Abstract
Severe Perkinsea infection (SPI) is an emerging disease of frogs responsible for mass mortalities of tadpoles across the United States. It is caused by protozoa belonging to the phylum Perkinsozoa that form a distinct group referred to as the Pathogenic Perkinsea Clade of frogs. In this work, we provide detailed description of gross and histologic lesions from 178 naturally infected tadpoles, including 10 species from 22 mortality events and 6 amphibian health monitoring studies from diverse geographic areas. On external examination, we observed abdominal distension (10, 5.6%), cutaneous erythema and petechia (3, 1.7%), subcutaneous edema (3, 1.7%), and areas of white skin discoloration (3, 1.7%). On macroscopic examination of internal organs, we found hepatomegaly (68, 38.2%), splenomegaly (51, 28.7%), nephromegaly (47, 26.4%), ascites (15, 8.4%), segmental irregular thickening and white discoloration of the intestine (8, 4.5%), pancreatomegaly (4, 2.2%), and pancreatic petechia (1, 0.6%). Histologically, over 60% of the liver (148/165, 89.7%), kidney (113/147, 76.9%), spleen (96/97, 99%), and pancreas (46/68, 67.6%) were invaded by myriad intracellular and extracellular Perkinsea hypnospore-like and trophozoite-like organisms. Numerous other tissues were affected to a lesser extent. Mild histiocytic inflammation with fewer lymphocytes or eosinophils was commonly observed in areas of infection that were not obscured by lympho-granulocytic hematopoietic tissue. In light of these observations, we suggest a logical pathogenesis sequence. Finally, we propose a “case definition” for SPI to promote standardized communication of results and prevent misdiagnosis with epidemiological and pathologically overlapping diseases such as ranavirosis.
Keywords
Nearly one-third of all known amphibian species are threatened and over 400 are critically endangered. 34 Since the 1970s, over 200 frog species have gone extinct, and hundreds more may face extinction in the coming decades. 1 Along with habitat degradation, emerging infectious diseases have an important role in this worldwide decline of frog populations. 1
In North America, chytridiomycosis and ranavirosis are responsible for mass mortalities of anurans. 11,16,25,36 Whereas chytridiomycosis mostly targets postmetamorphic frogs, 11,36 ranavirosis principally affects premetamorphic and metamorphic stages (hereafter referred as tadpoles). 16,25
In the last 2 decades, another infectious disease of tadpoles, severe Perkinsea infection (SPI), has been recognized as causing mass mortalities across the United States from boreal to subtropical areas. 6,9,20,22 The detection of SPI mortality in Alaska and other northern states suggests that this disease is likely distributed throughout much of the Nearctic region. 20 Like ranavirosis, 35 SPI can threaten susceptible frog species such as the critically endangered dusky gopher frog (Rana sevosa), by drastically reducing recruitment. 17,20
Severe Perkinsea infection is caused by protozoa belonging to the phylum Perkinsozoa (superphylum Alveolata). A recent phylogenetic analysis of isolates from SPI-affected tadpoles across the United States showed that all examined sequences of the small subunit of the ribosomal RNA-encoding gene clustered into a highly supported clade tentatively named Pathogenic Perkinsea Clade of frogs (PPC). 20 Although little is known about the etiologic agent of SPI, other Perkinsea species within the genus Perkinsus (eg, P. marinus, P. olseni), responsible for mass mortalities of mollusks and secondary economic losses, have been well studied. 12 These Perkinsus spp. have a direct life cycle, with a free-living aquatic zoospore (infective) stage. 2 After infection, the zoospore becomes a trophozoite that proliferates through host tissues by vegetative reproduction. In moribund or dead hosts, the trophozoite enlarges and develops a thick wall, thus becoming a hypnospore (resting stage) that endures outside the host in sediments. 27,37 Finally, zoosporulation occurs when the hypnospore is resuspended into the water column or ingested by a new host. 3
The relatively recent acknowledged threat that emerging infectious diseases represent for frog populations has led to increased monitoring of mortality events. 8 Yet, incomplete postmortem investigations may lead to misdiagnosis given the epidemiological and pathological similarities of some amphibian diseases and the existence of subclinical carriers. 10,16,20,28
Establishing a case definition is one route to standardize diagnostic results, promote consistent reporting of emerging diseases, and aid in differentiating diseases with overlapping diagnostic criteria. Although standard in human medicine, case definitions are uncommon for wildlife diseases. A recent case definition of the emerging amphibian chytridiomycosis of caudates caused by B. salamandrivorans was an important step in organizing diagnostic communications for a newly identified disease. 38 Case definitions typically include field, gross, histopathology, laboratory, and epidemiologic criteria for assigning a specific disease, condition, or presence of a pathogen, as well as confidence in a diagnosis given the current state of knowledge and evidence from available diagnostic criteria.
In previous publications, we provided a general outlook of frequent pathological changes associated with SPI. 19,20 In this article, we expand upon that previous work to provide more detailed macroscopic and histologic pathology descriptions of a large set (n = 178) of wild frogs with SPI, with special attention to frequency and severity of lesions in each organ. In light of these observations, we propose a plausible pathogenesis sequence. We also discuss the importance of complete pathologic evaluation during anuran disease investigations given the epidemiological and pathological similarities among SPI and other diseases of tadpoles such as ranavirosis. Finally, we propose a case definition of SPI based on current knowledge of the disease to standardize communications and reduce misdiagnosis.
