Abstract
Objectives:
Renal denervation is an interventional approach aiming to reduce high blood pressure. Its efficacy is subject of controversial debate. We analyzed autonomic function in patients undergoing renal denervation to identify responders.
Methods:
A total of 21 patients with treatment-resistant hypertension scheduled for renal denervation were included. Heart rate variability, pupillary function and sympathetic skin response were examined prior to intervention. Before and 1 or 3 months after intervention, 24-h ambulatory blood pressure readings were taken.
Results:
Patients were stratified according to sympathetic nervous system function. Sympathetic activity was reduced in 12 participants (group 1) and normal or enhanced in nine patients (group 2). The mean of daytime systolic blood pressure decreased in groups 1 and 2 from 168 to 157 mmHg (95% confidence interval for difference, 1–21 mmHg, p = 0.035) and from 166 to 145 mmHg (8–34 mmHg, p = 0.005), respectively. In a linear model, blood pressure reduction was 11.3 mmHg (0.3–22 mmHg) greater in group 2 than in group 1 (p = 0.045).
Conclusion:
Patients with preexisting reduced activity of the sympathetic nervous system benefited less from renal denervation.
Keywords
Introduction
Renal denervation (RDN) by radiofrequency ablation is a new therapeutic approach to reduce blood pressure (BP) in patients with treatment-resistant hypertension. 1 This catheter-based intervention has been thoroughly examined for its potential to reduce BP. The results of uncontrolled and controlled randomized studies with small case numbers indicated significant BP reductions.1,2
However, the first randomized controlled trial comparing RDN with a sham procedure did not show a significantly higher BP reduction in the intervention arm. 3 Azizi et al., 4 on the other hand, confirmed the BP-reducing effect in a recent randomized controlled trial comparing RDN with antihypertensive drug therapy.
The rationale of RDN is reduction in sympathetic nervous system (SNS) overdrive, thought to be an important contributor to the pathophysiology of essential and resistant hypertension.5,6 Indeed, sympathetic overactivity has been demonstrated in hypertensive patients by microneurographic recordings of postganglionic sympathetic nerve activity, or assessment of regional and body norepinephrine spillover, via isotope dilution. 7 An important implicit assumption underlying treatment with RDN intervention is that central sympathetic hyperactivity is transmitted to the kidneys through intact postganglionic sympathetic nerve fibers. If transmission is compromised, then RDN will be ineffective as denervation is being performed on peripheral sympathetic nerve fibers with an activity that has already been reduced.
A question that arises is whether there are patients who are more likely to respond to RDN than others and, if so, what would characterize them. Since the target of RDN is sympathetic nerve fibers of the renal arteries, responding to the intervention would require their contribution to the elevated BP, obviously requiring their functionality. Otherwise, RDN might only have a mild decreasing effect on BP or none at all.
Therefore, in this analysis, we addressed the question whether preexisting reduction in sympathetic activity is associated with reduced response to RDN.
Subjects and methods
This article presents the results of a retrospective subanalysis of data collected in a prospective, exploratory cohort study examining the change in autonomic function after RDN. This subanalysis addressed the question whether patients with detectable preexisting reduction in SNS function responded less to RDN.
Participants
Inclusion criteria were as follows: written and informed consent for the procedure and participation in neurophysiologic examination was required. Participants had to be at least 18 years old.
Exclusion criteria were atrial fibrillation, neuromuscular disorders, chronic inflammatory or malignant diseases, severe psychiatric diseases such as schizophrenia, borderline disorders or current depression, untreated thyroid dysfunction, drug or alcohol abuse, pregnancy and lactation, concomitant treatment with any antipsychotic or chemotherapeutic agents as well as non-compliance and concurrent participation in any other trial.
