Abstract
Equine dysautonomia (grass sickness) is characterized by autonomic neuronal degeneration and is often fatal. As outbreaks occur, rapid diagnosis is essential but confirmation currently requires histological examination. This study evaluated diagnostic accuracy of cytological examination of cranial cervical ganglion (CCG) scrapings for dysautonomia diagnosis. CCG smears from 20 controls and 16 dysautonomia cases were stained with May-Grünwald Giemsa (MGG), hematoxylin and eosin (HE), and cresyl fast violet (CFV), with HE-stained histological sections of CCG as gold standard for diagnosis. Examining all 3 stains together, the sensitivity and specificity were 100%. Occasional individual smears (4/107, 3.7%) were nondiagnostic due to low cellularity, and in a few individual smears the final diagnosis was correct but more tentative (CFV: 5/33 [15.1%], HE: 2/34 [5.9%], and MGG: 4/36 [11.1%]), due to low cellularity or suboptimal cell morphology. CCG cytology was considered reliable for rapid postmortem diagnosis of equine dysautonomia, particularly using MGG.
Equine dysautonomia (ED or equine grass sickness) is a disease of equids reported worldwide, but mainly recorded in Northern Europe, particularly the United Kingdom. 2 The disease is characterized by degenerative changes in the neurons of the autonomic system including the pre- and para-vertebral ganglia, enteric neuronal plexuses, and some brainstem nuclei. 5,6,12 These changes include chromatolysis, loss of Nissl substance, and nuclear pyknosis or loss. The disease occurs in acute, subacute and chronic forms, the first 2 being invariably fatal. Clinical signs of acute and subacute ED relate mainly to alimentary tract dysfunction (dysphagia, gastric distention with fluid, decreased intestinal motility, colonic impaction and colic), but skeletal muscle tremors and sweating are also evident. 11 The cause is unknown but an association with toxicoinfection with Clostridium botulinum type C has been suggested, 7 although a cause-effect relationship remains unproven. The disease almost always occurs in grazing horses and is strongly associated with particular premises. While single cases often occur, outbreaks are also common. 4
Currently, routine histological examination of ileal biopsies obtained at laparotomy, or autonomic ganglia at postmortem examination, are considered the gold standard for antemortem and postmortem diagnosis, respectively. 11 Histological examination may take 2-3 days for processing, and neuronal changes in more rapid cryostat sections are more difficult to interpret than in formalin-fixed, paraffin wax-embedded sections. 10 However, it is essential that diagnosis is as rapid as possible to allow management strategies to be implemented to avoid outbreaks occurring, 8 for example, removal of co-grazing horses from the pasture. The hypothesis of the current study was that cytological examination of neurons in scrapings of the cranial cervical ganglion (CCG) obtained at postmortem examination would provide a rapid diagnosis of ED.
Samples were collected from horse cadavers submitted to the Pathology service of the Royal (Dick) School of Veterinary Studies, University of Edinburgh, with approval of the University’s Veterinary Ethical Review Committee (approval no. 25 14). CCG were collected from 16 ED cases (9 acute, 4 subacute and 3 chronic, age range 10 months-17 years), and 20 control horses (age range 2.5-34 years) euthanized for a variety of reasons, including end of career horses (9 cases), fractured limb (3 cases), and 1 case each of chronic multilimb lameness, bladder paralysis (lower motor neuron bladder with secondary myogenic dysfunction), severe bilateral nephropathy, oesophageal rupture, and colic with ileal hypertrophy. In 3 cases the final cause for death was not definitively identified, but histopathological evaluation of CCG and ileum sections ruled out ED. The samples were collected between 30 minutes and 72 hours of death (median 16 hours). The CCG was sectioned longitudinally and the cut surface scraped using a scalpel blade held perpendicular to the surface. The material obtained was spread thinly onto 3 standard microscope slides and rapidly air-dried. For each animal, the cytological slides were stained with the Romanowsky-type stain May-Grünwald Giemsa (MGG), hematoxylin and eosin (HE), and cresyl fast violet (CFV), one slide for each stain, using standard methods. MGG was chosen as a standard cytological stain, HE for comparison with the HE-stained histological sections, and CFV because it specifically stains neuronal Nissl substance purple-blue. 13 The remainder of the ganglion was fixed in 10% phosphate-buffered formalin (pH 7.4) and processed to paraffin wax embedded blocks and HE-stained histological sections using standard methods.
