Abstract
Skeletal anomalies affect animal welfare and cause important economic problems in aquaculture. Despite the high frequency of skeletal problems in reared Solea senegalensis, there is lack of information regarding the histological features of normal and deformed vertebrae in this flatfish. The aim of this study was to describe the histopathological and radiographical appearance of vertebral body anomalies. Sixty-seven juvenile fish were radiographically examined 104 or 105 days after hatching. Through radiographic images, vertebral segments were selected and processed for histopathological examination from 7 normal and 7 affected fish. Alterations in bone shape and vertebral fusion were the most significant anomalies in the vertebral bodies. These alterations occurred most frequently between the last 3 abdominal vertebrae and the first 10 caudal centra. Radiographically, deformed vertebrae showed flattening of the endplates and narrowing of the intervertebral spaces. The radiographic findings concurred with the histological lesions where affected vertebrae exhibited irregular endplates and changes in trabecular bone. Radiolucent cartilaginous tissue was evident in the endplates of the deformed vertebra and, in some cases, the cartilaginous material extended from the growth zone into the intervertebral space. These changes were likely the primary alterations that led to vertebral fusion. Fused vertebrae were often reshaped and showed a reorganization of the trabeculae. The formation of metaplastic cartilage is frequent in a variety of anomalies affecting teleost species.
Skeletal anomalies constitute a significant problem in many cultured fish, 2 and Senegalese sole (Solea senegalensis) are no exception. 26 Since this flatfish species has high economic value, the production in Europe has almost doubled in just 1 year. 12 However, this cultured fish is highly susceptible to vertebral abnormalities both in natural and in experimental conditions. 20 Some studies indicate that 44% to 100% of fish have deformities in some lots, 16,19,20 and in some fish farms, the frequency of deformities reaches 80% to 100% of the fish. 7,8,32 Like in other fish species, these skeletal problems not only have animal welfare implications but also lead to severe economic losses as it decreases the market value and downgrades the final product. 2,14,25
Skeletal deformities in Senegalese sole have been associated with various causative factors, especially those involving environmental and nutritional conditions. 1,10,13,16,35,37 However, there is a paucity of information regarding the underlying mechanisms involved in skeletal deformities in commercial fish farming where multilevel factors interact during development. 3
Detection of malformations is primarily done by gross examination and palpation of the fish in Senegalese sole farms 38 and, to a lesser extent, by refined techniques such as double-staining technique, 8,19,21 computed radiography, 7,32 and even computed tomography, 31 as in other fish species. 11,24,42
Laboratory tools such as histopathology and molecular biology have been used to assess vertebral anomalies in Atlantic salmon (Salmo salar), an osteocytic bone species. 17,30,41,47,48,49 The morphological and mechanical properties of anosteocytic bone (ie, bone without osteocytes) in fish are incompletely understood. 9 Studies in gilthead seabream (Sparus aurata) and European seabass (Dicentrarchus labrax) showed some of the histopathological alterations in vertebrae. 15,28,33 However, scarce reports exist on the histological features of Senegalese sole bone. 4,5
The aim of this work was to evaluate and correlate the radiographic and histologic changes in the vertebrae of normal and deformed juvenile Senegalese sole.
Materials and Methods
A total of 67 juvenile Senegalese sole, 104 or 105 days after hatching and ranging from 5.9 cm to 9.8 cm (standard length), were used for this study. The animals came from a fish farm located in the northwest of Spain with a prevalence of skeletal anomalies around 40% at grading point. Fish were euthanized with an overdose of Tricaine methanesulfonate (MS-222; Sigma-Aldrich, Darmstadt, Germany) and fixed in 10% buffered formalin. Computed radiography was performed in fixed fish placed in 2 orthogonal projections according to previous reports 7,32 and using methacrylate plates to position the fish. Radiographic images were evaluated with a software program (RadiAnt DICOM Viewer 1.9.16.7446 Medixant, Poznań, Poland) to detect skeletal deformities. The assessment was focused exclusively on vertebral body anomalies (fusions or deformations) and spinal deviations (kyphosis, lordosis, and scoliosis). The study included 7 fish with radiographical evidence of vertebral deformities and 7 fish without evidence of vertebral deformity (control group). Vertebral segments containing the affected bone were trimmed, as well as the same anatomical areas for the control fish. The tissues were decalcified for 48 hours in Osteodec (Bio-Optica, Milano, Italy) and embedded in paraffin. Parasagittal sections of tissue were cut at 3 μm and stained with hematoxylin and eosin (HE) for routine histopathological examination. For the identification of acidic mucins and cartilage, tissue sections were stained with alcian blue/HE (AB-HE) and alcian blue/periodic acid–Schiff (AB-PAS). Also, Gallego’s trichrome (GT) stain was used to differentiate collagen fibers, muscle, and cartilage. 34 Finally, osteoid, mature bone, and cartilage were identified using a previously described modified tetrachrome osteoid stain (OS). 36 Slides were mounted using Coverquick 2000 mounting medium (BDH Prolabo, Singapore, Singapore) and observed with an Olympus BX51 microscope with a digital camera Olympus DP72 (Olympus, Hamburg, Germany). A comparative study was done by reviewing deformed and nondeformed vertebrae in the same fish, as well as with the corresponding segments of the control animals.
