Abstract
Beginning in 2015, athymic nude sentinel mice from conventional, medium-, and high-security facilities presented to the Comparative Pathology Laboratory (CPL) with weight loss, diarrhea, and/or rectal prolapse. Regardless of whether clinical signs were present or absent, the gross observation of ceco-colonic thickening corresponded histologically to pleocellular typhlocolitis with mucosal hyperplasia and lamina proprial multinucleated cells. A subset of affected sentinels exhibited granulomatous serositis and hepatosplenic necrosis with multinucleated cells. Initial suspicion of mouse hepatitis virus infection was excluded by polymerase chain reaction, electron microscopy, and serology. Multinucleated giant cells were confirmed as macrophages by positive immunoreactivity to Mac-3 and Iba-1 and negative immunoreactivity to pancytokeratin. From conventional and medium-security facilities, Helicobacter species were identified in 40 of 143 (27.9%) mice, with H. hepaticus accounting for 72.5% of identified Helicobacter species. Other agents included opportunistic bacterial infection (41/145, 28.3%), murine norovirus (16/106, 15.1%), and pinworms (2/146, 1.4%). From high-security facilities, only Enterobacter cloacae was identified (2/13, 15.4%), and no evidence of Helicobacter sp., murine norovirus, or pinworms was present. No potentially infectious disease agent(s) was identified in 71 of 146 (48.6%) affected nude sentinels from conventional and medium-security facilities and 11 of 13 (84.6%) affected nude sentinels from high-security facilities. No statistically significant differences in histologic lesion scores were identified between Helicobacter-positive and Helicobacter-negative mice. Thus, proliferative typhlocolitis with multinucleated giant cells was considered a nonspecific histologic pattern associated with a variety of primary and opportunistic pathogens in athymic nude mice.
Design and implementation of rodent health surveillance programs are essential for experimental quality assurance and timely detection of pathogenic agents within laboratory animal facilities. Rodent health surveillance programs are predicated on systematic investigations of population subsets to monitor and define pathogenic agents within a given population as a whole. 11,33,34,40,47 As mouse-based research is often conducted in a diverse array of academic and industrial settings, a single standardized approach to rodent health surveillance programs and monitored pathogens is impractical. Thus, specifications for these programs are frequently dictated by the needs and demands of individual research institutions. More generalized recommendations, however, provide beneficial guidelines regarding selection of sentinel animals used for testing, testing frequency, potential pathogenic agents, and interpretation of health surveillance results. 33
Traditional rodent health surveillance programs employ immunocompetent adult sentinels for pathogen detection. 33 However, the potential benefits (eg, early pathogen/opportunist detection) and risks (eg, a source of persistent infection) of utilizing immunodeficient sentinels in surveillance practices have been explored. 33 Beginning in 2014, the rodent health surveillance program at University of California, Davis (UC Davis) was redesigned to utilize both immunocompetent (Hsd: Athymic Nude-Foxn1nu heterozygotes, nu/+) and immunodeficient adult sentinels (Hsd: Athymic Nude-Foxn1nu homozygotes, nu/nu) for campus-wide pathogen detection.
During routine sentinel processing in 2015, nu/nu sentinels from conventional, medium-security, and high-security facilities at UC Davis presented with weight loss and diarrheal disease or were noted to have loose feces and thickened large intestines at necropsy examination. Histologically, cecal and colonic lesions were typified by proliferative typhlocolitis with scattered multinucleated cells within the lamina propria. Additional histologic lesions included occasional necrotizing splenitis and hepatitis as well as nodular granulomatous abdominal serositis, all with intralesional multinucleated cells. Given the medium- to high-security status of many affected immunodeficient sentinels in our cohort, pathogenic agents associated with proliferative typhlocolitis and multinucleated cells (possible syncytia) were of utmost concern.
Murine proliferative typhlocolitis is a well-established model for inflammatory bowel disease (IBD) in humans. 6 –9,13 –16,27,39 The pathophysiologic complexities of human IBD development are underscored by the broad array of chemical compounds, 39 genetic modifications, 8,9,12,26,27 and/or pathogens 1,2,4 –9,12,43,46 used to induce or contribute to murine proliferative typhlocolitis. Infectious pathogens of colony health concern that are known to result in proliferative typhlocolitis with or without multinucleated cells in immunodeficient mice include enterotropic strains of mouse hepatitis virus (MHV), 3,4,17,49 Helicobacter sp., 6 –9,12,38,43 Citrobacter rodentium, 2,3 pinworms, 3 and Escherichia coli. 3
Herein we describe proliferative typhlocolitis with multinucleated cells in immunodeficient sentinel mice. Through utilization of our rodent health surveillance screening as well as additional ancillary diagnostics, we aimed to (1) identify pathogenic and/or opportunistic agents associated with proliferative typhlocolitis in athymic nude sentinels and (2) determine the diagnostic utility of this unique histopathologic pattern of enteric inflammation.
Materials and Methods
Mice
Hsd: Athymic Nude-Foxn1nu homozygous (nu/nu) and heterozygous mice (nu/+) were purpose-bred for sentinel use in a closed colony at UC Davis. All experimental procedures were approved by the UC Davis Institutional Animal Care and Use Committee (IACUC). Research was conducted in compliance with the Public Health Service Policy on Humane Care and Use of Laboratory Animals and other federal statutes and regulations relating to animals and experiments involving animals. Research adhered to the principles stated in the 1996 and 2011 editions of the National Research Council’s Guide for the Care and Use of Laboratory Animals. The facility where this research was conducted is fully accredited by AAALAC, International.
The Hsd: Athymic Nude-Foxn1nu sentinel breeding colony was established in a high-security barrier facility by embryo rederivation. Once weaned, sentinels were segregated by sex and housed in groups of 4 per cage: 2 Hsd: Athymic Nude-Foxn1nu homozygotes (nu/nu) and 2 Hsd: Athymic Nude-Foxn1nu heterozygotes (nu/+). A single cage of 4 sentinel mice served approximately 80 to 100 colony animals. Sentinels had free access to water and standard laboratory chow (Purina laboratory chow, 5001). Sentinels were housed in a temperature-controlled facility with a 12-hour light/dark cycle. Between 3 to 6 weeks of age, nu/nu and nu/+ sentinel mice were distributed to campus facilities for sentinel surveillance. The number of sentinels utilized per room/rack was facility-dependent, as was the mode of cage ventilation. Sentinels remained within designated rooms for approximately 12 weeks (range, 10–14 weeks), at which time they were submitted to the Comparative Pathology Laboratory (CPL) for rodent health surveillance.
Sentinel Husbandry
Husbandry practices were dependent on the biosecurity level (Table 1) of the facility in which sentinels were housed. Dirty bedding exposure was routine. Briefly, 1 teaspoon to 1 tablespoon of soiled bedding was transferred directly into a new sentinel cage. The soiled bedding was gathered at each cage change from every cage being monitored by the set of sentinels. For individually ventilated cages (IVC), this was performed every 10 to 14 days and weekly for Microisolator (nonventilated) cages. Sentinels were transferred out of the previous cage and into the newly prepared cage with half clean and half soiled bedding.
Husbandry Requirements and Pathogen Status for Different Levels of Facility Biosecurity.