The cytological slides were examined blindly by one clinical pathologist (CP) and were classified as ED-affected or control based on the presence or absence of neuronal degeneration, recognized as a combination of chromatolysis, loss of Nissl substance, and pyknosis or loss of nuclei. This categorization was initially made for each slide and stain individually, then the results for all 3 slides from each case were considered collectively. Histological sections were used as gold standard for classification; in the histological sections, typical degenerative changes were evident in the ED cases, and normal neurons in the controls (Figs. 1–2).
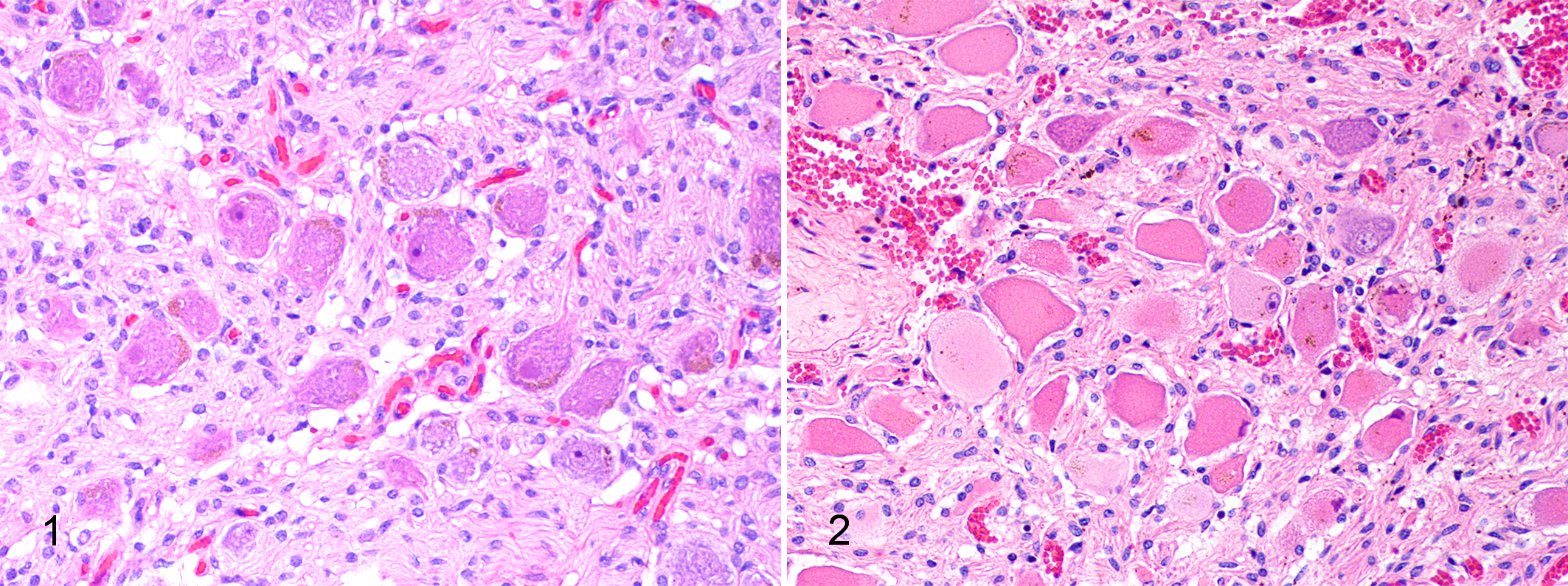
Normal cranial cervical ganglion, histological section, horse. Normal neurons have granular, basophilic Nissl substance and a large nucleus with open chromatin and a prominent nucleolus. Hematoxylin and eosin (HE).
