Abstract
Abomasal emptying defect (AED) is a disease syndrome that primarily affects Suffolk sheep and is characterized by distension and impaction of the abomasum. No histologic lesion has been consistently associated with this condition. There is no known etiology. In this study, nine cases of AED were identified by necropsy, including three rams and six ewes between 2 and 6 years of age. Four of the cases occurred sporadically, and five ewes were submitted on the same day from a single flock. Histologic examination of celiacomesenteric ganglia from six of the affected sheep revealed scattered chromatolytic or necrotic neurons, without inflammation. Chromatolytic neurons were observed more frequently in AED-affected sheep than in seven healthy Suffolk sheep (P < 0.08, weak statistical support). Neuronal necrosis was not observed in any of the healthy sheep. Lineage records of the flock that suffered an outbreak were incompatible with the possibility of a simple inheritance pattern for this disease; furthermore, the very occurrence of AED in outbreak form is inconsistent with transmission solely by inheritance. Only one of the six tested sheep showed concurrent immunohistochemical evidence of scrapie. The lesion pattern in celiacomesenteric ganglia is suggestive of a neurotoxicosis. Neuronal lesions of AED resemble dysautonomic diseases of humans and other animals.
Keywords
Abomasal emptying defect (AED), 1,13 abomasal impaction, 5 and abomasal dilatation and emptying defect 6,14 are terms that have been used to describe a disease syndrome of sheep. The defining feature is a markedly distended and impacted abomasum. Despite this characteristic gross lesion, no consistent histologic lesions have been described. The most frequently described clinical signs of AED are anorexia, progressive weight loss, and distension of the right ventral abdomen. Elevated ruminal chloride concentrations have been reported, presumably secondary to abomasal reflux. 13 Tachycardia and atrial fibrillation have also been observed in cases of AED. 1,5,6,8,14 The clinical duration of the disease ranges from days to months, and the prognosis is grave. 14
Causes of AED are not known. Thirty-nine of 43 cases reported in published articles occurred in Suffolk sheep of both sexes. 1,5,6,8,13,14,16,21 AED also has been reported in two Hampshire, 21 one Dorset, 1 and one Texel sheep. 16 Most cases of AED occur sporadically, 5,8,13,16,21 although there are some references to endemically affected flocks. 1,6 A single outbreak was reported previously in a flock of 92 Suffolk sheep, in which 14 ewes were diagnosed with AED during a 5-month period; 14 a lineage analysis of that flock did not reveal a simple inheritance pattern. 14 AED was reported in two sheep with scrapie; 16 however, scrapie has not been found in other reported cases of AED. 1,13,14
In this study, we describe a histologic lesion in the celiacomesenteric ganglia of AED-affected sheep. In addition, an outbreak of AED is documented in a flock of Suffolk ewes.
Materials and Methods
Pathology
Six cases of AED were diagnosed based on gross lesions found in sheep submitted to the Wyoming State Veterinary Laboratory (WSVL) between July 1993 and December 1996. An additional three cases that were examined by the Colorado Veterinary Diagnostic Laboratory (CVDL) were included in this study because these three sheep were submitted on the same day from the same flock as were two other sheep that were submitted to the WSVL. Specimens of cerebral cortex, thalamus, midbrain, medulla oblongata, and multiple levels of the gastrointestinal tract were collected for histologic examination.
In sheep, the celiac and mesenteric ganglia are commonly fused into a single celiacomesenteric ganglionic plexus. Celiacomesenteric ganglia were collected for histologic examination from the six sheep examined at the WSVL. The ganglion is normally quite small, and attempts to locate it in AED-affected sheep were complicated by its apparent atrophy. Dissection of the ganglion was facilitated by removing en bloc a section of the aorta that included the base of the celiac and cranial mesenteric arteries, fixing this tissue in 10% formalin for 1 day, and then performing further dissection on a benchtop. Any firm or fibrous structure that could be palpated or observed within the tissues surrounding the base of the celiac and mesenteric arteries was placed within one or more tissue cassettes. If the ganglion could not be positively identified grossly, then the connective tissues surrounding the base of those arteries were sliced into pieces and divided into tissue cassettes. Celiacomesenteric ganglia were also collected from seven healthy adult Suffolk sheep that were part of an unrelated study, to serve as negative controls.
All histologic specimens were fixed in 10% buffered formalin, processed through graded alcohols and xylene, embedded in paraffin, sectioned 5 µm thick, and stained with hematoxylin (HE).
