Abstract
Twenty-two feral rock pigeons (Columba livia) from 10 counties in California with ataxia, torticollis, and difficulty standing and flying were admitted to rehabilitation centers in late winter and spring of 2017 and died or were euthanized. Common necropsy findings included thin body condition, generalized deep red discoloration of organs, and hemorrhagic subcutaneous neck tissues. Meningoencephalitis was observed microscopically in 16 pigeons, and 3 also had protozoal schizonts in the brain. The most consistently affected regions of the brain were cerebellum and brainstem. Skeletal muscles, and less frequently the heart, contained large intrasarcoplasmic bradyzoites typically without inflammation. Fifteen of the 22 birds tested positive using pan-Sarcocystis polymerase chain reaction. The sequence of the amplicon was most closely related to S. calchasi, and the 8 subtyped sequences had 100% homology with S. calchasi. This investigation demonstrated the transcontinental and North American spread of S. calchasi causing a disease outbreak in free-ranging rock pigeons, thus warranting increased surveillance in susceptible native columbids.
Since its first identification as the cause of lethal encephalitis in racing pigeons (Columba livia f. domestica) in Germany in 2009, 7 Sarcocystis calchasi infection was described for the first time in the United States in 2011 in a feral rock pigeon from Minnesota 19 and in 2015 in a feral rock pigeon from Tokyo, Japan. 17 Subsequently in the United States, spontaneous S. calchasi infection has been reported sporadically in feral rock pigeons from Minnesota, in a racing pigeon from Missouri, 10 in psittacines from an enclosed aviary in California, 13 in captive roller pigeons in Pennsylvania, 16 and in free-ranging doves from Texas. 3 The life cycle of the parasite remains partially known; rock pigeons and cockatiels (Nymphicus hollandicus) have been demonstrated as intermediate hosts, 10 while the Northern goshawk (Accipiter gentilis) has been shown as the definitive host for the parasite in Europe, 8,9 likely with other European Accipiter hawks. 12 Mechanical transmission or transmission through feed and water between intermediate hosts is also speculated. 6
