Abstract
Mouse kidney parvovirus (MKPV), also known as murine chapparvovirus (MuCPV), is an emerging, highly infectious agent that has been isolated from laboratory and wild mouse populations. In immunocompromised mice, MKPV produces severe chronic interstitial nephropathy and renal failure within 4 to 5 months of infection. However, the course of disease, severity of histologic lesions, and viral shedding are uncertain for immunocompetent mice. We evaluated MKPV infections in CD-1 and Swiss Webster mice, 2 immunocompetent stocks of mice. MKPV-positive CD-1 mice (n = 30) were identified at approximately 8 weeks of age by fecal PCR (polymerase chain reaction) and were subsequently housed individually for clinical observation and diagnostic sampling. Cage swabs, fecal pellets, urine, and blood were evaluated by PCR at 100 and 128 days following the initial positive test, which identified that 28 of 30 were persistently infected and 24 of these were viremic at 100 days. Histologic lesions associated with MKPV in CD-1 (n = 31) and Swiss mice (n = 11) included lymphoplasmacytic tubulointerstitial nephritis with tubular degeneration. Inclusion bodies were rare; however, intralesional MKPV mRNA was consistently detected via in situ hybridization within tubular epithelial cells of the renal cortex and within collecting duct lumina. In immunocompetent CD-1 mice, MKPV infection resulted in persistent shedding of virus for up to 10 months and a mild tubulointerstitial nephritis, raising concerns that this virus could produce study variations in immunocompetent models. Intranuclear inclusions were not a consistent feature of MKPV infection in immunocompetent mice.
Keywords
Mouse kidney parvovirus (MKPV), also known as murine chapparvovirus (MuCPV), is a newly identified infectious agent that was first assembled using a metagenomics approach to assess inclusion body nephropathy in severely immunocompromised laboratory mice. 16 A similar virus was concurrently described by a separate group as part of the fecal virome of wild mice living in New York City, New York. 21 These viruses have since been identified as members of the same virus species, Rodent chaphamaparvovirus, 1,13 which have been observed in wild, laboratory, and pet mice with a worldwide distribution. 7,16,21 In the laboratory animal setting, prevalence of MKPV has been estimated at 5.1%—an estimate based on 178 positive samples of 3517 tested and representing 78 biomedical research institutions over a 7-month period. 7
MKPV contains nucleotide sequences with homology to an ancient lineage of highly divergent viruses with a broad range of host species, including bats, rats, birds, and pigs. 1,11 –13,15,20,23 The novel parvovirus group was provisionally named “chapparvovirus,” based on the acronym ChAP derived from the initially identified hosts, which included chiropteran, 1 avian, 15 and porcine 11 species. However, a significant reorganization of taxonomy within the Parvoviridae family has resulted in the addition of a new viral species, Rodent chaphamaparvovirus 1, to incorporate MuCPV and MKPV. 13 MKPV has remarkable tropism for the renal tubules and produces severe chronic renal disease in severely immunocompromised strains, including NSG (NOD.Cg-PrkdcscidIl2rgtm1Wjl /SzJ), B6.129P2-Cxcr6tm1LittRag1tm1Mom /J, and B6.Cg-Rag1tm1MomTyrp1B-w Tg (Tcra, Tcrb)9Rest/M strains. 7,16,18 Similarly, other chapparvoviruses can be found in the kidney of their hosts; 7 however, tropism for kidney has not been confirmed for these viruses and disease associations have not been described in species other than mice.
MKPV is genetically divergent from previously known mouse parvoviruses, which include minute virus of mice and mouse parvovirus. 14,16,21 Additional murine pathogens that can result in renal intranuclear inclusion bodies include adenovirus, cytomegalovirus, and polyomavirus. 8,14 Inclusion body nephropathy in laboratory mice has been sporadically observed and described by veterinary pathologists for decades; 2,9,14,18 however, previous efforts to determine the etiology were undermined by inconclusive ultrastructural features and a failure to detect known mouse pathogens. Following the discovery of the MKPV sequence in mouse kidneys affected by inclusion body nephropathy, a causal relationship between the virus and the disease was confirmed by fulfilling Fredricks and Relman’s criteria. 6,16
It is widely recognized that the use of severely immunocompromised mice can increase the risk of complications from pathogens that are considered incidental in immunocompetent mice. 5 CD-1 and Swiss Webster mice are stocks of genetically uncharacterized, outbred mice. Compared with inbred strains, outbred mice have increased phenotypic diversity, generally have increased reproductive performance, disease resistance, and are less expensive to produce. 3,4 While immunocompetent mice are generally considered less susceptible to infectious diseases, subclinical infections in immunocompetent mice can negatively influence research goals and colony management. With the increased use of severely immunocompromised mice in biomedical research, pathogens that are normally inconsequential can pose substantial complications.
This study evaluated mice naturally infected with MKPV in a research setting. We aim to describe the histologic lesions associated with MKPV in immunocompetent mice, evaluate shedding and persistence of MKPV infection in immunocompetent animals, and compare diagnostic sampling methods for the detection of MKPV.
Material and Methods
MKPV in NSG Mice and Patient-Derived Xenograft Material
MKPV was identified at Frederick National Laboratory (FNL) following histologic observations in NOD.Cg-Prkdcscid Il2rgtm1Wjl /SzJ (NSG) mice (n = 5) of severe renal disease with intranuclear inclusions in renal tubules between December 2018 and January 2019. These mice had been implanted with cryopreserved tumor fragments from patient-derived xenograft (PDX) material obtained from an outside biomedical research institution and mice were undergoing quarantine in a negative-pressure semi-rigid isolator located in a dedicated quarantine animal facility. Ten isolators were present in the affected building. The NSG hosts originated from a dedicated FNL breeding facility at the Biological Testing Branch of the National Cancer Institute, which is in a dedicated, stand-alone building with dedicated staff. Following the initial cases of MKPV in the quarantined PDX hosts, the breeding facility was tested and found to be negative for MKPV by PCR on fecal pellets from the cage floor, kidney tissue, cecal contents, and environmental samples; mice were also submitted for pathology evaluation.
Quantitative Polymerase Chain Reaction (PCR)
A quantitative PCR assay was developed for MKPV viral DNA detection. The primers and probe sequences were designed to target the nonstructural protein NS1 region of the virus. Sequences were obtained from GenBank (https://www.ncbi.nlm.nih.gov/genbank/) with 100% match to Memorial Sloan Kettering Cancer Center (GenBank: MH670588) and Centenary Institute strains (GenBank: MH670587). 16 DNA was extracted from feces, urine, swab of wet and dirty bedding, blood, formalin-fixed paraffin-embedded tissue, or xenograft tumor fragment samples using Qiagen DNA mini kits. The real-time PCR was performed using forward primer: 5′-GAG GTG GTC GGA TCG GTT T-3′, reverse primer: 5′-GGG ACA GAG GTT CCG AGA-3′, and probe: 5′-FAM- AGC TTT GTC TAA GGT TTG –MGB-3′. Cycling conditions were as follows: 95 °C for 10 minutes, followed by 45 cycles at 95 °C for 15 seconds, and annealing/extension at 60 °C for 1 minute. The fluorescence output signal was measured during the extension step. The copy number was calculated by interpolation against a standard curve of known copy number. Cutoff Ct values were determined by the standard curve of known copy number of the targets that could be confirmed by gel end point assay with a detectable band.