The study was approved by the Ethics Committee of the Faculty of Medicine of the Leipzig University, and the trial was conducted in accordance with the Declaration of Helsinki. Following publication of the Simplicity HTN-3 study, the Ethics Committee requested stopping the trial before enrollment was complete. Furthermore, the Ethics Committee denied approval of new prospective studies on RDN. Therefore, we were left to perform the analysis on the subset of patients who had already been enrolled.
Intervention
The RDN procedure was performed by four interventionalists (three at the University Heart Centre and one at the Department of Cardiology, Leipzig University) as previously described. 8 Briefly, an ablation catheter was inserted in the renal arteries near the hilus via puncture of the femoral artery. Gradually pulling the catheter in a helical fashion back toward the abdominal aorta, the electrode at the catheter tip was repeatedly placed on the arterial wall and heat was applied via radiofrequency. Per artery, three to eight ablation points were set depending on vessel size and the catheter. If there were duplicate renal arteries on one side, both arteries were treated, so long as the vessel size was adequate. Patients who were treated at the Leipzig University Heart Center (n = 16) underwent the procedure with 6F Simplicity Flex™ catheter (Medtronic, Santa Rosa, CA, USA); those treated in the Department of Cardiology and Angiology of University Hospital Leipzig (n = 5) were denervated by 8F EnligHTN™ (St. Jude Medical, Saint Paul, MN, USA).
Assessment of BP
To assess BP levels, we analyzed means of daytime BP values obtained in 24-h ambulatory BP measurements. These were performed at the Leipzig University Heart Centre and the Department of Cardiology and Angiology of the University Hospital Leipzig before, as well as 1 (n = 5) or 3 months (n = 16) after, RDN. In two of five patients treated at the Leipzig University Hospital, we received pre- or post-interventional measurements via referring cardiologists. For the remaining three patients, random ambulatory measurements as pre-interventional values were provided by their primary care physicians. To avoid missing values in a small cohort, we calculated means of values recorded in a 6-month period prior to intervention. In accordance with the results by Conen et al., 9 we corrected the values by −5 mmHg in two patients age 60–70 years and −13 mmHg in one patient age 76 years.
Autonomic nervous system function
For assessment of autonomic nervous system (ANS) function (SNS and parasympathetic nervous system (PNS) function), the following three methods were applied, as previously described. 10
Heart rate variability
Cardiac autonomic function was determined by heart rate variability (HRV) analysis via the computer-based system ProScicard (Medset Medizintechnik GmbH, Hamburg, Germany).
Analysis of HRV at rest was performed more than 5 min in supine position. As previously described in more detail, the following time- and frequency-domain indices of HRV were calculated: root mean square of successive differences (RMSSD), coefficient of variation (CoV) and power spectrum analysis in the very low frequency spectrum: 0.01–0.05 Hz (ln(VLF)), low frequency spectrum: 0.05–0.15 Hz (ln(LF)) and high frequency spectrum: 0.15–0.5 Hz (ln(HF)) as well as the low frequency/high frequency ratio (ln(LF/HF)).10–12
Additionally, cardiovascular function tests were performed: HRV after actively standing up was tested (“maneuver,” defined as ratio of the longest R-R interval and the shortest interval (30/15 ratio)). With the patient sitting, HRV under deep breathing at 6 breaths/min was recorded. The ratio of the longest R-R interval during expiration to the shortest interval during inspiration (E/I ratio) was calculated.
“Valsalva’s maneuver” was performed maintaining a pressure for 10 s. The Valsalva’s ratio, defined as the highest heart rate during the maneuver divided by the lowest rate within 30 s after the end of the maneuver, was calculated. 13 Normal values of all HRV parameters refer to the values described by Ziegler et al. 12 which accord to unpublished normative data obtained in our own laboratory.
