Abstract
A field study addressing causes of mortality in freshly dead northern elephant seals (Mirounga angustirostris, Gill, 1866) was conducted on San Miguel Island, California, in February 2015. Necropsies were performed on 18 pups ranging in age from stillbirths to approximately 7 to 8 weeks. The primary gross diagnoses in these pups included trauma, myopathy, starvation/emaciation, infections, congenital anomalies, and perinatal mortality. However, 6 (33%) had a previously unrecognized myopathy characterized by multiple white streaks that were most obvious within the inner layer of the abdominal wall and the small innermost ventral intercostal muscles. Following histological examination, 2 more pups from San Miguel Island and 6 pups from The Marine Mammal Center (Sausalito, California) were found to have similar lesions. Histologically, the lesions within the skeletal muscles were characterized by a multifocal polyphasic, mild to severe, acute to subacute necrotizing myopathy with mineralization. Acute necrosis and degeneration characterized by pyknotic nuclei, eosinophilic cytoplasm and cytoplasmic vacuolization were found in smooth muscle myocytes within the urinary bladder and digestive system. Degeneration of myocytes was present in the tunica media of a few small- to medium-sized vessels and was characterized by a vacuolar degeneration and occasionally necrosis. This condition has been termed multifocal necrotizing myopathy. A cause of this myopathy was not identified.
Elephant seals are the most massive pinnipeds in the world. 18,19 There are 2 species in the genus Mirounga (Gray, 1927): the northern elephant seal, Mirounga angustirostris (Gill, 1866), and the southern elephant seal, Mirounga leonina (Linnaeus, 1758). Northern elephant seals occupy rookeries along the western coast of North America from mid–Baja California, Mexico, to central California and undergo 2 annual pelagic migrations to forage in the North Pacific as far north as the Aleutian Islands of Alaska. 3,30
Northern elephant seals were presumed extinct by 1892 primarily due to commercial harvesting that began in the early 1800s to obtain blubber for rendering into oil. A small, residual breeding colony survived on Isla de Guadalupe, Baja California, Mexico, and grew rapidly through the early 1900s with legal protection from further hunting. The northern elephant seal population is currently estimated to be 210,000 to 239,000 animals; approximately 179,000 animals are found on California rookeries. 20,32 At least 10 northern elephant seal rookeries are located in California, the largest being on San Miguel Island, which is the most northwestern of the 8 Channel Islands of Southern California within the Channel Islands National Park. In addition to northern elephant seals, 5 species of pinnipeds currently occupy the island: California sea lions (Zalophus californianus), northern fur seals (Callorhinus ursinus), harbor seals (Phoca vitulina), Steller sea lions (Eumetopias jubatus), and Guadalupe fur seals (Arctocephalus townsendi). California sea lions, harbor seals, northern elephant seals, and northern fur seals have viable breeding populations there. Guadalupe fur seal females have come to the island in low numbers in recent years and given birth; thus, they may be recolonizing their breeding sites on San Miguel Island where they did historically. 14 Steller sea lions occasionally visit the island but do not currently breed on San Miguel Island.
Northern elephant seals recolonized San Miguel Island in the 1950s. Their population on San Miguel Island is estimated to be approximately 62,000 animals based on counts through 2010. The number of pups born on San Miguel Island alone accounts for approximately 40% of all elephant seal pups born in California. The population of northern elephant seals on San Miguel Island is currently increasing. 3,14,20,32 Despite extensive studies addressing ecology and dynamics of northern elephant seal populations, little information on causes of mortality in pups inhabiting rookeries has been published. The initial purpose of this study was to investigate general causes of mortality in northern elephant seals on rookeries on San Miguel Island, California. However, this article addresses a previously unrecognized condition affecting pups characterized grossly by multiple white streaks in the abdominal and intercostal muscles and histologically by an acute to subacute necrotizing myopathy. This condition has been called multifocal necrotizing myopathy (MNM), denoting the gross and histological lesions observed.