In most of the cytology slides, sufficient neurons were present for a diagnosis to be made. There was a clear difference in neuron morphology between the ED and control cases (Figs. 3-8). In the MGG-stained smears, Nissl substance and large nuclei with dispersed chromatin were evident in the controls whereas in the ED cases, the cytoplasm appeared smooth with loss of Nissl substance and the nuclei, where present, were pyknotic and often marginated. Similar changes were seen in the CFV and HE-stained sections, although CFV stained cellular structures other than Nissl substance less clearly than the other stains. A subset of 4 cases (2 controls, 1 acute ED, and 1 subacute ED) was also stained with an aqueous-based, Romanowsky-type quick stain available for use in veterinary practice (Rapi-Diff II Stain Kit®, Atom Scientific LTD, Manchester, UK) and gave identical staining properties to the MGG-stained smears.
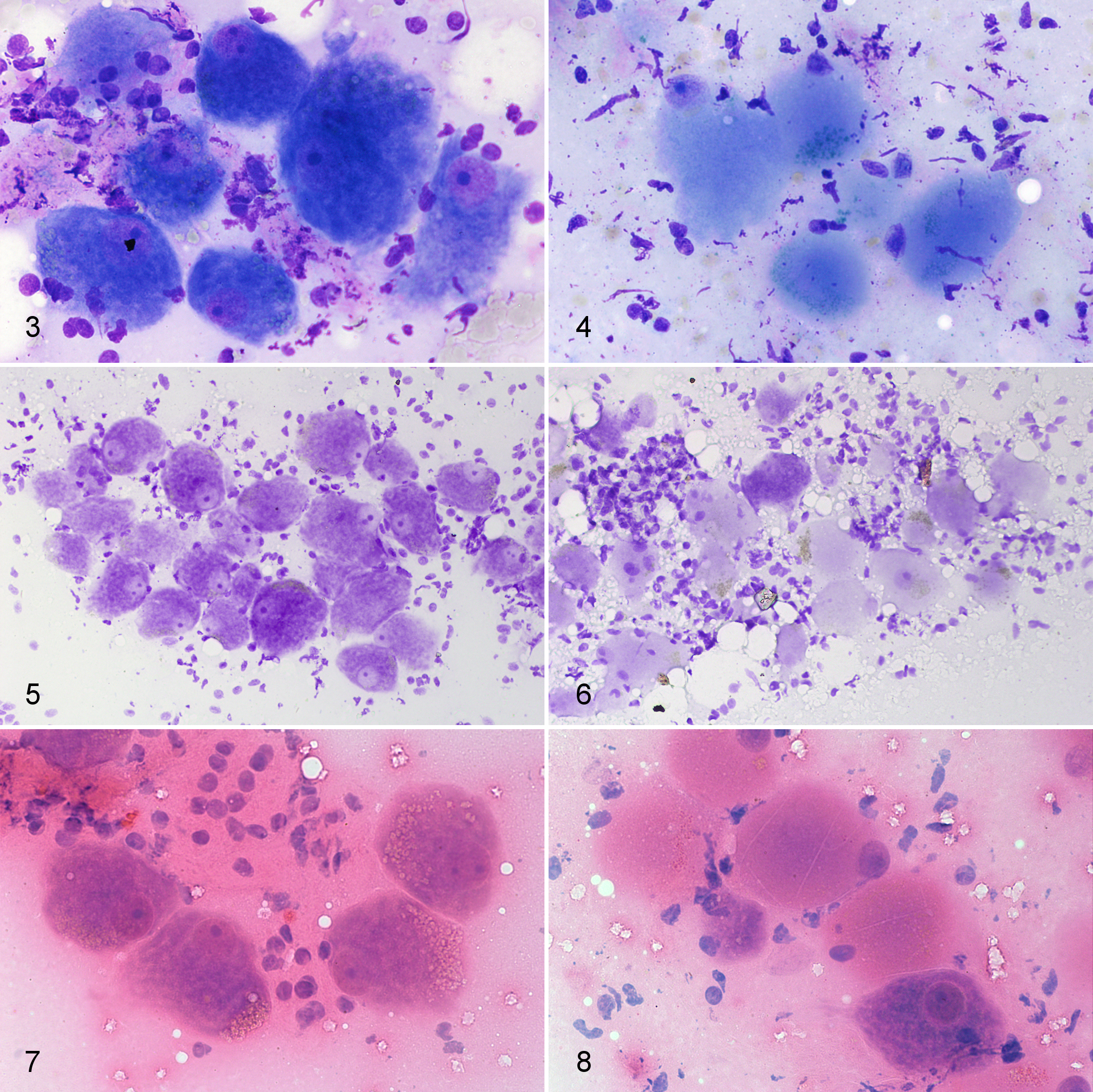
Considering all 3 stains together, a correct diagnosis was made in all 36 cases (sensitivity and specificity 100%). One HE-stained smear was lost during processing. A small number of individual smears (4/107 from 2 controls and 2 acute ED cases; 3.7%) were nondiagnostic due to very low cellularity and were excluded from the calculation of the results for the individual stains. Considering individual smears, a correct diagnosis was achieved for each diagnostic slide (103/103). However for a small number of individual slides, specifically 5/33 (15.1%) CFV-stained, 2/34 (5.9%) HE-stained, and 4/36 (11.1%) MGG-stained smears, a correct diagnosis was still made but the diagnostic confidence of the pathologist was lower due to low cellularity (for all MGG smears) or suboptimal morphological detail of the neurons (especially in the CFV-stained slides). Diagnostic accuracy for reaching a certain diagnosis for each stain (calculated as [definitive true positive + definitive true negative results]/number of diagnostic slides) was 84.8% for CFV, 94.1% for HE, and 88.8% for MGG.
