Abstract
Neurotropism is a striking characteristic of bornaviruses, including parrot bornavirus 2 (PaBV-2). Our study evaluated the distribution of inflammatory foci and viral nucleoprotein (N) antigen in the brain and spinal cord of 27 cockatiels (Nymphicus hollandicus) following experimental infection with PaBV-2 by injection into the pectoral muscle. Tissue samples were taken at 12 timepoints between 5 and 114 days post-inoculation (dpi). Each experimental group had approximately 3 cockatiels per group and usually 1 negative control. Immunolabeling was first observed within the ventral horns of the thoracic spinal cord at 20 dpi and in the brain (thalamic nuclei and hindbrain) at 25 dpi. Both inflammation and viral antigen were restricted to the central core of the brain until 40 dpi. The virus then spread quickly at 60 dpi to both gray and white matter of all analyzed sections of the central nervous system (CNS). Encephalitis was most severe in the thalamus and hindbrain, while myelitis was most prominent in the gray matter and equally distributed in the cervical, thoracic, and lumbosacral spinal cord. Our results demonstrate a caudal to rostral spread of virus in the CNS following experimental inoculation of PABV-2 into the pectoral muscle, with the presence of viral antigen and inflammatory lesions first in the spinal cord and progressing to the brain.
Keywords
The family Bornaviridae of the order Mononegavirales includes mammalian viruses including Borna disease viruses (BoDV-1 and 2) and a novel zoonotic species transmitted by variegated squirrels (variegated squirrel bornavirus 1; VSBV-1) 47 as well as viruses of reptiles and aquatic, psittacine, and passerine birds. 1 Parrot bornaviruses (PaBVs), including Parrot bornavirus-2 (PaBV-2), are avian bornaviruses that belong to the species Psittaciform 1 or 2 orthorbornavirus and are the causative agents of proventricular dilatation disease (PDD), a lethal disease of captive psittacine birds. 2,25,31
Several aspects of the pathogenesis of PDD are still not well understood, including its natural route of infection. 26,33,43,53 Experimental inoculations using mixed routes of infection (oral, oculonasal, subcutaneous, and intramuscular) 15,37,41,46 or single routes (intramuscular, intravenous, or intracerebral) 18,42,43 were able to induce clinical disease and characteristic lesions. However, histopathology and immunohistochemistry were implemented to analyze late timepoints of infection when the animals presented clinical signs or at the end of the experiments. Therefore, crucial information about lesion distribution and viral progression in the early timepoints was consequently missed. Nevertheless, recent studies using only oculonasal or oral routes of infection were not able to establish persistent infection of PaBV-2 in experimentally inoculated cockatiels. 21
Neurotropism is an important feature of mammalian bornaviruses and has been extensively studied in natural and experimental cases of Borna disease. 20,34,48,55 BoDV-1 infection occurs in horses and sheep in central Europe 49 and causes a lymphoplasmacytic meningoencephalomyelitis with predilection for the gray matter. It affects primarily the hippocampus, caudate nucleus, substantia nigra, midbrain, and hypothalamus. 52,54 Eosinophilic intranuclear inclusion bodies, known as Joest-Degen bodies, are a sporadic finding in the neurons of animals affected by Borna disease and particularly prominent in the hippocampus. 4 Additionally, retinal degeneration and lymphoplasmacytic optic neuritis have been reported as a cause of blindness in horses infected with BoDV-1. 5,14 BoDV-2 was isolated from a horse with severe neurological disease in Austria, and although it differs from BoDV-1 by more than 15% of nucleotide identity, the clinical and pathological presentation were identical to BoDV-1 infection. 38
The distribution of inflammatory lesions of natural and experimental bornaviral diseases in other mammals such as variegated squirrels, humans, and immunocompetent rodents are similar to those observed in horses and sheep. 16,52 In contrast, neonatal rodents experimentally inoculated with BoDV-1 lack evidence of encephalitis but suffer significant developmental injury in the central nervous system (CNS), particularly in the hippocampus and cerebellum, including dentate gyrus involution and the loss of cerebellar granular cells, respectively. 28,54,55
Neurons, glial, and ependymal cells are the main target cells of the brains infected by both mammalian and avian bornaviruses. 8 –11,58 Immunolabeling is present in the nuclei alone or nuclei and cytoplasm of these cells, a reflection of the intranuclear replication of bornaviruses, 27 a characteristic shared among only 3 other families of animal RNA viruses: the Orthomyxoviridae, Retroviridae, and Nyamiviridae. 12,23,54
Although neurotropism is a prominent characteristic of PaBVs, studies on the precise localization of inflammatory lesions and viral distribution are limited and lack a systematic approach. 40,44 The aim of this study was to analyze the distribution of inflammatory lesions and PaBV nucleoprotein antigen localization in a chronologic manner to map the CNS areas affected by this virus.
Material and Methods
Ethics Statement
All procedures in this study were conducted using protocols approved by the Texas A&M Biosafety and Animal Use Committees (IACUC 20150-0045) and Institutional Biosafety Committee (IBC2015-021 and IBC2015-142) and meet all federal requirements as defined in the Animal Welfare act (AWA), Public Health Service Policy (PHS), and Humane Care and Use of Laboratory Animals.