Materials and Methods
This study was conducted during a 1-week period in February 2015, at 4 rookeries on San Miguel Island (34°02′N and 120°23′W): West Cove, North West Cove, Adams Cove, and Judith Cove. Each morning, freshly dead pups were collected from the rookeries, dragged approximately 30 to 50 m from the rookery, and necropsied using standard procedures. 27
Pups were assigned to 1 of 4 age categories: stillbirths, neonates, and black-coated or silver-coated pups. Age estimation of pups was based on aeration within the lungs, meconium in the rectum, fresh or dried umbilicus, and color of pelage with degree of molt. Stillbirths had no or only minimally aerated lungs, fresh umbilicus, and meconium filling the rectum. Neonates (birth to 24 hours of age) had fully aerated lungs, fresh umbilicus, and meconium in the rectum. Black-coated pups had fully aerated lungs, dried umbilicus, no meconium in the rectum, and a black pelage. Ages of black-coated pups were from 2 days to approximately 4 to 5 weeks. Pups are usually weaned around 28 to 30 days; therefore, some of the black-coated pups were weaned and some were not. Following weaning, pups stay on the rookery for approximately 2 months. Black-coated pups begin to moult between 3 and 4 weeks of age, and the black coat is replaced with silver-gray pelage within a couple of weeks. Silver-coated pups are weaned and have a shiny silver pelage and are usually older than 6 weeks. 31 Body condition of pups was scored by midsternal blubber thickness and assigned to 1 of 4 body condition categories: excellent (>15 mm of sternal blubber), good (10–15 mm), poor (5–9 mm), and emaciated/starving (<5 mm). 29
Tissues from all major organs were collected during necropsy, placed in individual 1-gallon plastic Zip-Loc bags, and taken to a small field laboratory for further processing. Selected tissues were preserved in 10% neutral buffered formalin, and fresh tissues were frozen for ancillary diagnostic studies. Fixed tissues were processed routinely and stained with hematoxylin and eosin. Special stains, including Brown-Hopps modified Gram, Gomori’s methenamine-silver, von Kossa, Luna, and Masson’s trichrome, and immunohistochemistry (IHC) for macrophages (CD18) and for muscle/satellite cells (desmin) were performed on selected tissues. 23 Selected tissues from 10 pups (skeletal muscle, heart, diaphragm, adrenal gland, kidney, pancreas, and urinary bladder) were immunohistochemically labeled for Epstein-Barr virus using a rabbit monoclonal antibody (Epstein-Barr Virus [MRQ-47]; Cell Marque, Rocklin, CA, USA; Sigma-Aldrich, St. Louis, MO, USA) and Refined Red Detection (Leica, Buffalo Grove, IL, USA) with an automated stainer. This immunohistochemical method was used because it was previously reported to detect gammaherpesvirus in northern elephant seal pups. 15 Tissues from 8 pups analyzed with this immunohistochemistry procedure were collected from pups inhabiting San Miguel Island. Tissues from 2 pups from Año Nuevo State Reserve, California, were used as negative controls because this myopathy had not been observed in pups from this rookery. 29
Following histological examination, selected formalin-fixed tissues (skeletal muscle and urinary bladder) from 2 affected animals were further fixed in 2% (v/v) glutaraldehyde in 0.1 M sodium cacodylate and postfixed in osmium tetroxide. Samples were embedded in Poly/Bed812 (Polysciences, Warrington, PA). Sections (1 μm thick) were cut and stained with toluidine blue for initial light microscopic examination and to locate areas of interest for ultrastructural evaluation. Thin sections (60–80 nm) were cut from selected areas and stained with uranyl acetate and lead citrate and evaluated using a JEOL JEM 1200EX transmission electron microscope (JEOL USA, Inc., Peabody, MA, USA).
Five pieces of fresh-frozen skeletal muscle from 3 affected elephant seal pups from San Miguel Island were submitted to the Wyoming Diagnostic Laboratory, Laramie, for examination using negative staining electron microscopy. Submitted tissues were ground with sand and water in a mortar and pestle, clarified by centrifugation at 3440 × g for 20 minutes, and pelleted by centrifugation at 39,700 × g for 1 hour. The resulting pellet was resuspended in 1% phosphotungstic acid stain and viewed on a Hitachi H-7650 transmission electron microscope (Hitachi High Technologies America, Clarksburg, MD).