Methods
We reviewed pathology reports and hematoxylin and eosin-stained histologic slides of 178 tadpoles from 22 SPI-associated mortality events and 6 amphibian health monitoring studies (mortality not specifically tracked or estimated) in 13 states of the United States from 1999-2017. Severe Perkinsea infection was confirmed in each of these 178 tadpoles by Isidoro-Ayza et al. 19,20 Postmortem investigation took place at the USGS-NWHC (Madison, Wisconsin). Confirmation of SPI was based on histological observation of pathological changes in tissues with presence of Perkinsea-like organisms in at least 1 organ, including brain, eyes, gastrointestinal tract, gills, heart, liver, lungs, kidney, pancreas, skeletal muscle, skin, spinal cord, spleen, and thyroid gland.
Anuran species and developmental stage identification was confirmed based on external morphological features. 14,29,33
For histopathology, samples were fixed in 10% formaldehyde for at least 48 h. Specimens with partially or completely ossified skeleton, were decalcified in formic acid-sodium citrate mixture overnight. Fixed samples were dehydrated with a graded ethanol series, embedded in paraffin and sectioned at a thickness of 4-μm with a rotary microtome, then stained using Mayer’s hematoxylin and eosin. For each carcass, the overall extent (as percentage) and distribution of the histologic lesions on hematoxylin and eosin-slides, the distribution of the Perkinsea-like organisms in different organs (including trophozoite-like and hypnospore-like organisms), and the estimated ratio of trophozoite-like to hypnospore-like structures in liver were assessed by a board-certified veterinary pathologist (MIA).
For electron microscopy, a fragment of liver and spleen from 1 SPI-affected tadpole (USGS-NWHC identification 28207-015); and kidney from another SPI-affected tadpole (28207-012) were preserved in paraformaldehyde-glutaraldehyde solution (Karnovsky’s fixative), after which they were dehydrated in a graded ethanol series, infiltrated with epoxy propylene oxide, and embedded in epoxy resin. The epoxy block was sectioned with an ultra-microtome at a thickness of 1-μm to produce semithin sections that were thereafter stained with uranyl acetate followed by lead citrate and examined with a transmission electron microscope equipped with a digital photomicrograph (Hemmamtsu ORCA HR Camera). Electron microphotographs of liver from 2 SPI-affected tadpoles (4957-288 and 16406-009) 20 were reviewed for the present study.
In previous studies, 19,20 PPC was confirmed by PCR amplification and sequencing of the small subunit of the ribosomal RNA-encoding gene in 19 of these 178 tadpoles, including at least 1 tadpole from 12 of 19 Counties with histologically confirmed SPI events (10 out of 13 states) (Supplemental Table S1). In addition, previous to fixation, kidney, chilled or frozen and thawed liver, or pooled internal organs of 44 of these 178 tadpoles from 14 SPI mortality events and 1 amphibian health monitoring study were tested for frog virus-3 as previously described, 23,24 with only 1 tadpole being positive. 19,20
Fresh liver or spleen from 27 of the 178 tadpoles from 8 SPI mortality events and 2 amphibian health monitoring studies were processed for bacterial testing. Briefly, aseptically collected specimens were inoculated onto blood and MacConkey agar plates and incubated at 37C. All isolates were identified using API strips (BioMerieux, Marcy l’Etoile, France) or the OmniLog microbial identification system (Biolog Inc, Hayward, CA, USA).
All samples used for this study were from tissue archives and originated from wildlife disease investigations conducted on amphibian carcasses from 1999 to 2017. Euthanasia of frogs included in this study was approved by USGS National Wildlife Health Center Institutional Animal Care and Use Committee (IACUC) protocol EP080707.
Results
Life Stage and Body Condition
Tadpole life stage 14 was determined and reported in 166 of 178 SPI-affected tadpoles (93.3%). Thirteen (7.8%) were late hatchlings (Gosner stage 25), 135 (81.3%) were larval stages (Gosner stages 26-41) and 18 (10.8%) were metamorphs (Gosner stages 42-46). No adult frogs were diagnosed with SPI. Body condition was reported in 113 tadpoles and was good in 85 (75.2%) and poor in the remaining 28 (24.8%). 20
Macroscopic Lesions
On gross examination, a subset of 10 tadpoles (5.6%) presented marked abdominal distension. Skin lesions were observed in 9 tadpoles (5.1%), including multifocal, irregular redness of the skin (erythema) and pinpoint hemorrhages (petechia) (3, 1.7%) (Fig. 1); subcutaneous bilateral symmetric edema of the abdominal region, base of the tail, inguinal area and thighs (Fig. 2) (3, 1.7%); and randomly distributed 2 to 10-mm areas of white discoloration (3, 1.7%). On internal examination, 15 tadpoles (8.4%) presented moderate to abundant accumulation of serous fluid in the coelomic cavity (ascites); 68 tadpoles (38.2%) presented enlargement of the liver with rounded edges (hepatomegaly) and mottled pale discoloration (Fig. 3); 51 tadpoles (28.7%) presented enlarged spleen (splenomegaly) with mottled pale or red discoloration (Fig. 3); 47 tadpoles (26.4%) presented increased size of the kidney (nephromegaly), including pronephros and mesonephros, with pale or red discoloration (Fig. 3); the intestine of 8 carcasses (4.5%) presented segmental to coalescing areas of irregular thickening and white discoloration of the intestinal wall (Fig. 4); and 5 tadpoles (2.8%) presented lesions in the pancreas were noted, including moderate enlargement of the organ (pancreatomegaly) and diffuse orange discoloration (Fig. 3) (4, 2.2%), or multifocal petechial hemorrhages (1, 0.6%).