Quantitative pupillography
This was measured using a pupillograph (AMTech Pupilknowlogy GmbH, Weinheim, Germany). Pupil diameter in darkness (PDD) was assessed, and pupillary light reflex was measured over a period of 2 s. Afterward, relative light reflex amplitude (A), latency (L), constriction velocity (CV) and early re-dilation velocity (RDV) were determined as previously described. 10 The mean value of two stable measurements of left or right eye was used for further analyses. 14 Normal values used as reference were taken from the trial of Keller. 15
Sympathetic skin response
For assessment of sympathetic skin response (SSR), all patients were examined in a silent room in supine position with their eyes closed. Following a short rectangular electric stimulus (10–20 mA, 0.1 ms duration) over the glabella, SSR was recorded with surface electrodes (palmar and plantar) with a dedicated device (Keypoint, Nautus Europe GmbH, München, Germany). 10 The test was considered abnormal if an increase in potential could not be distinguished from the signal noise of baseline within 3.5 s of the electric stimulation.
To assess SNS function, the following parameters were considered: ln(VLF) and ln(LF) of HRV, PDD and presence or absence of the SSR in upper (SSRu) and lower (SSRl) extremity. These parameters have documented validity in assessing sympathetic fibers.11,14,16–19 SNS function was regarded as reduced if at least one of the following parameter was abnormal: ln(LF) or ln(VLF) of HRV, PDD of pupillography or if SSR was absent. Participants whose results were consistent with this definition were assigned to group 1 (reduced SNS activity). All remaining patients were assigned to group 2 (normal or enhanced SNS activity). Of note, the tests used are not qualified to distinguish between normal sympathetic activity and sympathetic overdrive.
PNS function was assessed by the following parameters: RMSSD, CoV, ln(HF), 30/15 ratio, E/I ratio, Valsalva’s ratio, latency, amplitude and CV of the pupillary light reflex. PNS function was not considered for group assignment.
Statistics
Because of low expected counts, Fisher’s test was used for contingency tables. Given the small sample size, a group comparison was performed using the exact Wilcoxon–Mann–Whitney test. There were too few females to include sex in the linear models, but age and baseline values as a covariate were used in linear models and terms were dropped from a nested model based on Akaike’s information criterion. The choice of this analysis strategy follows recommendations for pre- and post-treatment scenarios, in which adjustment for the baseline values results in a gain of statistical power, 20 and the choice of a small number of covariates is in line with general recommendations, for example, of the European Medicines Agency. 21 Tests were considered significant for p < 0.05.
Results
Participants
A total of 34 patients were included in the study, and 13 participants were excluded or did not return for follow-ups due to atrial fibrillation (n = 3), withdrawal of consent (n = 3), disability to attend follow-up due to an aneurysm at the puncture site requiring therapy (n = 1), non-standardized intervention such as unilateral denervation or use of a catheter other than Simplicity Flex™ or EnligHTN™ (n = 3), non-compliance (n = 2) and migration (n = 1).
We analyzed all 21 patients who completed the follow-ups of the original trial. The initial flow of participants is illustrated in Figure 1. The baseline characteristics of the study cohort are presented in Table 1.

Flow of participants between baseline and follow-ups. Baseline: T0 (5 h to 6 days before RDN); follow-ups: T1 (1–5 days after RDN), T2 (1 month after RDN), T3 (6 months after RDN).
Baseline characteristics.