MKPV Surveillance of Animal Facilities
PCR diagnostic testing was performed at NCI facilities in Bethesda and Frederick. Between the period of January 3, 2019, and January 28, 2020, 8944 specimens were submitted for MKPV testing to the Animal Diagnostic Laboratory at FNL, which included both direct colony testing and sentinel testing of dirty bedding. For comparison of MKPV positivity in different sample types from individual mice, cecal contents, and kidney tissue were submitted for PCR testing following necropsy from 899 sentinel mice. Sentinel mice included Crl: CFW(SW) Swiss Webster and Crl: CD1(ICR) CD-1 mice. To compare other diagnostic samples, multiple diagnostic specimens were compared from the same time points and cages by PCR; these specimens included fecal pellets from the cage, swabs of wet and dirty bedding, urine, and/or renal and urinary bladder tissues, and cecal content.
CD-1 Study Populations
A cohort of 33 female, Crl: CD1(ICR) (CD-1) mice that were commercially bred at Charles River Laboratories were identified by cage-pooled fecal PCR to be positive for MKPV from a single building at FNL. All mice were born between January 2, 2019, and February 12, 2019, and arrived at a dedicated mouse modeling transgenic core facility at 35 and 42 days of age as recipients for embryo transfer. The embryo transfer procedure occurred at 49 to 56 days of age. Positive MKPV PCR tests of cage-pooled fecal material was observed for these mice between 48 and 76 days of age; no previous MKPV testing was performed. After the pups were weaned, the recipient CD-1 mice from MKPV positive cages were transferred to individual housing for further evaluation (n = 30) or euthanized for PCR of renal tissue and histopathology (n = 3). Individually housed CD-1 mice were maintained in a specific pathogen free animal facility, which excluded the following agents: mouse parvoviruses 1–5, mouse hepatitis virus, sendai virus, lymphocytic choriomeningitis virus, hantaan virus, K virus, mouse adenovirus 1 and 2, ectromelia virus, minute virus of mice, reovirus type-3, mouse theilovirus (Theiler’s murine encephalomyelitis virus, strain GDVII), pneumonia virus of mice, murine cytomegalovirus, mouse rotavirus (epizootic diarrhea of infant mice), mouse thymic virus, polyoma virus, Clostridium piliforme, cilia-associated respiratory bacillus, Spironucleus muris, Citrobacter rodentium, Helicobacter species, Mycoplasma pulmonis, ectoparasites, endoparasites, and pathogenic bacteria (other than Pseudomonas aeruginosa, Klebsiella pneumoniae, and Klebsiella oxytoca). Nonpathogenic protozoans consistent with Entamoeba muris and flagellated protozoa (eg, trichomonads) have occasionally been found in sentinel mice from this animal facility. Testing for Helicobacter spp. has been historically negative in this facility. Animal health monitoring was based on dirty bedding transfer sentinels and direct colony testing. These CD-1 mice were housed in a limited access barrier facility in which personnel wear dedicated clothing in addition to disposable personal protective equipment, including head cover, mask, and gloves, all required prior to entering the animal facility. All mice were housed in static microisolator cages that were changed in biosafety cabinets. All animals werehoused on autoclaved corn cob bedding and received an autoclaved diet and reverse osmosis water.
To compare diagnostic sampling for MKPV, fecal pellets and dirty cage swabs were tested by PCR at 100 (n = 30) and 128 days (n = 30) after the initial positive PCR test. At these time points, blood and urine were also collected for evaluation by PCR for all 30 CD-1 mice. From each mouse, 6 to 10 fecal pellets were collected from the cage using disposable forceps and disposable gloves. For urine samples, 100 to 200 µl was collected using LabSand (Coastline Global Inc), a nonabsorbent bedding, to facilitate urine collection and to prevent fecal contamination. To ensure adequate hydration while housed on LabSand, mice were provided HydroGel (ClearH2O). Cage swabs were passed through the dirty bedding including the wet corners when present. Blood samples were collected via submandibular method. Clinical signs were not observed. CD-1 mice were submitted for scheduled humane euthanasia via carbon dioxide followed by necropsy, MKPV PCR of renal tissue, and histopathology between 225 and 274 days of age (n = 10; 177 or 198 days post initial positive test) and between 366 and 394 days of age (n = 20; 318 or 319 days post initial positive test). Complete blood counts (CBC) and serum chemistry were evaluated for mice submitted between 225 and 274 days of age using a VetScan VS2 Chemistry analyzer (Abaxis) and a HEMAVET950 (Erba Diagnostics). References ranges for mice were derived from available inbred strains data accessed from the Mouse Phenome database for female mice (https://phenome.jax.org/search/). In addition, 3 MKPV-positive CD-1 mice from the initial cohort described above were submitted directly to necropsy at 99 or 100 days of age, 11 days following the initial positive MKPV test. All experimental procedures were approved by the FNL Institutional Animal Care and Use Committee. Research was conducted in compliance with the Public Health Service Policy on Humane Care and Use of Laboratory Animals and other federal statutes and regulations relating to animals and experiments involving animals. The facilities where this research was conducted are fully accredited by AAALAC, International.
Pathology, RNA In Situ Hybridization, and Immunohistochemistry
Thorough necropsies were performed including evaluation of all organ systems for CD-1 mice (n = 33). In addition, aged Crl: CFW(SW) Swiss Webster sentinel mice with a history of positive MKPV PCR tests (n = 11) and negative age-matched controls (n = 6) were submitted for histopathology. Both kidneys were bisected longitudinally. One longitudinal section of renal tissue was submitted for MKPV PCR testing for all CD-1 and Swiss Webster mice to confirm MKPV status; the other 3 sections were submitted for pathology. All organs were fixed in 10% neutral buffered formalin for 72 hours and tissues were routinely processed for histology. Tissues evaluated for this study included both kidneys as a minimum; additional tissues submitted included liver, gall bladder, lung, spleen, urinary bladder, and skin. For 10 CD-1 mice, kidney, urinary bladder, heart, liver, pancreas, salivary gland, glandular and nonglandular stomach, duodenum, jejunum, ileum, cecum, and colon were examined histologically.