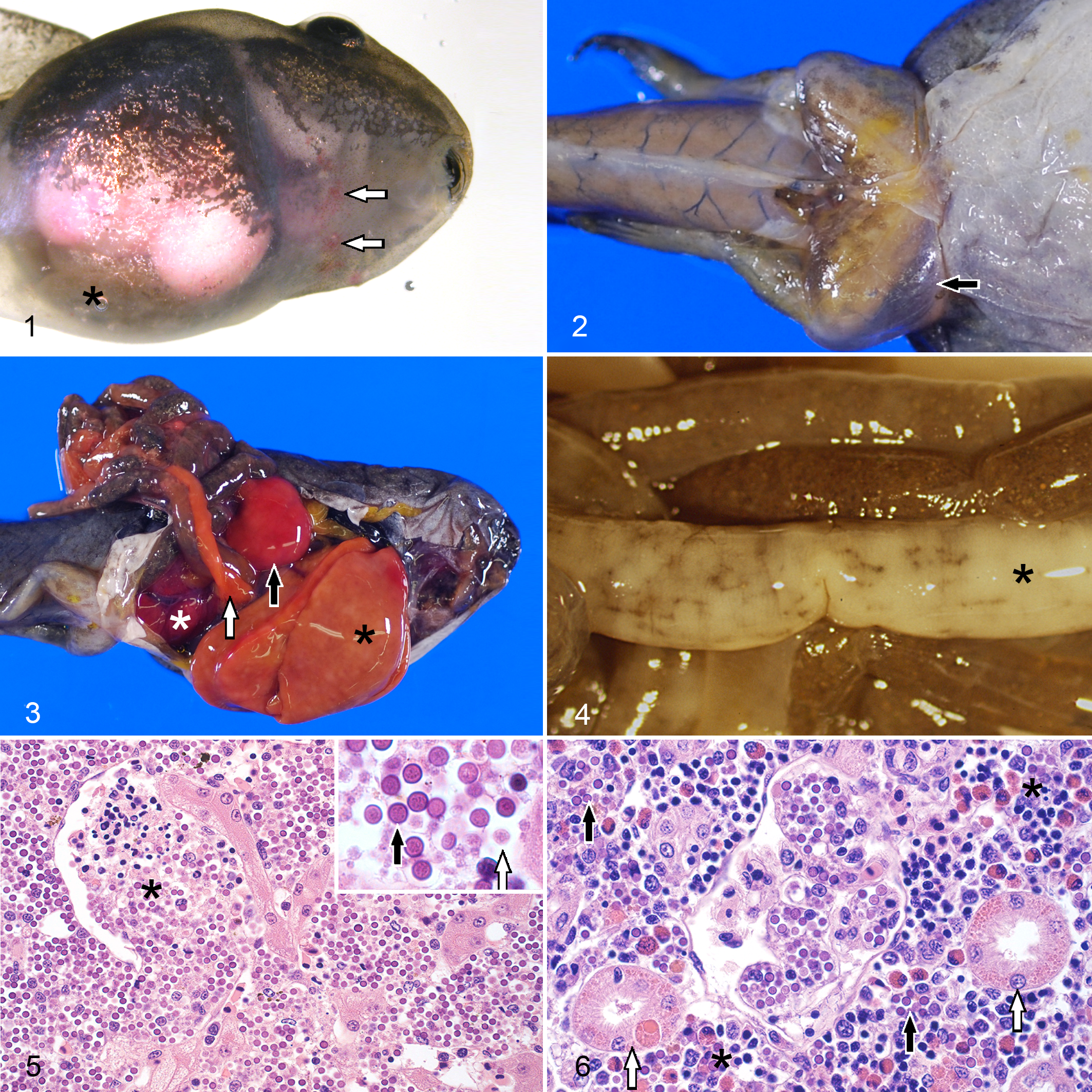
Severe Perkinsea infection (frog virus-3 negative), frog.
Histologic Lesions
Histologic lesions were associated with invasion of numerous tissues by myriads of intracellular and extracellular Perkinsea-like organisms. All the organs routinely examined presented variable invasion by these organisms and secondary tissue damage. The most extensively affected organs were liver, spleen, kidney and pancreas. The extent of the lesions in these 4 organs from the 178 tadpoles included in this study is summarized in Supplemental Table S2. In addition, 1 or more of the following tissues were invariably affected but to a smaller extent; cartilage, central nervous system, gastrointestinal tract, gills, gonads, heart, peritoneum, skeletal muscle, skin, and thyroid glands.