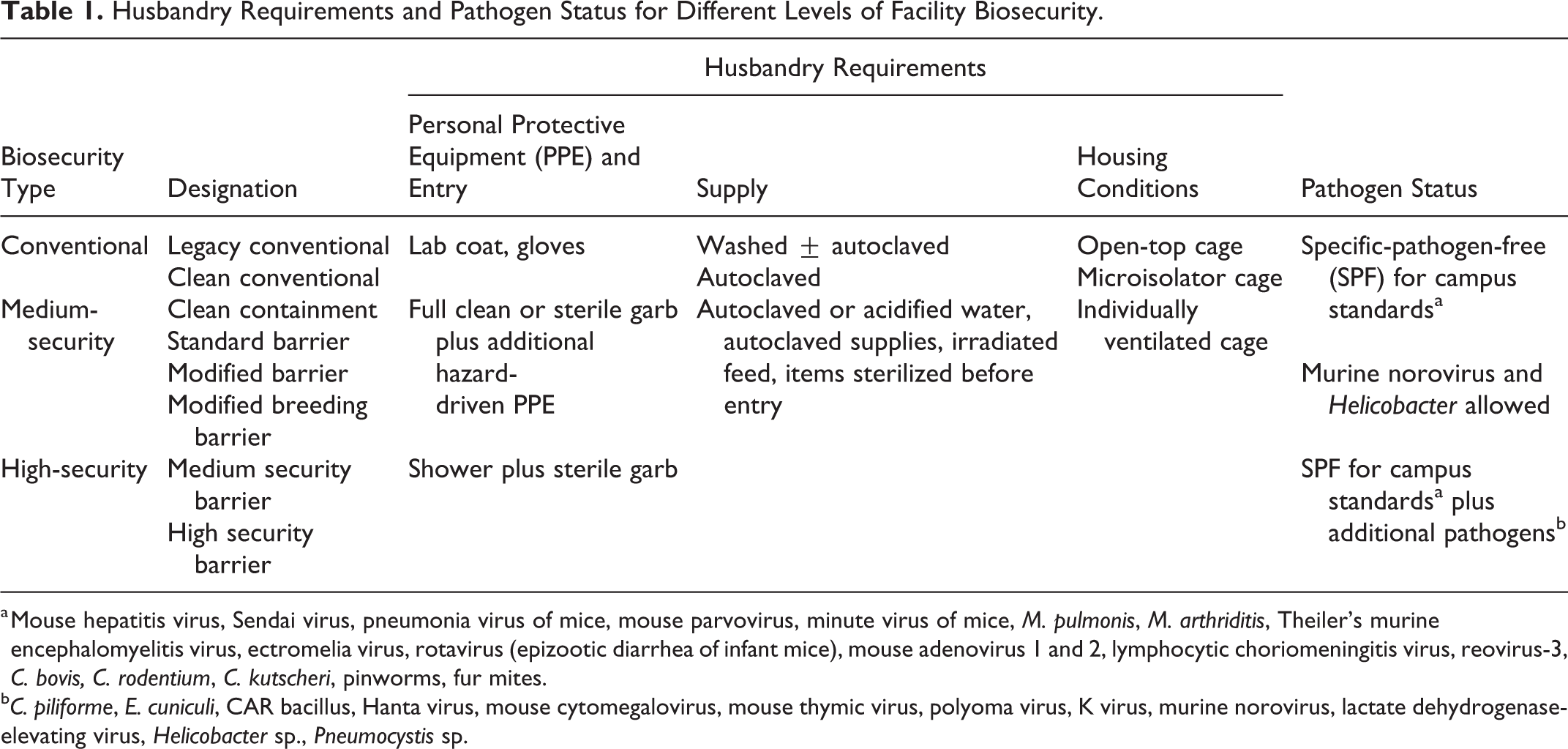
a Mouse hepatitis virus, Sendai virus, pneumonia virus of mice, mouse parvovirus, minute virus of mice, M. pulmonis, M. arthriditis, Theiler’s murine encephalomyelitis virus, ectromelia virus, rotavirus (epizootic diarrhea of infant mice), mouse adenovirus 1 and 2, lymphocytic choriomeningitis virus, reovirus-3, C. bovis, C. rodentium, C. kutscheri, pinworms, fur mites.
b C. piliforme, E. cuniculi, CAR bacillus, Hanta virus, mouse cytomegalovirus, mouse thymic virus, polyoma virus, K virus, murine norovirus, lactate dehydrogenase-elevating virus, Helicobacter sp., Pneumocystis sp.
Due to a wide variety of facilities and biosecurity levels at UC Davis, several bedding types were utilized during the duration of our observational study. However, the most frequently utilized was autoclaved corn cob bedding. Non-autoclaved corn cob bedding was used infrequently within conventional security facilities. Other bedding types sporadically used in conventional security facilities included Sani-Chips, CareFresh, and per request of the investigator, ALPHA-dri. Autoclaved bedding was used in all medium-and high-security facilities.
Similar to bedding variability, drinking water sources were facility-dependent. Sources included tap water, deionized water, autoclaved water, and chlorinated Hydropacs. Chlorinated Hydropacs were tested for aerobic growth 1 month after preparation. Autoclaved water was tested every 1 to 3 months (spore ampule test) depending on its primary use.
Health Surveillance
Sentinel mice submitted for rodent health surveillance were euthanized by CO2 asphyxiation and cardiac exsanguination. If clinical signs were observed (ie, weight loss, diarrhea, and/or rectal prolapse), individual sentinel mice were submitted early, ahead of the scheduled screening date. Health surveillance was performed based on the biosecurity level of the facility from which the sentinels were derived (Table 1). Briefly, sentinel mice were routinely processed for gross examination and varying combinations of serology, parasitology (ie, evaluation of pelage and intestinal contents for fur mites and pinworms, respectively), microbiology (ie, aerobic culture of nasopharynx and ceco-colic contents), and molecular diagnostics (ie, including but not limited to qPCR for Helicobacter sp. and pinworms and RT-qPCR for MHV). Cecal wall samples were utilized for Helicobacter sp. qPCR if individual sick animals were submitted early due to disease. In contrast, pooled fecal pellets were utilized for Helicobacter sp. qPCR from both nu/nu and nu/+ sentinels in the absence of clinical and/or gross disease. If positive for generic Helicobacter sp. assay, Helicobacter speciation was performed using qPCR on the PCR product for the following organisms: H. hepaticus, H. typhlonius, H. bilis, and H. rodentium. Based on gross findings, select tissues were processed for histopathology as needed.
The pathogens tested for in serological profiles included MHV, Sendai virus, pneumonia virus of mice, mouse parvovirus, minute virus of mice, Mycoplasma pulmonis, Mycoplasma arthriditis, Clostridium piliforme, Theiler’s murine encephalomyelitis virus, ectromelia virus, mouse rotavirus (epizootic diarrhea of infant mice), mouse adenovirus 1 and 2, lymphocytic choriomeningitis virus, Encephalitozoon cuniculi, cilia-associated respiratory bacillus, Hanta virus, reovirus 3, mouse cytomegalovirus, mouse thymic virus, polyoma virus, K virus, and murine norovirus (MNV).
Histopathology
If clinical signs were observed, all major organ tissues were processed for histopathology. If gross abnormalities were noted in routinely submitted sentinels lacking clinical signs, only select tissues were harvested and submitted for histopathology. Because of rodent health surveillance volume, tissues from sentinel mice lacking clinical signs and/or gross abnormalities during sentinel processing were not routinely archived or submitted for histopathology. However, a small cohort of mice were selected to evaluate for microscopic disease (ie, non-clinical, no gross lesions) in nu/+ cage mates of affected (ie, clinical or gross lesions) nu/nu sentinels. Thus, the nu/+ cohorts in 12 cages of sentinels containing affected nu/nu mice (n = 24 nu/+) were evaluated histologically.
Tissues were immersion-fixed in 10% neutral-buffered formalin for 72 hours. Formalin-fixed tissues were processed routinely, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. Select sections were stained with Gram and acid-fast (Ziehl-Neelsen and Fite’s) stains. Steiner’s silver stain was run to evaluate for the presence of intralesional argyrophilic spiral bacteria. Intestinal tracts from n = 15 mice (n = 8 Helicobacter PCR-positive and n = 7 Helicobacter PCR-negative) were blindly evaluated by a single pathologist (D. M. Imai) for the presence or absence of argyrophilic spiral bacteria.