Kidney sections were evaluated for inclusions and graded by 3 board-certified veterinary pathologists (EFE, LLB, NMP) for inflammatory infiltrates and tubular changes. Inflammatory infiltrates were graded as follows: 0, within normal limits; 1, minimally increased inflammatory infiltrates composed predominantly of lymphocytes and plasma cells often focally forming 3 to 5 cell thick perivascular cuffs; 2, mildly increased inflammatory infiltrates that were multifocal; 3, moderately increased inflammatory infiltrates that form thick perivascular cuffs multifocally and were prominent with the 4× objective; 4, markedly increased inflammatory infiltrates that widely separated vessels from adjacent renal parenchyma. Tubular changes were graded as follows: 0, within normal limits; 1, minimal tubular degeneration that was often focal; 2, mild tubular degeneration present with multifocal regions containing evidence of tubular degeneration and regeneration and focal necrosis; 3, moderate tubular changes containing tubular degeneration and multifocal necrosis; 4, marked multifocal tubular changes including tubular necrosis. Grading was based on the most severe kidney section. Intranuclear inclusions were noted if observed. Karyomegalic cells were defined using hematoxylin-eosin (H&E)-stained sections as cells containing nuclei at least 2× larger than adjacent normal renal tubular epithelial nuclei.
For MKPV RNA in situ hybridization, longitudinal sections of kidney (CD-1, n = 9; NSG, n =1); lung, liver, gall bladder, spleen, and xenograft tissue (NSG, n = 1); and liver, gall bladder, pancreas, salivary glands, nonglandular and glandular stomach, small intestinal gut roll (closed, including ingesta), and cecum colon, and rectum gut roll (closed, including ingesta; CD-1, n = 5) were routinely processed and embedded. Five-micrometer-thick sections were deparaffinized twice in xylene for 5 minutes each and then twice in 100% ethanol for 3 minutes each. Tissue pretreatment consisted of boiling 15 minutes in 1X Target Retrieval Reagent (ACD) followed by a 30-minute Protease Plus (ACD) incubation at 40 °C. MKPV detection by chromogenic in situ hybridization (CISH) was performed with the RNAscope 2.5 LS Probe-V-MKPV-Cap (ACD #571638), a 20 “ZZ” probe targeting 1996-3085 of GenBank accession number MH670588.1, using the manual RNAscope 2.5 HD Assay-RED (ACD #322350). Mus musculus peptidylprolyl isomerase B (Ppib) RNAScope probe was used as a positive control, and the Bacillus subtilis dihydrodipicolinate reductase (dapB) RNAScope probe was used as negative control. Known positive tissues included PCR-positive MKPV infected renal tissue from a female NSG mouse. Dual RNAScope and Iba-1 immunohistochemistry were performed on liver sections. Following RNAscope, a rabbit polyclonal, anti-Iba-1 (Biocare, #CP 290, 1:500 dilution) was used for chromogenic immunohistochemistry on liver sections following 20 minutes of heat-induced epitope retrieval in a citrate buffer. A Bond Polymer Refine detection kit (Leica #DS9800) was used minus the post primary reagent. Positive controls for Iba-1 included normal mouse spleen; negative controls involved the replacement of the anti-Iba1 antibody with a nonspecific antibody of the same species and isotype (Abcam #ab37415).
Transmission Electron Microscopy
Tissue sample preparation for electron microscopy involved removing 2-mm cores from formalin-fixed paraffin-embedded kidney blocks, de-paraffinizing, re-hydrating, osmicating, dehydrating, and re-embedding in Epoxy resin. 10 De-paraffinizing was accomplished as follows: immersion of tissue cores in 1 ml of 100% xylene at room temperature for 15 minutes twice, followed by immersion in 1 ml of 100% ethanol at room temperature for 5 minutes twice at which time samples were allowed to air dry for 15 minutes. Kidney tissues were then fixed in sodium cacodylate buffer (0.1 M, pH 7.4) containing formaldehyde (4%, v/v) and glutaraldehyde (2%, v/v) for 2 hours. Post fixation was made in 1% osmium tetroxide (1%, v/v) in same buffer for 1 hour. The tissues were en bloc stained in 0.5% (w/v) uranyl acetate in acetate buffer (0.1 M, pH 4.5) for 1 hour followed by ethanol dehydration (eg, 35%, 50%, 70%, 95%, 100%) and propylene oxide. Epoxy resin infiltration was made in an equal mixture of resin and propylene oxide overnight. The tissue was embedded in a pure epoxy resin in a plastic mold and cured in 55 °C oven for 48 hours; 70 to 80 nm thin sections were cut with an ultramicrotome and a diamond knife. Thin sections were mounted on copper grids and stained in uranyl acetate and lead citrate. The grids were examined and imaged in an electron microscope operated at 80 kV and digital images were taken by CCD camera.
Statistical Analyses
Comparisons of histologic grades were made between age groups and between MKPV-positive mice and age-matched control MKPV-negative mice. Results are expressed as mean + the 95% confidence interval, and comparisons were made by one-way ANOVA to evaluate the effects of time post-exposure on renal histologic lesion severity in MKPV-positive mice and multiple ANOVA to include infection status and mouse stock as independent variables in MKPV-positive mice and age-matched controls. All statistical analyses were performed in the R environment (http://cran.r-project.org).
Results
MKPV in Immunocompromised Mice
The initial cases of MKPV were identified by histologic observation of severe renal lesions with abundant intranuclear inclusion bodies in NSG mice; affected mice had been implanted with cryopreserved PDX material obtained from an outside biomedical institution. Unused vials containing tumor fragments from this model were subsequently submitted for PCR testing and found to be positive for MKPV. The NSG breeding facility—located in a separate building at FNL—was found to be negative for MKPV by PCR on samples of fecal pellets from the cage floor, kidney tissues, cecal contents, and environmental samples. Only mice cohoused in the 2 isolators or that shared the anesthesia induction box with the initial PDX model NSG mice were found to be MKPV-positive. The other 8 isolators, housing NSG mice implanted with different xenografts, were found to be negative for MKPV by PCR.
NSG mice developed severe renal lesions with pitting of the kidney (Fig. 1a). Chromogenic in situ hybridization (ISH) for RNA encoding MKPV capsid protein revealed widespread labeling throughout all regions of the kidney (Fig. 1b). Histologic lesions included severe tubular degeneration, necrosis, and loss; papillary necrosis; and karyomegalic tubular epithelial cells with prominent intranuclear inclusion bodies (Fig. 2a) that were positively labeled by ISH (Fig. 2b). Cores were taken from formalin-fixed paraffin-embedded kidney for transmission electron microscopy from one severely affected NSG mouse. Inclusion bodies were composed of flocculent, electron-lucent material (Fig. 3); characteristic viral ultrastructure was not observed (Fig. 4). The spleen, liver, and lungs from the same severely affected NSG mouse were also evaluated for the presence of viral RNA using chromogenic ISH. Although no labeling was observed in the spleen or lungs, the liver contained multifocal ISH-positive cells consistent with hepatocytes; labeling was cytoplasmic and punctate. Double-labeling using MKPV ISH and Iba1 immunohistochemistry revealed no labeling of Iba1-positive cells (ie, Kupffer cells; Fig. 5) and ISH labeling for MKPV was predominantly cytoplasmic in cells consistent with hepatocytes.