The data analyzed in this study are not available as Supplemental Materials.
Results
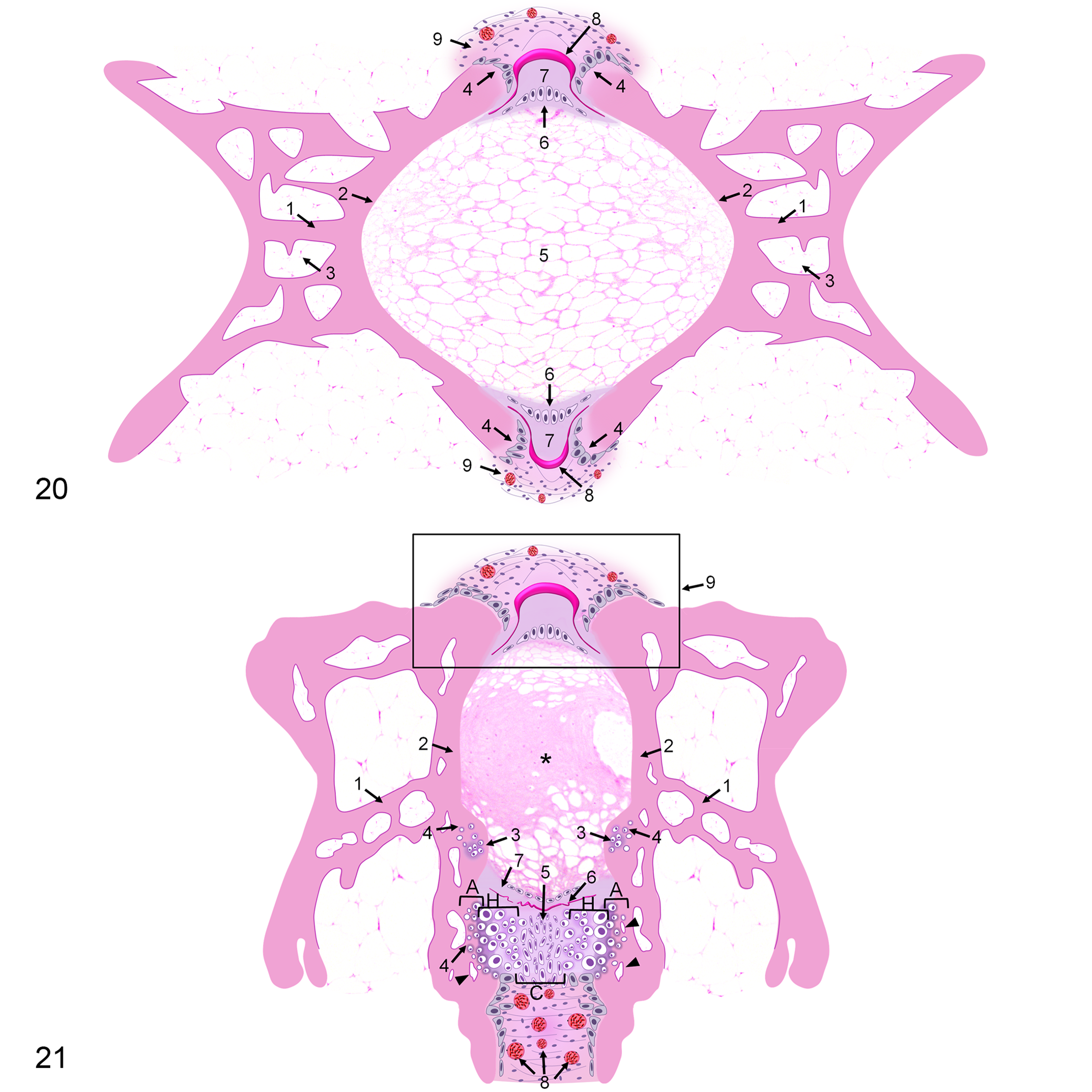
The radiographic approach used in juveniles allowed the detailed assessment of the vertebral column, although in the smaller fish, the definition of the caudal-most vertebral bodies (preurals and urostyle) was impaired. Radiographic images of control fish showed an aligned rachis, composed of succeeding vertebrae along the abdominal, caudal, and caudal complex region (Fig. 1a,b). The vertebrae showed an X-shape internal structure separated by radiolucent intervertebral spaces (IVSs) (Fig. 2). Histologically, vertebral bodies showed a symmetrical amphicoelous form and followed a line interposed by IVSs filled with the notochord (Fig. 3). Supplemental Table S1 shows the characteristic colors of the vertebral structures using different stains. Vertebrae composed of anosteocytic osseous tissue had trabecular bone between the 2 endplates of laminar bone (Figs. 3, 4). A layer of osteoid-secreting osteoblasts was observed in the growth zone of the endplates covered by external intervertebral connective tissue (Fig. 4). In the IVSs, the notochord was composed of vacuolated chordocytes in the center and chordoblasts in the notochordal epithelial layer (Fig. 4). Depending on the parasagittal section, some chordocytes appeared less vacuolated in a denser area in the notochord center (Fig. 3). Notochordal cells were encased in a collagenous layer (notochordal sheath) and the external elastic membrane (Fig. 4). OS revealed mature bone in the vertebral centrum, except for the endplates, where osteoid was the dominant component (Fig. 5).