Viral Culture and Inoculum Preparation
Parrot bornavirus 2 (PaBV-2 CK1-34) was inoculated into duck embryo fibroblasts (DEF, Schubot Exotic Birds health center laboratory cell collection) cultured for 7 passages in minimum essential medium (MEM) supplemented with 10% fetal bovine serum (FBS, Gibco, ThermoFisher Scientific, Walthan, MA), and subsequently maintained in MEM supplemented with 2% FBS, as previously described. 19 The cell debris were removed by centrifugation at 3000 × g for 10 minutes. Serial 10-fold dilutions of 3 stocks of virus aliquots were analyzed by focus-forming assays to determine viral titration. Titers above 8 × 104 focus forming units per milliliter (FFU/ml) were considered acceptable for the experimental inoculation as previously described. 18,19
Experimental Animals, Virus Inoculation, and Infection Timeline
Thirty-four cockatiels (Nymphicus hollandicus) originated from 2 PaBV-negative aviaries were used for experimental inoculations of PaBV-2 (CK1-34), as previously described. 33 Briefly, cockatiels were divided into 12 groups that corresponded to different euthanasia timepoints (5, 10, 20, 25, 30, 35, 40, 60, 80, 95, 100, and 114 days post-inoculation; dpi). Each group had 3 experimental cockatiels, except for timepoints 5 dpi (2 cockatiels), 10 dpi (2 cockatiels), 25 dpi (4 cockatiels), 35 dpi (5 cockatiels), 95 dpi (1 cockatiel), and 100 dpi (2 cockatiels), as described elsewhere. 33 Twenty-seven cockatiels were inoculated with PaBV-2 while 7 cockatiels were inoculated only with PaBV-free DEF and served as negative controls. Negative controls were distributed in groups correspondent to timepoints for 20 dpi, 25 dpi, 30 dpi, 35 dpi, 40 dpi, 60 dpi, and 114 dpi (1 negative control for each of these timepoints). Cockatiels were inoculated using intramuscular injection in the right pectoral muscle with 4 × 104 FFU of virus. Any clinical signs observed during the experimental period were documented. At the corresponding timepoints, cockatiels were anesthetized with isoflurane (IsoThesia, Henry Schein, Melville, NY) and then euthanized with carbon dioxide (CO2). The results of the histological and immunohistochemical analyses of other organs from the same experimentally infected animals are reported elsewhere. 33
Histology
Brain and spinal cord of each bird were placed in 4% neutral formaldehyde for 48 hours. Coronal sections of the brain were obtained from the cerebrum at the level of the hyperpalium and nucleus basorostralis (Level I), cerebrum at the level of the striatum (Level II), and cerebrum at the level of the arcopalium, thalamus, and midbrain (Level III). Additionally, a sagittal section was taken encompassing the hindbrain and cerebellum (Level IV) (Supplemental Figure S1). Three transversal serial samples from cervical spinal cord between C1 and C13, thoracic spinal cord between T1 and T7, lumbosacral spinal cord at the level of the synsacrum, and the 5 caudal vertebrae (Supplemental Figure S2) were collected from all birds. Samples of spinal cord were collected with intact vertebrae and decalcified, which prevented the destruction of the adjacent spinal ganglia during the trimming process. All samples were processed routinely for histologic examination, and when necessary, decalcification was performed using Formical-4 (American master tech, Lodi, CA) as recommended by the manufacturer. The histological evaluation was done blindly by a board-certified pathologist (R. R. Rech).
The neuroanatomic location of each inflammatory focus was mapped in a diagram, and the description of the regions of the brain was adapted for this purpose and followed what has been suggested by the avian brain nomenclature consortium, 45 avian brain atlases, 7,36 and avian medicine researchers (Fig. 1). 13,39,51 A single blood vessel surrounded by layers of lymphocytes and plasma cells (lymphoplasmacytic perivascular cuffing) or a focus of inflammation in the neuroparenchyma (glial nodule) was interpreted as a single inflammatory focus (Figs. 2, 3) and indicated by a dot in the diagram. The severity of inflammation was represented by 4 different dot sizes (minimal, mild, moderate, or severe), and each bird with inflammatory lesions was represented by a different color. Lesions were considered minimal when 1 single layer of perivascular cuffing and no more than 1 glial nodule was in the analyzed section, mild when vessels had 2 layers of perivascular cuffing and/or 2 to 3 glial nodules, moderate when vessels had 3 layers of perivascular cuffing and/or 4 to 7 glial nodules, and severe when vessels had 4 or more layers of perivascular cuffing and/or more than 7 glial nodules. All sections were analyzed separately for each bird and then overlapped to generate the final diagram using a graphic software (Photoshop elements, Adobe systems, San Jose, CA). Lymphoplasmacytic inflammation was scored as follows: – (absent), + (minimal), ++ (mild), +++ (moderate), ++++ (severe).
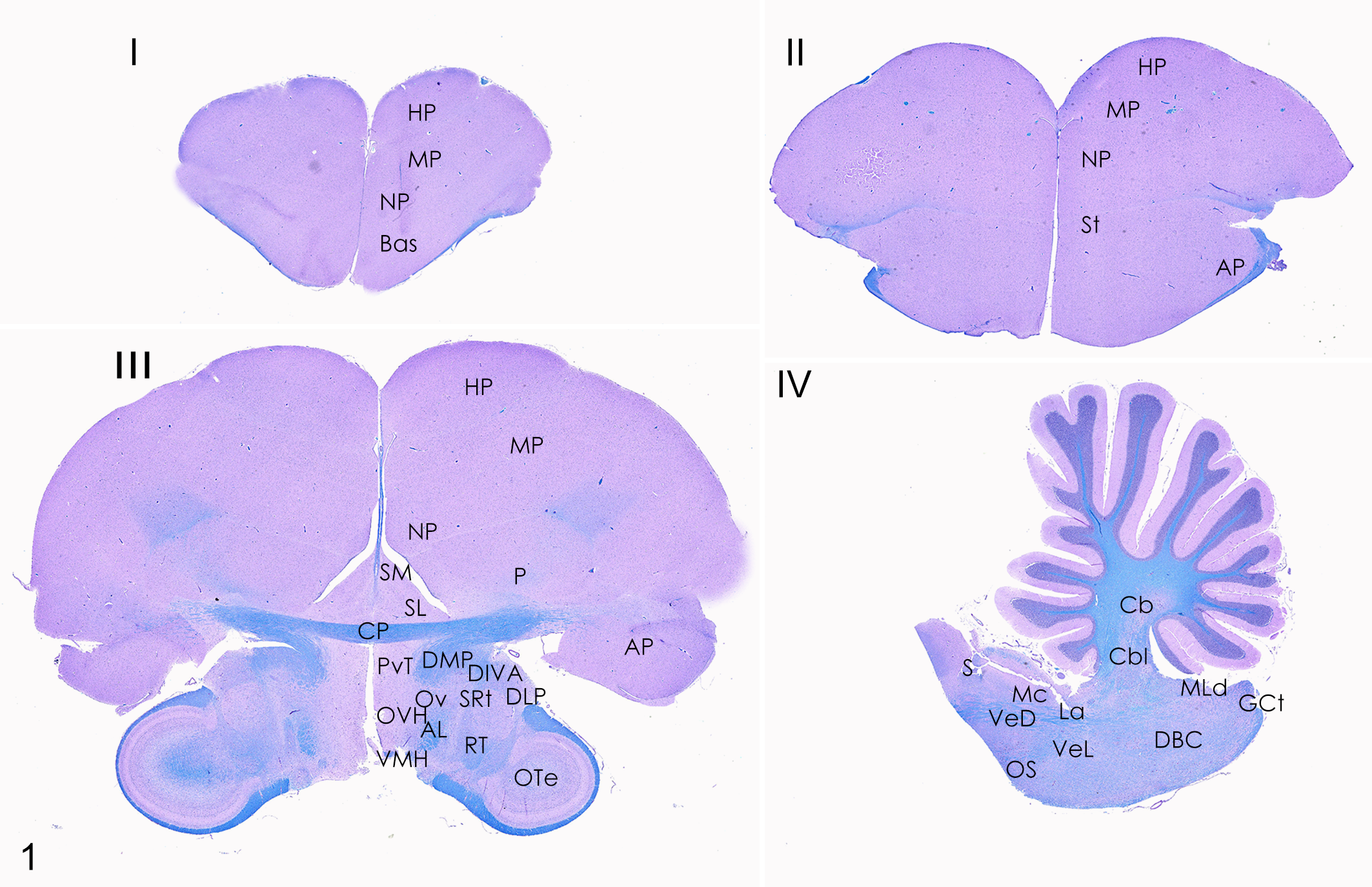
Brain, cockatiel. Schematic map of histological landmarks in the 4 selected brain levels (I, II, III, IV). Luxol-fast blue/periodic acid-Schiff (PAS) stain. Abbreviations: AL, ansa lnticularis; AP, arcopallium; Bas, nucleus basorostralis; Cbl, nucleus cerebellaris intermedius; CbM, nucleus cerebellaris intermedius; CP, comissura posterior; DBC, nucleus decussationis brachiorum conjunctivorum; DIVA, dorsalis intermedius pars ventralis anterior; DLP, nucleus dorsolateralis posterior thalami; DMP, nucleus dorsomedialis posterior thalami; GCt, substantia grisea centralis; HP, hyperpallium; La, nucleus laminaris; Mc, nucleus magnocellularis; NP, nidopallium; OS, nucleus olivaris superior; Ote, optic tectum; Ov, nucleus ovoidalis; OVH, organum vasculosum of the hypothalamus; P, pallidum; PvT, paraventricular nuclei of the thalamus; Rt, nucleus rotundus; S, nucleus solitarius; SL, nucleus septalis lateralis; SM, nucleus septalis medialis; SRt, nucleus subrotundus; St, striatum; VeD, nucleus vestibularis descendens; VeL, nucleus vestibularis lateralis; VMH, ventromedial nucleus of hypothalamus.