Liver concentration levels of vitamin E, mercury, and selenium were determined in 11 of 18 pups from San Miguel Island using routine methods at the Colorado State University Veterinary Diagnostic Laboratory, College of Veterinary Medicine, Fort Collins, as follows. Liver vitamin E was determined using a Waters (Milford, MA, USA) Model 2695 HPLC following extraction with hexane. Vitamin E (wet weight) was detected with a fluorescence detector, excitation wavelength of 295 nm and emission wavelength of 325 nm. Tissue selenium (dry weight) was determined using a Varian (Mulgrave, Victoria, Australia) 1275 atomic absorption spectrophotometer with hydride generation and detected at 196 nm. Tissue mercury (wet weight) was determined with a cold vapor technique using a Varian 1275 atomic absorption spectrophotometer at 254 nm. The Paleontological Statistics Software was used for statistical analysis of the results obtained. 17 Nonparametric Mann-Whitney test (U) was used to compare differences in average vitamin E, selenium, and mercury levels in northern elephant seal pups with and without gross evidence of MNM. 17
In addition to the pups examined from San Miguel Island, 21 northern elephant seal pups between 3 and 4 months of age rescued by The Marine Mammal Center (TMMC), Sausalito, California, between 2010–2012 and 2014–2016 were included in this study. These pups were necropsied at TMMC. Similar formalin-fixed tissues as examined from the pups collected from San Miguel Island were processed histologically and examined by one of the authors (T.R.S.).
Results
Characterization of the Myopathy
In this field investigation, 18 freshly dead pups (11 females and 7 males) were found on rookeries of San Miguel Island and necropsied. Body condition of these pups varied from excellent to emaciation. The gross lesions were categorized as blunt trauma, emaciation, infections (gross lesions suggestive of an infectious agent), perinatal mortality (stillbirths and neonates), congenital anomalies, and a novel myopathy. Most pups in all body condition scores had multiple gross and histological lesions.