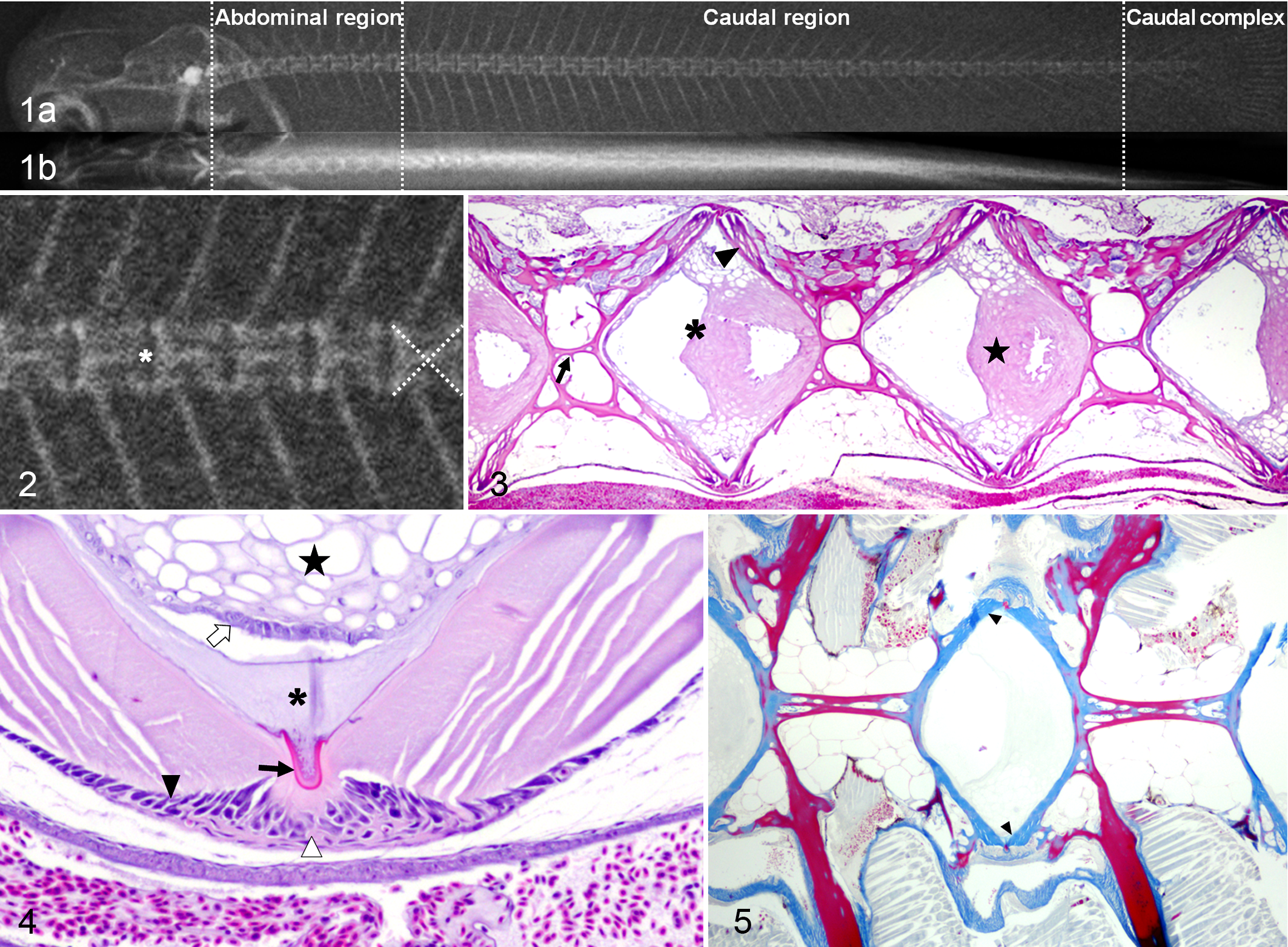
Spinal anomalies were found radiographically present in 7 of 67 (∼10%) fish. Vertebral deformation and fusion were the 2 major anomalies, affecting 6 of 7 and 3 of 7 of the selected fish, respectively. These lesions were most frequently located between the last 3 abdominal vertebrae (A7–A9) and the first 10 caudal centra (C1–C10). The most common radiographic features in the deformed vertebrae were flattened and irregular endplates, which often appeared more radiodense than those in the adjacent vertebrae (Fig. 6). Overall, the IVS was narrow (Fig. 6) and sometimes with an irregular outline. Microscopic alterations in the endplate paralleled those changes observed radiographically. The deformed vertebral bodies also showed alterations of the osseous internal trabeculae. The main longitudinal trabeculae were oblique, sometimes symmetrically, resembling a concave or convex (Fig. 7) structure. In 16 of 23 (∼70%) of the deformed vertebrae, a variable number of cells resembling chondrocytes were present in the endplates, toward the IVS. These cells were particularly prevalent in the dorsal or ventral aspect of the endplate, near the growth zone. Usually, when opposed centra trabeculae formed a convex structure, chondrocytes were located ventrally (Fig. 7). Some vertebrae had large numbers of chondrocytes separated by abundant extracellular cartilaginous matrix (ECM). The chondrocytes resided inside lacunae and were surrounded by a halo of ECM (Fig. 8).