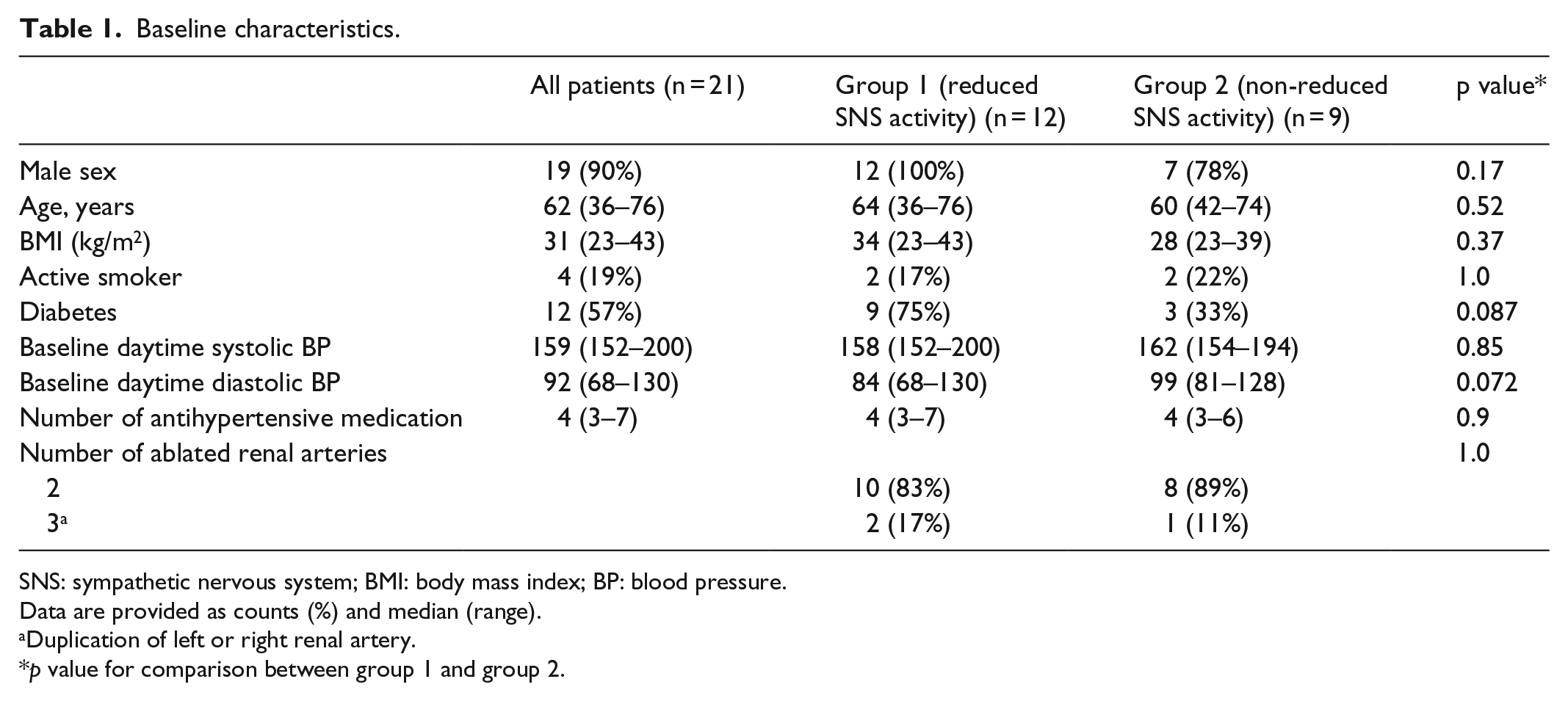
SNS: sympathetic nervous system; BMI: body mass index; BP: blood pressure.
Data are provided as counts (%) and median (range).
Duplication of left or right renal artery.
p value for comparison between group 1 and group 2.
ANS function
An overview of abnormal ANS parameters and BP reductions in all participants is displayed in Table 2. SNS activity was reduced in 12 patients who were assigned to group 1. In 6 of the 12 patients of group 1, reduced SNS activity was detected by pupillography, in 1 by HRV analysis and in 2 by SSR. The remaining three patients had various combinations. Eight of the 12 participants with reduced SNS activity also had reduced PNS activity.
Abnormal SNS, PNS parameters and BP reduction in all participants.