Most sections of liver examined presented random, multifocal-coalescing replacement of over 60% (148/165, 89.7%) of the hepatic parenchyma and varying disruption of Glisson’s capsule by myriads of Perkinsea-like organisms (Fig. 5). Two life stages were observed: 4-µm diameter spherical structures with thick deep basophilic wall, and granular pale basophilic cytoplasm (hypnospore-like); and 2-µm diameter, amoeboid, pale basophilic organisms (trophozoite-like) (Fig. 5, inset). The hypnospore-like stage was the more abundant (>50% of the organisms in 3 random 100X fields) in 126/162 (77.8%). The Perkinsea-like organisms were embedded in variable amounts of necrotic debris, fibrin, and erythrocytes. Sparse macrophages with occasional intracytoplasmic Perkinsea-like organisms and varying numbers of lymphocytes and eosinophils were observed in the affected tissues. Granulopoietic tissue with predominance of the eosinophil series, characterized by a nonlobed to bilobed eccentric nucleus and numerous large round bright eosinophilic cytoplasmic granules (Supplemental Fig. S1), was commonly observed in portal spaces. In less severely affected areas, sinusoids were multifocally expanded and variably obliterated by Perkinsea-like organisms. The hepatic cords were dissociated and in many instances hepatocytes contained up to 4 Perkinsea-like organisms in their cytoplasm. Endothelial cells in sinusoids and small blood vessels were occasionally missing, and clusters of the same organisms, fibrin, necrotic debris and erythrocytes were attached to the exposed subendothelial surface (thrombi) (Fig. 5). In some instances, medium to large areas of the organ showed coagulative necrosis and occasional presence of nearby thrombi. Infected and uninfected hepatocytes presented hydropic degeneration or atrophy. Medium- and large-size blood vessels and bile ducts showed variable cholestasis.
In the kidney (pronephros and mesonephros), Perkinsea-like organisms infiltrated the intravascular and perivascular interstitial space and the glomerular tufts, in most instances replacing over 60% (113/147, 76.9%) of the renal tubules and glomeruli (Fig. 6). Remaining tubules showed variable attenuation, hydropic degeneration or intracellular eosinophilic, hyaline, droplets (protein reabsorption) and necrosis of the epithelial cells, and in many cases, intracytoplasmic invasion by the infectious agent. Multifocal, interstitial lympho-granulopoietic tissue with dominant eosinophil series was prominent in most instances, and inflammatory infiltrates were generally not assessable.
In the spleen, mostly affecting the red pulp with extension into the white pulp, Perkinsea-like organisms infiltrated the intravascular and perivascular spaces often replacing 60% of the organ (96/97, 99%), and embedded in variable amounts of necrotic debris. The same scattered inflammatory infiltrates described in the liver were also observed in this organ.
In the pancreas, intravascular and extravascular Perkinsea-like organisms expanded the interstitium and replaced the exocrine epithelium of over 60% of the organ in most tadpoles (46/68, 67.6%). Perkinsea-like organisms were occasionally observed in the cytoplasm of exocrine pancreatic epithelial cells which showed variable degrees of hydropic degeneration. Scattered histiocytic and lympho-eosinophilic inflammatory infiltrates similar to those described in the liver and spleen were also observed in the affected portions of pancreas.
The branchial and oral lamina propria and the gastrointestinal lamina propria and submucosa were expanded by intravascular and perivascular Perkinsea-like organisms and mild to moderate perivascular edema with variable attenuation, degeneration and necrosis of the lining and glandular epithelium (Figs. 7 and 8). Frequently, Perkinsea hypnospore-like organisms were also present in the lumen of the buccal cavity and gastrointestinal tract. Mild histiocytic inflammation with fewer lymphocytes or eosinophils was occasionally observed in affected areas. In some instances, granulopoietic tissue with predominant eosinophil series was observed in the mesenteric vessels.