Parrot bornavirus 2 infection, brain, cockatiel. Figure 2. Lymphoplasmacytic perivascular cuffing. HE. Figure 3. Glial focus in the gray matter. HE.
Immunohistochemistry
Immunohistochemistry was performed using polyclonal antibodies against a specific region of the PaBV N-protein, as previously described, 33 on serial tissue sections, mounted on charged slides, and examined by light microscopy. Due to the intranuclear replication of PaBVs, only cells with intranuclear or intranuclear and intracytoplasmic immunolabeling were considered positive. Cells with only intracytoplasmic immunolabeling were considered negative. Immunolabeling was scored as previously described, 33 and the progression for each CNS section was recorded in schematic plates. Briefly, slides were scored based on the number of N-protein positive cells in the whole section of brain or spinal cord as follows: – (absent), + (1%–3% affected cells), ++ (4%–10% affected cells), +++ (11%–25% affected cells), and ++++ (more than 25% affected cells). The immunohistochemical evaluation was done blindly by a board-certified pathologist (A. Rodrigues-Hoffmann).
Results
Distribution of the Inflammatory Infiltrate
In the spinal cord, lesions were equally distributed among the 3 segments with wide distribution within ventral and dorsal horns (Fig. 4). For infected birds, inflammation was more prominent in the central core of the brain, mainly in the thalamus and hindbrain (Fig. 5).
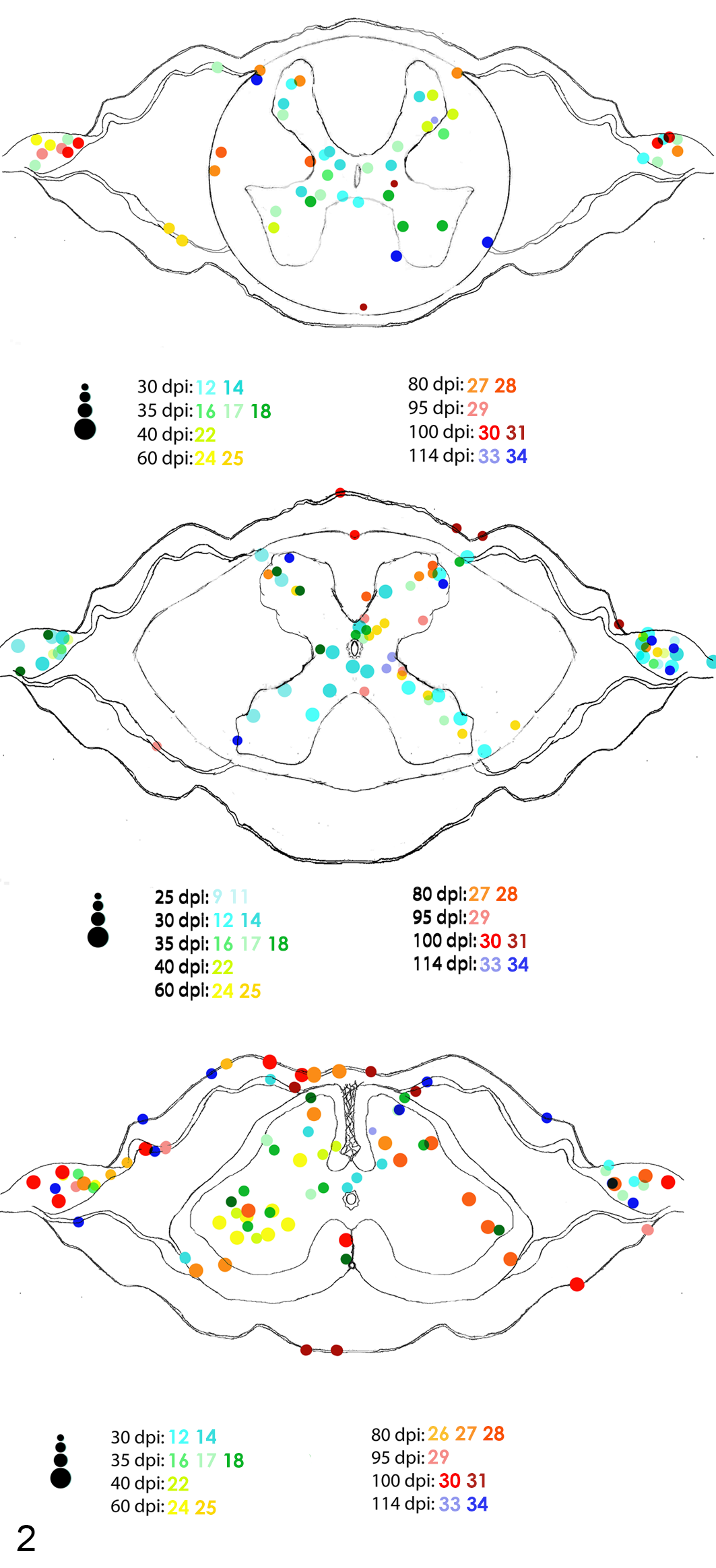
Distribution and severity of inflammation in the cervical (top), thoracic (middle), and lumbosacral (bottom) spinal cord of PABV-2-inoculated cockatiels. Animals with inflammatory lesions are represented by different colored dots and numbers. Each dot represents an inflammatory focus, and the 4 different dot sizes represent different degrees of severity (minimal, mild, moderate, and severe).