Immunohistochemistry
Because of a previous report of scrapie in two sheep with AED, 16 celiacomesenteric ganglia from affected sheep were stained immunohistochemically for PrPres, 22 using monoclonal antibody (MAb) F89/160.1.5. Only PrPres, the abnormal protease-resistant prion protein, is detected in this procedure because normal prion protein is destroyed by autoclaving. Dewaxed sections on adhesive-coated glass slides were placed in formic acid solution for 5 minutes, rinsed in water, immersed in an antigen retrieval solution (Dako), autoclaved for 20 minutes at 122 C (Biocare Medical), and cooled in antigen retrieval solution for 25 minutes. Sections were immunostained with the MAb for 32 minutes at 37 C using an automated machine (Ventana Medical Systems) followed by addition of biotinylated anti-mouse secondary antibody, a streptavidin–alkaline phosphatase conjugate, a stain enhancer followed by substrate chromogen (Fast Red A followed by Fast Red B), and a HE counterstain with bluing. Positive controls consisted of brain and tonsil from a sheep with scrapie.
Site visit
The ranch from which five sheep with AED were submitted (on 9 April 1996) was visited by the attending veterinarian (D. E. Christensen) on 28 March and by two of the investigators (D. E. Christensen and M. M. McAllister) on 14 April. Pastures, feedstuffs, water sources, and lambing records were examined, and the owners were interviewed.
Lineage analysis