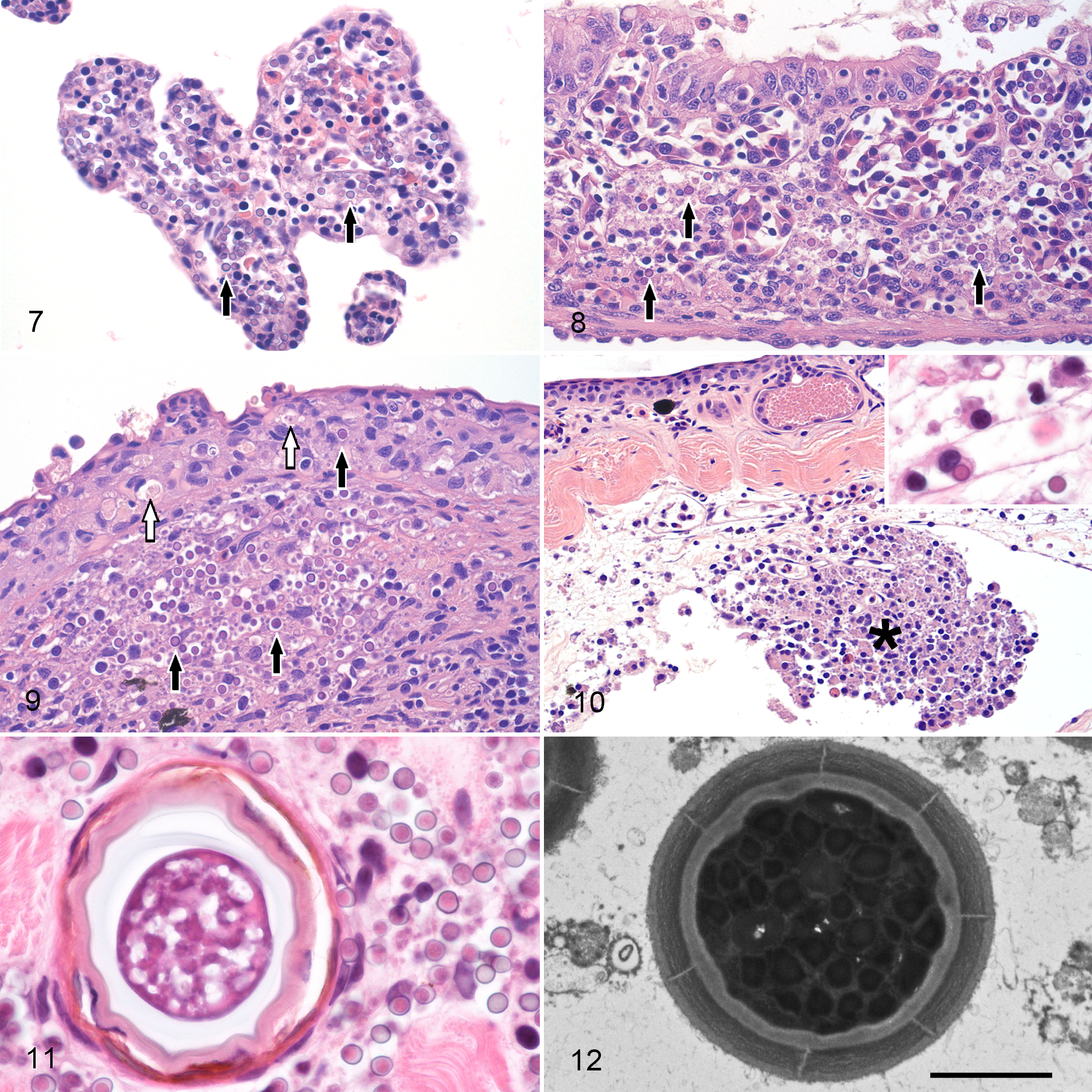
Severe Perkinsea infection.
Intravascular and perivascular, small to extensive infiltrates of Perkinsea-like organisms were observed in the superficial dermis. The epidermis was rarely invaded by Perkinsea-like organisms and presented variable degeneration and necrosis of the affected keratinocytes (Fig. 9). In some tadpoles, the endothelium lining subcutaneous lymphatics was disrupted and replaced by clusters of Perkinsea-like organisms and necrotic debris that were partially obliterating the lumen (Fig. 10). With the same angiocentric pattern, infiltrates of this parasite were also observed in the endomysium and perimysium of the skeletal muscle (Fig. 11), endocardium and myocardium, peritoneum, leptomeninges and choroid plexus of the brain, and very rarely in the capillaries of the neuroparenchyma and vascular layer of the retina (choroid coat). In all these tissues, presence of the organism was associated with variable perivascular to diffuse edema, endothelial activation and loss, as well as degeneration and necrosis of the myocytes, myocardiocytes, mesothelial cells, or choroid cells. The same type of inflammatory infiltrates observed in the gastrointestinal tract and gills was occasionally observed in dermis, skeletal muscle, and peritoneum.
On transmission electron microscopy, hypnospore-like and trophozoite-like organisms ultrastructurally similar to those previously described 20 were seen free in extracellular and virtual necrotic spaces, and intracellularly in hepatocytes, renal epithelial cells, endothelial cells, and within the cytoplasm of macrophages, endothelial cells and epithelial cells (hepatocytes and tubular epithelial cells) (Fig. 12).
Coinfections and Concomitant Pathology
Fifty-nine SPI-affected tadpoles (33.1%) presented concomitant infections. In 37 tadpoles (20.8%), there were normal intestinal burdens of intestinal metazoa including pinworm morphologically compatible with Gyrinicola sp. (33), intestinal cestodes (1) or both (3). Metacercariae encysted in different tissues were observed in 24 tadpoles (13.5%). In 21 tadpoles (11.8%), metacercariae in the mesonephroi were identified as Echinostoma sp., and in 6 tadpoles (3.37%) metacercariae in the dermis were identified as Riberoia ondatrae. Twenty-three (12.9%) presented ectoparasitic infestations with Leptotheca sp. (19), Trichodina sp. (3), or Epistilis sp. (1). Ichthyophonus sp. cysts with no or mild associated granulomatous myositis were observed in 8 tadpoles (4.5%). Seven tadpoles presented mild focal hyperkeratosis of the oral disc with intralesional chytrid-like thalli (zoosporangia) (3.9%). Necrotizing dermatitis and stomatitis caused by Saprolegnia spp. was diagnosed in 4 tadpoles (2.2%). We previously 20 isolated and amplified frog virus-3 DNA in 1 of 44 tadpoles that were tested for this pathogen (2.3%). Myxidium sp. infections were detected in the liver of 2 tadpoles (1.1%). Routine culture of liver (25) or spleen (2) from 27 tadpoles resulted in no bacterial growth indicative of ante-mortem bacterial infection. Deformities of the hind limbs including absence (amelia) or underdevelopment (ectromelia), or extra number of limbs (polymelia), were observed in 3 individuals (1.7%).