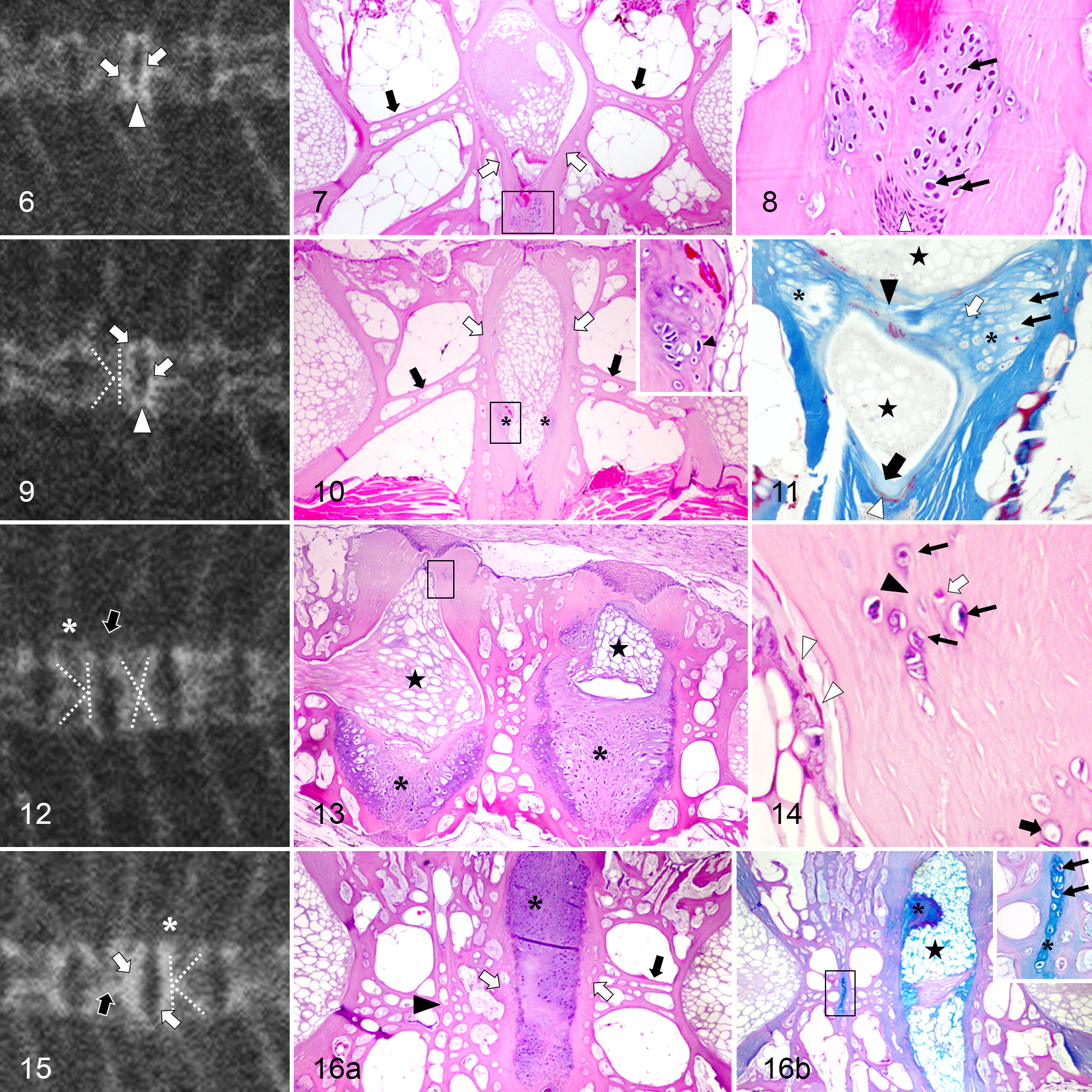
In 1-sided compressions, the radiographic image of the centrum presented a “K”-like shape (Fig. 9), which microscopically corresponded with a flattened endplate from 1 side and oblique trabeculae (Fig. 10). In addition to the growth zone, chondrocytes were observed in the articular bone. Occasionally, aggregates of chondrocytes formed protuberances, which protruded bilaterally and symmetrically toward the notochord in the IVS (Fig. 10 and inset). In 2 of 6 deformed fish, these protuberances joined together, forming a bridge among the vertebral bodies (Fig. 11). The thickness and height of some vertebral bodies were increased compared to the normal adjacent vertebrae (Fig. 12). Microscopically, cartilaginous tissue was invading the IVS in 2 of 6 fish (Fig. 13). The extension of this material ranged from 30% to 100% of the IVS, depending on the depth of the section, apparently pushing the notochord (Fig. 13).