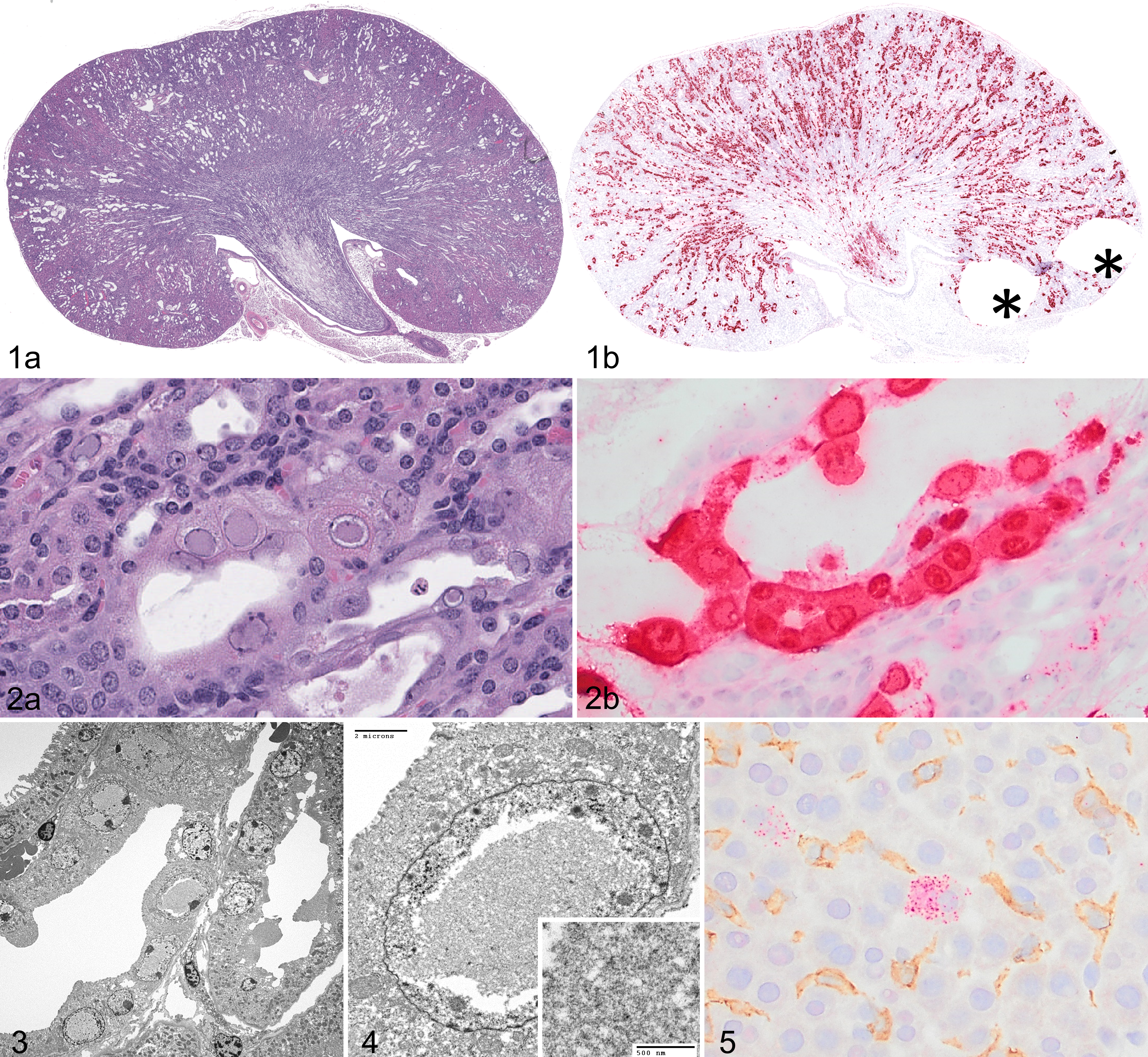
Mouse kidney parvovirus (MKPV), kidney, NSG Mouse.
MKPV in Immunocompetent Mice
Thirty-three female CD-1 mice were identified from MKPV positive cages between 48 and 76 days of age; 3 of these mice were euthanized for renal tissue PCR and histopathology and the remaining 30 were individually housed for evaluation of disease progression, diagnostic sampling, and pathologic characterization (Fig. 6). All mice were clinically normal throughout the duration of the study based on the visual assessment of their appearance, posture, activity, and body condition. MKPV infection was identified in the first 3 CD-1 mice by PCR testing of renal tissue. At 100 days following the initial positive PCR test, testing for MKPV in urine, blood, fecal pellets, and cage-swab samples were compared using PCR; this procedure was repeated at 128 days following the initial positive PCR test and confirmatory PCR was performed on renal tissue at the time of euthanasia.
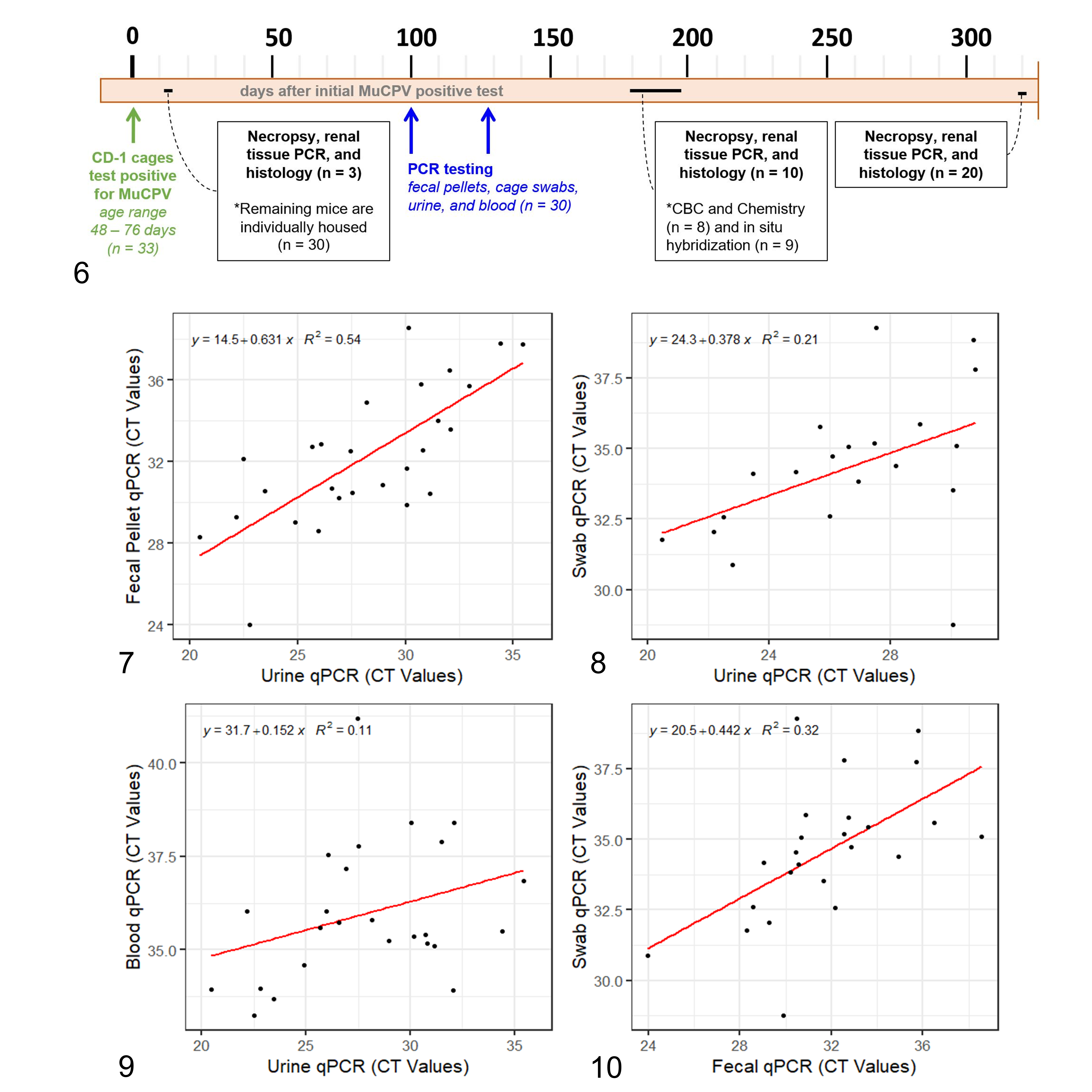
Timeline of CD-1 mouse studies and diagnostic sampling, starting at the initial positive test for mouse kidney parovirus (MuCPV).