Discussion
Since first detected in 1999, SPI events have been increasingly identified as the cause of mass mortalities of tadpoles across the United States 9 ,19, 20,22 and represent, after ranavirosis, the second most frequently diagnosed life threatening infectious disease of tadpoles in the United States. 20 Like ranavirosis, 35 SPI can cause over 95% mortality in tadpole populations and has the potential to drive fragmented frog populations to extinction by substantially reducing recruitment. 17,20
SPI Pathology
Most SPI-affected tadpoles presented with well-developed fat bodies in the coelomic cavity (good body condition), which suggests that the course of SPI is rapid and does not lead to prolonged anorexia or negative energetic imbalance. 20 Eighty SPI-affected tadpoles that were examined lacked gross abnormalities (44.9%). When present, the most common SPI associated gross lesions were hepatomegaly, splenomegaly and nephromegaly. Rarely pancreatomegaly was noticed. Organomegaly corresponded histologically with extensive necrosis and replacement of preexistent tissues by myriads of Perkinsea-like organisms. Tissue necrosis may be caused by the invasion and presumed replication of Perkinsea-like organisms in epithelial cells, including hepatocytes, tubular epithelial cells, pancreatic exocrine epithelium, keratinocytes, enterocytes, branchial epithelium or thyroid epithelium. In some instances, Perkinsea-like organisms were observed in the cytoplasm of endothelial cells. This finding, along with the observation of endothelial loss and thrombosis suggests that vascular damage induced by the protozoa might partially contribute to hypoxic degeneration and necrosis. Eosinophilic granulopoiesis was frequently noticed in kidney, liver, and mesenteric tissues, while lymphopoiesis was mostly observed in kidney. In addition, SPI-affected tadpoles presented with increased numbers of circulating eosinophils (eosinophilia). In immature stages of some frog species, elevated production and deployment of eosinophils might be a physiological change that responds to the high demand of eosinophils necessary for tissue remodeling. 13 The exuberant extramedullary hematopoiesis and eosinophilia observed in many of the examined SPI-affected tadpoles probably concealed underlying mild inflammatory infiltrates. For infected tissues in which the cellular inflammatory response was not obscured by hematopoietic tissue, such as skin, specific inflammatory infiltrates were minimal or absent. When present, inflammatory cells were scattered histiocytes with occasional intracytoplasmic Perkinsea-like organisms, fewer lymphocytes and eosinophils (Inset Fig. 10). Contrary to adult frogs which appear to confine the Perkinsea-like organisms in granulomas, 21 widespread presentation of SPI in tadpoles suggests inability of the tadpole immune system to control the infection. The high susceptibility of tadpoles to SPI might be partially explained by incomplete development of their adaptive immune system or down-regulation of immune responses during metamorphosis. 30 Moreover, environmental and ecological factors such as water temperature might impair immune response of tadpoles to PPC. 4 Nonetheless, mild but efficient granulomatous tissue confinement of metazoan and other intracellular protozoa such as Ichthyophonus sp. observed in SPI-affected tadpoles suggests that tadpoles are capable of mounting an immune response to pathogens and that PPC might use alternative mechanisms to evade the host immune response (Fig. 11).
Two other gross findings observed in SPI were ascites and subcutaneous edema. In SPI, edemas may be partially explained by a decrease of the oncotic pressure secondary to hypoalbuminemia following extensive acute hepatic necrosis. 7 Likewise, acute renal failure secondary to glomerular and tubular damage probably contributes to the formation of edemas by eliciting protein loss, 7 which was observed in this study as eosinophilic protein droplets in the renal tubular epithelium of SPI-affected tadpoles. In addition, edemas might be caused in part by massive sinusoidal and vascular obliteration by Perkinsea-like organisms, which significantly increases sinusoidal and systemic hydrostatic pressure. 7 Endothelial damage and increased vascular permeability may also partially contribute to the formation of edemas and occasionally observed petechial hemorrhages in skin and pancreas.