Six of these 18 pups (33%) had gross lesions characterized by multiple white streaks that were most obvious within the muscles of the abdominal wall (Musculus rectus abdominis and M. transversus abdominis) and the thoracic wall (M. intercostalis externus and M. transversus thoracis) (Fig. 1). 21 On gross examination, limb muscles appeared to be mildly affected, but diaphragm and neck muscles appeared to be spared.
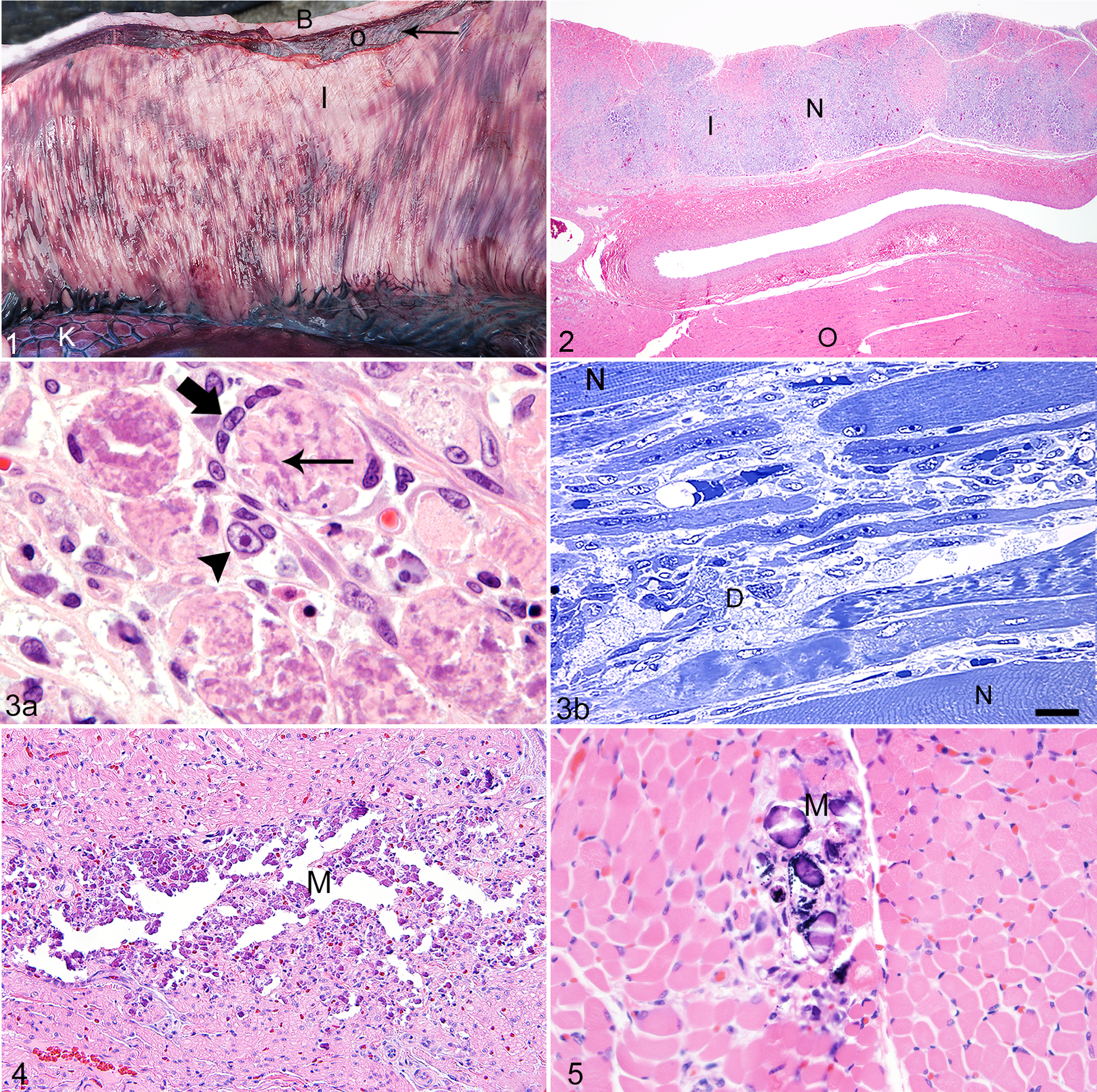
Histologically, the lesions within the skeletal muscles were characterized by a multifocal polyphasic, mild to severe, acute to subacute necrotizing granulomatous myopathy with or without mineralization (Fig. 2). In many areas, the basal lamina and satellite cells were still intact; in other areas, the satellite cells had undergone hyperplasia. In other regions, satellite cells were necrotic (Fig. 3a,b). There was mild evidence of regeneration. Small areas of myopathy also could be found in other muscles, even ocular and laryngeal muscles, but were most severe in skeletal muscles within the abdominal and thoracic walls. A few small foci of necrosis with mineralization were present in the ventricular and atrial walls of the heart and in the diaphragm in 4 pups (Figs. 4, 5) but not necessarily in the same pup. These lesions were slightly more severe in the atria compared to the ventricles. Occasionally, necrosis of Purkinje cells of the heart was present.
Mild acute necrosis and degeneration were present in smooth muscle bundles of the urinary bladder and, to a lesser degree, in the muscular tunics of the digestive system. The lesions within smooth muscle myocytes were characterized by pyknotic nuclei, eosinophilic cytoplasm, and mild to moderate vacuolar degeneration of the cytoplasm (Fig. 6). A few small- to medium-sized vessels in areas affected by skeletal muscle necrosis had mild vacuolar degeneration of the myocytes in the tunica media and necrosis of endothelial cells (Fig. 7). Other histological lesions present in affected pups included small focal randomly distributed areas of necrosis in the adrenal cortex and pancreas, as well as small foci of fibrosis within skeletal muscle (Table 1).