After the initial positive PCR test, 28 of 30 (93%) mice were found to be persistently positive for MKPV by urine testing (Supplemental Table S1). At the 100-day time point, 27 mice were identified as positive by urine testing by PCR; of these, 26 (96%) had positive fecal pellets, 22 (81%) had positive cage swabs, and 24 (89%) had positive blood samples. Cycle threshold values for qPCR were positively correlated for all sample types (Figs. 7–10); the strongest correlation was observed between data for fecal pellet and urine samples at this time point (Fig. 7). Testing at 128 days following the initial PCR test produced similar results; however, one previously MKPV-negative mouse was detected as positive (Supplemental Table S1). Out of the 30 CD-1 mice individually housed, 2 mice (7%) were negative for all tests, including urine, blood, cage swab, and fecal pellets tested at both time points and renal tissue tested at necropsy.
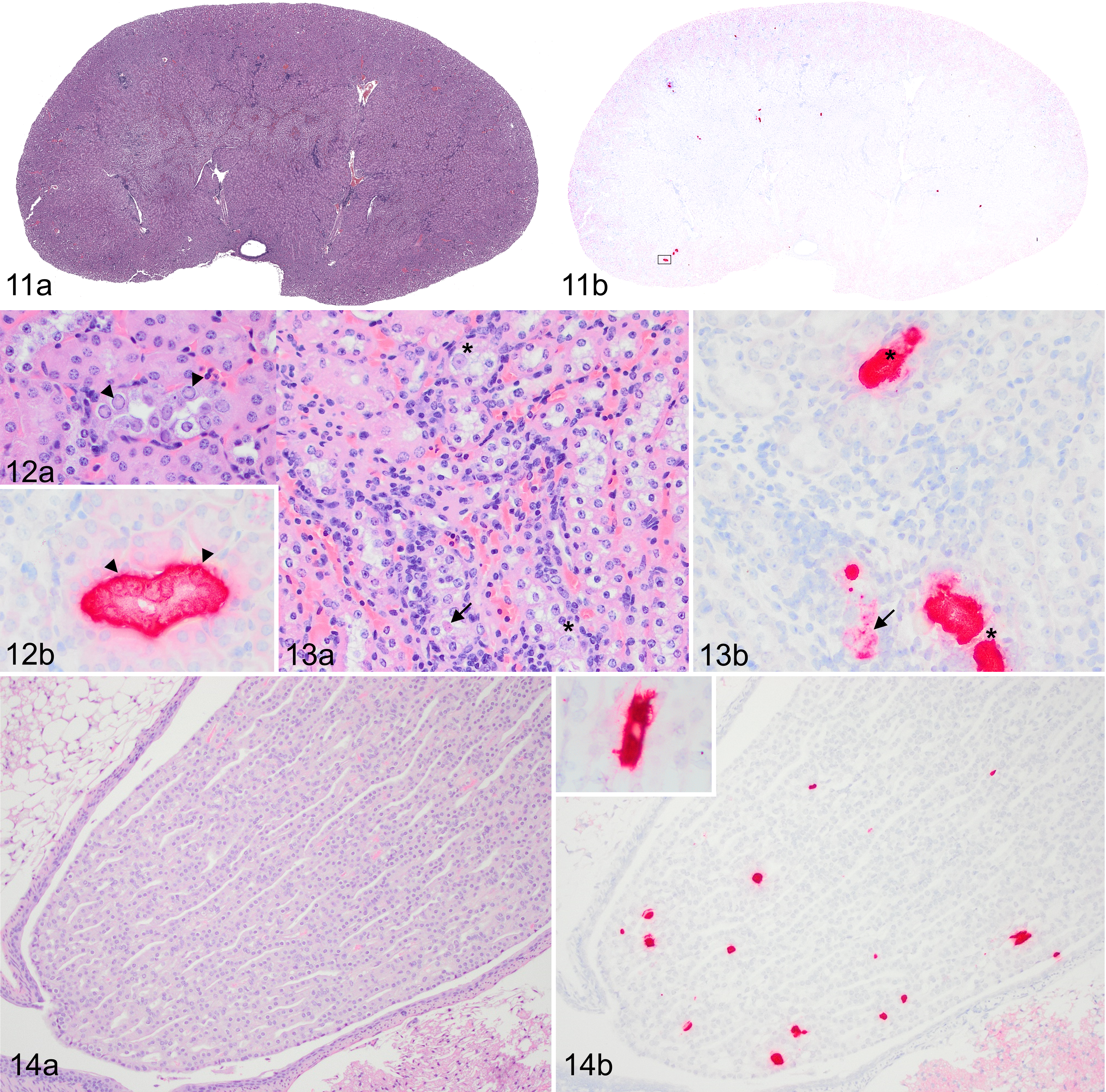
Mouse kidney parvovirus (MKPV) infection, kidney, CD-1 mouse.