Twenty homozygous athymic nude sentinel mice diagnosed with proliferative typhlocolitis were randomly selected from archived necropsy records. Ten mice were positive, and 10 were negative for Helicobacter sp. using qPCR. All mice were negative for MHV (RT-qPCR) as well as other pathogens of significance using bacterial culture, qPCR, and direct fecal examination. An ordinal histopathologic grading scale (score 0–4) was designed to evaluate cecal and colonic mucosal hyperplasia, degree of inflammation, multinucleated giant cell number, and mitotic figures. A binary histopathologic grading scale (score 0 or 1) was used to evaluate for crypt abscessation, ulceration, lymphoid aggregates, goblet cell loss, and the presence or absence of stomach and small intestinal involvement. Each feature was evaluated and graded continuously across all mice in a blinded manner before evaluating the remaining features.
Immunohistochemistry
The following immunohistochemical protocols for pancytokeratin, Mac-3, Iba-1, and F4/80 (respectively) were used to further characterize intralesional multinucleated cells. Antigen retrieval was performed with proteinase K (Dako S3020, Carpinteria, CA, USA) at room temperature for 7 minutes or EDTA (Ventana CC1) at 95°C to 100°C. Primary antibodies included pancytokeratin (PCK-26, Abcam, Cambridge, MA, USA, ab6401, diluted 1:500), Mac-3 (clone M3/84, BD Biosciences, USA, Catalog #553322, diluted 1:200), Iba-1 (Wako Pure Chemicals Industries, Ltd., Osaka, Japan, Catalog #09-19741, diluted 1:200), and F4/80 (CL: A3 -1, Abcam, Cambridge, MA, USA, Catalog #ab6640, diluted 1:100). Secondary antibodies and detection were achieved with the Biocare Medical 4+ Detection System (mouse link HM606), Biocare Medical Streptavidin-HRP (HP604), and NovaRed for peroxidase (Vector SK-4800, Burlingame, CA, USA), OmniMap anti-rat HRP (Ventana 760-4457) and ChromoMap DAB detection system (Ventana 760-159 or 760-124), OmniMap anti-rabbit HRP (Ventana 760-4311), or ChromoMap anti-rat HRP (Ventana 760-4456). Sections were counterstained in Mayer’s hematoxylin (Sigma-Aldrich, St. Louis, MO, USA), air dried, and coverslipped. Internal positive controls included epithelial cells (ie, gastric and/or intestinal epithelial cells) and intralesional macrophages, respectively.
Electron Microscopy
Targeted samples from formalin-fixed paraffin-embedded tissues were processed routinely for ultrastructural evaluation. The area of interest was removed from a paraffin block with 100% xylene for clearing overnight. 23 The sample was rehydrated to a mixed aldehyde and processed using a standard protocol. 19 The sample was embedded in an Epon/Araldite resin mixture. Ultrathin sections were cut on a Leica Ultracut UCT (Leica, Austria) and stained with uranyl acetate and lead citrate before viewing on a Philips CM120BT electron microscope (FEI, Hillsboro, OR, USA made in Eindhoven, The Netherlands). Images were acquired with a Gatan BioScan, model 792, digital camera (Pleasanton, CA).
Statistical Analysis
Continuous data are presented as mean ± SEM. Ordinal data are presented as median and range. Group comparisons were performed using Kruskal-Wallis analyses using GraphPad Prism (San Diego, CA). Categorical data were analyzed using a Fisher’s exact test. Differences were considered significant at P ≤ .05.
Results
Epidemiology
From February 2015 through August 2016, there were 6703 Hsd: Athymic Nude-Foxn1nu sentinel mice submitted for health surveillance screening at the CPL. Out of these, 3363 sentinels were nu/nu (50.2%), and 299 (8.9%) of these nu/nu sentinels were processed for histopathology based on clinical or gross evidence of a variety of diseases. Of the 299 nu/nu sentinels, 159 (53.2%) were diagnosed with proliferative typhlocolitis (Supplemental Table S1). Of the 159 cases of proliferative typhlocolitis, 30 (18.9%) were clinically ill and submitted early due to any combination of nonspecific weight loss, abdominal distension, diarrhea, and/or rectal prolapse. The remaining 129 (81.1%) cases were not recognized as clinically ill and were submitted for histologic processing following observation of ceco-colonic thickening, serosal nodules, and/or hepatosplenic foci of pallor (see Gross Findings) during routine sentinel screening. Of the 159 cases of proliferative typhlocolitis, most came from medium-security (81/159, 50.9%) and conventional (65/159, 40.9%) facilities, with far fewer coming from high-security (13/159, 8.2%) facilities. As sentinel mice were co-housed 4 per cage (2 nu/nu and 2 nu/+), a small subset of cages was evaluated to assess concurrent disease presence in nu/+ cage mates (ie, non-clinical, no gross lesions) of affected nu/nu sentinels. In 3 out of 24 (12.5%) nu/+ mice (evaluated in n = 12 cages), only mild evidence of proliferative typhlocolitis was identified. All 3 mice with mild evidence of proliferative typhlocolitis were positive for both Helicobacter sp. and E. coli.
Gross Findings
Affected nu/nu sentinels exhibited a constellation of gross findings secondary to gastrointestinal disease, including perianal fecal staining, decreased body condition scores, and dilation of the intestinal tract visible through the ventral and ventrolateral abdominal walls (Fig. 1). Grossly, cecal and colonic walls were opaque, mottled pale-tan to pink, and mildly to severely thickened (Fig. 2). Distal colonic segments lacked formed fecal pellets and contained soft, pasty, light-brown fecal material. Mesenteric lymph nodes and spleens were frequently enlarged. Occasionally, multiple pinpoint to 0.2-cm diameter foci of pallor were visible on the splenic or hepatic capsular surface, with extension into the subjacent parenchyma. Less frequently, 0.1- to 0.3-cm, pale tan, smoothly contoured, exophytic nodules were present on the serosal surface of the intestine or within the mesentery.