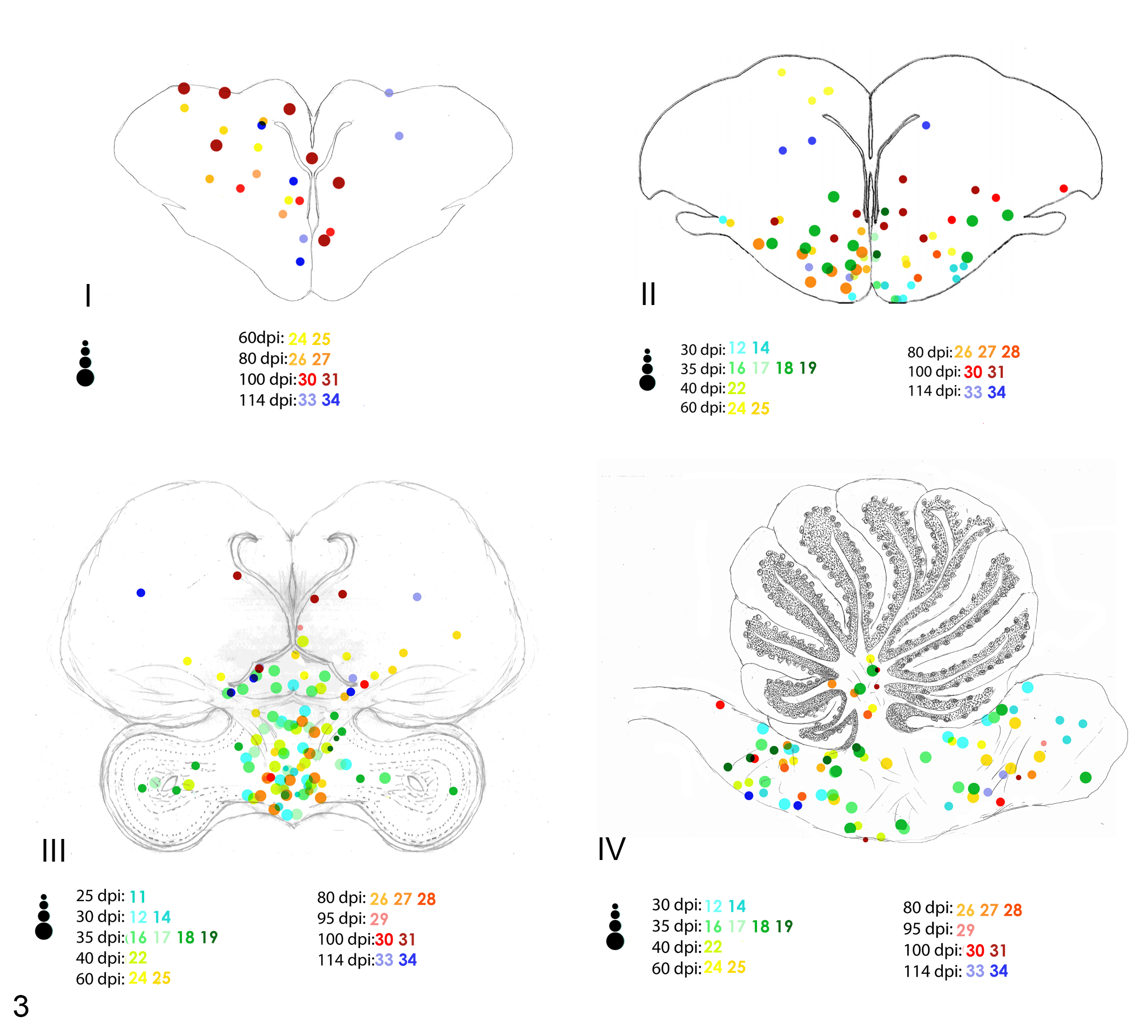
Distribution and severity of inflammation in 4 selected levels (I, II, III, and IV) of the brain of PABV-2–inoculated cockatiels. Animals with inflammatory lesions are represented by different colors. Each dot represents an inflammatory focus, and the 4 different dot sizes represent different degrees of severity (minimal, mild, moderate, and severe).
The progression and severity of the inflammation across different timepoints in the examined CNS sections are summarized in Table 1. The first spinal cord segment to develop inflammatory foci was the thoracic spinal cord at 25 dpi. Inflammation was predominant in the ventral horns accompanied by ganglioneuritis until 35 dpi, when it started to spread to the dorsal horns. Both cervical and lumbosacral segments of the spinal cord first developed inflammation at 30 dpi, affecting both dorsal and ventral horns as well as adjacent spinal ganglia. All sections presented inflammation, primarily in the gray matter, throughout all timepoints until 114 dpi. Although less frequent and severe than inflammation in the gray matter, inflammatory foci in the white matter were first observed close to nerve insertions of the ventral funiculus at 30 dpi, followed by the dorsal funiculus at 80 dpi. Then white matter inflammation spread to ventral, dorsal, and lateral funiculi until 114 dpi. Ganglioneuritis was observed at all timepoints from 25 to 114 dpi, with the following exceptions: 25 and 114 dpi in the cervical segment, 95 and 100 dpi in the thoracic, and 40 dpi in the lumbosacral spinal cord. Ganglioneuritis was more severe and frequent in the lumbosacral spinal segment. Meningitis in the spinal cord was only observed at 80, 95, 100, and 114 dpi, and it was also more frequent and severe in the lumbosacral segment. No inflammation was observed in the glycogen body.