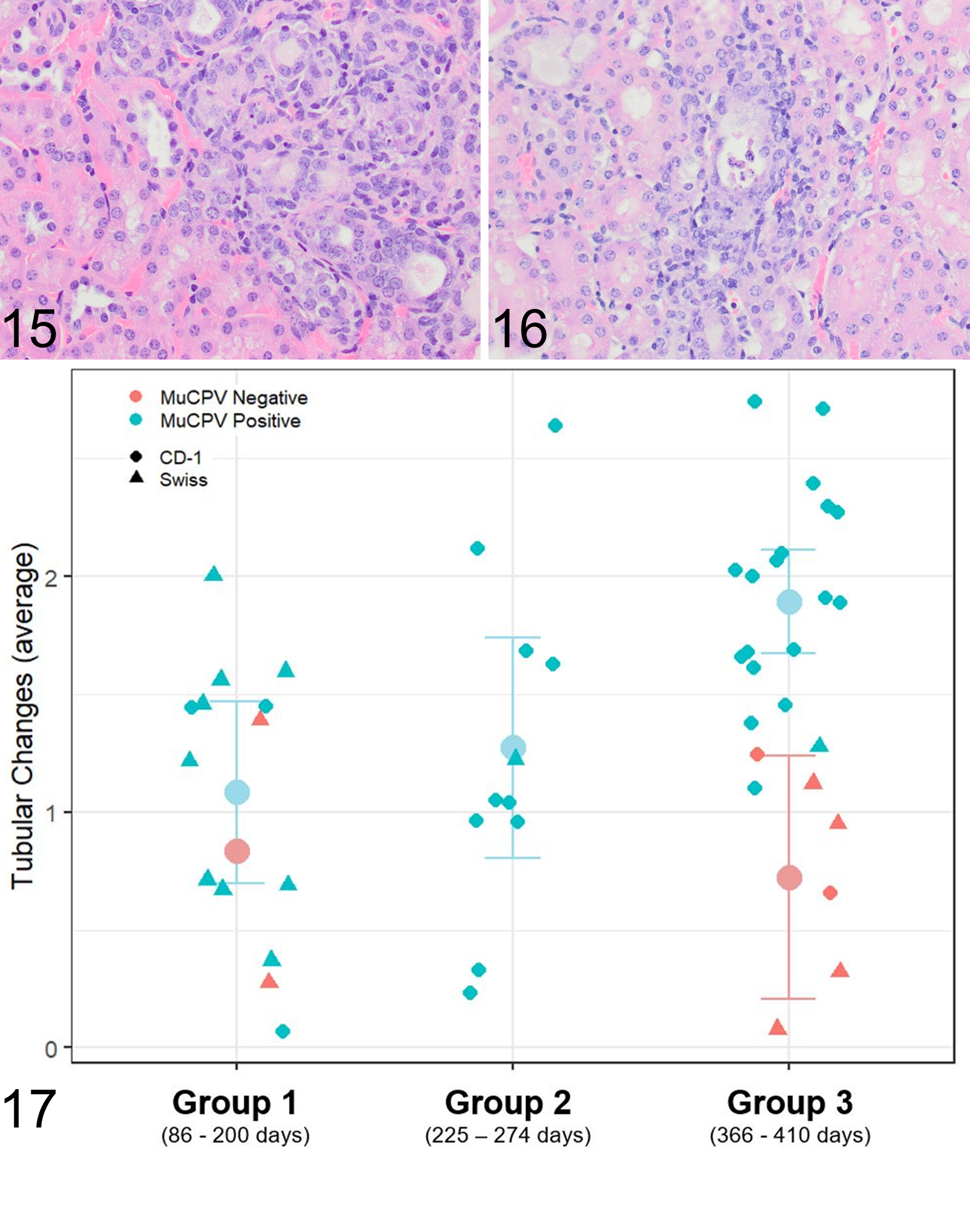
CD-1 mice were submitted for necropsy and histopathology in 3 age groups (Fig. 6). Significant gross lesions were not observed. CBC and serum chemistry (n = 8) were evaluated for mice from age group 2, 177 or 198 days after the initial positive MKPV test, and no consistent changes were observed. Serum urea and creatinine were within normal limits. In persistently MKPV-infected mice, mild to moderate interstitial nephritis was observed in 29 of 31 (94%) CD-1 mice and 8 of 11 (73%) Swiss Webster sentinel mice (Supplemental Table S2). Histologic renal lesions included inflammatory infiltrates in the renal cortical interstitium (Fig. 11a) and chromogenic ISH labeling for MKPV RNA was often sparse and multifocal in the renal cortex (Fig. 11b). Pale eosinophilic inclusion bodies marginating chromatin were observed for 1 CD-1 mouse (Fig. 12a); serial sections of the tubule containing inclusion bodies was strongly positive for MKPV RNA by ISH (Fig. 12b). Tubules displayed degenerative changes characterized by cell swelling with increased vacuolation and karyomegalic cells (Fig. 13a), which were positive for MKPV RNA by ISH on serial section (Fig. 13b). Histologically normal renal papilla (Fig. 14a) also had multifocal MKPV ISH labeling within collecting ducts (Fig. 14b). Tubular changes were minimal (Fig. 15) to mild (Fig. 16), and histologic grades for tubular changes increased with time post-exposure for CD-1 mice (P ANOVA < .001; Fig. 17). Inflammatory cells formed mild (Fig. 18) to moderate (Fig. 19) cuffs in the perivascular spaces, separated tubules in the cortex and corticomedullary junction, and were composed predominantly of lymphocytes and plasma cells with fewer neutrophils. Histologic grade for inflammatory infiltrates increased with duration of MKPV infection for CD-1 mice (P ANOVA = .006; Fig. 20). MKPV RNA was not typically observed by ISH in regions containing inflammatory infiltrates. Histologic grades for inflammatory infiltrates (P ANOVA = .007) and tubular changes (P ANOVA = .004) were significantly elevated in MKPV-positive mice compared to control mice.
Mouse kidney parvovirus (MKPV) infection, kidney, Swiss Webster mouse. Renal tubules are lined by vacuolated epithelial cells (renal tubular degeneration).