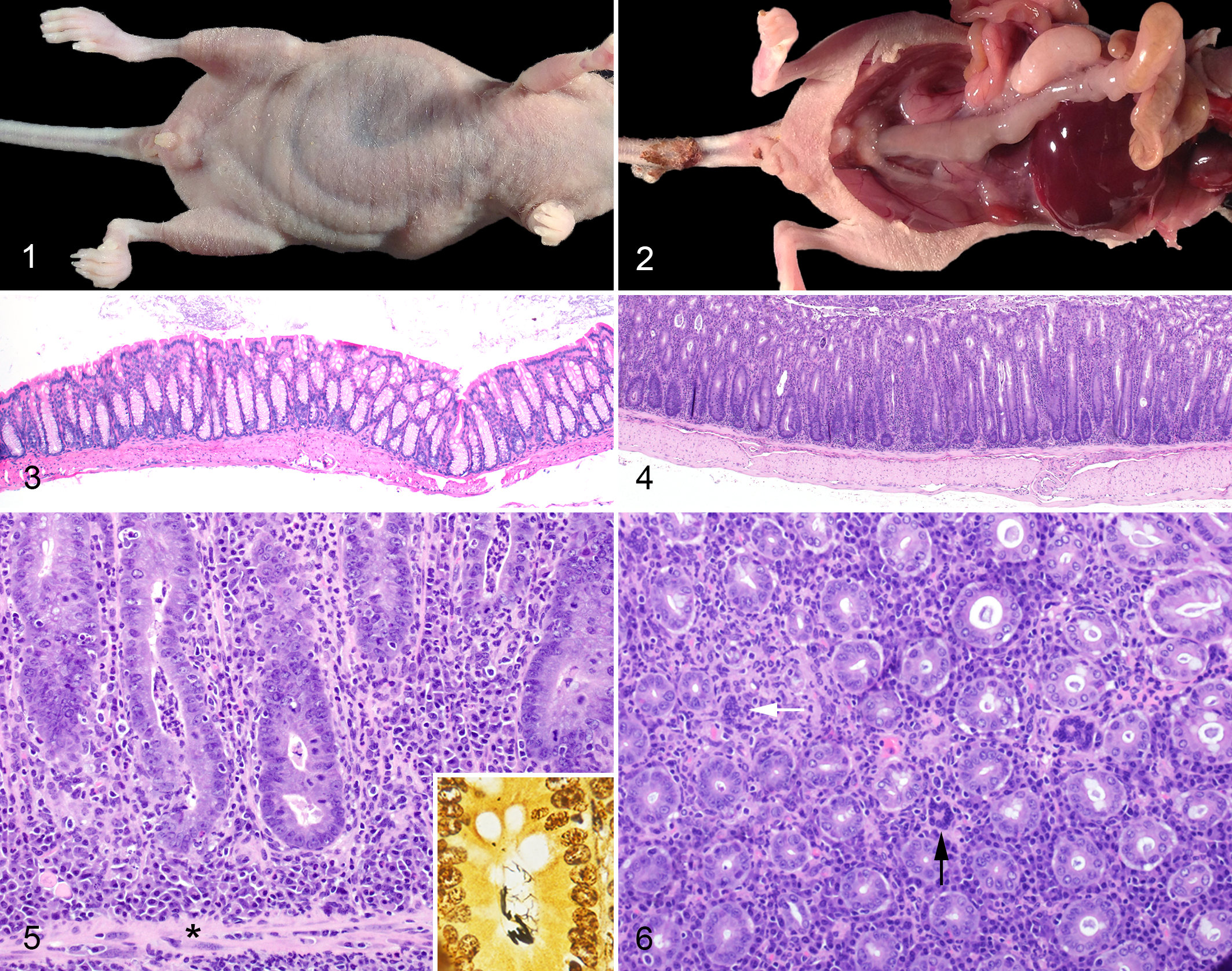
Histopathology Findings
Compared to unaffected cagemates (Fig. 3), affected cecocolonic segments were characterized by diffuse hyperplastic typhlocolitis and decreased gastrointestinal contents (Fig. 4). Proliferative lesions were most frequently noted within the colon and cecum, infrequently within the duodenum, and only rarely within the other parts of the small intestine and/or stomach. The typhlocolitis was characterized by a predominantly neutrophilic and plasmacytic inflammatory infiltrate, admixed with fewer lymphocytes and histiocytes. Inflammatory cells expanded the lamina propria, elevated mucosal crypts, and occasionally extended beyond the muscularis mucosae to include the submucosa, tunica muscularis, and serosa (Fig. 5). The mucosal proliferation was characterized by pronounced crypt elongation, densely crowded columnar mucosal epithelial cells, and an increased mitotic rate. Multinucleated cells were identified within the lamina propria in 131 of 159 (82.4%) mice with proliferative typhlocolitis. If segregated by barrier type, multinucleated cells were present histologically in 11 of 13 (84.6%) cases from high barrier facilities, 70 of 81 (86.4%) cases from medium barrier facilities, and 50 of 65 (76.9%) cases from conventional facilities. Typically, multinucleated cells were round to irregularly shaped with randomly oriented nuclei or poorly demarcated nuclear aggregates (Fig. 6). Crypts were occasionally dilated and contained sloughed epithelial cells admixed with neutrophils. Slender, argyrophilic spiral bacteria were identified in many mucosal crypts using Steiner’s silver staining (Fig. 5, inset). Argyrophilic spiral bacteria were only observed within the intestinal tract of animals that tested positive for Helicobacter sp. by qPCR. Additional but less frequent ceco-colic lesions included superficial mucosal ulceration, goblet cell loss, and mucosal and submucosal lymphoid aggregates. No additional organisms were identified within the tissues (including within multinucleated cells) using Gram or acid-fast (Ziehl-Neelsen and Fites) stains.
Foci of hepatic and splenic pallor corresponded to multifocal areas of necrosis (Figs. 7, 8.) composed of central eosinophilic acellular debris peripherally rimmed by abundant basophilic karyorrhectic debris, scattered histiocytes, and frequent multinucleated cells (see aforementioned description). Similarly, intestinal serosal nodules were composed of necrosis admixed with transmural aggregates of histiocytes, neutrophils, and multinucleated cells embedded within a fibrous connective tissue stroma (Fig. 9). A subset of enlarged lymph nodes was effaced by sheets of neutrophils, histiocytes, plasma cells, and multinucleated cells (Fig. 10). Lymph node enlargement was predominantly due to cortical lymphofollicular hyperplasia and marked medullary plasmacytosis. Splenic enlargement was due to a combination of lymphofollicular hyperplasia, plasmacytosis, and extramedullary hematopoiesis (not shown).
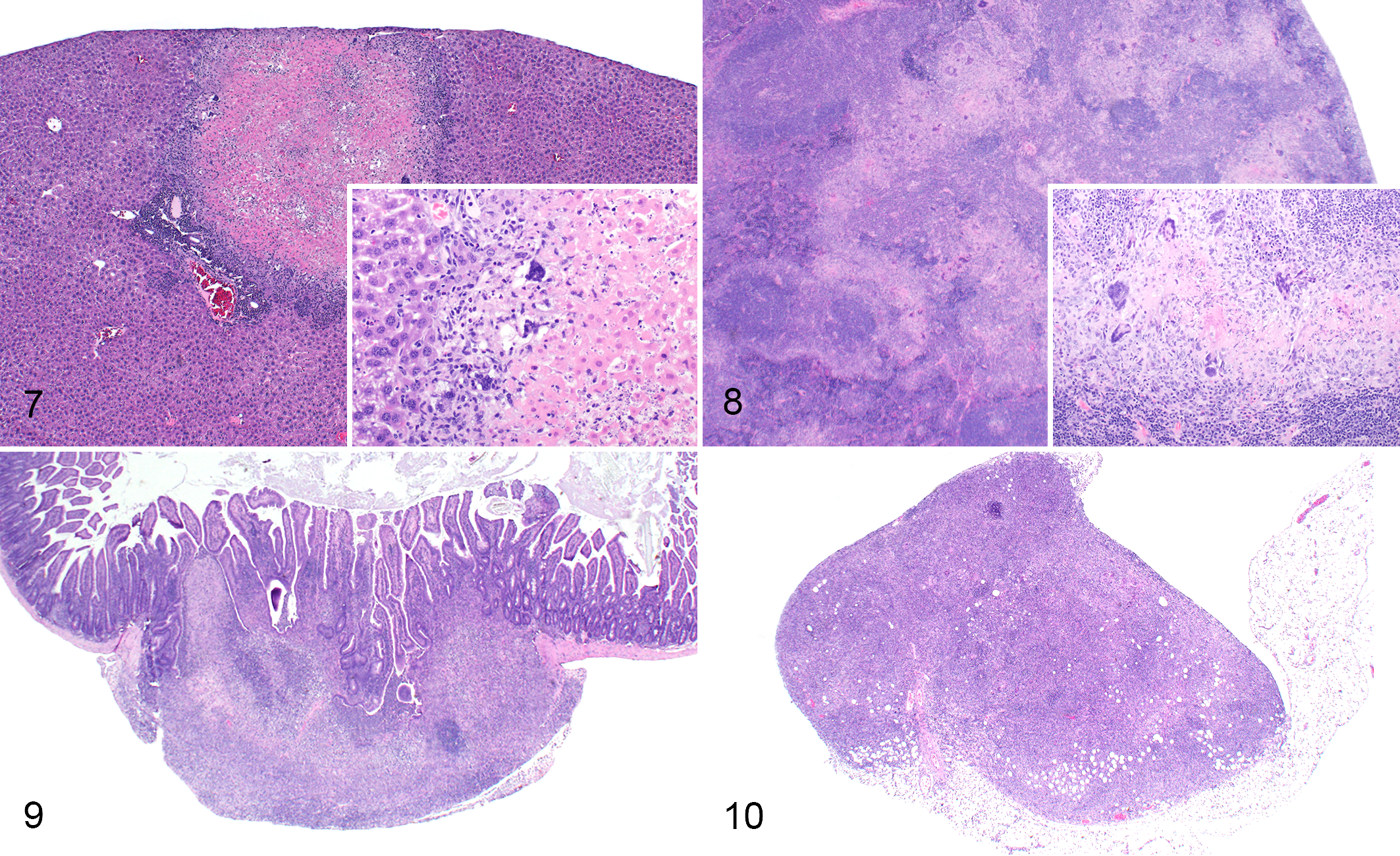
Exclusion of Mouse Hepatitis Virus as a Causal Agent
Susceptibility to MHV is dependent on host immune status and viral strain. Certain strains of MHV have been shown to induce proliferative typhlocolitis, hepatosplenic necrosis, and/or peritonitis with intralesional multinucleated cells (epithelial syncytia) in immunodeficient mice. 4,17,49 Thus, ancillary diagnostics were employed to rule out MHV as a potential pathogen. In addition to proliferative typhlocolitis, 8 nu/nu sentinels from 2 separate facilities and 4 rooms exhibited gross and/or histologic lesions suggestive of potential MHV infection. Lesions included pyogranulomatous to necrotizing hepatitis, splenitis, peritonitis, and lymphadenitis with multinucleated cells (Figs. 7–10). Serologic testing of nu/+ cagemates for MHV was negative in all cases, as were additional sentinels submitted per request from affected rooms. Fecal and lymph node tissues were negative for MHV using RT-qPCR performed at CPL. Negative status of fecal and lymph node tissues was confirmed using RT-qPCR at Charles River Laboratories. Furthermore, RT-qPCR for MHV was negative on scrolls of formalin-fixed paraffin-embedded tissues exhibiting MHV-like histologic lesions. Ultrastructural evaluation of multinucleated cells did not reveal any viral particles or bacteria.