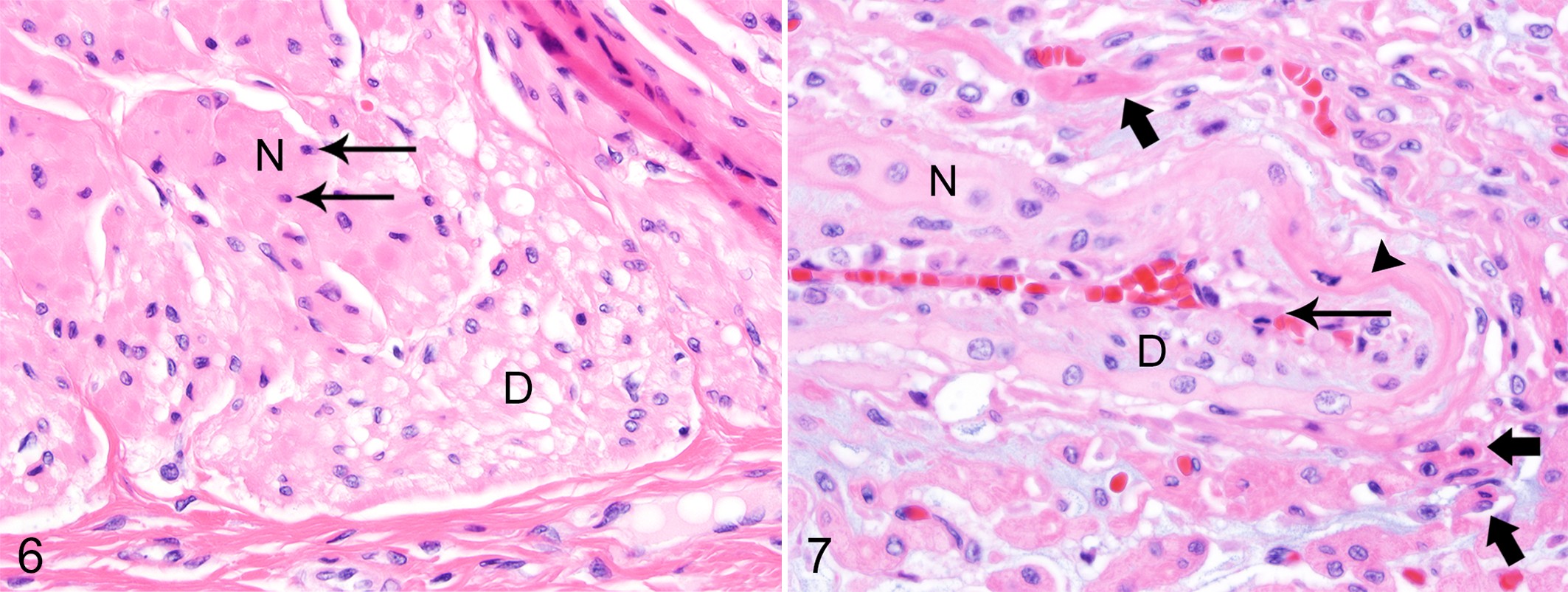
Histological Lesions Found in 39 Northern Elephant Seal Pups From San Miguel Island and The Marine Mammal Center (TMMC).a
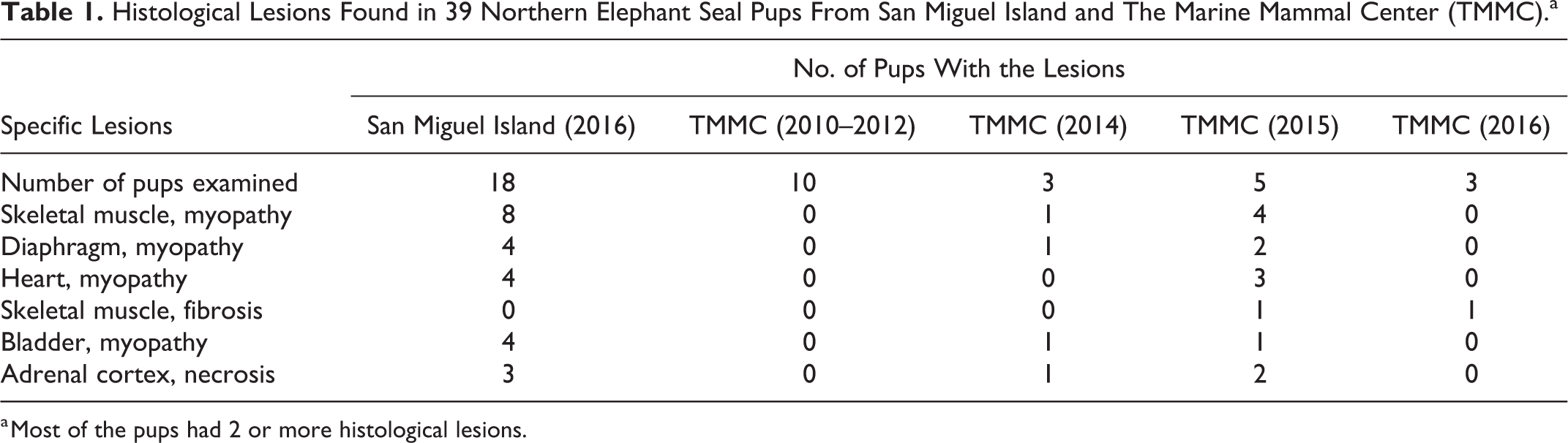
a Most of the pups had 2 or more histological lesions.