Pedigree records of the above flock were examined to determine whether the affected sheep were closely related.
Examination of ganglionic neurons
In a blinded procedure, celiacomesenteric ganglia were examined histologically for neuronal pathology. Ganglia were categorized as abnormal if any chromatolytic or necrotic neurons were observed, or they were categorized as normal if no neuronal lesions were observed. A directional Fisher Exact Test 19 was used to compare results between the group of six affected sheep and the control group of seven healthy adult Suffolk sheep.
Results
Nine cases of AED were diagnosed by gross necropsy. Eight sheep were Suffolks, and one was a Suffolk-mix. There were six females and three males. The sheep were between 2 and 6 years of age, and clinical signs had been observed from 10 days to 8 months. All sheep had a history of anorexia and weight loss. The sheep with the longest duration of clinical signs had a history of regurgitation.
The affected sheep came from four flocks. Two flocks had one affected ram each. One ram and one ewe came from a flock of purebred Suffolks that had a history of endemic AED; these two animals were examined 22 months apart. Five affected ewes were submitted on the same day; these ewes came from a flock that was having an outbreak of disease.
Pathology
The affected sheep were in poor body condition. Abomasae were distended, extended caudally as far as the pelvic inlet (Figs. 1, 2), and were estimated to contain two to five times the normal volume of ingesta. Abomasal contents were weighed from two sheep and equaled 8.6 and 14 kg. Abomasal ingesta was dry, contained forage with long fiber lengths (which is abnormal in the abomasum), and often contained undigested whole corn and oats (Fig. 2). Greatly distended omaso-abomasal orifices, up to 12 × 14 cm in diameter, were observed in five of the six sheep that were necropsied at the WSVL (Fig. 3) and the sixth appeared normal (i.e., it was closed). Atrophy of ruminal papillae was noted in two sheep. The ram with the longest history of clinical illness had the most severe gross lesions, including an enlarged flaccid esophagus, an enlarged omasum, a distended reticulo-omasal orifice, and an enlarged impacted abomasum.
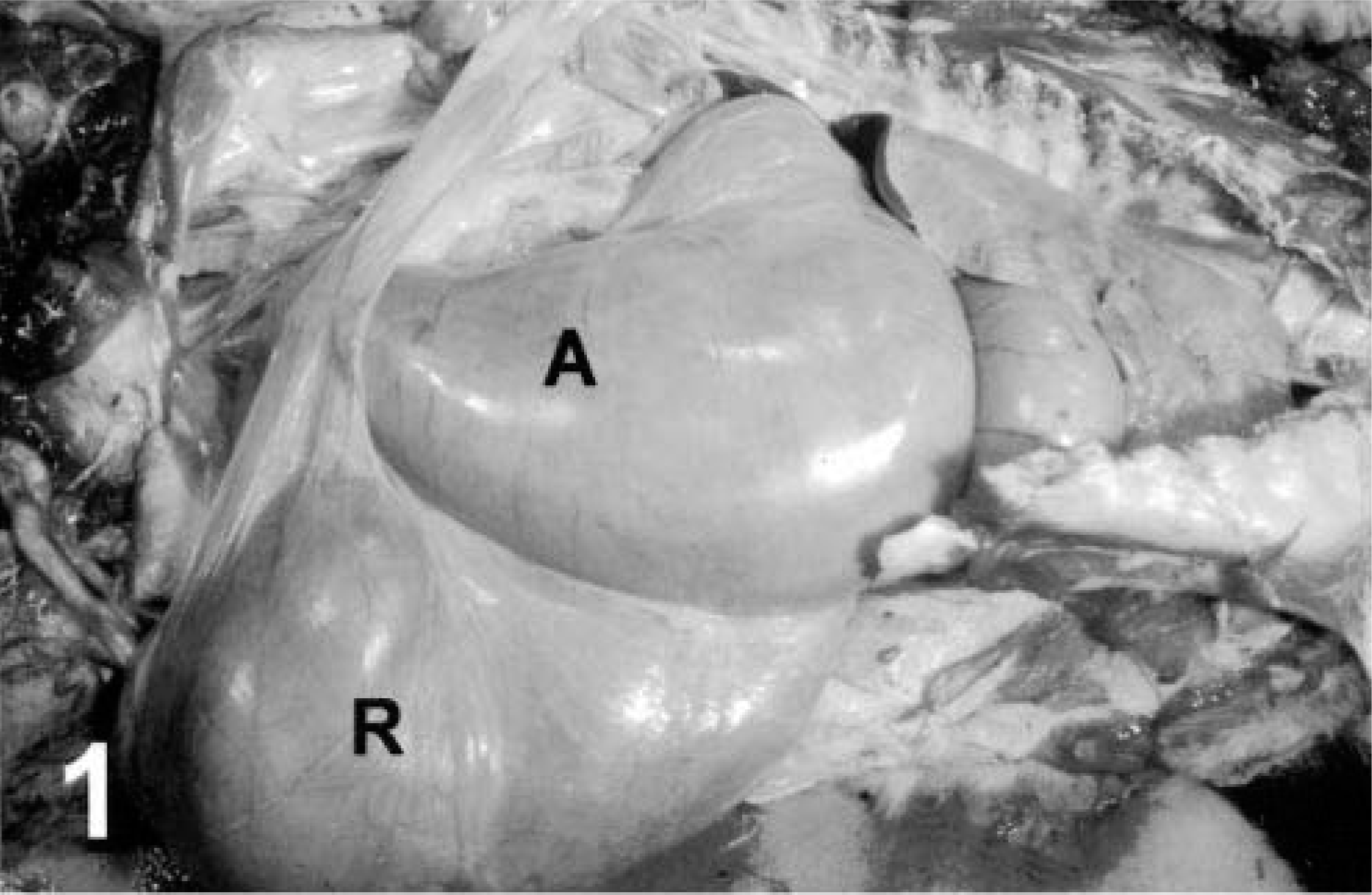
Abomasum, AED; Suffolk-cross sheep. Note the greatly distended abomasum (A), which is almost as large as the rumen (R).
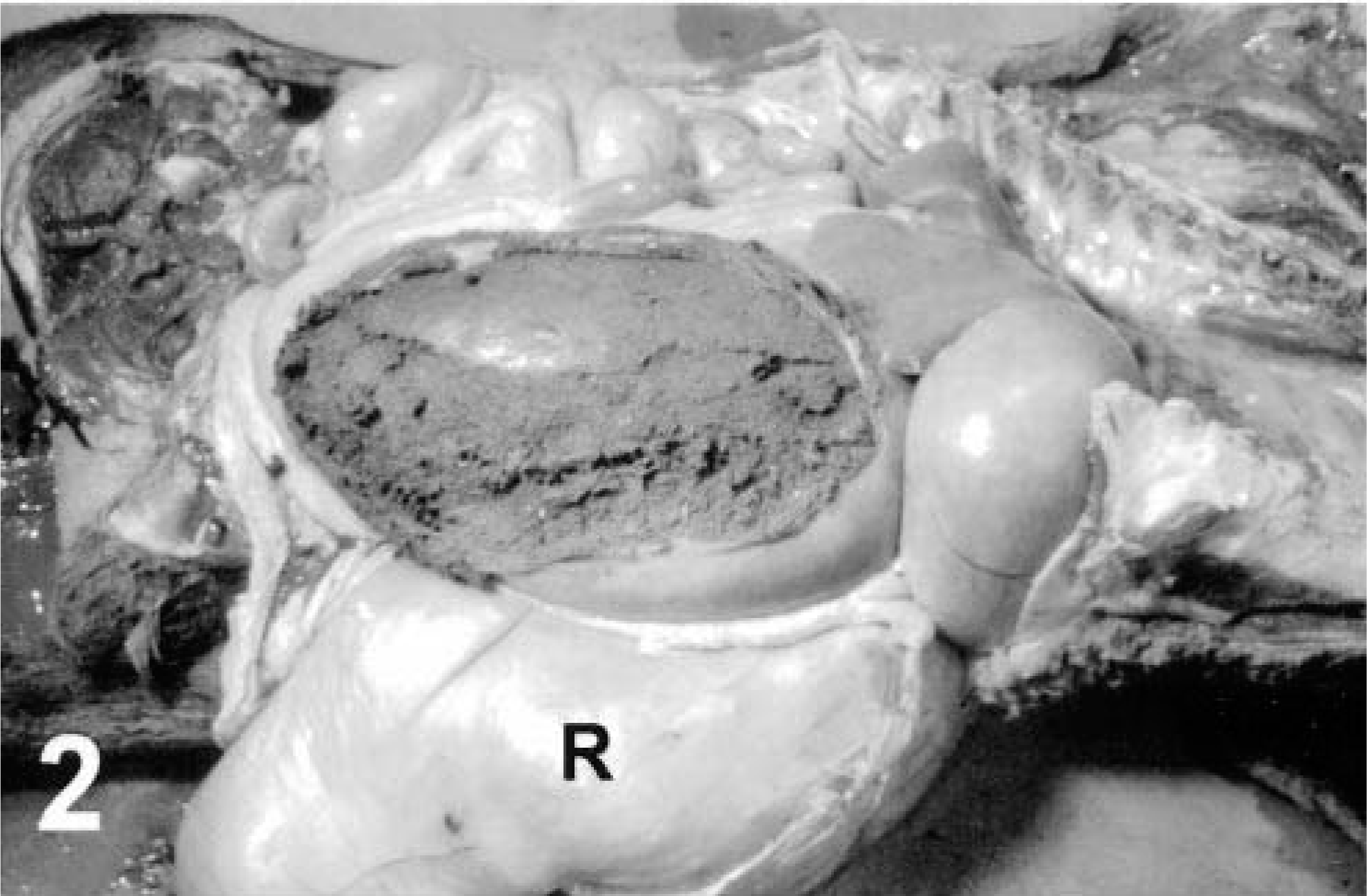
Abomasum, AED; purebred Suffolk sheep. The right wall of the greatly distended abomasum has been resected to reveal rumenlike ingesta that is densely compacted and dry. The abomasum extends caudally to the pelvic inlet and is about the size of the rumen (R).
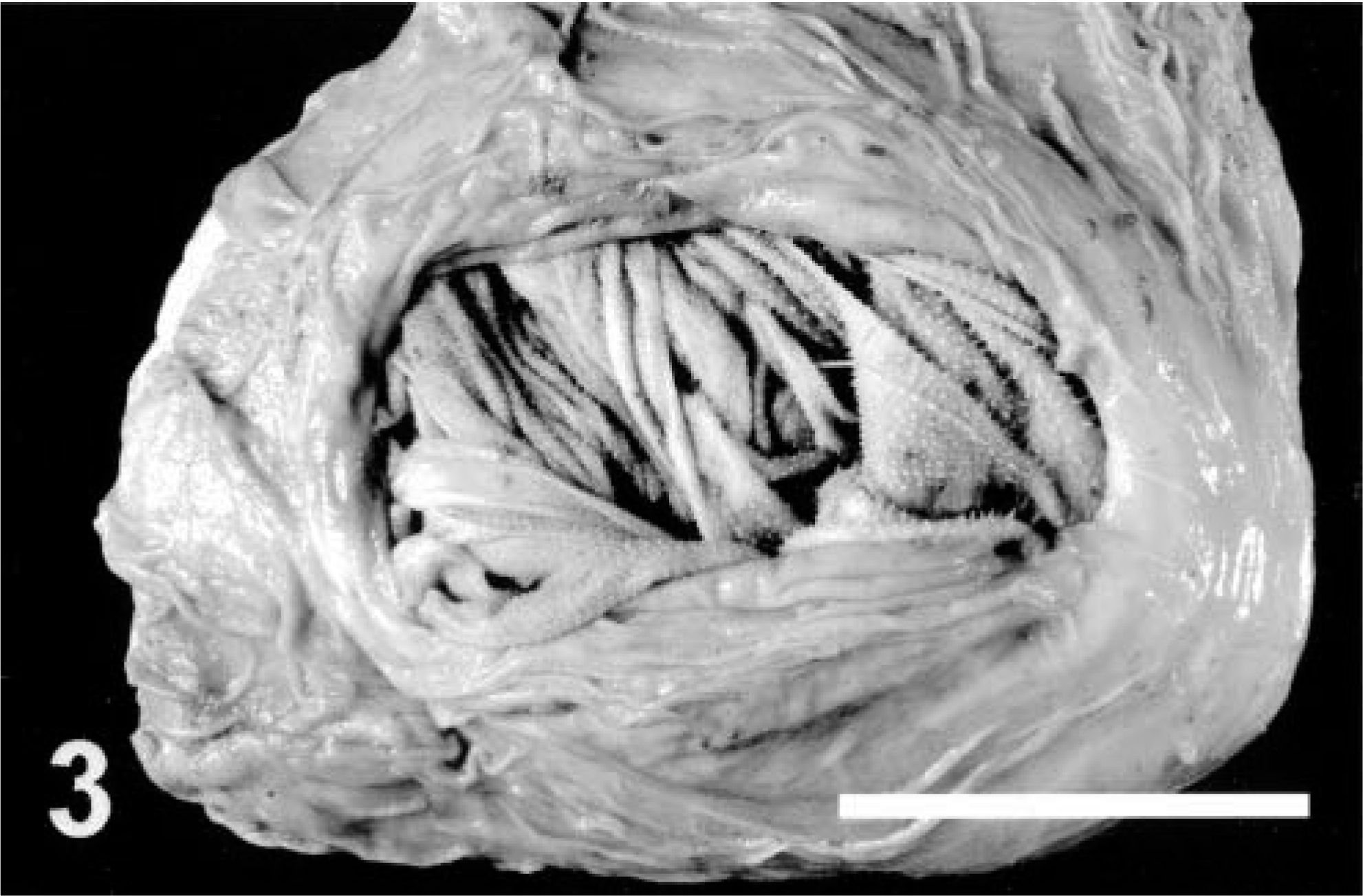
Abomasal mucosa with view of the omaso-abomasal orifice, AED; Suffolk sheep (same animal as in Fig. 2). Note dilatation of the orifice, revealing omasal leaves on the opposite side. Bar = 5 cm.