Almost all the trabecular and articular lesions showed a mirror image involving 2 consecutive vertebrae. When the affected area had only a few chondrocytes, these cells were isolated and embedded in a bone matrix, often forming a tissue compatible with chondroid (Fig. 14). Deeper into the endplate, the chondrocytes appeared smaller (Fig. 14).
More severe anomalies were observed, combining fusions and deformations (Fig. 15). These vertebrae had excessive cartilaginous tissue between fused vertebrae and the opposing centra in histological sections (Fig. 16a). However, in a serial section, chondrocytes disappeared, and the IVS was occupied by notochordal structures with a small amount of ECM (Fig. 16b) related to the presence of chondrocytes in the endplate.
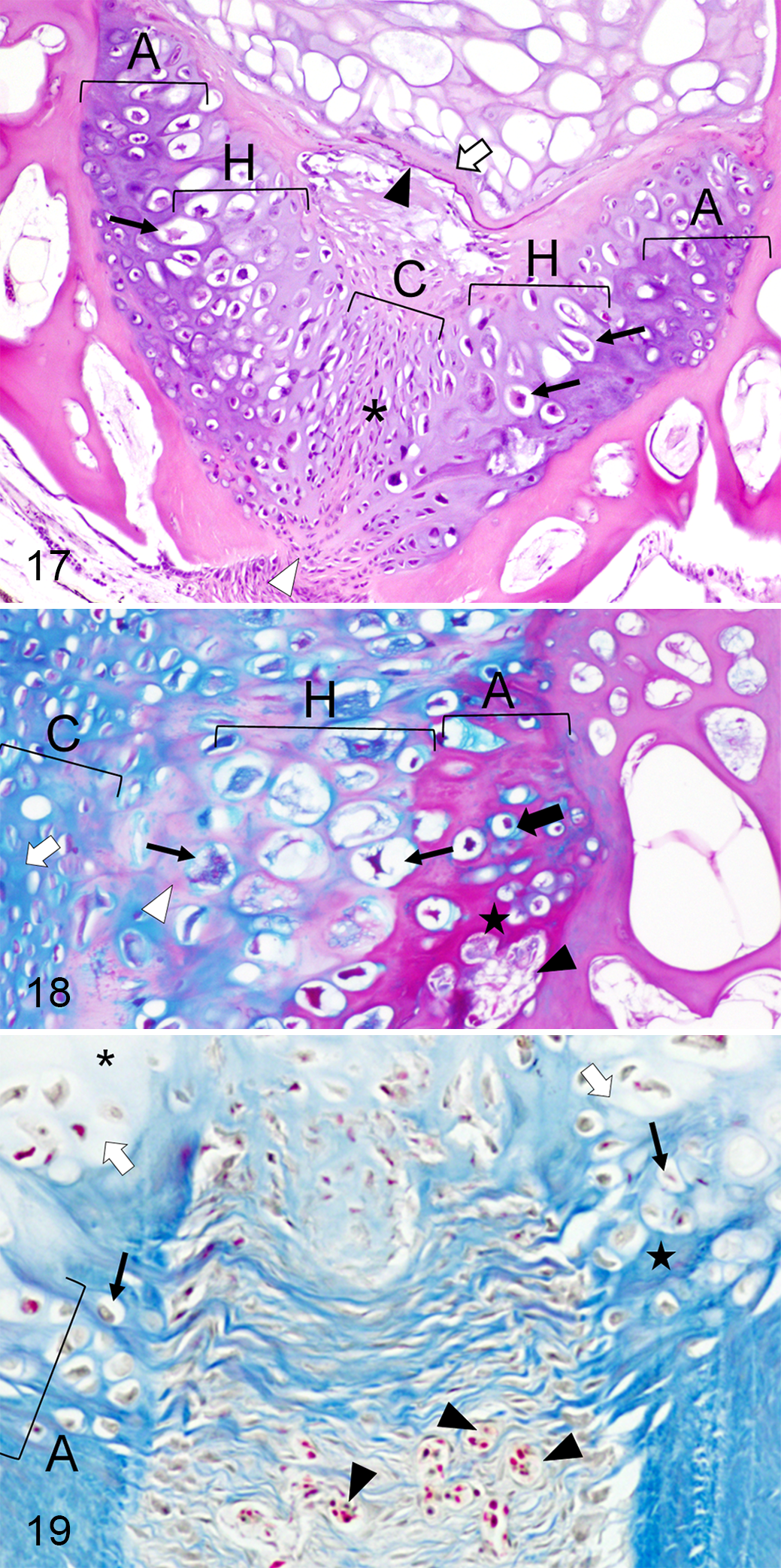
Cartilaginous tissue within the IVS was rich in chondrocytes surrounded by abundant ECM (Figs. 17, 18), compatible with cell-rich hyaline cartilage. Chondrocytes had different degrees of maturation depending on their location in the IVS (Fig. 17). The tissue was composed of a central area occupied with small slightly elongated chondrocytes (Figs. 17, 18). At both sides of this area, there were rounded and hypertrophied chondrocytes and a zone where cartilaginous tissue was associated with the endplates (Fig. 17, 18). In the former region, the matrix involving some groups of chondrocytes changed its affinity for the dye (Figs. 17–19). Occasionally, it was difficult to distinguish the boundaries between the cartilage in the IVS and the endplate as both looked alike.