The predominant protozoal stage identified in natural SPI was the hypnospore-like stage. However, in 7/15 (46.7%) of the SPI-affected tadpoles that presented with less than 60% of liver affected (earlier stages), trophozoite-like organisms were the dominant stage. Unfortunately, most tadpoles collected in passive surveillance studies are in terminal stages of the disease (moribund or dead), which hampers assessment of the disease progression. In the better studied Perkinsus spp. of mollusks, hypnospore life stages are only observed in moribund or dead specimens. 3,27 The transformation from trophozoite to hypnospore is triggered by chemical changes in the decaying tissues such as decreased oxygen tension and pH. 5 Likewise, in SPI, extensive necrosis and multiorgan failure might elicit massive and rapid transformation of the trophozoite-like stage into hypnospore-like stage. Thereafter, the hypnospore-like stage presumably persists under variable conditions in the decaying tissues, in the environment (water or sediments) or in the digestive tract and feces of a paratenic host. To confirm this hypothesis, controlled infections for serial durations followed by light and electron microscopy need to be conducted.
Despite limitations associated with the opportunistic nature of our samples, a logical proposed course of SPI involves infection of the tadpole by zoospores through the oral mucosa and gills, gastrointestinal tract or percutaneous; local replication of trophozoite-like organisms and systemic active or passive migration within leukocytes (most likely monocytes) through blood vessels and lymphatics (Inset Fig. 10) as reported for other Perkinsea such as Perkinsus marinus, which infects and replicates within the hemocytes of infected oysters; 32 and secondary replication in epithelial and endothelial cells of multiple organs causing degeneration and necrosis. Mild histiocytic inflammation and phagocytosis of organisms by local macrophages is probably evaded by these Perkinsea organisms as described for other well-known Perkinsozoa. 32 Extensive disruption of liver and kidney cause hepatic and renal failure, occasionally leading to generalized edema. Edema might also be aggravated by the endothelial damage of blood vessels and lymphatics. Severe pancreatic necrosis likely contributes to death by release of activated proteolytic enzymes and proinflammatory cytokines into the blood stream causing a systemic inflammatory response syndrome and shock. 18 The moribund tadpoles are easily predated or scavenged after death by other tadpoles or paratenic hosts, and hypnospore-like organisms are eventually released into the environment where they zoosporulate under appropriate conditions. Tadpoles that survive initial infection and become adults may successfully contain the parasite in localized granulomas. 21
SPI Case Definition
To have an accurate knowledge of the progression and real impact of emerging diseases such as SPI on frog populations, we must use consistent surveillance protocols and methods. Underdiagnosis of diseases of tadpoles in sites with low herpetological or recreational activity is expected given the rapidity with which predators and scavengers remove sick or dead tadpoles from the environment. 15,20 However, misdiagnosis can represent another major data gap if mortality investigations are based solely on epidemiological information, gross pathology, and molecular tests. Mortality investigations that lack complete pathological examination may incorrectly connect the presence of a pathogenic agent in an individual (infection) with a deleterious health effect in this individual (disease). 10,31
In the particular case of SPI, its epidemiology, clinical presentation and gross pathology overlaps in many aspects with those observed in ranavirosis of anurans in North America (Table 1). Moribund tadpoles from SPI and ranavirosis outbreaks show similar and often unspecific clinical signs suggestive of systemic disease. 24 In addition, external lesions or abnormalities in SPI and ranavirosis are frequently absent 26 or are not perceptible due to postmortem changes. When external changes are observed, in many instances they are similar, namely: abdominal distension, subcutaneous edema, cutaneous erythema and petechia, or patchy pale discoloration of the skin. 24 One or more of these external lesions were observed in 19 (10.7%) SPI-affected tadpoles included in this study. Finally, occurrence of subclinical infections and coinfections with either Perkinsea organisms or frog virus-3 16, 20,22,24 may increase the risk of misdiagnosing these 2 diseases if histopathology is not conducted.
Comparison of the Epidemiology and Pathology of Natural Severe Perkinsea infection (SPI) and Ranavirosis in Tadpoles in North America.