Histopathology of celiacomesenteric ganglia revealed scattered, individual, chromatolytic or necrotic neurons throughout the ganglia of the affected sheep, without leukocytic infiltration (Figs. 4, 5). Affected neurons were usually sparsely distributed, but the sheep with the longest history of illness and the most severe gross lesions had several affected neurons per 100× field and an apparent decreased density of neurons. Swelling of neuronal processes was not observed within ganglia and nerve fibers of the autonomic plexus. Fine vacuolation of neurons also was observed in a single case that also had immunohistochemical evidence of scrapie.
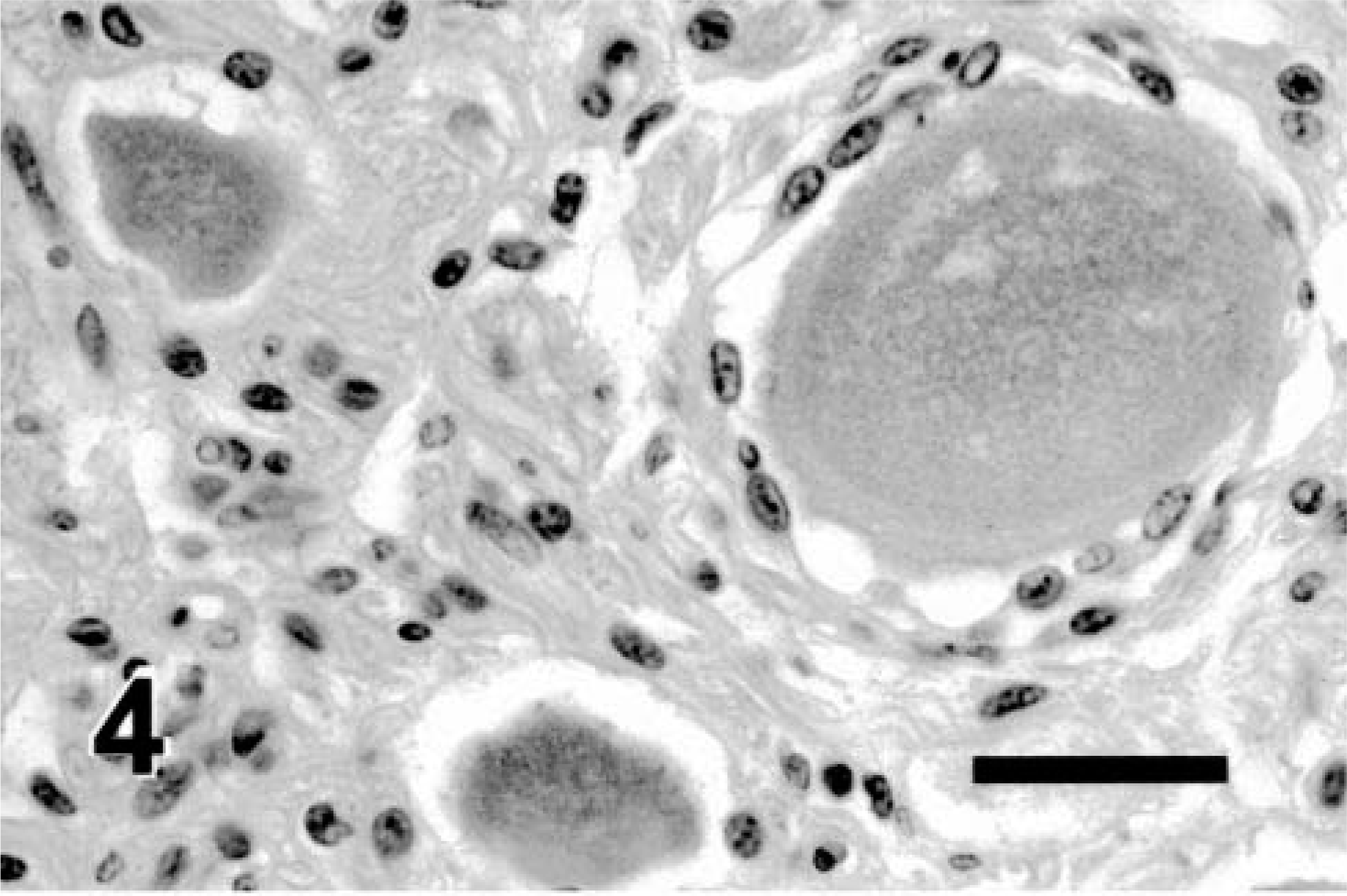
Celiacomesenteric ganglion, AED; Suffolk sheep. Note the swollen neuron with pale cytoplasm and lack of visible Nissl substance, characteristic of chromatolytic degeneration. HE. Bar = 50 µm.