Progression of the inflammatory lesions and immunolabeling for parrot bornavirus-2 in the central nervous system of infected cockatiels across 12 different timepoints.*
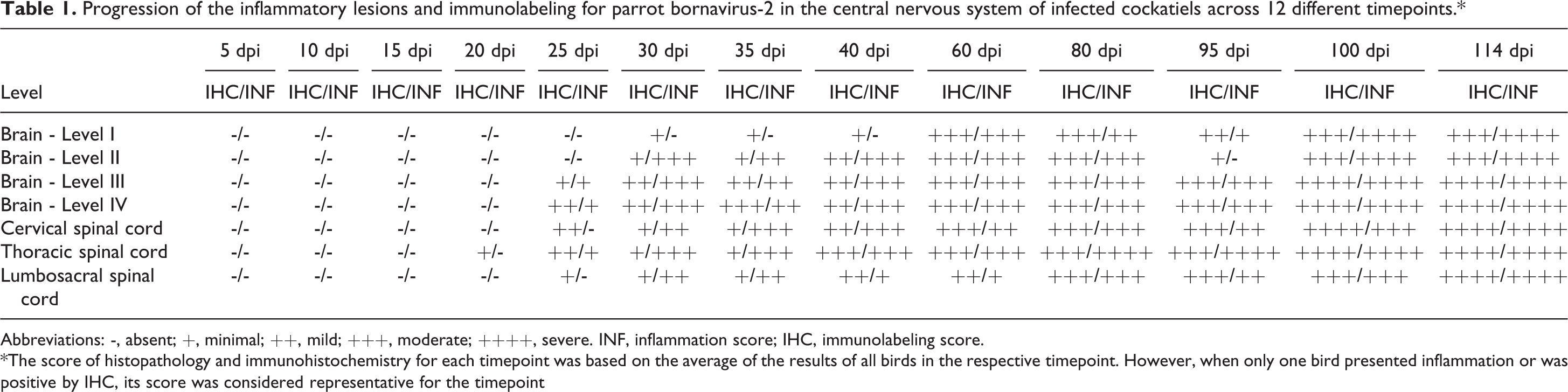
Abbreviations: -, absent; +, minimal; ++, mild; +++, moderate; ++++, severe. INF, inflammation score; IHC, immunolabeling score.
*The score of histopathology and immunohistochemistry for each timepoint was based on the average of the results of all birds in the respective timepoint. However, when only one bird presented inflammation or was positive by IHC, its score was considered representative for the timepoint
At 25 dpi, the first areas of the brain to present inflammation were the hindbrain and the thalamus. More specifically, encephalitis was more prominent in the gray matter and first spreading through the nucleus magnocellularis, nucleus olivaris superior, and nucleus laminaris in the hindbrain and through the paraventricular nucleus of the thalamus. By 30 dpi, inflammation had spread laterally through thalamic nuclei, including the nucleus ovoidalis and nucleus rotundus, and dorsally to the commisura posterior, which was the main region of the white matter affected by inflammation. The nuclei septalis and scattered areas in the cerebrum at the level of the striatum and nidopallium were also affected at 30 dpi. At 35 dpi, inflammatory foci reached the optic tectum of the midbrain. Inflammation in the hindbrain was consistently present in the areas corresponding to the nucleus olivaris superior, nucleus solitarius, nucleus laminaris, and nucleus decussationis. Only 5 had inflammatory foci in the cerebellum, which only started at 40 dpi and were concentrated at the nucleus cerebellaris intermedius and nucleus cerebellaris internus. Inflammation reached the most dorsal areas of the caudal cerebrum comprising the mesopallium and hyperpallium at 40 dpi and cranial cerebrum at 60 dpi affecting both gray and white matter. Hindbrain and midbrain continuously presented inflammatory foci throughout 60, 80, 95, and 100, until 114 dpi. The cerebrum did not show inflammation at 95 dpi. At 60, 80, 100, and 114 dpi, inflammatory foci were mainly concentrated in the mesopallium and hyperpallium. Meningitis in the brain was only observed at 60, 100, and 114 dpi, and its severity ranged from minimal to mild.
Distribution of Viral Antigen
Immunolabeling was observed in the nuclei and/or nuclei and cytoplasm of predominantly neurons, but glial and ependymal cells were also intermittently positive (Figs. 6–8). Immunolabeling was observed as early as 20 dpi unilaterally in the neurons and glial cells of the ventral horn and adjacesnt ganglia of the thoracic spinal cord and at 25 dpi in the cervical and lumbosacral segments. At 40 dpi, the immunolabeling spread to the ependymal cells, adjacent neurons, and glial cells, including cells in the dorsal horns in all 3 segments of the spinal cord. Immunolabeling in the glial cells of the white matter was first observed at 80 dpi. In all following timepoints (80, 95, 100, and 114 dpi), a diffuse labeling involving neurons, glial, and ependymal cells in both gray and white matter as well as neurons of the adjacent ganglia was observed (Fig. 9).
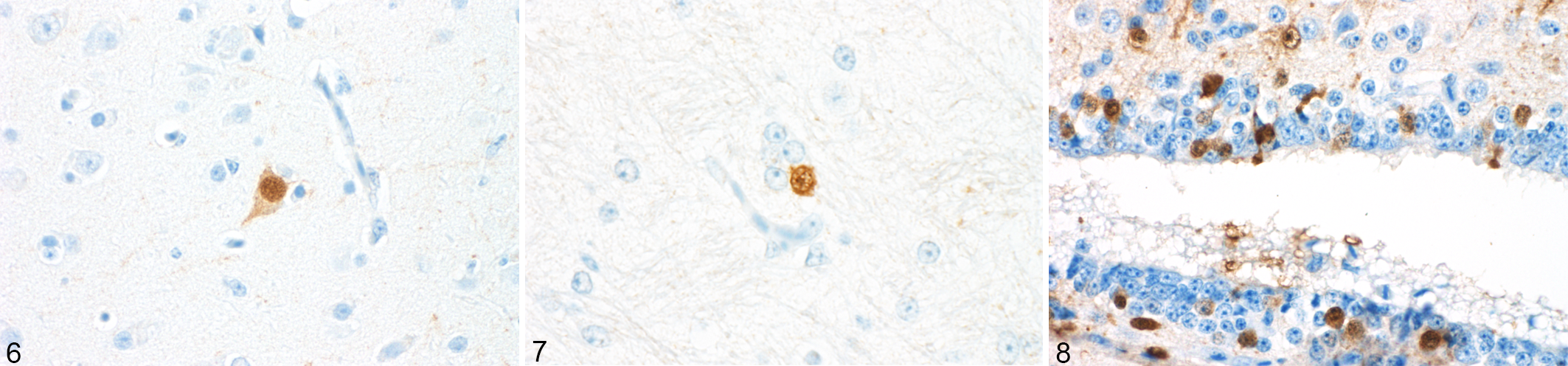
Parrot bornavirus 2 infection, cerebrum, cockatiel, immunohistochemistry (IHC) for PaBV-2. Figure 6. Intranuclear and intracytoplasmic immunolabeling for PaBV-2 nucleoprotein in neurons. Figure 7. Intranuclear immunolabeling for PaBV-2 nucleoprotein in glial cells. Figure 8. Intranuclear immunolabeling for PaBV-2 nucleoprotein in ependymal cells.