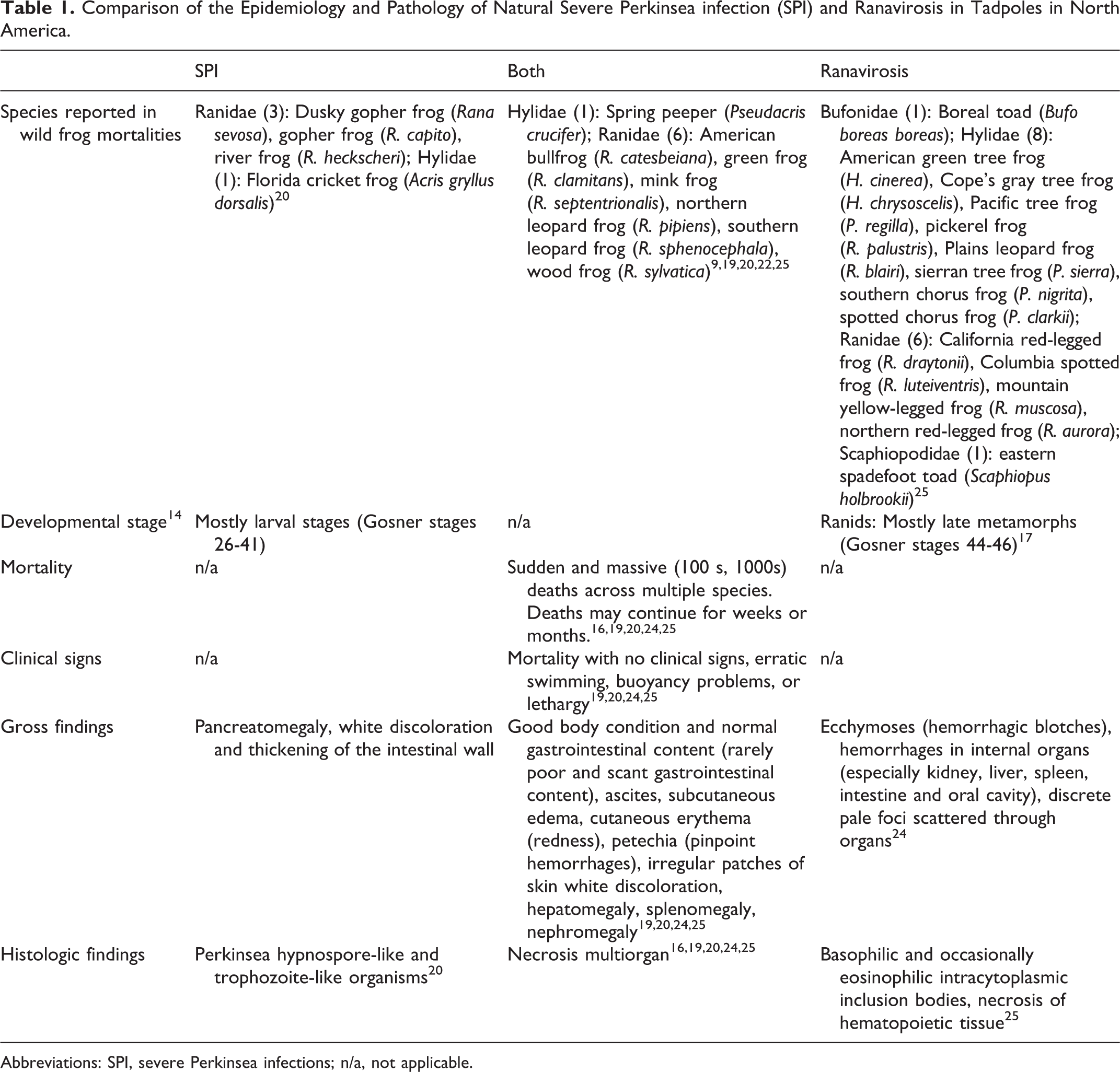
Abbreviations: SPI, severe Perkinsea infections; n/a, not applicable.
Establishing a case definition is 1 approach to standardize SPI diagnostic results, promote consistent reporting and prevent misdiagnosis with either well-known or novel diseases of frogs. With the current information available on SPI, we present a disease definition summarized in Table 2. This definition summarizes background information on this novel pathogen and clinical, gross and histological presentation of the disease as well as specific criteria required for its diagnosis. Based on this case definition, confirmation of SPI as cause of death requires: 1) histological evidence of pathological changes in 1 or more organs with presence of Perkinsea-like organisms; and 2) molecular detection and identification, from at least 1 affected organ, of Perkinsea protozoa belonging to the Pathogenic Perkinsea Clade. 6,20 Finally, in this definition we propose additional terminology to be used when the diagnostic “gold standard” cannot be reached. However, given the early stage of the study of this disease, these criteria might change following development of more specific molecular diagnostic techniques. At the same time, variations in presentation might accompany geographical, environmental, and host expansion of this disease.
Abbreviations: SSU rRNA, small subunit of the ribosomal RNA-encoding gene.
a Potentially worldwide.
b Potentially all year round.
Severe Perkinsea infection is a potentially emerging threat for frogs in North America. Further consistent and active surveillance is warranted to fully assess the impact of SPI on wild populations. In this article, we provide a complete review of the pathology of natural SPI, suggest a plausible pathogenesis sequence, and propose a case definition of SPI to standardized criteria for diagnosis and communications of this disease. This information will also be useful to increase awareness about the importance of broader comprehensive postmortem disease investigations including histopathologic assessment.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818798132 - Pathology and Case Definition of Severe Perkinsea Infection of Frogs
Supplemental Material, DS1_VET_10.1177_0300985818798132 for Pathology and Case Definition of Severe Perkinsea Infection of Frogs by Marcos Isidoro-Ayza, Daniel A. Grear, and Aurélie Chambouvet in Veterinary Pathology
Footnotes
Acknowledgements
We recognize the technical assistance provided by all the personnel of the USGS-NWHC, particularly David Earl Green for conducting necropsies and compiling pathology data; Kimberli Miller and Emily W. Lankau for providing a standardized format for case definitions of wildlife diseases; Jeffrey M. Lorch and Rebecca Cole for their critical revision of the manuscript; and Stephanie Steinfeldt for her long-term assistance on amphibian necropsies. We also recognize the personnel of the USGS-ARMI for their field work over the last 16 years. This is contribution number 620 of the USGS-ARMI.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Aurélie Chambouvet was supported by the project PARASED ANR-16-ACHN-0003. Any use of trade, product, or firm names is for descriptive purposes only and does not imply endorsement by the US Government.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