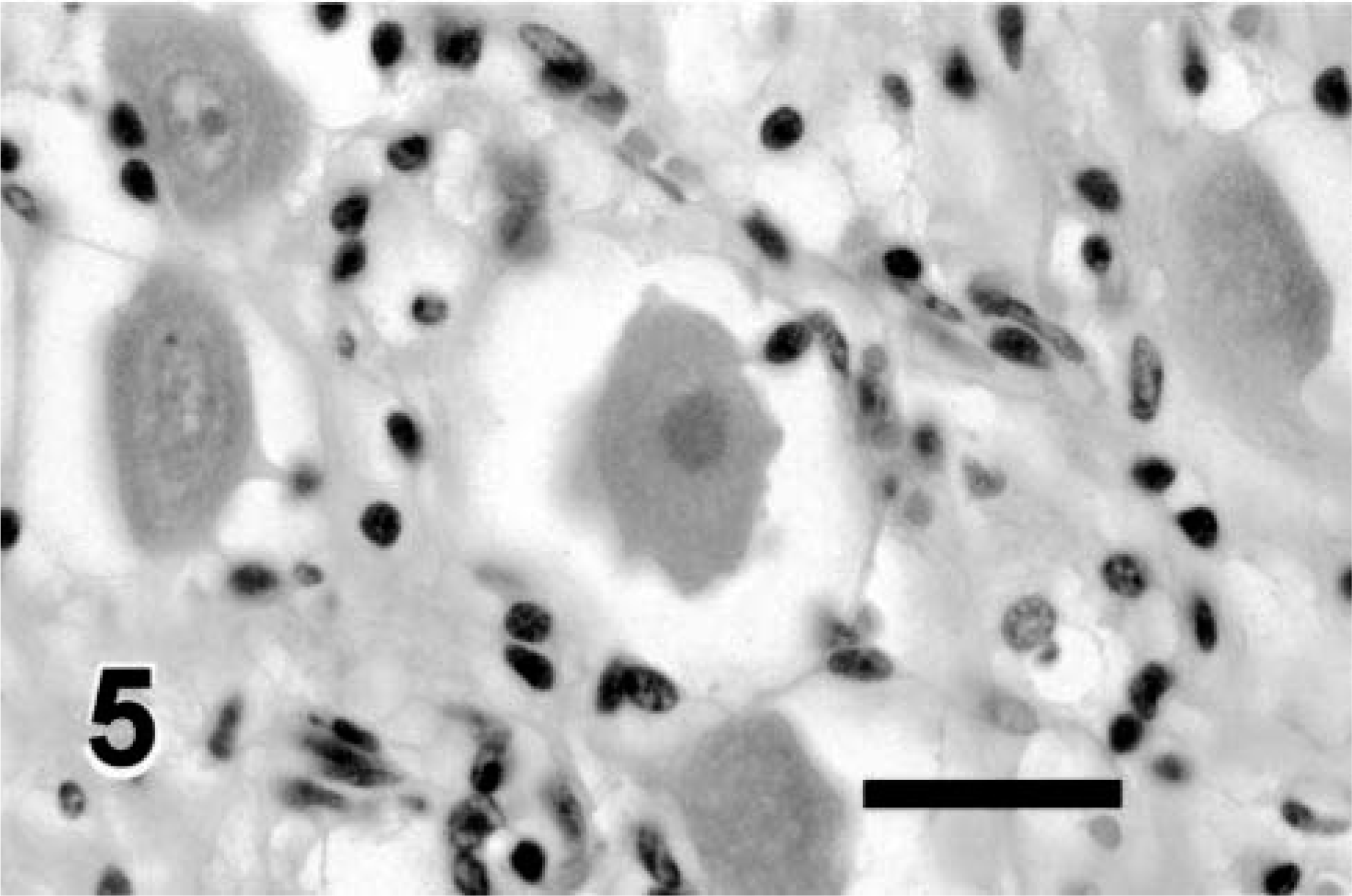
Celiacomesenteric ganglion, AED; Suffolk sheep. Note the neuron with karyolysis, homogeneous cytoplasm, and crinkled cell border, indicative of necrosis. The cytoplasm was deeply eosinophilic. HE. Bar = 50 µm.