Following histological examination of these 18 pups, 2 pups that did not have gross lesions within skeletal muscles were found to have similar but smaller histological lesions of a necrotizing granulomatous myopathy, making a total of 8 (44%) pups from San Miguel Island with lesions of myopathy.
The Brown-Hopps modified Gram stain and Gomori’s methenamine-silver stain did not demonstrate any bacteria or fungi within the lesions. Van Kossa stain confirmed the presence of extensive mineralization. Masson’s trichrome stain confirmed a few small areas of fibrosis in pups from TMMC. IHC for CD18 and desmin confirmed the presence of macrophages and muscle satellite cells, respectively. IHC for Epstein-Barr virus was positive in the kidney tubules, acinar cells of the pancreas, and skeletal muscle myocytes in pups from San Miguel Island, including those affected and unaffected by MNM. IHC for Epstein-Barr virus was also positive within the renal tubules and acinar cells of the pancreas from pups from Año Nuevo State Reserve, where no evidence of MNM was found in 2012, but all skeletal muscles slides from these pups were negative.
Skeletal muscle and urinary bladder samples from 2 affected northern elephant seal pups (1 from San Miguel Island and 1 from TMMC) were evaluated using electron microscopy. Similar lesions as described with the hematoxylin-eosin tissue slides were present in the skeletal and smooth muscle myocytes. An etiological agent could not be confirmed using electron microscopy.
The average vitamin E, selenium, and mercury levels in the 6 pups from San Miguel Island with gross lesions of MNM compared to 5 pups from San Miguel Island without gross or histological lesions of MNM (used as controls) were not significantly different (Table 2). Normal reference values for vitamin E, selenium, and mercury concentrations in northern elephant seal pups could not be found in the literature. Reported levels in the liver of other pinnipeds are selenium levels of 5 to 7 ppm in adult northern fur seals, 2 to 7 ppm in adult ringed seals, and 0.5 to 5.0 ppm in adult harbor seals, as well as mercury levels of 8 to 12 ppm in adult northern fur seals, 2 to 5 ppm in adult ringed seals, and <0.1 to 15 ppm in adult harbor seals. 4,24 The values for selenium and mercury in the northern elephant seal pups were not statistically different from levels reported in northern fur seals, ringed seals, and harbor seals.
Sex, Body Condition, Gross Lesions, Vitamin A (Liver, Wet Weight), Vitamin E (Liver, Wet Weight), Selenium (Liver, Dry Weight), and Mercury (Liver, Wet Weight) Concentrations in 11 Northern Elephant Seal Pups From San Miguel Island.