Deformed vertebral segments also showed alterations in notochordal structures. The external elastic membrane was usually scattered and irregular in the converging area of 2 endplates showing chondrocytes (dorsally or ventrally) (Figs. 11, 14, 17). The collagenous layer associated with the damaged membrane was occasionally less evident or thinner (Figs. 11, 17). A high number of blood capillaries were sporadically present in the connective tissue, externally to the layer of osteoblasts (Fig. 19). A schematic summary of the histological features in control and deformed vertebrae is illustrated in Figs. 20 and 21.
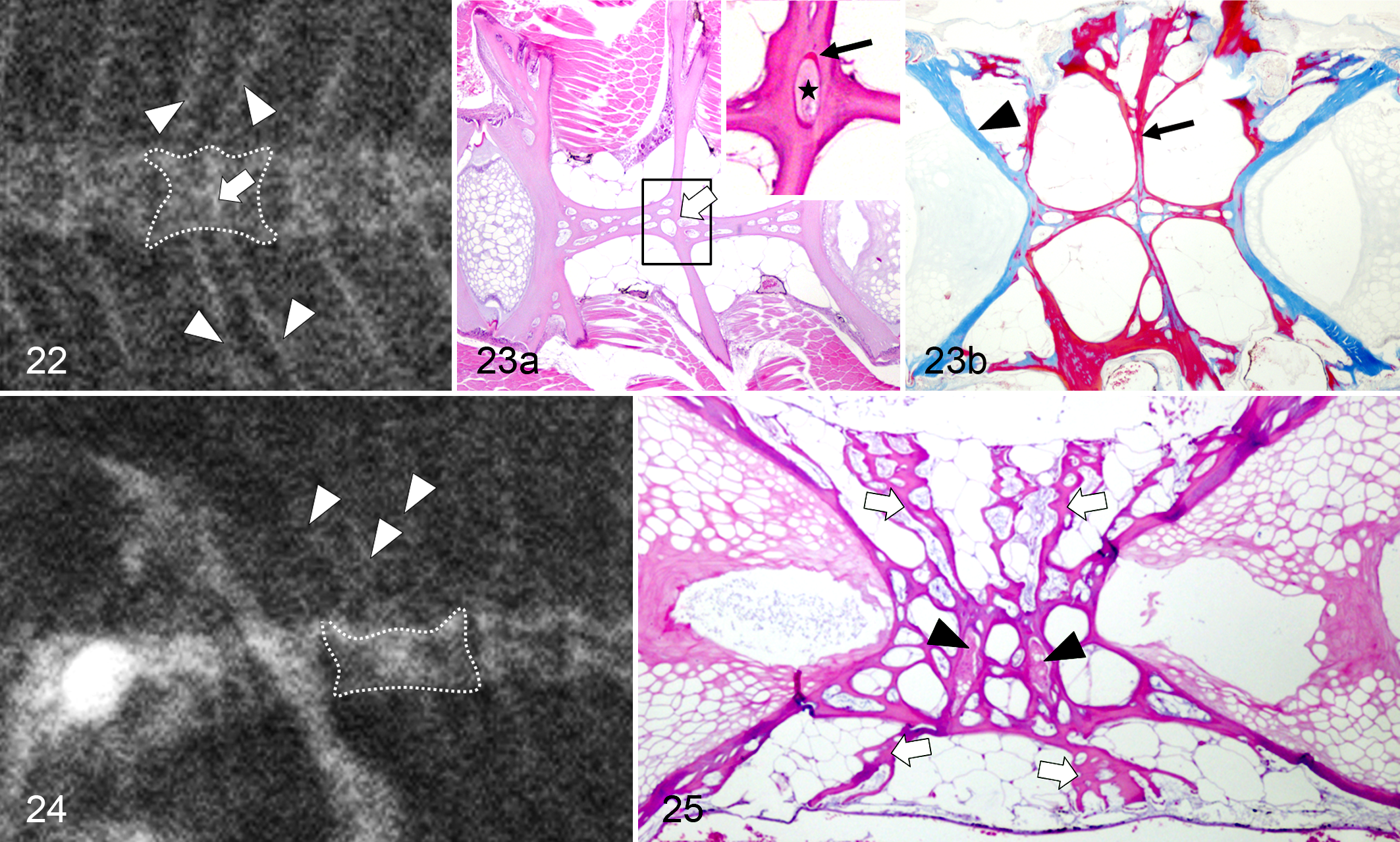
Fused vertebrae were radiographically recognized by the presence of extra neural and hemal elements (Fig. 22). In many cases, the union was reshaped, showing almost the same length as any normal vertebra (Fig. 22). Histopathologic findings in fused vertebrae consisted of the reorganization of the internal longitudinal trabeculae, but the fusion line persisted (Fig. 23a). In serial sections, remnants of the notochord and the external elastic membrane were detected in the center of the anomaly (Fig. 23a, inset). In 1 case, a small portion of cartilaginous tissue was observed in a similar location (Fig. 16b, inset). OS stain revealed mature bone in the central region of most of the fused vertebrae (Fig. 23b). Fusion involving 3 vertebral bodies was also detected (Fig. 24), showing 2 small notochord portions in the corresponding location of the former IVSs (Fig. 25).