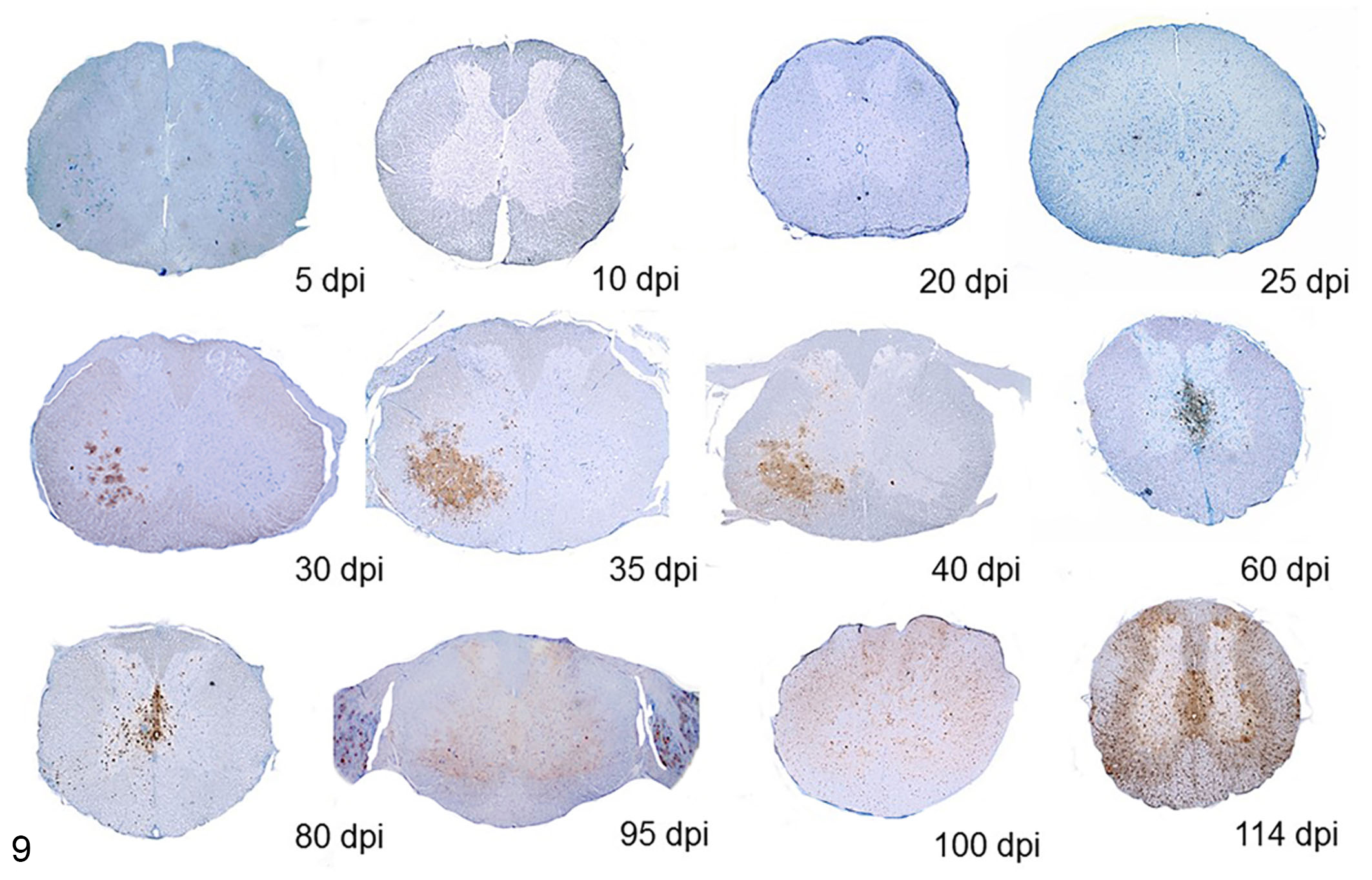
Spinal cord, cockatiel. Progression of immunolabeling for PaBV-2 nucleoprotein in the thoracic spinal cord. Immunolabeling was observed as early as 20 dpi unilaterally in few neurons and glial cells of the ventral horn and adjacent ganglia of the thoracic spinal cord. At 30 dpi, immunolabeling was more intense and affecting more neurons and glial cells but still focally and unilaterally concentrated in the ventral horns. Immunolabeling progressively involved all areas of white and grey matter by 80 dpi. IHC for PaBV-2.
In the brain, the thalamic nuclei and the hindbrain were simultaneously the first areas of the brain with PaBV immunolabeling at 25 dpi. From 25 to 30 dpi, scattered neurons of the paraventricular nuclei of the thalamus and glial cells scattered throughout the thalamus were positive. At 30 dpi, few neurons of the nucleus basorostralis and striatum in the cerebrum were positive. At 35 dpi, the labeling rapidly spread throughout the central core reaching the nuclei septalis in the subpallium and neurons of the nuclei cereberallis and also in the granule cell layer of the cerebellum. Ependymal cells were also positive at 35 dpi, particularly in the organum vasculosum of the hypothalamus, surrounding the third ventricle. At 40 dpi, immunolabeling was strongly positive in the pallial areas, particularly prominent in the arcopallium and nidopallium. Also at 40 dpi, PaBV-positive neurons and glial cells were identified in the optic tectum and hyperpallium. The most widespread immunolabeling for hindbrain was first observed at 60 dpi. A diffuse pattern affecting virtually all regions of the cerebrum was observed from 60 to 114 dpi. A widespread pattern affecting not only the cerebellar nuclei but also the white matter and the Purkinje cells was first observed at 80 dpi and remained positive until 114 dpi (Fig. 10).