In late winter and spring of 2017, there was an outbreak of neurologic disease in which free-ranging rock pigeons presented with ataxia and torticollis. Postmortem diagnostic investigations were carried out on 22 birds from 10 California counties (Fig. 1). Of the birds examined, 19 were adults and 3 (bird Nos. 8, 9 and 19) were juveniles; 12 were males and 10 were females. The birds were typically in thin to fair body condition. The most consistent gross lesions were diffusely red internal organs (17), dark red expansion of the neck subcutis (14), and ocular hyperemia (12). Additional findings included enlarged spleens (3), diarrhea (3), severe diffuse liver pallor (1), few gray-tinged pinpoint pulmonary foci (1), fungal plaques on air sacs (1), and hemorrhagic proventricular glands (1).
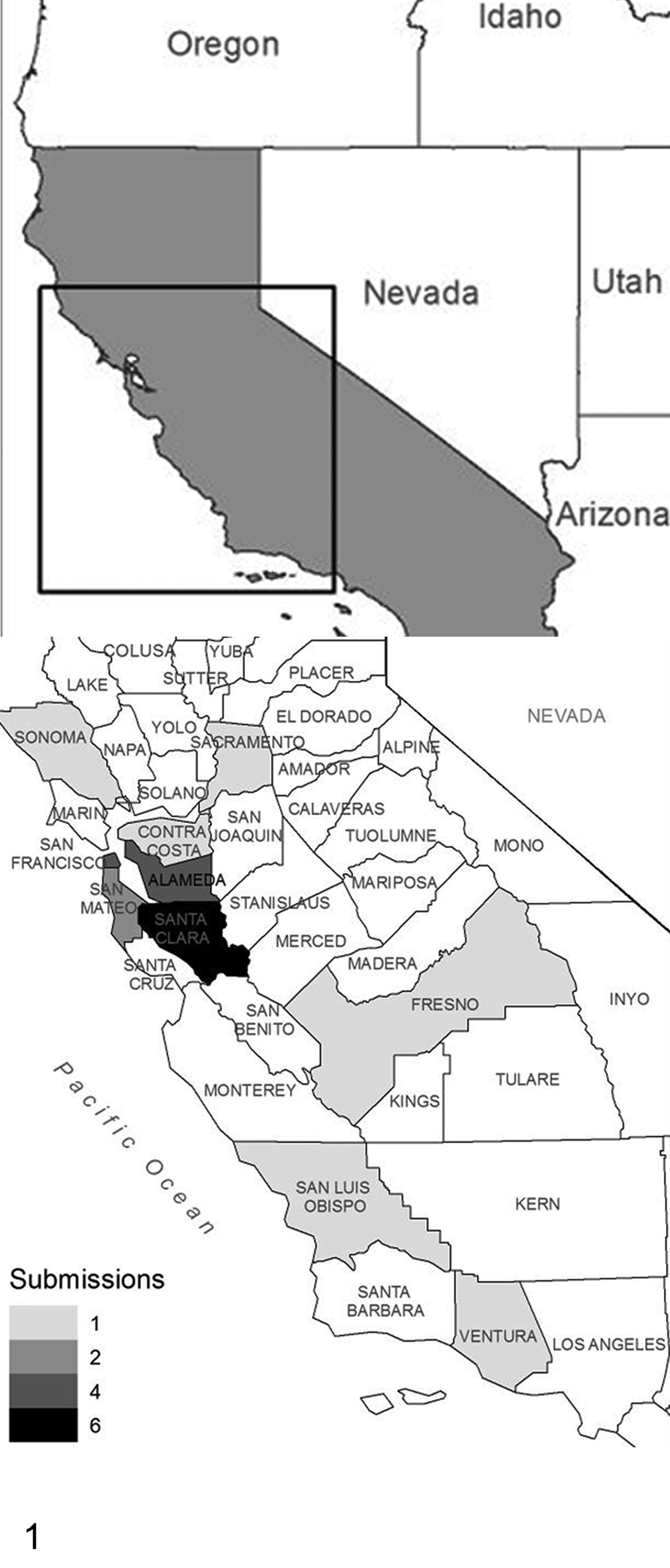
The geographic distribution within California of 22 feral rock pigeons with neurologic disease attributed to Sarcocystis calchasi infection.
Significant histologic lesions were restricted to the brain and skeletal muscle, consisting of inflammation with or without protozoal organisms. The inflammatory lesions were scored as “+” for mild, “++” for moderate, and “+++” for severe infiltrations and as positive or negative for protozoal organisms (Suppl. Table S1). In the brain of 16 pigeons, there was perivascular and generalized lymphohistiocytic necrotizing meningoencephalitis (Figs. 2, 3), which involved the cerebrum and cerebellum in 14 and was limited to the cerebellum and brainstem in 2. Lesions overall were predominated by T cells (Fig. 4), and there were scattered foci of histiocytic nodules (Fig. 5). Occasional vessels had mural inflammatory cells and fibrinoid change (Fig. 6), and bird No. 10 also had few heterophils and occasional Mott cells. In bird Nos. 18 to 22, a reliable histopathological evaluation could not be made based on hematoxylin and eosin (HE) alone, and small groups of CD3-immunopositive T lymphocytes confirmed the encephalitis in bird No. 21. Schizonts were observed in the brains of 3 pigeons, mostly adjacent to or unassociated with inflammatory regions (Fig. 7).