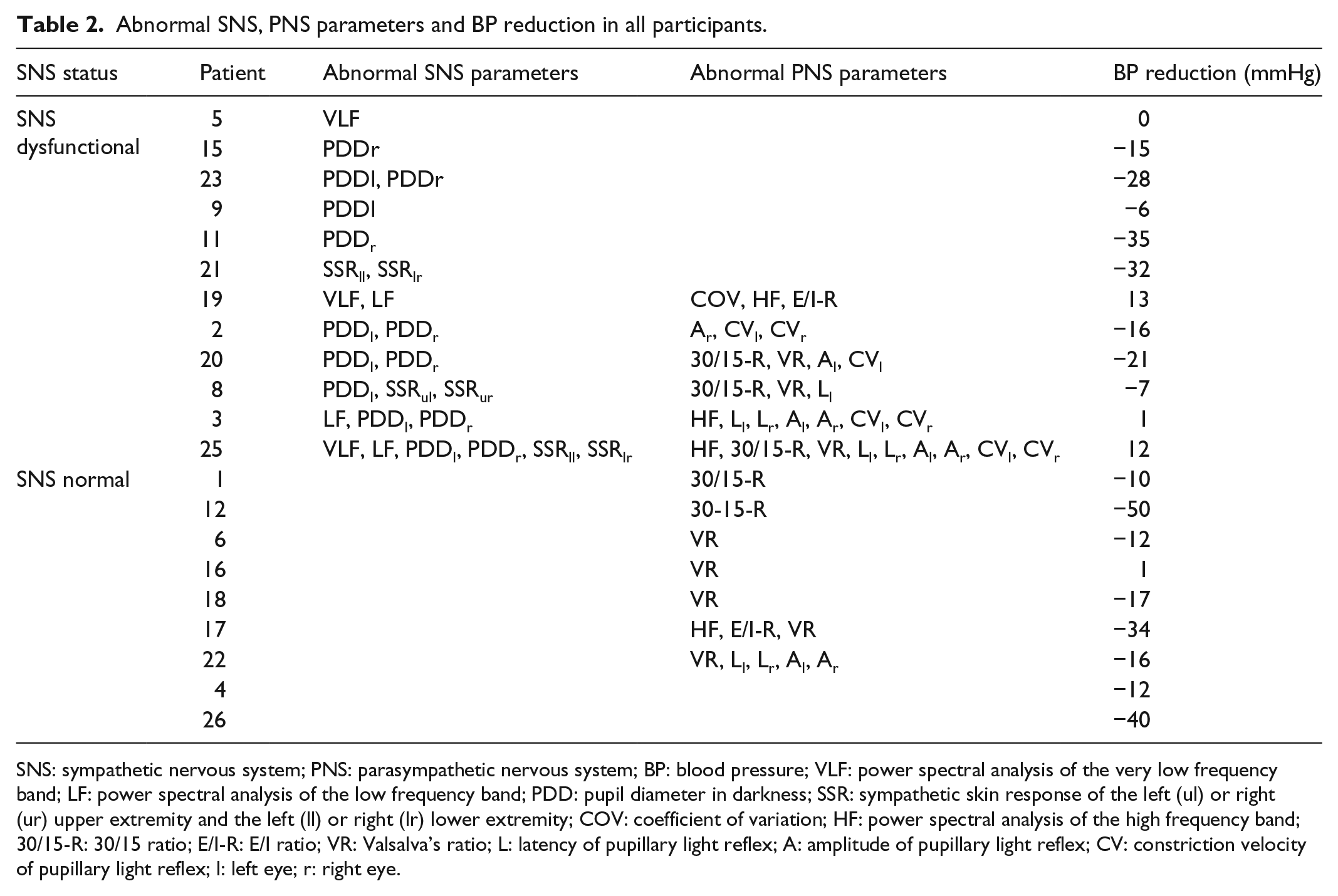
SNS: sympathetic nervous system; PNS: parasympathetic nervous system; BP: blood pressure; VLF: power spectral analysis of the very low frequency band; LF: power spectral analysis of the low frequency band; PDD: pupil diameter in darkness; SSR: sympathetic skin response of the left (ul) or right (ur) upper extremity and the left (ll) or right (lr) lower extremity; COV: coefficient of variation; HF: power spectral analysis of the high frequency band; 30/15-R: 30/15 ratio; E/I-R: E/I ratio; VR: Valsalva’s ratio; L: latency of pupillary light reflex; A: amplitude of pupillary light reflex; CV: constriction velocity of pupillary light reflex; l: left eye; r: right eye.