Multinucleated Cells are of Macrophage-Origin
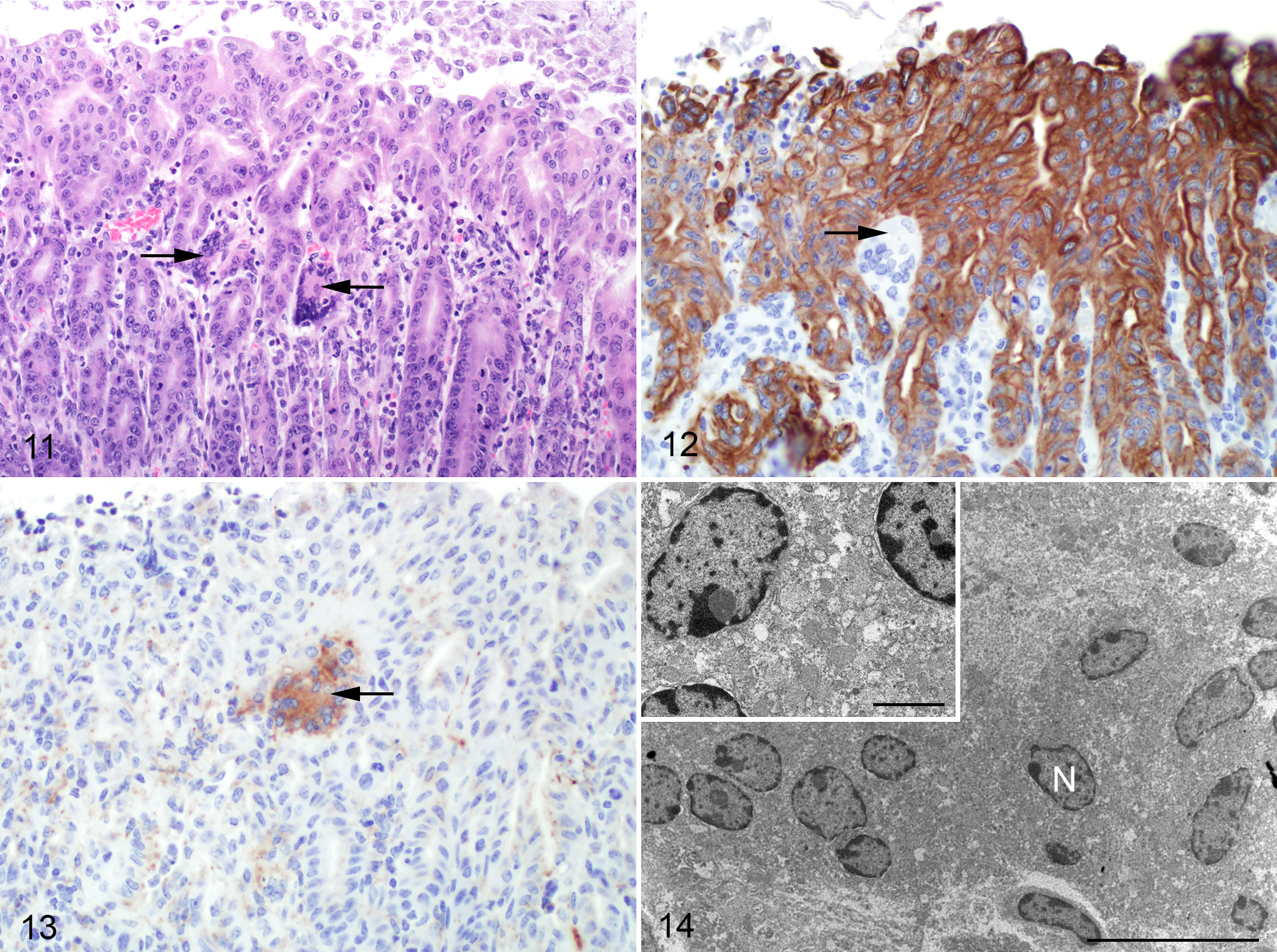
Given the varied morphologic appearance of multinucleated cells (Figs. 6, 11) throughout the intestinal, splenic, hepatic, and serosal lesions, immunohistochemistry and electron microscopy were employed to differentiate multinucleated giant cells of macrophage origin from epithelial-derived syncytia. Multinucleated cells exhibited diffuse, dark-brown, cytoplasmic immunoreactivity to Mac-3 (Fig. 12) and membranous immunoreactivity to Iba-1 (not shown) and lacked immunoreactivity to both F4/80 (not shown) and pancytokeratin (Fig. 13), thus confirming their macrophage lineage. Electron microscopic features of multinucleated cells (Fig. 14) included multiple nuclei with varied amounts of euchromatin, heterochromatin, and occasional nucleoli. Cytoplasmic features included mitochondria, smooth and rough endoplasmic reticulum, and free ribosomes. Neither epithelial characteristics (ie, cytokeratin filaments, desmosomes, etc) nor intracellular infectious organisms were evident.
Identified Agents Differ Between Conventional, Medium-Security, and High-Security Facilities
Of the nu/nu sentinels with proliferative typhlocolitis, 40 of 156 (25.6%) were PCR-positive for Helicobacter sp. (Fig. 15). The most common Helicobacter species identified were H. hepaticus (29/40, 72.5%) and H. typhlonius (4/40, 10.0%). The remaining 6 of 40 (15.0%) sentinels were PCR-positive for Helicobacter agents not routinely speciated at CPL. All Helicobacter-infected sentinels were from conventional or medium-security facilities, not high-security facilities. Seropositivity to MNV was detected in 16 of 106 (15.1%) conventional and medium-security housed mice. In 13 of 16 cases (81.3%), MNV-seropositive mice with proliferative typhlocolitis were co-infected with either Helicobacter sp. (3/13, 23.1%), lactose-fermenting E. coli (3/13, 23.1%), Aspiculuris tetraptera (1/13, 7.8%), or a combination of Helicobacter sp. and other opportunistic bacteria (6/13, 46.2%).