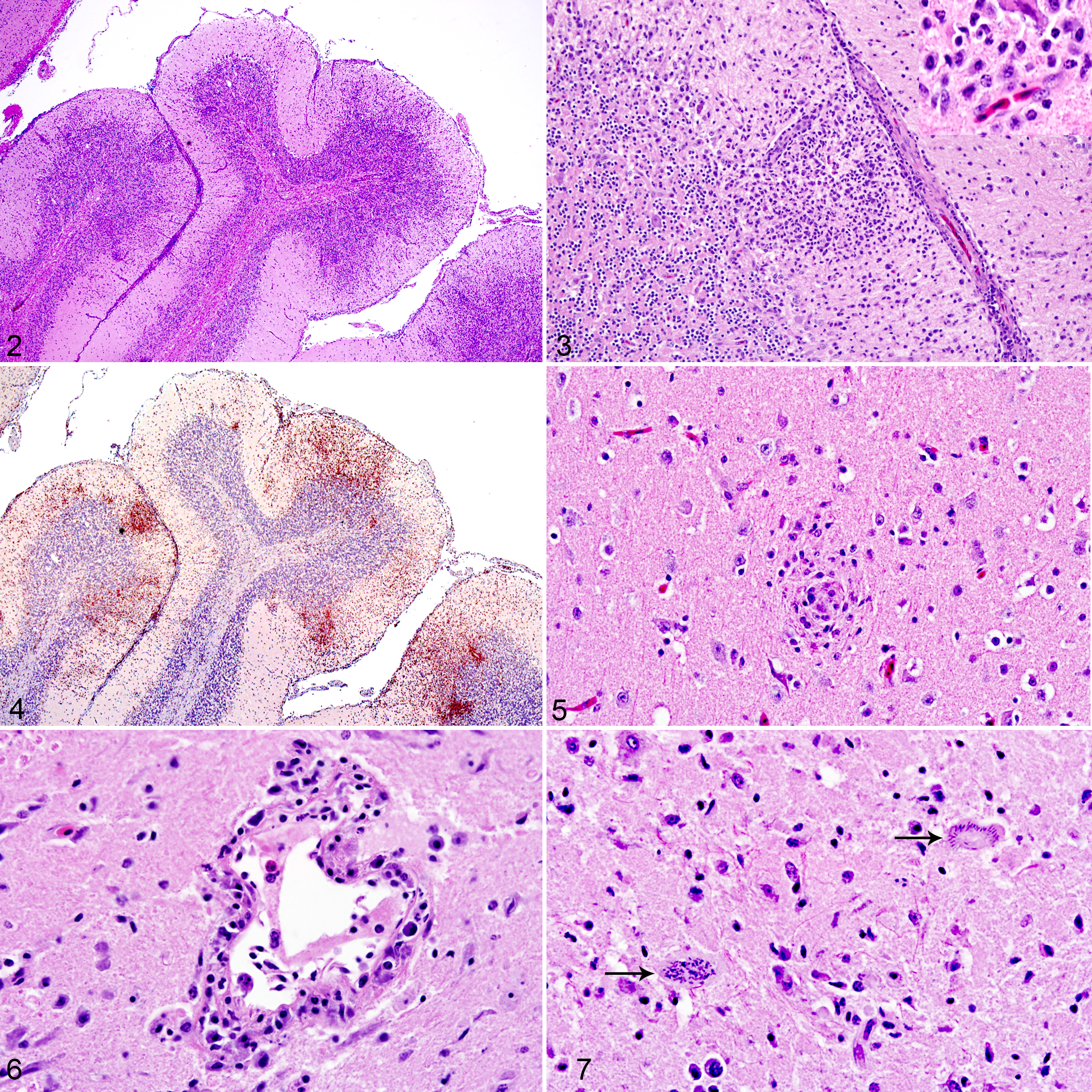
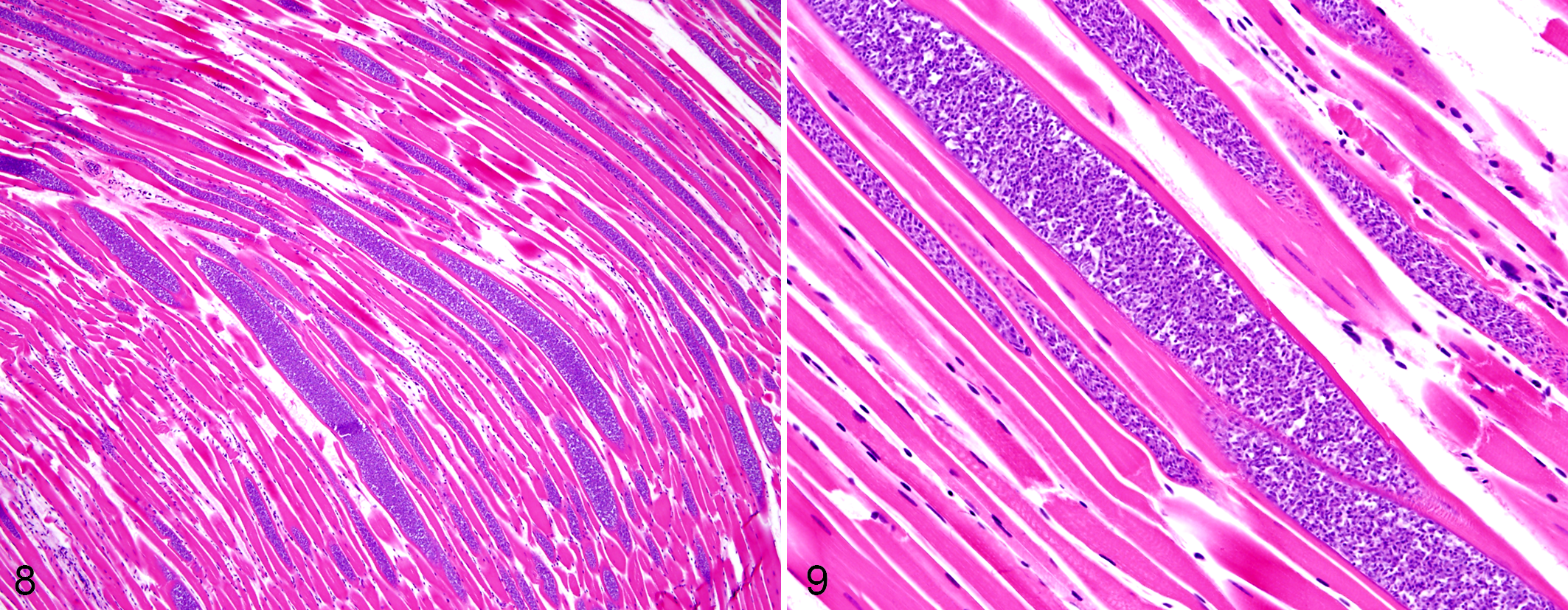
Muscle lesions included lymphohistiocytic rhabdomyositis and rhabdomyonecrosis observed in 3 of the 17 birds, while intrasarcoplasmic protozoal cysts (Figs. 8, 9) were present in 10 pigeons. Bradyzoites were also observed in the heart of 9 birds, none associated with inflammatory lesions. In bird Nos. 5 and 6 where skeletal muscles were unavailable, the heart contained mild to moderate numbers of cysts. Lesions and protozoa were absent in the skeletal muscle and heart of bird Nos. 18 to 22. Pectoral and thigh muscle sections had consistently similar histopathological scores; only pectoral muscle results are shown (Suppl. Table S1).
Other histopathologic findings were severe diffuse lipidosis in the 1 bird with a grossly pale liver, caseonecrotic pneumonia with intralesional bacteria (n = 1) in which rare Salmonella sp. was isolated, fungal air sacculitis and pneumonia (n = 1), and Tetrameres sp. infection in the hemorrhagic proventricular glands (n = 1). The neck subcutis demonstrated ectatic superficial to deep dermal blood vessels with multifocal perivascular fibrin and hemorrhages, as well as occasional protozoal cysts in the deep skeletal muscles.
Frozen brain tissue from all pigeons and pectoral muscle tissues from 17 pigeons were tested for Sarcocystis by polymerase chain reaction (PCR). DNA was extracted as previously described, 4 and a 521-bp portion of the 18S ribosomal RNA gene was amplified and sequenced. 1 The PCR was positive in 19 of 22 pigeons in either muscle (88%) or brain (68%) or both (59%) tissues (Suppl. Table S1). Sequences from 15 pigeons had 100% sequence identity to both S. calchasi and the closely related Sarcocystis columbae genomes. 2 For subtyping, 364 bp of the internal transcribed spacer (ITS) region were amplified by PCR 18 in 8 of the 15 pigeons and sequenced. All samples had 100% sequence identity to S. calchasi (GenBank accession no. KC733715) (Suppl. Fig. S10). Sequences were submitted to GenBank under accession numbers MG273727 (18S) and MG283135 (ITS).