BP
Across all participants, mean reduction (95% confidence interval (CI)) in systolic daytime BP was 15.4 mmHg; CI (7.8, 23.0) mmHg, p < 0.001).
At baseline, BP correlated negatively with age (r = −0.62; CI (−0.83, −0.26), p = 0.003). However, in a linear model with BP at follow-up as the dependent variable and age, the baseline value of BP and the SNS group as independent variables, age was no longer relevant and was dropped from the model. In this simplified model, patients of group 2 had their BP lowered by 11.3 mmHg (CI (0.3, 22.3) mmHg, p = 0.045) more than those of group 1 (reduced SNS activity). Eight of the nine (89%) patients in group 2 had their BP lowered by at least 5 mmHg compared to 8 of 12 (75%) patients in group 1, a non-significant difference, p = 0.34 (Figure 2). BP was lowered by 10 mmHg in eight of nine (89%) patients in group 2 versus 6 of 12 (50%) patients in group 1, p = 0.16.

Daytime SBP at baseline and follow-up plotted for patients with reduced and non-reduced SNS activity. The dashed line indicates a reduction in BP by 5 mmHg.
Discussion
This study generates the hypothesis that patients with preexisting reduced sympathetic activity displayed a lower reduction in BP than patients with non-reduced sympathetic nerve activity. The mean BP reduction exceeded 20 mmHg in the group exhibiting non-reduced SNS function and was 11 mmHg greater than in the group with reduced SNS activity.
Due to the small sample size and because there was no group undergoing a control (e.g. sham) intervention, we cannot draw any safe conclusions about the presence and magnitude of a BP-reducing effect of RDN. The reduction in BP by on average 15.4 mmHg seems to be somewhat larger than observed in the HTN-3 trial which revealed reductions of 6.75 mmHg in 24-h average systolic blood pressure (SBP) after 6 months. 3 However, these results cannot be compared directly with our findings, as we used the daytime average of the 24-h measurement. Our data are consistent with the HTN-1 and -2 trials which showed a substantial BP reduction in office-based BP measurement 1 month (14/10 mmHg) and 6 months (32/12 mmHg) after RDN, respectively, which endured or even increased over the course of further follow-up.1,2,22,23 Finally, the reduction found in this study is of the same order of magnitude as that reported by Azizi et al., 4 who recorded a reduction in daytime ambulatory SBP by 15.8 mmHg after 6 months in the RDN group. The presence of BP reduction in this magnitude may have enhanced chances of detecting a modulating effect of SNS function.
We found that response to RDN was strongly correlated to baseline SBP, with greater reduction after RDN being associated with higher baseline BP values. Again, this is in line with previous studies employing office SBP recordings or ambulatory SBP measurements.24–26 Supporting these findings, a recent randomized controlled trial on patients with only mild hypertension revealed no significant BP reduction 6 months after RDN. 8 Baseline SBP values between the reduced and non-reduced SNS activity groups were similar, rendering it highly unlikely that BP differences between groups have confounded the results. In contrast to SBP, baseline diastolic BP (DBP) is unrelated to BP response to RDN. 24 Therefore, the fact that DBP tended to be higher in the group with reduced SNS activity is unlikely to have confounded our results.
The discordant value of one patient revealing a BP reduction of −50 mmHg is hard to explain and can be due to several unconsidered variables. Discordant values of similar magnitude have also been seen in a controlled trial. 4 Strikingly, this patient does not reveal any reduced sympathetic function in the subtests conducted, which could lead one to the assumption that this patient, in contrast to others, might have had sympathetic overactivity prior to the intervention. This remains speculative, and a physiological or an undetected reduced function remains a possibility. Removing this value in the spirit of a sensitivity analysis results in a group difference of 9.0 mmHg (−2.0 to 20 mmHg, p = 0.10).