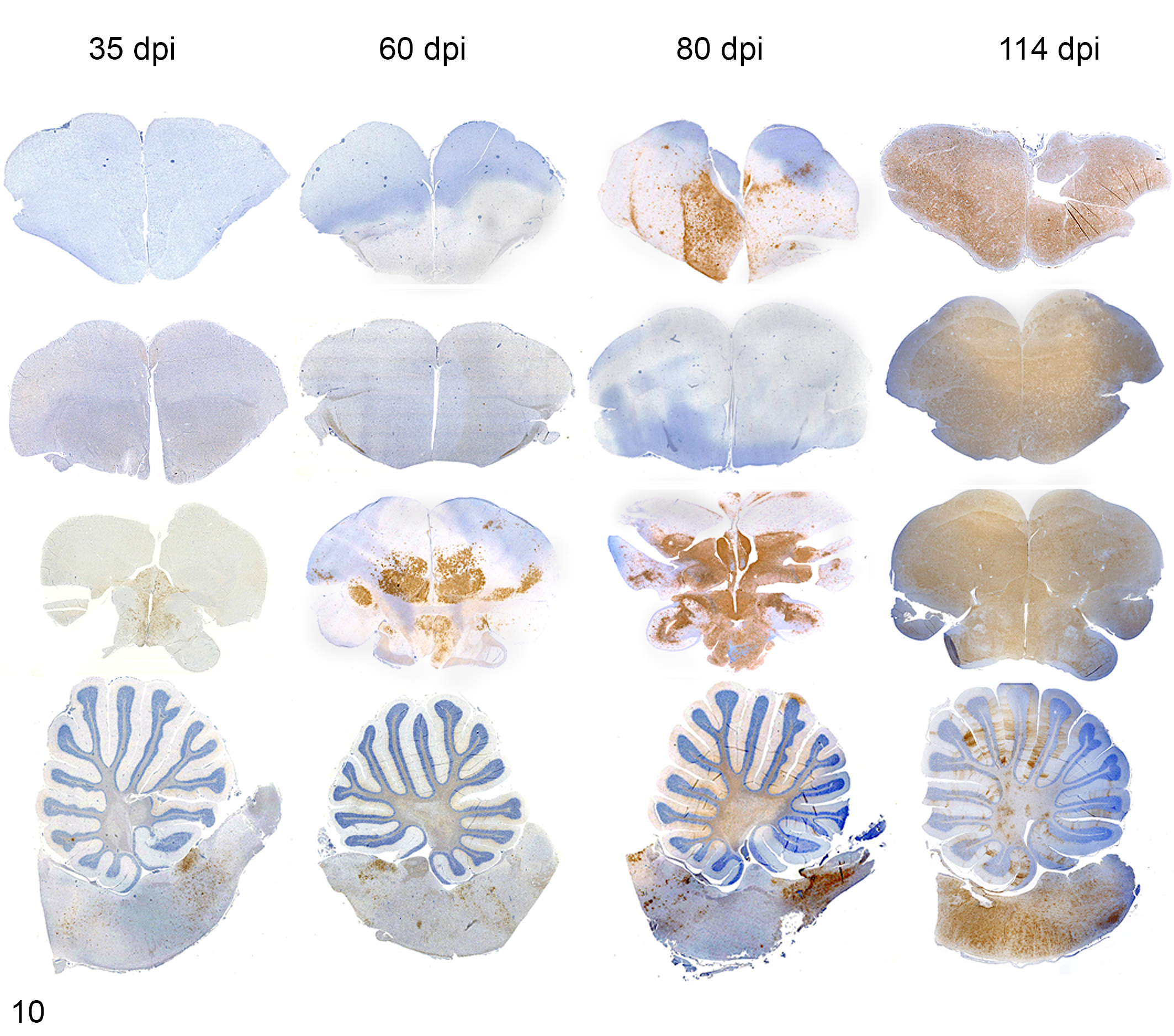
Brain, cockatiel. Progression of immunolabeling for PaBV-2 nucleoprotein in 4 brain levels at 4 timepoints. Immunolabeling was readily visible at 35 dpi in nuclei of the central core, which can be observed at levels III and IV of the brain. The immunolabeling in these areas progressively spread throughout these sections until 114 dpi. Immunolabeling in sections I and II were more prominent only starting at 80 dpi and remained widespread until 114 dpi. IHC for PaBV-2.
No inflammatory foci or antigen immunolabeling were observed in uninfected control birds. Individual data of the inflammatory infiltrates and viral antigen distribution for the CNS of each one of the experimental cockatiels are available in Supplemental Table S1.
Discussion
After 20 dpi, all infected birds experimentally inoculated with PaBV-2 in the pectoral muscle developed a lymphoplasmacytic myelitis or lymphoplasmacytic encephalitis or a combination of both. This was associated with consistent intranuclear immunolabeling of PaBV N protein antigen mainly in neurons, compatible with what has been described in natural cases of PDD. 40 Although previous studies have been able to identify PaBV N-protein antigen and inflammatory lesions in the CNS of natural and experimental cases of PDD, 15,18,41,42,43,46 the distribution of the inflammation and/or immunolabeling were broadly determined with minimal neuroanatomic mapping. 40,44 In this study, immunolabeling for PaBV N-protein preceded the development of inflammation throughout all timepoints. The ventral horns of the thoracic spinal cord were the first positive CNS areas at 20 dpi. The hindbrain and the thalamus were the first areas of the brain to simultaneously present viral antigen and inflammation at 25 dpi. The interval between 25 and 60 dpi was crucial for the viral spread; during this period, several areas in the gray and white matter were progressively and consistently affected, and both inflammation and viral distribution were more concentrated to the ventral areas of the brain.
PDD has been often described as a gastrointestinal disease, and signs may include proventricular dilation, regurgitation, crop impaction, anorexia, weight loss, and undigested seeds in the feces. 53 Experimental studies in cockatiels have shown gastrointestinal signs prior to the development of neurological signs, 42,43 and ganglioneuritis has been reported as a more consistent finding than meningoencephalomyelitis. 15,42,43,46 However, it is important to note that PDD is primarily a neurological disease, and gastrointestinal signs are in fact caused by ganglia and nerve dysfunction. 53 As previously demonstrated, 33 lesions in the CNS may develop prior to peripheral lesions, although this does not necessarily mean that the affected bird will have clinically evident neurological disease, especially in the early timepoints of infection. In experimental studies, the route of inoculation, tissues analyzed, and methodology implemented must be considered to explain the discrepancy between the development of neurological and gastrointestinal signs.
The anatomical localization of lesions in the CNS is a crucial step for developing differential diagnoses of neurological diseases 13 and consequently, the understanding of the pathogenesis of neurotropic viruses. Lesions in different anatomical regions of the CNS may lead to distinct neurological conditions, 34 hence the importance of mapping the main areas affected in neurological diseases such as PDD. Although the correlation between affected areas and clinical presentation of PaBV-infected birds has not been thoroughly explored, the neuroanatomical localization of lesions in rodents experimentally infected with BoDV has provided a good understanding of the clinical presentation. In Lewis rats, experimental infection of BoDV usually causes a widespread lymphoplasmacytic meningoencephalitis with viral antigen dissemination in all brain regions, which results in a biphasic neurobehavioral disorder. However, when the rats are infected with the BoDV-obese variant, inflammation and viral antigen localization is restricted to the septum, hippocampus, amygdala, and ventromedial tuberal hypothalamus, which results in a neuroendocrine disorder and consequent obesity. 22
The most common neurological signs seen in birds affected by PDD correspond to both behavioral and motor disorders, including lethargy, depression, ataxia, seizures, paresis, and paralysis. 26,53 In our study, the concentration of inflammatory lesions in viscero-limbic components (eg, ventral striatum and pallidum), associated nuclei (eg, nucleus dorsolateralis posterior thalami, nucleus dorsomedialis posterior thalami), and nuclei involved in reward and motivation (eg, paraventricular nuclei of thalamus) 32 are in agreement with the regions that are usually affected by behavioral disorders that involve the limbic system in mammals. Lesions in these areas are usually associated with lethargy, depression, obtundation, and stupor. 13
In mammals, the widespread inflammatory lesions in the cerebrum may explain signs such as seizures, while lesions in the cerebellum might be associated with ataxia. Although not well documented in birds, similar circumstances might explain these clinical manifestations in PDD cases. Purkinje cell degeneration and necrosis have been reported in some PDD cases 3,40 but were not observed in our study. Glial foci were sporadically seen in the CNS of our experimental cockatiels, which might have been associated with neuronal death at some point of the course of the disease and therefore might play a role in the pathogenesis of neurological manifestations commonly seen in PDD cases. The wide distribution of lesions in the gray matter of the spinal cord seen in our study explain clinical signs such as paresis and paralysis, sporadically seen in PDD cases. Additionally, the high diversity of parrot bornaviruses can potentially be associated with variable target cells and lesions and consequently, distinct clinical presentations. 42
In this study, the inflammatory lesions bear some similarities to what has been described for mammalian bornaviruses. Lymphoplasmacytic meningoencephalomyelitis with predilection for gray matter, including areas such as the thalamus and hypothalamus, and relative sparing of the cerebellum and dorsal cerebrum was our main histological finding as reported for horses, sheep, variegated squirrels, humans, and immunocompetent rodents. 16,20,21,38,44,55 In contrast, the predilection for olfactory bulb and hippocampus in mammals, a feature that fits the olfactory route of infection proposed for BoDV pathogenesis 8 and follows a rostral-caudal progression of BoDV infection, was not observed in our study.