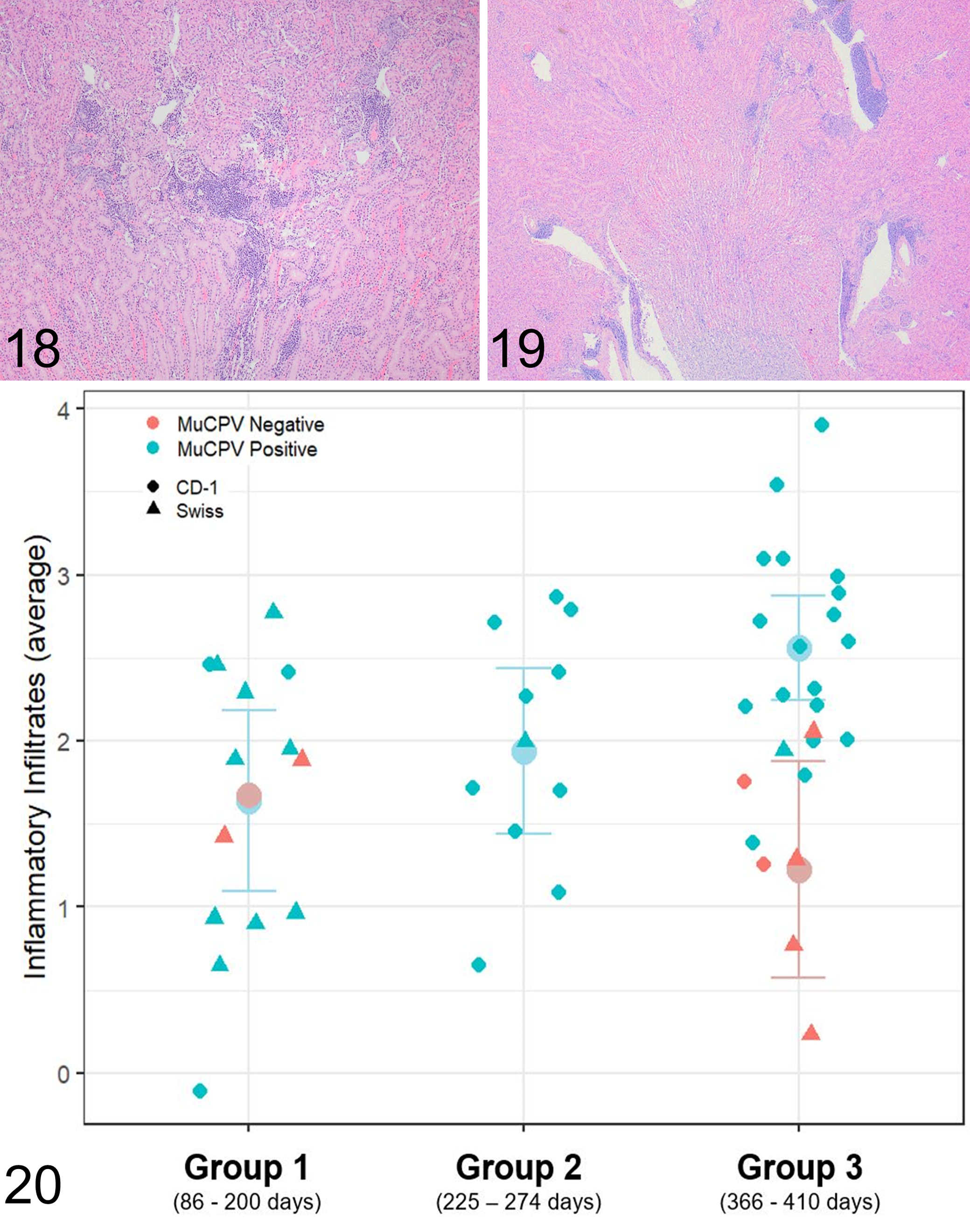
Mouse kidney parvovirus (MKPV) infection, kidney, CD-1 mouse. Mild inflammatory infiltrates expand the renal interstitium. HE.
MKPV Surveillance in Animal Facilities
Prevalence of MKPV in Bethesda NCI was evaluated over 5 separate vivarium facilities. Two different testing modalities were performed: (1) direct colony testing of 10 randomly selected colony cages per rack (pooled) and (2) sentinel testing of dirty bedding. For direct colony PCR testing, 1.4% (4/294) of racks were found to be positive. For sentinel testing, 16.6% (26/157) of racks were observed to be positive. From the 400 submissions with multiple diagnostic samples submitted for simultaneous evaluation by PCR (fecal pellets and either swabs of dirty bedding, kidney, or bladder), 37 were positive for at least one specimen. Concordance was observed for 17 of 37 (46%) samples, in which both the fecal pellets and additional sample were positive for MKPV. Of the 20 cases with discordant results, 10 cages with positive fecal pellets contained mice with either negative kidney (n = 4) or negative bladder (n = 6) tissue. The other 10 cages had negative fecal pellets and either positive dirty bedding (n = 3) or mice with positive kidney tissue (n = 7). Thus, among the 37 cages believed to be positive, negative PCR results were observed in 4 of 24 (16%) kidney tissue samples, 10 of 37 (27%) fecal pellets, and 6 of 8 (75%) urinary bladder samples. Dirty bedding samples consistently produced positive PCR results (n = 5) for any cage associated with an MKPV-positive result from a different sample.
For 899 individual mice, both cecal content and renal tissue were submitted for MKPV PCR testing. For 825 mice (91.8%), both specimens were negative. For 74 samples (8.2%), at least one specimen was positive as follows: both kidney and cecal content were positive (41 of 74; 55%), only kidney was positive (26 of 74; 35%), or only cecal contents were positive (7 of 74; 10%).
Distribution of MKPV Viral RNA in Tissue Sections
By PCR testing in both NSG and CD-1 mice, MKPV was consistently detected in the kidney, liver, and cecal contents (n = 3 per strain). Because cecal contents were found to be PCR-positive, we evaluated a range of tissues associated with the digestive tract to determine whether additional tissues may serve as a reservoir for replication and shedding via the feces. Using chromogenic ISH, we evaluated salivary glands, glandular and nonglandular stomach, pancreas, liver, gall bladder, duodenum, jejunum, ileum, cecum, and large intestine. Labeling was not observed in any tissues other than the kidney in CD-1 mice (Supplemental Table S3).
Discussion
MKPV is widespread in mouse populations, and we show that infections in immunocompetent animals in the laboratory setting often persist for over 6 months. In female CD-1 mice persistently infected with MKPV, clinical signs were not observed and serum chemistry revealed no elevations in urea or creatinine despite shedding of virus for up to 10 months post-exposure. Histologic findings were consistently observed in the kidneys of CD-1 and Swiss Webster mice infected with MKPV and lesion severity increased with time post-exposure for CD-1 mice. For Swiss Webster mice, duration of MKPV infection was not investigated with serial MKPV PCR tests in a consistent manner, so it is unclear whether lesions increase with severity based on increased time post-exposure. Lesions of tubular degeneration and necrosis were associated with MKPV RNA by in situ hybridization in CD-1 mice.
The histologic findings in CD-1 and Swiss mice associated with MKPV ranged from minimal to moderate lymphoplasmacytic interstitial nephritis with minimal to mild degeneration of tubular epithelium and rare intranuclear inclusion bodies. Although MKPV has a high prevalence in wild and pet mice, 7,21 the histologic lesions in these populations have not previously been described. Intranuclear inclusion bodies along with lymphoplasmacytic infiltrates have been previously reported in immunocompetent laboratory mice, including sentinel Swiss Webster 16 and ICR mice; 2 however, the cause was not determined 2 or there were small numbers of mice with limited evaluations. 16 We found that inclusion bodies are not a common feature of MKPV infection in immunocompetent mice and, therefore, MKPV should be considered as a potential differential diagnosis for interstitial nephritis in mice despite the absence of inclusion bodies.
