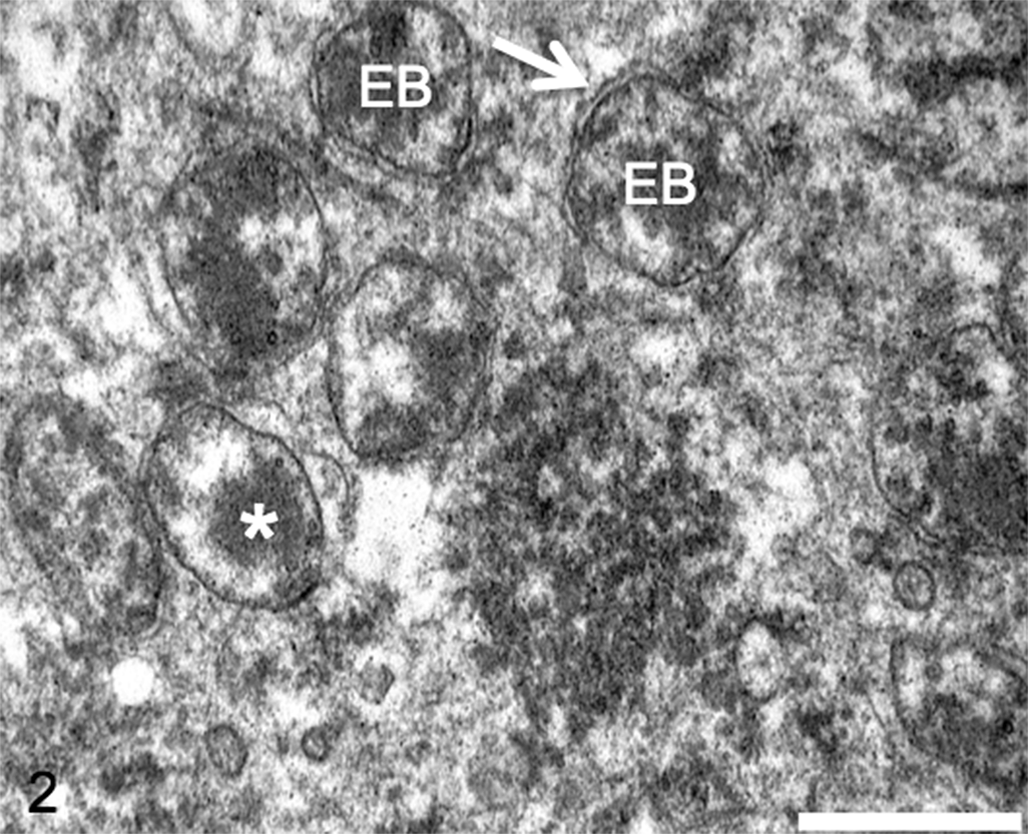
Testis; koala, case No. 1. Chlamydia-like elementary bodies (EBs) within Sertoli cells. Note the chlamydial double membrane (arrow) and the dense eccentric nuclear material (*) of the EBs. Transmission electron microscopy. Bar = 500 nm.
The clear association of Chlamydia with lesions, the absence of Chlamydia in normal koala testes and epididymides, and the absence of any other visible bacterial pathogen on Gram-stained sections strongly implies that Chlamydia plays a pathogenic role in these lesions. Our inability to amplify C. pecorum or C. pneumoniae by qPCR from the second case (case No. 2), despite amplifying koala DNA and detecting chlamydial antigen by IHC and the Clearview test, might indicate an alternative chlamydial species is involved in this case. Alternatively, chlamydial DNA might have been selectively degraded by macrophages; the morphology of IHC labeling in case No. 2 was more consistent with phagocytized chlamydial antigen in macrophages, in contrast to the large number of well-defined inclusions of apparently viable Chlamydia in Sertoli cells of case No. 1.
Lesions were consistent with the range of chronic-active and granulomatous inflammatory lesions that represent different stages of inflammation in chlamydial disease of other mammals. 5,8,9 It could not be determined whether the route of infection in these cases was hematogenous or ascending, although the range of lesions suggested a progression from discrete, well-defined inclusions in cells of the tubular epithelium in association with mild inflammation to interstitial granulomatous inflammation and phagocytosis of chlamydial antigen by interstitial macrophages following loss of epithelial integrity. The mild inflammation in intact seminiferous tubules was consistent with the seminiferous tubules being an immune-privileged tissue; although complete degeneration of spermatogenic cells may occur in some cases, interstitial orchitis is unlikely to occur as long as the basement membrane of the infected tubule is intact. 1,4 It is evident that chlamydial infection of the koala prostate and urethra does not ascend to the epididymis and testis in all cases; in this study, orchitis and epididymitis occurred twice among 8 koalas with chlamydial prostatitis. Further investigation of factors that determine the progression of infection to the testis and epididymis in the koala is clearly warranted.
Given the apparent impact of the chlamydial infection on spermatogenesis described in this study, future investigations should focus on the impact of the infection (clinical and subclinical) with respect to sperm quality and the venereal transmission of viable EBs. Chlamydial infections in human males may affect fertility by directly fragmenting sperm cell DNA and affecting motility, concentration, and morphology. 1 Scope exists to examine mechanisms of chlamydial male infertility in the koala, as reliable techniques for assessing sperm DNA fragmentation in the koala have been developed and natural chlamydial infection of the koala is very common in some populations. 6,10 As previous studies have considered only the consequences of female koala sterility as a result of chlamydiosis, our findings are an important contribution to the understanding of this disease’s effect in relation to the reproductive potential of koala populations. The lack of previous reports of orchitis and epididymitis in the koala is surprising and may be related to the less obvious gross pathology associated with disease in testicular tissue, when compared to cystitis and prostatitis and female koala reproductive pathology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was supported by a research donation provided by San Diego Zoo, San Diego, California, USA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