Abnormal results of HRV analysis, pupillography and SSR recordings indicate the presence of peripheral autonomic neuropathy.12,19,27,28 Our results, therefore, suggest a lower responsiveness in patients with preexisting autonomic sympathetic neuropathy. Autonomic neuropathy, characterized by damage of thinly myelinated A-delta fibers of small bore and non-myelinated C-fibers, is a frequent complication of diabetes mellitus.29–31 In line with this, we found that 9 of 12 patients (75%) in whom SNS function was reduced, but only 3 (33%) of 9 patients in whom SNS function was not reduced, suffered from diabetes. The implication is that diabetes may reduce responsiveness to RDN by virtue of its association with autonomic sympathetic neuropathy. However, peripheral autonomic neuropathy can also develop in individuals with merely impaired glucose tolerance, well before diabetes becomes manifest clinically and as a consequence of obesity, in the presence of normal glucose metabolism.10,32
Of note, 44% of the HTN-3 trial participants were diabetics, and the mean body mass index (BMI) was 34 kg/m2, corresponding to class I obesity according to the World Health Organization (WHO) classification of obesity.3,33 Given the high prevalence of reduced peripheral SNS activity in diabetes and obesity, it was likely present in a large group of HTN-3 participants and, hence, may have contributed to therapeutic failure in HTN-3 trial. Consequently, it might be beneficial to investigate obese and diabetic patients for reduced peripheral sympathetic activity carefully.
The parameters included in this work have documented sensitivity to reduced function of postganglionic sympathetic nerve fibers.11,19,29
Measurements of HRV used in this study are well established and can be used to detect cardiac autonomic denervation seen in early diabetic neuropathy.12,27 We used spectral power in low- and very low frequency bands of HRV as markers with a proven sympathetic component.11,16 According to the current studies, it still remains uncertain to what extent SNS and PNS contribute to LF oscillations. 11 However, both parameters were shown to be of clinical importance: Whereas reduced power in all spectral bands, including LF and VLF, characterizes diabetic autonomic neuropathy, VLF was shown to be a predictive parameter for survival after myocardial infarction. 11 The PDD, in contrast, has been proven to be dominantly mediated by the sympathetic nerves.18,17 Furthermore, several studies report on reduced PDD under various pathological conditions directly affecting the sympathetic fibers.29,34,35 Likewise, SSR is altered in several diseases with SNS dysfunction. 19
Group allocation was based on a comprehensive physiological evaluation of SNS function. However, our inclusion criteria specified that a small number of abnormalities were sufficient to assign patients to the group with reduced SNS activity. Although there is substantial cross-correlation between the results of tests of SNS function (e.g. association between HRV and pupillography results and between HRV and SSR findings), the correlation to renal sympathetic nerve function, if any, is unknown.28,36 Interestingly, among five non-responders, four of them had abnormal ln(LF) and ln(VLF) in HRV analysis, whereas pupillomotor abnormalities or absent SSR alone were not associated with RDN failure. This may suggest that reduced activity of cardiosympathetic fibers has special significance as a marker for poor RDN responsiveness. However, as our trial was not statistically powered to detect correlations between RDN response and single parameters or their combinations, the question of whether individual tests may have superior predictive capacity remains to be addressed in future studies.
Limitations
This study suffers from some limitations. First, data analysis was retrospective. Therefore, assessment of 24-h ambulatory BP was not available for all patients. In three patients, either pre- or post-interventional 24-h ambulatory measurements could not be obtained and had to be replaced by corrected random ambulatory measurements. 9 The impact of the random ambulatory data was examined in a sensitivity analysis. Excluding those three patients, one finds a slightly larger BP reduction between groups 1 and 2 of 13.9 mmHg (3.1–24.6 mmHg, p = 0.015). Furthermore, kidney function remained unconsidered.