The current study described mice infected with a virus of the species Rodent chaphamaparvovirus 1, which includes 2 viruses: MKPV, initially described in laboratory mice with renal lesions, 16 and MuCPV, a virus initially described as part of the fecal virome of wild mice. 21 Revised taxonomy for the family Parvoviridae has recently been approved by the International Committee on Taxonomy of Viruses, 13 which includes significant reorganization for viruses previously described as “Chapparvoviruses.” Sequencing would be required to distinguish between MKPV and MuCPV.
One limitation of this study is that our observations were restricted to female mice from only 2 immunocompetent stocks of mice: CD-1 and Swiss Webster. The mice used for this analysis were not obtained for the purpose of MKPV evaluation. The female CD-1 mice were initially utilized as recipients for embryo transfer and the Swiss Webster mice were initially sentinel mice for the NIH animal health program. Although the initial FNL cases of MKPV were found in NSG mice associated with cryopreserved tumor fragments obtained from an outside university, there was no plausible link to the CD-1 mice. Rather, 2 more likely possibilities are that (1) the CD-1 mice were infected with MKPV prior to arrival from the vendor or (2) the CD-1 mice acquired the infection in our facility from routine husbandry procedures between the time of arrival and the detection of MKPV by the first PCR performed, 2 to 5 weeks later. The CD-1 mice that were used in this study arrived at our facility when they were 5 to 6 weeks of age. Within 1 to 2 weeks of their arrival these mice were used as recipient moms for the embryo transfer procedure by the FNL mouse modelling core. The embryo transfer procedure was an unlikely source of contamination as the embryos were washed prior to implantation.
We have shown that MKPV was present in the urine and feces of immunocompetent mice. Roediger et al reported 2 immunocompetent exposure experiments with inconsistent results: a co-housing experiment which resulted in MKPV detection in feces but not urine, and a bedding-transfer experiment which resulted in detection of MKPV in urine. 16 Here we show that immunocompetent CD-1 mice had productive MKPV renal infections with viral nucleic acid persistently detectable in the urine, blood, and feces.
While fecal pellets were an effective sample for screening MKPV-positive cages, disease surveillance at NCI found that no single sample type identified all positive cages. This was likely related to stage of infection and individual variability. Sentinel samples of dirty bedding-tested for MKPV by PCR demonstrated discordance in positivity when multiple samples were submitted from the same cage and that cage-swabs of wet and dirty bedding identified more positive cages than fecal pellet sampling alone. Therefore, while diagnostic strategies to test for MKPV in the laboratory animal setting are still evolving, testing moist fecal pellets together with a swab or samples of urine-soaked bedding from live animals and testing renal tissue from sentinel animals submitted for necropsy may represent the best surveillance methods for MKPV. When positive cages are present, repeated screening of MKPV-negative cages would be necessary to ensure their negative status.
MKPV-positive samples from CD-1 mice were detected for over 10 months following the initial MKPV-positive PCR test, suggesting that even immunocompetent mice may not clear the MKPV infection. Also, not all the CD-1 mice potentially exposed to MKPV became persistently infected when individually housed, which suggests there is variability in susceptibility to renal infection following MKPV exposure.
Parvoviruses have been shown to have immunomodulatory properties in mice, 22 which may affect the reproducibility of research involving mice. We show that MKPV produces chronic, subclinical kidney disease in immunocompetent mice, and although less severe than inclusion body nephropathy in immunocompromised mice, such infections in immunocompetent mice have the potential to compromise the interpretation of data. Furthermore, persistently infected mice may serve as a source of infection of other strains. Advanced MKPV infections in severely immunocompromised mice have been associated with expansion of activated macrophage populations. 16 Complete blood counts were evaluated for the persistently MKPV-infected mice in this study and were within normal limits for CD-1 mice; other measurements of immune system function were not performed.
Given uncertainties surrounding the long-term effects on colony health and research results associated with MKPV infection, deciding whether to exclude MKPV will depend on the individual needs of each research program. Typical parvoviruses are generally difficult to remove from the environment; 17,19 however, data on the environmental stability of MKPV—a distantly related, atypical parvovirus—is not currently available. At this time, most commercial vendors do not claim to supply MKPV negative animals, which may pose an additional challenge for institutions seeking to exclude or eradicate MKPV.
MKPV RNA was not observed in tissues other than the kidney in CD-1 mice by ISH despite frequent positivity of cecal contents in MKPV-positive sentinel mice (65%). Given the lack of viral RNA in the stomach, intestine, salivary glands, pancreas, liver, and gall bladder, we suspect that PCR positive cecal contents are likely a result of ingested virus, potentially via coprophagy, rather than replication and excretion into the gastrointestinal tract or associated digestive organs. However, evidence of viral nucleotide sequences has been reported in the cecal mucosa of a single NSG mouse, 7 and we have shown MKPV viral RNA within hepatocytes of a single NSG mouse. It is unclear whether feces, in the absence of urine, are a significant part of transmission.
We demonstrate that MKPV can be consistently detected in the feces, blood, and urine of immunocompetent mice and that MKPV infection in these mice produces chronic shedding. The histologic lesions associated with MKPV infection in immunocompetent CD-1 and Swiss Webster mice include a mild to moderate interstitial nephritis and intranuclear inclusion bodies occur rarely.
Supplemental Material
Combined_supplemental_materials-Edmondson_et_al - Naturally Acquired Mouse Kidney Parvovirus Infection Produces a Persistent Interstitial Nephritis in Immunocompetent Laboratory Mice
Combined_supplemental_materials-Edmondson_et_al for Naturally Acquired Mouse Kidney Parvovirus Infection Produces a Persistent Interstitial Nephritis in Immunocompetent Laboratory Mice by Elijah F. Edmondson, Wang-Ting Hsieh, Josh A. Kramer, Matthew W. Breed, Melody E. Roelke-Parker, Julie Stephens-Devalle, Nathan M. Pate, Laura L. Bassel, Melinda G. Hollingshead, Baktiar O. Karim, Donna O. Butcher, Andrew C. Warner, Kunio Nagashima and Jatinder Gulani in Veterinary Pathology
Footnotes
Acknowledgements
We thank the technical staff of the Molecular Histopathology Lab, Animal Diagnostic Lab, and Laboratory Animal Medicine departments of the Laboratory Animal Science Program at Frederick National Laboratory for Cancer research, particularly Tamara Morgan, Angela Stahl, Stacy Manan, Melanie Martin, Jackie Stewart, Mei-Chen Tseng, Min Mo, Jayati Bera, William Aguilar, Rick Moxley, and Mark Shrader for their assistance, knowledge, and support. We would also like to thank Patricia Pesavento, DVM, PhD, and Charles Everett Alex, DVM, PhD, for assistance with viral taxonomy.
Declaration of Conflicting Interests
The author(s) declare no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded in whole or in part with federal funds from the National Cancer Institute, National Institutes of Health (NIH), under Contract No. 75N91019D00024. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US government. This research was supported in part by the Developmental Therapeutics Program in the Division of Cancer Treatment and Diagnosis of the National Cancer Institute, NIH.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