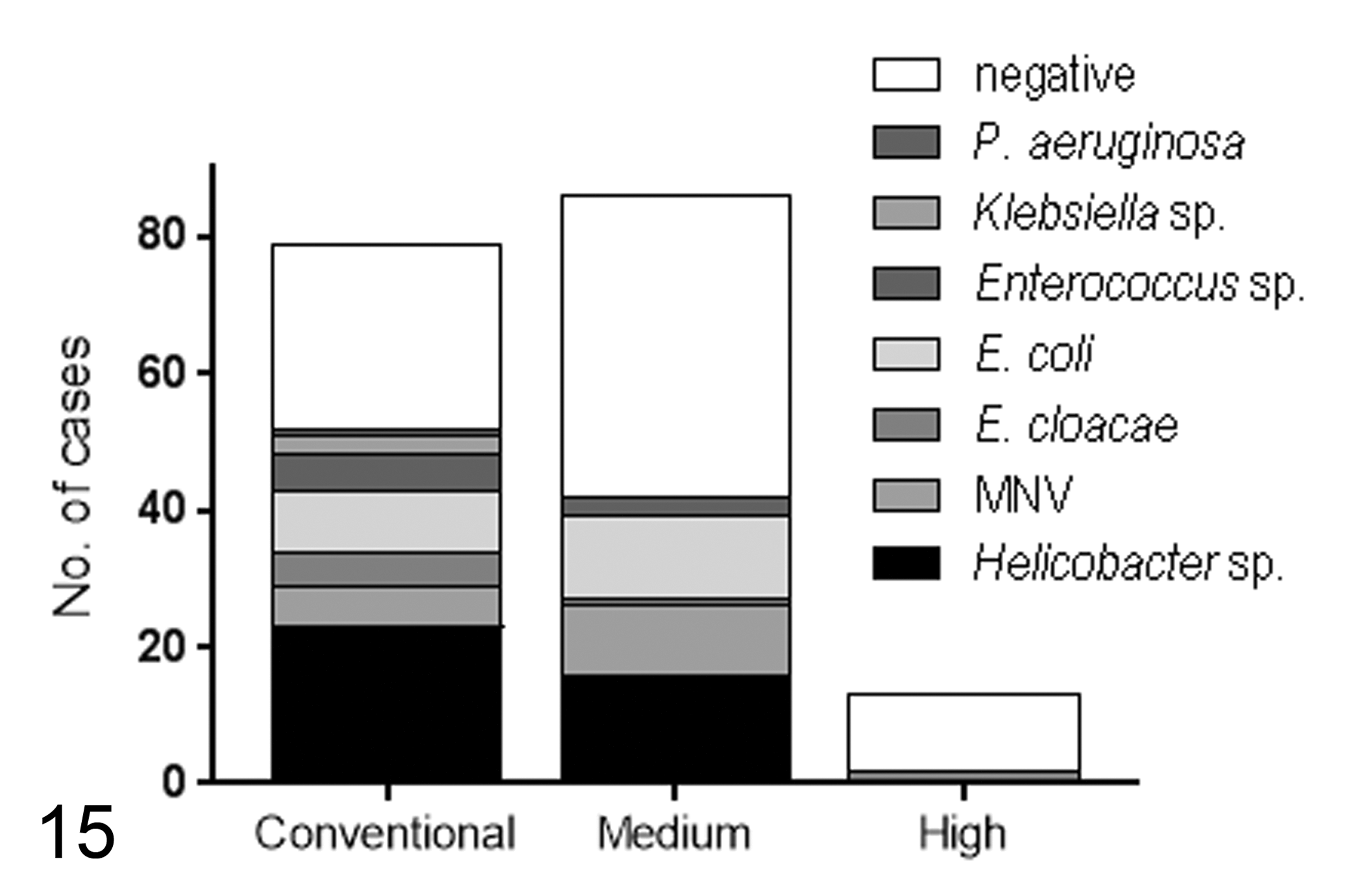
Pathogens associated with proliferative typhlocolitis in nu/nu sentinels by facility type. Isolated agents within high-security facilities were limited to E. cloacae, while a variety of primary and opportunistic agents were identified in nu/nu sentinels within medium-security and conventional facilities. Note the high number of cases for which a causative agent was not identified (negative) across all facility types. MNV, murine norovirus.
In conventional and medium-security facilities, additional identified agents included E. coli (21/145, 14.5%), Enterobacter cloacae (6/145, 4.1%), Aspiculuris tetraptera (2/146, 1.4%), Klebsiella sp. (3/145, 2.1%), Enterococcus faecalis or Enterococcus sp. (8/145, 5.5%), Acinetobacter sp. (1/145, 0.7%), Pseudomonas aeruginosa (1/145, 0.7%) and Ralstonia picketti 45 (1/145, 0.7%). In high-security facilities, E. cloacae was recovered from 2 of 13 (15.4%) affected nu/nu sentinels. No organisms were identified in 71 of 146 (48.6%) affected nude sentinels from conventional and medium-security facilities and 11 of 13 (84.6%) affected nude sentinels from high-security facilities.
Multinucleated Giant Cell Formation Is Not Agent-Dependent
Once intralesional multinucleated cells were confirmed to be of macrophage origin, we next assessed their association with isolated viral, bacterial, and parasitic pathogens (Table 2). There were no statistical associations between the histologic presence or absence of multinucleated giant cells and the most frequently identified agents (Helicobacter sp. P = .1381, E. coli P = .1381, MNV P = .1863, Enterobacter sp. P = .1481). Statistical analyses were not performed on the following agents due to low prevalence: Klebsiella sp., pinworms, R. picketti, P. aeruginosa, Acinetobacter sp.
Intralesional Multinucleated Giant Cells (MNGC) Are Not Agent-Dependent.
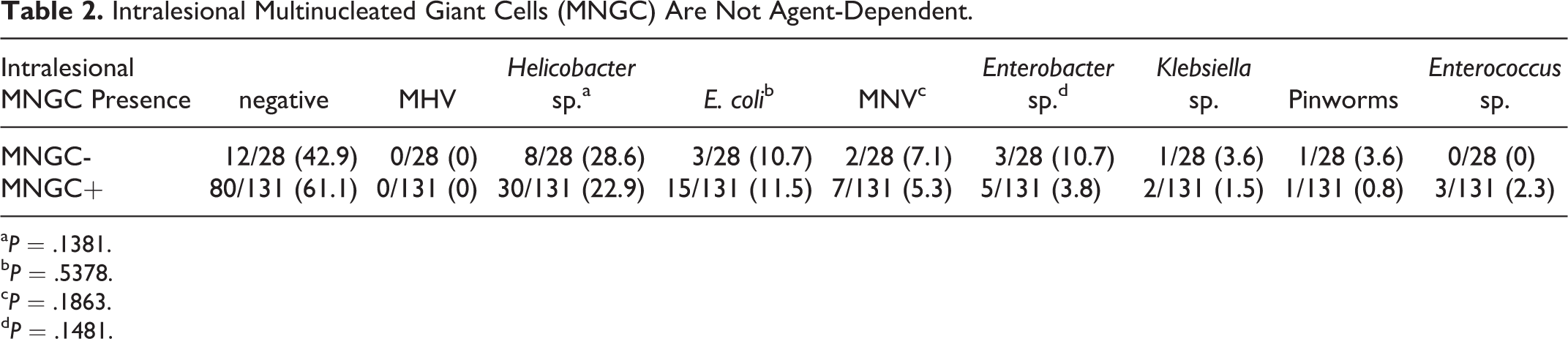
a P = .1381.
b P = .5378.
c P = .1863.
d P = .1481.
We further compared the histologic features of our most frequently identified infectious disease agent, Helicobacter sp. Histologic features of 10 Helicobacter-positive nu/nu sentinels were compared against 10 Helicobacter-negative nu/nu sentinels. There was no statistically significant difference in multinucleated giant cell presence or number noted between Helicobacter-positive and Helicobacter-negative nu/nu sentinels (Fig. 16). No statistical difference was noted between the 2 groups in degree of gastric involvement, small intestinal involvement, severity of inflammation overall, cecal mucosal hyperplasia, or colonic mucosal hyperplasia (Fig. 16). Furthermore, no statistical difference was noted between the 2 groups in degree of inflammation, crypt abscessation, mucosal ulceration, lymphoid aggregates, mitotic figures, or goblet cell loss (data not shown).