The natural route of infection for PaBVs remains to be elucidated. Experimental studies have been able to reproduce clinical disease using different routes of inoculation, including single intramuscular, intracerebral, or intravenous routes, 18,42, 43 and also by combined intramuscular, oral, oculonasal, and subcutaneous routes. 15,37,41 Nonetheless, experiments using combined routes have nebulous results in regards to which one of the routes was responsible for causing clinical disease. Recently, experimental studies using oral and oculonasal routes of infection have failed to induce disease and/or PDD lesions in cockatiels after 6 months or experiment, 21 which may indicate that these routes are unlikely to be associated with PaBVs’ natural route of transmission. Additionally, skin injuries due to squirrel bites and scratches have been reported by family members of the 3 patients who died with variegated squirrel bornavirus-1 (VSBV-1)–induced encephalitis in Germany. 24 The successful experiments using intramuscular routes of inoculation in psittacine birds and the recent data about skin injuries in VSBV-1 cases reiterate the importance of investigating the association of tegument injuries and intramuscular lesions as a port of entry for PaBVs. However, it is important to note that although intramuscular inoculation of PaBV-2 successfully caused PDD in cockatiels of our experiment, there is still not enough data to support that this is the route of infection in natural PDD cases.
A potential intramuscular route of infection (as was done in our experiment 33 ) creates the possibility of PaBVs being spread first to the spinal cord and only reaching the rostral areas of the late in the course of infection, resulting in a caudal-rostral progression of infection and explaining the reduced inflammation and presence of viral N-protein antigen seen in the more rostral areas during the late timepoints.
Although the hippocampus is associated with spatial memory in both birds and mammals, its localization and structure differ enormously between these classes of animals. 50 The mammalian hippocampus is located rostral to the midbrain and ventral to the thalamus, while the avian hippocampus has a dorsal location in the pallial region of the cerebrum. 29 This area was not affected in the cockatiels of this study, probably due the predilection of the virus for the central core of the brain and consequent inflammation concentrated in the ventral areas of the brain. Additionally, birds lack a distinguishable dentate gyrus, which makes the progressive cell death of the granule cells that is reported in the hippocampus of BoDV-infected rats difficult to assess in birds. 30,35
The pathognomonic feature of mammalian BoDV infection, the eosinophilic intranuclear inclusion bodies known as Joest-Degen bodies, 4 were not observed in this study. Although these inclusion bodies have been reported in few PDD cases, 6,18,57 demonstration of the inclusion bodies in hematoxylin and eosin–stained sections as observed for mammals 56 has not been consistent, and a careful analysis of the morphological and staining characteristics of the inclusion bodies must be taken in consideration to differentiate them from fragmented nucleoli or artifacts. The immunohistochemical pattern observed in samples from humans, squirrels, shrews, horses, sheep, and rodents infected with mammalian bornaviruses 17,24,56 has a similar presentation to the immunolabeling in the nuclei, cytoplasm, and neuronal and glial processes noted in the CNS of birds infected with PaBVs. 40,44,58
Our results suggest that the common practice of collecting and assessing only a single section of the brain could lead to a false-negative diagnosis in PaBV-infected birds, especially in the early timepoints of infection. We observed inflammation in the hindbrain and midbrain from early to late timepoints (25–114 dpi). On the other hand, the cerebrum at the level of the hyperpalium remained as the least affected area until 100 dpi. Therefore, sampling limited to rostral areas of cerebrum would be prone to miss the vast majority of inflammatory lesions, which might result in the misleading assumption that gastrointestinal ganglioneuritis was present prior to meningoencephalomyelitis.
In conclusion, our results suggest that thalamus and hindbrain were considered the most appropriate sections of the brain for sampling to determine PaBV-2 infection spread and inflammation. Little or no difference was observed between the 3 segments of the spinal cord in our study of experimental PaBV-2 infection, based on distribution of viral antigen and inflammatory lesions. Further research is needed to understand whether a similar process happens in naturally infected birds and elucidate the mode of viral spread from animal to animal as well as more fully understand the physiologic and behavioral effects of this temporal progression in the distribution of viral antigen and inflammation. We advocate sampling the spinal cord with the vertebral bodies in birds because this results in outstanding visualization of the spinal ganglia and the decalcification process does not interfere with viral antigen detection.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818798112 - Distribution of Viral Antigen and Inflammatory Lesions in the Central Nervous System of Cockatiels (Nymphicus hollandicus) Experimentally Infected with Parrot Bornavirus 2
Supplemental Material, DS1_VET_10.1177_0300985818798112 for Distribution of Viral Antigen and Inflammatory Lesions in the Central Nervous System of Cockatiels (Nymphicus hollandicus) Experimentally Infected with Parrot Bornavirus 2 by Jeann Leal de Araujo, Aline Rodrigues-Hoffmann, Paula R. Giaretta, Jianhua Guo, Jill Heatley, Ian Tizard, and Raquel R. Rech in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the excellent technical assistance provided by Debra Turner, Luan Henker, Cintia Queiroz, Caitlin Older, Courtney Smith, Anna Blick, Mackenzie Branco, Kelly Mallet, Brittany Kerr, Robin Katchko, Randi Gold, Sierra Guidry, and Kristen Streeter.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Avian Health Complex, the Schubot Center, Texas A&M University, and the CAPES-Science without borders program for supporting this study.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