Discussion
The present study shows the most common vertebral body anomalies affecting Senegalese sole applying radiographic and histopathologic methods. As far as we know, this is the first histopathologic description of fused and deformed vertebrae unrelated to axis deviations in this flatfish species.
The radiographic study revealed that deformations and fusions are the 2 major types of vertebral body abnormalities in juvenile Senegalese sole. Similar anomalies were previously described in the caudal complex vertebrae of larvae, using a double-staining technique for cartilage and bone, with a prevalence of 19% and 25%, respectively. 8,20 As reported in several fish species, 5,30,33,41 Senegalese sole also develop vertebral deformation with alterations of longitudinal bony trabeculae and flattened endplates. Microscopically, the deformed vertebral centra display cartilaginous alterations in the endplates and IVS, but these alterations are not evident in the radiographic images. The symmetrical characteristics of these abnormalities may indicate that the vertebral lesions likely start in the region comprising intervertebral tissue and opposing endplates. 30 In curveback lineage of guppies (Poecilia reticulata), vertebral deformations have been primarily described in the concave side of lordotic and kyphotic vertebrae. 22 This study also showed that the chondrocytes were present in the concave aspect of centra displaying an oblique symmetric orientation of the trabecular bone. Fused centra were radiographically detected by the presence of extranumerary neural and hemal structures. Also, microscopic findings suggest that trabecular remodeling or modeling occurr and remnants of intervertebral notochord persisted in the fusion center. In 1 fish, a cartilaginous residue was found in the fusion line (just in the intersection of perpendicular trabeculae), corresponding to the location of the IVS. Similar findings were observed in Atlantic salmon in intermediate and final stages of fusion. 47,49
The formation of cartilage constituents can be present in both anomaly types, deformations and fusions, and in distinct fish species. 4,41,47 These may constitute an ongoing process of aggravation of the lesions in which some deformed segments could be already showing early signs of fusion. 18,47 Also, 2 fused centra showed cartilage in the adjacent IVS, indicating the progression into a 3-vertebrae fusion. 47 It has been suggested that adjacent asymmetrically compressed vertebrae would fuse, while homogeneously compressed vertebrae might represent a more “stable alteration.” 40,41,42,47 Therefore, it would be interesting to evaluate vertebral anomaly signaling pathways as well as environmental factors involved in the containment and aggravation of the problem. 47
The development of chondroid and cartilaginous tissue observed in skeletal anomalies is not well understood in fish. Anosteocytic teleost bone is able to perform adaptive, plastic responses to loading conditions. 9,28,29 The presence of cartilaginous tissue could attenuate mechanical stress at the autocentrum. 5 Also, chondroid tissue seems to meet the demand for an accelerated local growth rate and the need for a shear-resistant support. 27,33
Chondrocytes were located in the endplates, as reported in other studies, 5,28,30,33,41 especially near the growth zones constituted by osteoblasts and mesenchymal cells. This distribution can be based on the availability of oxygen and nutrients in the peripheral areas. 5 Our results showed the proliferation of exuberant cartilaginous tissue visibly from the growth zones into the IVS. These findings may indicate that chondrocytes could arise from metaplastic mesenchymal cells in the growth plate, 5 as a result of altered mechanical load, such as suggested for vertebral curvatures. 4 Mesenchyme cells may differentiate into chondroblasts rather than osteoblasts in response to high rates of change in compression 23 or in a low-oxygen environment during fracture repair. 6 Although cotranscription of some osteogenic and chondrogenic markers may occur in the osteoblasts in the growth zone during the fusion process, 49 the mesenchymal source of osteoprogenitor cells has not been clearly identified in fish. 46 Moreover, osteogenic cells can transform into osteocytes or into chondrocytes in the Atlantic salmon kype skeleton as they become entrapped in the bone matrix. 43 Therefore, the possible involvement of osteoblasts should not be disregarded, nor the metaplastic transformation of the notochord area into cartilage. 41
Some authors hypothesized that once the chondrocytes differentiate in the growth zone, they can be entrapped by the newly deposited osteoid matrix and eventually die due to lack of nutrients and hypoxia, with only those near the endplate surviving. 5 Another hypothesis is that cartilage could be developing into the IVS, stuck on the endplate, causing alterations in the endplate shape and compressing the notochord. The previously described hypotheses are not mutually exclusive and might represent the same subsequent process whereby cartilage is replaced by osseous tissue, as occurring in later steps of fusion. 47