Multiple levels of brain were examined histologically from eight sheep. Four animals had no detectable lesions, one had a mild to minimal, focal lymphocytic meningitis in the midbrain, and two cases had mild to minimal, multifocal perivascular cuffing, which consisted of small clusters of lymphocytes, plasma cells, and macrophages adjacent to occasional vessels. Equivocal microscopic lesions consistent with scrapie were observed in one animal, consisting of neuronal vacuolation in a paraventricular nucleus in the thalamus; this was the same sheep that was immunohistologically positive for PrPres in the celiacomesenteric ganglion. Vagal nerves from two sheep were examined and appeared normal.
The gastrointestinal tracts from all nine affected sheep were examined histologically. Seven cases lacked obvious histologic lesions in the gastrointestinal tract. One sheep had multifocal, moderate to dense infiltrates of lymphocytes in the submucosa or lamina propria at the level of the lower esophageal sphincter, rumeno-reticulum, omasum, omaso-abomasal junction, and abomasum. In this case, some neurons within small submucosal ganglia exhibited cytoplasmic eosinophilia suggestive of chromatolysis. In one sheep, no myenteric or submucosal ganglia were observed throughout the abomasum or omaso-abomasal junction (severe numerical atrophy), although myenteric and submucosal ganglia were present in the small intestine and colon. Also, in this sheep, there were clusters of lymphocytes and plasma cells within the mucosa and submucosa of the abomasum, scattered smooth muscle cells in the abomasum had pale vacuolated cytoplasm, and the omaso-abomasal fold had a focus of smooth muscle necrosis.
Immunohistochemistry for scrapie
There was positive immunohistochemical staining of PrPres in the celiacomesenteric ganglion of one of the six sheep that were examined. Approximately 5% of ganglionic cells in the celiacomesenteric ganglion of this sheep had discrete punctate areas of positive staining in satellite cells or on the plasmalemma of neurons, or both. Following this result, samples of colon and medulla oblongata from this sheep also were tested for PrPres. There was positive staining in gut-associated lymphoid tissue, but results in the medulla were negative.
Lineage analysis
Pedigree records were examined from the flock that had an outbreak of AED. Eighteen of 160 ewes that had recently lambed (11%) died between 26 February and 9 April after exhibiting anorexia and weight loss. On 9 April, two of these ewes (one dead and one alive) were submitted to the WSVL and three ewes (one dead and two alive) were submitted to the CVDL and at necropsy were diagnosed with AED. These five ewes had five different dams and five different sires and had been purchased at various times from three different flocks. Pedigree records were examined from 17 of the ewes that died (including the five diagnosed with AED by necropsy), and no evidence of inbreeding was found. These 17 ewes had 16 different dams, 11 different sires, and were either born into the flock or purchased at various times from four other flocks. These 17 ewes were between 2 and 7 years of age.
Site visit
The ranch suffering the disease outbreak was visited by the attending veterinarian on 28 March and by two of the investigators on 14 April (5 days after submission of five ewes for necropsy). These site visits did not identify an obvious cause of the disease outbreak. The flock consisted of 200 ewes, which were mostly Suffolks but also included a few Hampshires. Ewes were pregnancy examined in December 1995 using ultrasound. Forty ewes were open (not pregnant), and 160 ewes were pregnant. Sheep were fed native grass hay and received a mineral-protein-urea supplement until 1 January 1996. At that time, pregnant ewes were fed pasture grass supplemented with mixed alfalfa and grass hay and 0.45 kg of shell corn per day. Approximately one-third of the ewes were corralled on 10 January, in preparation for lambing, and the remaining pregnant ewes were corralled as their lambing dates approached. Corralled ewes were fed second-cutting alfalfa hay cut to a 2.5 cm length, 0.45 kg cracked corn, and a molasses-based mineral supplement that did not contain copper or urea. After lambing, the corn ration was doubled. There was no history of water deprivation.
According to the owner's records, ewes became ill between 3 days and 3 weeks after lambing. Affected ewes were anorexic and died after 10 days to 3 weeks of illness. The owner believed that the ewes that were the most aggressive eaters were the ones that became affected. None of the open ewes became ill (these ewes had remained on pasture and were not fed chopped alfalfa or corn). The corn and chopped alfalfa hay that were fed to the ewes had been consumed before the 14 April site visit and, therefore, were not available for analysis.