Ancillary tests ruled out avian paramyxovirus 1, Sarcocystis neurona, Sarcocystis falcatula, avian influenza virus, bacterial disease, and heavy metals and selenium abnormalities.
The overall findings in this outbreak of neurologic disease in feral rock pigeons by S. calchasi are similar to previous studies. 5,7 Pigeons here did not have specific or significant gross lesions, as is typical of the described encephalitic form of the disease. 6 Nevertheless, a consistent gross finding was the engorged and sometimes hemorrhagic neck vessels, likely associated with the plexus venosus collaris and agonal changes. Similar gross observations were present in roller pigeons with the hepatitis form of disease, although the histopathological findings were not provided. 16
Hallmarks of S. calchasi infection have been a high prevalence of encephalitis with absent or low numbers of intralesional protozoa, along with abundant cysts in skeletal muscles without inflammation. 3,7,8,10 The latter can be explained by the natural life cycle of the protozoa, and the discrepancy between the presence of encephalitis and the absence of organisms has been attributed to delayed-type hypersensitivity reaction in the brain rather than direct protozoal affect. 5,11 Schizonts were observed in only 3 pigeons, although S. calchasi immunohistochemistry (IHC) may have revealed additional organisms. 10,11
Several pigeons had localized lesions primarily in the brainstem and cerebellum, similar to the initial study of S. calchasi. 7 Thus, examination of ample number of brain sections, in particular the caudal brain, may be essential to make the diagnosis. The same holds for bird No. 19: this was the only pigeon that had negative results on histopathology and PCR, but it had minimal brain tissue available. In other birds where the caudal brain was not available or there was no encephalitis observed in the sections (bird Nos. 18, 20, 22), PCR was positive in the pectoral muscle tissue. This variability of lesions, presence of protozoal organisms, and PCR results are typical for S. calchasi infections under experimental conditions. 5,10,11 Bird Nos. 8 and 9 were negative by PCR on brain tissue and histopathology, but muscle tissues were unavailable and only very small sections of cerebrum with no inflammation were tested, and thus their diagnosis remains inconclusive.
The outbreak described here has ecological and geographic implications. Over the past decade, the infection and disease has been reported in increasing numbers from Europe and the United States. With this outbreak, it has emerged along the West Coast of the United States, causing a disease outbreak in the feral pigeon population in California. The definitive host in North America remains unknown; possible candidates are the Northern goshawk along with other accipiters such as Cooper’s hawks (Accipiter cooperii) and sharp-shinned hawks (Accipiter striatus). Given the wide geographic range of feral rock pigeons and the potential definitive hosts, spillover of S. calchasi to native columbids is a real concern. While rock pigeon populations are not expected to be affected by this disease, infection may pose a potential threat to already declining populations of native columbids in California and elsewhere, including mourning doves (Zenaida macroura) and band-tailed pigeons (Patagioenas fasciata). 14,15
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818794262 - Sarcocystis calchasi Outbreak in Feral Rock Pigeons (Columba livia) in California
Supplemental Material, DS1_VET_10.1177_0300985818794262 for Sarcocystis calchasi Outbreak in Feral Rock Pigeons (Columba livia) in California by Aslı Mete, Krysta H. Rogers, Rebecca Wolking, Daniel S. Bradway, Terra Kelly, Melanie Piazza, and Beate Crossley in Veterinary Pathology
Footnotes
Acknowledgements
We thank P. Hogan with Peninsula Humane Society and SPCA, A. Kinney with Wildlife Center of Silicon Valley, A. Reams with Wildlife Care of SoCal, Dr. S. Riggs with Pacific Wildlife, and K. Snapp and A. Avila with Wildlife Care Association and for submitting carcasses for this investigation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