Three main mechanisms of bone formation have been described in teleosts, depending on the species and skeletal structures: intramembranous, perichondral, and endochondral. 2 Of these, the latter two usually involve a cartilaginous template, 2 although vertebral bodies form through intramembranous ossification. 4 Hypertrophic chondrocytes have an important role in the final phases of higher vertebrate endochondral ossification since they modify ECM, allowing matrix mineralization, vascular invasion, and posterior bone formation. 6,39 Similarly to other studies, hypertrophic chondrocytes were located proximal to the endplate, 4 although the most peripheral were embedded in a more basophilic matrix (HE), which corresponded with osteoid. Endochondral and perichondral ossification, as well as chondral bone, needs further investigation to better understand the structural and chemical processes occurring in different fish species. 2 Moreover, a chondroidal mechanism of ossification could be taking part in these processes, as suggested for Atlantic salmon and European seabass. 28,49
Concerns still exist on how to identify the specific pathogenic mechanisms involved in the onset of skeletal deformities, especially in anosteocytic bone fish. The histopathologic changes observed in fish with deformed and fused vertebrae further support the importance of identifying which cells are involved in the pathogenesis of vertebral problems. In mammals, osteocytes are believed to detect strain and mechanical load and to regulate cellular events implicated in the formation and remodeling of bone. 9,39,45 The fact that fish with anosteocytic bone may respond to loading conditions may indicate an alternative load detection mechanism. 9,45 Some reports postulate that the osteoblasts and bone-lining cells are likely candidates for sensing mechanical load in anosteocytic bone. 44,45 The particular location of the cartilage in the growth zones may favor this hypothesis, although it requires further investigation. Moreover, other authors proposed that chondrocytes could serve as mechanosensors in most compressed areas. 5
Studies with Atlantic salmon revealed an irregular and fragmented external elastic membrane in fish with fused and compressed vertebrae. 30,50 The rupture of this membrane may be involved in the progression of spinal anomalies and likely coevolves with other structural changes of the notochordal sheath. 50 In fact, the collagenous layer associated with the fragmented membrane was occasionally less evident or thinner. According to other studies, these lesions may decrease flexibility and possibly disturb nutritional transportation across the notochord and could be related to the development of spinal fusions. 50
In summary, the histological techniques complemented considerably the radiographic studies. Lesions consisted of alterations of the shape and orientation of bony trabeculae and vertebral endplates. The presence of chondrocytes in the endplates and within the IVS was a common response in different types of anomalies affecting teleost species with either osteocytic or anosteocytic bone. Some of the alterations in deformed vertebrae correspond with those seen in the initial stages of fusion and, in 1 case, with the aggravation of a primary fusion process. The cartilage proliferation from the growth zones into the IVS suggests that the chondrocyte mesenchymal origin could arise from metaplastic cells in the growth area as a result of altered mechanical load. In later stages, the hyaline cartilage appears to be replaced by osseous tissue. Alterations in the external elastic membrane and the collagenous layer are presumably linked to vertebral deformities. Further work is needed to investigate the molecular pathways that may be involved in mechanosensor and local effector mechanisms, thus promoting the development of cartilage in anosteocytic bone anomalies.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818800027 - Skeletal Anomalies in Senegalese Sole (Solea senegalensis), an Anosteocytic Boned Flatfish Species
Supplemental Material, DS1_VET_10.1177_0300985818800027 for Skeletal Anomalies in Senegalese Sole (Solea senegalensis), an Anosteocytic Boned Flatfish Species by Ana Manuela de Azevedo, Ana Paula Losada, Andrés Barreiro, Sonia Vázquez, and María Isabel Quiroga in Veterinary Pathology
Footnotes
Acknowledgements
We thank Stolt Sea Farm (especially A. Riaza and I. Ferreiro) for providing fish samples and technical support, A. López for the constructive editorial contributions to the manuscript, J. D. Barreiro for assistance with the radiographic procedures, Prof. C. Boglione and Dr. L. Prestinicola for their contributions on the AB-HE staining protocol, and S. Maceiras for her excellent technical work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was funded by “Consellería de Economía e Industria” of Xunta de Galicia (10MMA020E) and by “Programa de Consolidación e Estructuración de Unidades de Investigación Competitivas GPC2015/034,” Spain. A. M. de Azevedo held a University Professorship Formation (FPU) grant from the Spanish Ministry of Education.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