The cytological examination of neurons is undertaken relatively infrequently for disease diagnosis although the morphology of neurons in smears have been described and our findings in the normal neurons are similar to previous reports. 3 The control cases in this study represented a variety of conditions; we did not include subsets of specific disorders in the group, such as neurological diseases, as the reported histopathological changes in ganglia are considered characteristic for ED. 5 Previous studies included controls with specific diseases (botulism, 9 tetanus and ragwort poisoning 1 ), but the typical features of degeneration were not seen in the neurons of those cases.
To the authors’ knowledge, cytological examination of neurons has not been previously described in ED. The morphological changes that characterize the disease in standard histological sections were also evident in the cytology smears. Although a diagnosis could be made using MGG, CFV and HE-stained smears, diagnostic confidence was slightly higher with MGG- or HE-stained smears. Particularly, MGG appeared to be the stain that showed more obvious morphological differences between ED and control cases. This might be partly due to familiarity with the use of this standard cytological stain, but the morphological features also appeared clearer, particularly in comparison with CFV. MGG staining is more likely to be readily available for cytological staining in diagnostic clinical pathology laboratories and it is recommended that this stain is used when assessing neuron cytology in ED. As identical morphological features could be identified when using a rapid stain used in general practice (Rapi-Diff II Stain Kit®), it is possible that the technique could be undertaken by suitably trained veterinary practitioners in the field as well as by diagnostic laboratories. It was notable that the neuronal morphology was well preserved and the differences within control and ED cases were still clearly evident at least 72 hours after death, since a delay before postmortem examination is a common problem in equine practice. In addition, by using cytology, the time delay between sample collection and diagnosis, due to sample processing, was reduced from 2-3 days for histological sections to less than 1 hour.
A few problems were encountered during the study, mainly related to technical issues. A minority of slides were nondiagnostic due to very low cellularity. It is important that adequate deep scraping of the ganglion cut surface is undertaken, with careful smearing to prevent cell lysis. However, overall, the technique was easy and quick to perform with a very high degree of accuracy in suitably prepared samples. Also, suboptimal morphology of the cells hampered a completely confident interpretation for a few slides. Poor morphology of neurons was mostly observed in CFV-stained smears, and this could reflect inconsistent staining properties of CFV on cytology slides.
A limitation of the study is that the evaluation of diagnostic performance of cytology for ED diagnosis was based on examination of the smears by only one clinical pathologist. However, in different settings, the diagnostic performance of cytology may vary due to interpersonal variation amongst pathologists, level of experience, and familiarity with histopathological diagnosis of this specific disease.
Use of the ganglion cytology scrapings could have considerable benefits in suspected ED cases in that management changes could be implemented sooner, reducing the chance of further cases occurring.
Footnotes
Acknowledgements
The authors thank the owners, primary clinicians, and staff of the Dick Vet Equine Hospital and Pathology Departments for their help. We thank Dr Jorge Del Pozo for his help with figures.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Equine Grass Sickness Fund [grant number B30227].