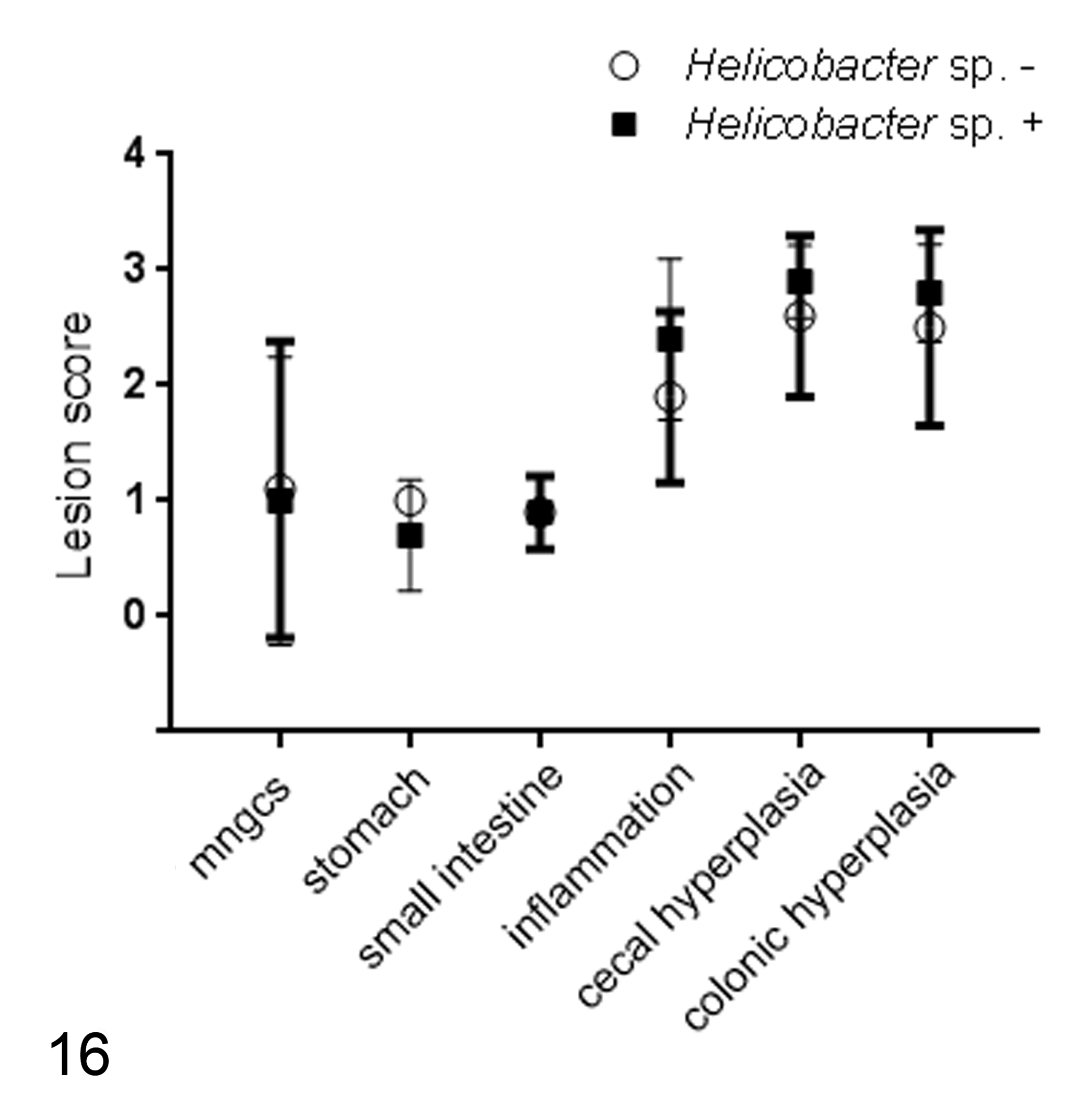
Anatomic distribution and histopathologic features of proliferative typhlocolitis, including multinucleated giant cells, do not correlate with Helicobacter sp. infection.
Discussion
A syndrome of proliferative typhlocolitis was identified in nu/nu sentinels across multiple rodent animal housing facilities at UC Davis. Diagnostic testing determined that facility biosecurity status (ie, conventional, medium-security, or high-security facilities) was directly related to identified agent(s). In conventional or medium-security facilities, Helicobacter sp., MNV, and opportunistic bacteria were most frequently identified in cases of proliferative typhlocolitis. In contrast, in high-security facilities, only opportunistic non-Helicobacter agents were identified in sentinels exhibiting proliferative typhlocolitis. Regardless of which organisms, if any, were identified in affected nu/nu sentinel mice, the unique histopathologic pattern (eg, colonic hyperplasia, pleocellular typhlocolitis, multinucleated giant cells, etc) was not specific for any 1 agent or type of agent. Rather, this histologic pattern should be used as an indicator for additional Helicobacter sp. and MNV testing, particularly in conventional and medium-security husbandry conditions.
Murine models of inflammatory bowel disease encompass a variety of contributory factors, including genetic strain and intestinal microflora. Helicobacter-induced proliferative typhlocolitis is a well-established model of inflammatory bowel disease. 6 –9,15,16,43 Of the enterohepatic Helicobacter sp., several are documented to cause proliferative typhlocolitis in immunodeficient strains, such as IL10 KO 8,27,43 and SCID mice. 7,31 Helicobacter hepaticus is the most frequently implicated species 7,9,15,16,48 ; however, many other Helicobacter species such as H. bilis, 22,32,44 H. rodentium, 8,36,38,44 H. typhlonius, 8,36 H. mastomyrinus, 12,36 H. apodemus, 36 and most recently, H. japonicum 43 have been associated with murine typhlocolitis, rectal prolapse, and/or carcinoma development. Akin to what others have found, Helicobacter hepaticus was the most frequently identified species in our nu/nu sentinels (29/40, 72.5%), followed by Helicobacter typhlonius (4/40, 10.0%). At CPL, speciation of Helicobacter is limited to H. hepaticus, H. typhlonius, H. bilis, and H. rodentium. In our cohort, 6 of 40 (15.0%) Helicobacter sp. positive sentinels were PCR-positive for Helicobacter agents not routinely speciated at CPL. Thus, these sentinels could reflect infection with more recently identified species of Helicobacter such as H. mastomyrinus, 12,36 H. apodemus, 36 and/or H. japonicum. 43
More importantly, characterizing the role of Helicobacter sp. in proliferative typhlocolitis development is challenging within the context of our observational study. A large percentage (48.6% in conventional and 84.6% in medium-security facilities) of mice exhibited proliferative typhlocolitis in the absence of any pathogen screened for within our rodent health surveillance program. This calls into question the causal relationship between our isolated pathogens (including Helicobacter sp.) and inflammatory disease. In our cohort, however, the presence and absence of argyrophilic spiral bacteria within inflamed and hyperplastic large intestinal tissue was perfectly correlated with Helicobacter PCR positivity and negativity. This finding, coupled with the vast Helicobacter-induced proliferative typhlocolitis literature, lends support to the role of Helicobacter as 1 cause of large intestinal inflammation within our sentinel population.
Following the discovery of MNV in 2003, 25 several studies have demonstrated the variable impacts of MNV infection in mice. Disease susceptibility is widely dependent on viral strain, mouse immune status, intestinal microbiota, and co-infection with opportunistic pathogens such as Helicobacter. 5,20,21,24,29,30 More specifically, the contributory role of MNV was demonstrated in coinfection studies of Mdr1a-/- mice with MNV4 and Helicobacter bilis, 30 where co-infected mice developed greater weight loss and IBD scores. In contrast, MNV4 monoinfection, or coinfection of MNV4 and Helicobacter, in Smad3-/- 29 and IL10-/- mice 20 did not induce IBD or impact its severity. In our affected nu/nu sentinels, a low overall level (13.1%) of MNV seroconversion was detected and only detected in conventional (6/48, 12.5%) and medium-security sentinels (10/58, 17.2%). Most of the MNV seropositive sentinels (81.3%) were co-infected with 1 or more of the following agents: Helicobacter sp., E. coli, E. cloacae, E. faecalis, and Aspiculuris tetraptera. Thus, while MNV does not appear to act as a primary cause of proliferative typhlocolitis in nu/nu mice, its role as a co-pathogen is possible.