The owners stated that the flock had suffered several vaginal and rectal prolapses in December. Ewes had been treated with ivermectin and Clostridium perfringens C&D toxoid on 20 December.
Statistical comparison of ganglionic lesions
In a blinded fashion, celiacomesenteric ganglia were examined from the six cases submitted to the WSVL and compared with ganglia collected from seven healthy adult Suffolk sheep. There was weak statistical evidence (P < 0.08) that neuronal lesions occurred more frequently in sheep with AED. Rare, mildly chromatolytic neurons were observed in two of the healthy control sheep, which, therefore, had been classified as abnormal for the purpose of statistical testing. Neuronal necrosis was not observed in any of the control sheep.
Discussion
Although most cases of AED are sporadic, 5,8,13,16,21 this investigation includes the second documented account of a disease outbreak in a flock of ewes. 14 The occurrence of multiple cases of AED in a flock within a 1-month period and the lack of a close familial relationship among affected ewes in that outbreak indicate that AED is not a strictly inheritable trait. In addition, the wide age range of affected sheep in this and other studies (at least 2–6 years in this study) is not a typical finding of inherited diseases. Nevertheless, Suffolk sheep clearly are predisposed to this condition compared with other breeds.
Previously, no histologic lesion has been consistently associated with AED. In this investigation, we found scattered chromatolytic and necrotic neurons within the celiacomesenteric ganglion; this lesion suggests that AED may be caused by altered autonomic innervation. Previous reports of arrhythmia in affected sheep could also be consistent with alteration of the autonomic nervous system. 1,5,6,8,14 Neuronal degeneration without evidence of axonal swelling, as observed in this investigation, is indicative of primary degeneration of the soma; the absence of axonal swelling is inconsistent with the possibility of peripheral damage to axons and subsequent retrograde neuronal degeneration. Celiacomesenteric ganglia were the only ganglia that were collected in this study, although chromatolysis or atrophy of submucosal and myenteric ganglia were observed in the forestomachs and abomasa of two sheep.
No evidence of inflammation was observed in affected celiacomesenteric ganglia, which is not typical of diseases that are caused by infectious agents. Therefore, we speculate that AED may be incited in susceptible sheep (primarily Suffolks) by an unknown toxin. Because only sympathetic nerve fibers synapse within the celiacomesenteric ganglion, we further speculate that an excitotoxin could overstimulate sympathetic neurons, thereby causing immotility and subsequent impaction of the abomasum, followed by neuronal necrosis. Increased sympathetic tone could also initiate tachycardia that has been observed in cases of AED. 8 The occurrence of an outbreak of neurodegeneration and necrosis, affecting a specific neuronal target, is reminiscent of diseases caused by excitotoxins. Domoic acid is an example of an excitotoxin that targets a specific population of neurons and has caused an outbreak of Parkinson's disease in humans who consumed contaminated freshwater mussels. 10
Other possible inciting causes of AED, such as viral infections, cannot be excluded. We observed minor lymphoplasmacytic infiltrates, possibly consistent with a viral etiology, in the central nervous system of three sheep, and similar but more extensive inflammation was observed in the stomachs of one sheep. Either a toxic or a viral etiology could explain the occurrence of AED outbreaks.
In 1987, two sheep with AED also were found to have scrapie. 16 However, there has been no further support for this association. 1,13,14 In this investigation, only one of the six sheep that were examined immunohistochemically had positive staining for PrPres in the celiacomesenteric ganglion. The presence of PrPres in this ganglion is consistent with the pattern seen in early neuroinvasive stages of scrapie. 22 In the absence of evidence of scrapie in the other five animals, the most plausible explanation is that this animal was coincidentally affected by both scrapie and AED.
Degeneration and necrosis of neurons within autonomic ganglia occur in dysautonomia syndromes that occur in several species of animals and humans. In Europe, “grass sickness” of horses is characterized by gastrointestinal hypomotility and colic and is associated with degenerative lesions in autonomic ganglia. 7,9 Although usually sporadic, small outbreaks of grass sickness have occurred 23 and may even be associated with a similar disease in hares on the same premises. 3,23 Forms of dysautonomia also occur in dogs 12,15,24 and cats, 2,11,17,18 and there has been one report of an affected llama. 4 Both sporadic and familial forms of dysautonomia affect humans 20,25 and may occur more frequently in certain ethnic groups. 20
In this investigation, we provide evidence that AED of sheep may be an acquired form of dysautonomia. Pathologists should collect and examine the celiacomesenteric, submucosal, and myenteric ganglia and perhaps other autonomic ganglia from sheep that have this condition. Researchers interested in acquired diseases of the autonomic nervous system may regard outbreaks of AED as rare opportunities to search for etiologic clues. If a cause of AED can be determined, this could potentially benefit our understanding of acquired dysautonomic diseases of humans and other animals.