Second, the interval between intervention and assessment of post-interventional BP reduction was short lasting only between 1 (n = 5) and 3 months (n = 16). Hence, the existence of possible transient effects cannot be ruled out. However, as BP-reducing effects of RDN are likely to remain stable or even increase in the long-term, this limitation is unlikely to affect our main conclusion.22,23
Third, the assignment to the groups and, therefore, the evaluation of reduced function of the SNS was based on clinical electrophysiological tests providing only indirect information on sympathetic nerve fiber damage. Unfortunately, to our knowledge, there are no non-invasive means of measuring functionality of the sympathetic nerve fibers to the kidney. Whether reduced sympathetic output to heart, pupils and skin is indicative of reduced sympathetic output to the kidneys remains speculative. This is due to the lack of a method that can directly measure the function of renal sympathetic nerves in living human beings. Indirect measurements such as microneurographic recordings of postganglionic sympathetic nerve activity or measurement of norepinephrine spillover are invasive, require special expertise and hence cannot be applied on a broad scale. Therefore, electrophysiological tests are the best compromise available, currently.
Fourth, only two participants were female. A previous study did not report-gender specific differences in responsiveness to RDN, 3 suggesting that gender distribution is unlikely to have confounded our results.
Fifth, two different catheters have been employed. However, proof of principle studies, equal in patient numbers and baseline BP levels, revealed similar BP changes 6 months after procedure for both catheter systems.1,37 Indeed, the number of ablation points differed between patients due to individual anatomical conditions and tended to be lower if the Simplicity catheter was applied. We cannot rule out that this has confounded our results.
Sixth, the lack of a control group means we cannot rule out the possibility that the changes in BP and better response of the group without reduction in SNS activity were unrelated to the RDN therapy.
Finally, this study is based on a small number of cases. The small sample size means that the results are particularly sensitive to the analysis methods used. This in conjunction with the exploratory nature of the analysis implies that the group difference we observe should be interpreted as a reason to conduct further research and not as a demonstration of a physiological difference.
Future research
As mentioned above, we think that electrophysiological testing is the best choice we have to assess peripheral sympathetic nerve function in the clinical setting. This is why its possible role in predicting success of RDN should be further investigated.
Because a method powered to examine a correlation between our tests and actual function of renal sympathetic nerve fibers will be hard to find, future research should address the following points:
First, the authors may consider using the combination of parameters chosen in this trial, as they represent an easily applicable set of tests with reasonable physiological validity.
Second, it would be advisable to conduct trials with larger cohorts in advance to confirm normal values.
Third, it might be interesting, in preliminary stages, to examine the correlation between those tests and measurements of sympathetic overactivity such as noradrenaline spillover or microneurographic recordings of postganglionic sympathetic nerve activity.
Finally, a trial examining our thesis should consist of a randomized and controlled design with a larger sample size. Based on our data, about 160 patients would be needed for a trial with 90% power.
Conclusion
In conclusion, our exploratory findings provide evidence suggesting that peripheral reduction in sympathetic activity is associated with poorer response to RDN. To avoid ineffective procedures and detect contraindications, future studies should try to verify this hypothesis prospectively and to identify a combination of subtests that is both reliable and feasible.
Footnotes
Acknowledgements
The authors would like to thank Patrick Meyer, Anke Schimansky, Nadine Krekow and Christoph Böhler for the conduction of autonomic function tests as well as Ulrike Tetzlaff for her support in organization. Especially, they are thankful for the patients who made an effort to come to their department and spend their precious time undergoing the examinations.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.L. has received speakers’ fees from Medtronic. The remaining authors declare that there is no conflict of interest.
Ethical approval
Ethical approval for this study was obtained from the ethics committee of the medical faculty of the Leipzig University (012-12-07112011).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