In our facilities, a clear distinction arose between agents associated with disease in conventional or medium-security facilities versus high-security facilities. Per design, biosecurity designations for sentinels at UC Davis are stratified according to tiers of husbandry requirements and pathogen exclusion. At minimum, our conventional facilities exclude the following pathogens: MHV, Sendai virus, pneumonia virus of mice, mouse parvovirus, minute virus of mice, M. pulmonis, M. arthriditis, Theiler’s murine encephalomyelitis virus, ectromelia virus, rotavirus (epizootic diarrhea of infant mice), mouse adenovirus1 and 2, lymphocytic choriomeningitis virus, reovirus-3, Corynebacterium bovis, Citrobacter rodentium, Corynebacterium kutscheri, pinworms, and fur mites. Our high-security facilities additionally exclude the following: C. piliforme, E. cuniculi, CAR bacillus, Hanta virus, mouse cytomegalovirus, mouse thymic virus, polyoma virus, K virus, MNV, lactate dehydrogenase-elevating virus, Helicobacter sp., and Pneumocystis sp. Thus, despite the presence of proliferative typhlocolitis in high-security facilities, these excluded pathogens were not identified. Rather, opportunistic bacterial pathogens within the normal microbiota (eg, E. cloacae) may be contributing to lower bowel disease. This suggestion is supported by additional reports of commensal-driven inflammatory bowel disease in various strains of immunodeficient mice. 1,18,26,37,46 Furthermore, when nu/+ cage mates of affected nu/nu sentinels were evaluated histologically, 3 out of 24 (12.5%) were found to have only mild evidence of proliferative typhlocolitis. All 3 nu/+ sentinels were positive for both Helicobacter sp. and E. coli. This suggests that documented causes of proliferative typhlocolitis may induce subclinical microscopic disease in immunocompetent sentinels, whereas commensals are unlikely to drive subclinical microscopic disease in this population.
In addition to proliferative typhlocolitis, a small proportion of affected animals exhibited lesions suggestive of MHV infection (eg, necrotizing splenitis and hepatitis, granulomatous serositis and lymphadenitis, intralesional multinucleated cells). MHV produces characteristic gross and histologic lesions 4,49 depending on strain-tropism (eg, polytropic MHV versus enterotropic MHV) and mouse immune status. Atypical presentations have been demonstrated in IFNγ-KO mice inoculated with MHV alone 17 or in combination with H. hepaticus. 10 In these cases, histiocytic to lymphohistiocytic pleuritis and peritonitis predominated, with the former exhibiting intralesional multinucleated epithelial syncytia. Given the immune status of our animals, exclusion of MHV as a potential cause was essential. Serologic testing of heterozygote cagemates was negative in all instances, as was PCR on fecal, lymph node, and lesioned tissues. Furthermore, immunohistochemical and electron microscopic analysis confirmed that intralesional multinucleated cells were of macrophage origin and not viral-induced epithelial syncytia. All affected sentinels had concurrent proliferative typhlocolitis. Thus, the lesion distribution (ie, serosal, mesenteric, hepatic, splenic) points toward systemic dissemination of ceco-colic opportunistic pathogens.
Intralesional multinucleated giant cell formation was an unanticipated feature of proliferative typhlocolitis. Despite differences in morphology and mechanistic development, Langhans-type giant cells and foreign body–type giant cells can be found concurrently within lesions. Fusion of foreign body–type giant cells are typically mediated by T-cell induced cytokines such as IL-4 and IL-13, while Langhans-type giant cells are mediated by IFNγ and a macrophage maturation factor. 35 Despite the partial reliance on T-cell–mediated cytokine production, multinucleated giant cell formation has been documented in athymic nude 41,42 and SCID mice 28 but only rarely within the context of murine inflammatory bowel disease. 28 Despite pathophysiologic differences in giant cell formation, distinct functional roles for Langhans-type giant cells and foreign body–type giant cells are not well defined. Perhaps the formation of multinucleated giant cells in athymic mice is a nonspecific mechanistic adaptation to a variety of microbial organisms based on a limited T-cell–dependent immune response.
As an observational study constructed largely within the confines of the sentinel program infrastructure, there are inherent limitations. Most significantly, we are unable to state the full epidemiology of proliferative typhlocolitis within our sentinel program. Because of sample processing volume, wet tissues are neither archived nor processed for histopathology if a sentinel lacks clinical signs and gross lesions during sentinel processing. Thus, we cannot draw comparisons between all nu/nu sentinels with and without proliferative typhlocolitis or compare isolated pathogens in affected versus nonaffected nu/nu sentinels. Rather, we aimed to compare the presence or absence of identifiable pathogens as well as histopathologic features within nu/nu sentinels displaying proliferative typhlocolitis. Additionally, identification of bacterial agents relies on aerobic culture, serology, and PCR. Thus, our screening program would not detect bacteria that are nonculturable, require special media, require anaerobic or microaerophilic conditions, or are not included in our serological or PCR panels. These limitations aside, the health surveillance panels are designed to detect known pathogens of colony health concern and include methods to detect all recognized infectious disease agents that can cause proliferative typhlocolitis in laboratory mice. Similarly, sample bias (either cecal wall or feces, depending on the number of sentinels per cage) may prevent or inhibit detection of Helicobacter sp. that exhibit intermittent to absent shedding through the intestinal tract. Finally, the immunocompetence of the nu/+ mice could potentially be compromised by heterozygosity for Foxn1nu , but the vendor explicitly repudiates any evidence of immunodeficiency or partial expression of the nu/nu phenotype and does recommend these heterozygotes for use as sentinels.
In summary, proliferative typhlocolitis in nu/nu sentinel mice is a nonspecific disease process that occurs regardless of facility biosecurity level. Proliferative typhlocolitis was seen in association with Helicobacter hepaticus (with or without concurrent murine norovirus seropositivity) within conventionally housed and medium-security nu/nu sentinels. In contrast, only sporadic opportunistic bacteria were identified as potential causes in high-security nu/nu sentinel mice. Pathogens of high colony health concern that are known to cause similar lesions in immunocompromised hosts (eg, MHV) were definitively excluded. Histopathologic lesions, including multinucleated giant cell formation, were indistinguishable between affected Helicobacter-positive and Helicobacter-negative sentinels. Together, these findings suggest that proliferative typhlocolitis with multinucleated giant cells is a nonspecific aberrant inflammatory response to a variety of commensals and/or pathogens in athymic mice.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818798106 - Proliferative Typhlocolitis With Multinucleated Giant Cells: A Nonspecific Enteropathy in Immunodeficient Sentinel Mice
Supplemental Material, DS1_VET_10.1177_0300985818798106 for Proliferative Typhlocolitis With Multinucleated Giant Cells: A Nonspecific Enteropathy in Immunodeficient Sentinel Mice by Kerriann M. Casey, Amanda L. Johnson, Melea N. Hunrath, Jenelle K. Fraser, Nicole C. McCowan, Katherine Wasson, Rosalinda A. Doty, Stephen M. Griffey, and Denise M. Imai in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Acknowledgements
We thank Niki DeGeorge and Cynthia Alcazar from the Comparative Pathology Laboratory for their technical contributions within the rodent health surveillance program. We also like to recognize Christopher Kwong, Felicia DonRusello, Kurt Takahashi, Amber Villareal, and Michael Manzer at the Histopathology Laboratory in the UC Davis Veterinary Medical Teaching Hospital for their expertise with histology and immunohistochemistry. Pat Kysar performed electron microscopy processing and image acquisition. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