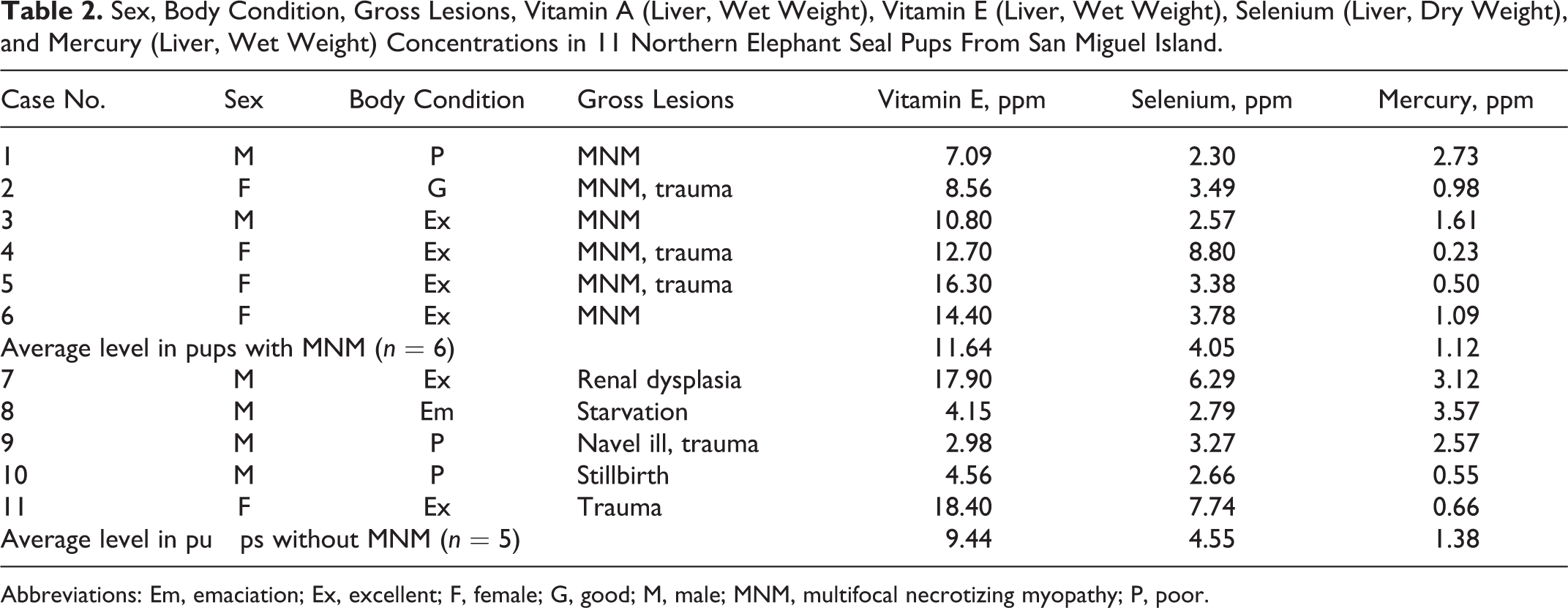
Abbreviations: Em, emaciation; Ex, excellent; F, female; G, good; M, male; MNM, multifocal necrotizing myopathy; P, poor.
Consideration of the Causes of Death
Of the 6 pups with gross lesions of MNM, the body condition was excellent in 4, good in 1 and emaciated in 1. The emaciated pup had extensive gross and histological lesions of MNM, as well as the typical histological lesions commonly found in emaciated/starved pups; this pup may have never nursed. For 3 of the 5 pups with excellent or good body condition, the immediate cause of death was blunt-force trauma resulting in multiple fractures of the liver and hemoabdomen in 2 pups and hemothorax in the other. The other 2 pups in excellent body condition only had gross and histological lesions of MNM, and other potential causes of death were not found. The 2 pups from San Miguel Island that had no gross lesions of MNM (only histological lesions) were in excellent body condition, and the immediate cause of death was blunt-force trauma to the abdomen causing multiple fractures of the liver and hemoabdomen.
One female pup that had extensive gross lesions of MNM on gross necropsy had been found alive and observed for 2 days prior to death. She was bright and alert but only moved about 10 m each night, which is an extremely short distance for a pup of her age. Her tracks in the sand were abnormal and not symmetrical as seen in normal healthy pups of similar size and age.
The finding of trauma as a cause of death in 6 of the 8 pups (75%) with lesions of MNM and the clinical signs in the 1 pup observed alive suggest that this myopathy might affect mobility and reduce the ability of affected pups to escape from subadult and adult seals. Trauma to these pups usually comes in 3 different forms. The first and most common involves subadult male elephant males trying to breed pups, causing severe blunt trauma to the chest and abdomen. The second involves aggressive females occasionally biting and throwing pups that are not theirs. Finally, pups are sometimes trampled by moving or fighting territorial bulls.
Four of the pups from San Miguel Island had small regions of myocardial necrosis with mineralization, and 1 pup had mild necrosis of a few Purkinje cells that might have had an impact on heart function or resulted in arrhythmias. These myocardial lesions were not as severe as described in California sea lions with domoic acid toxicity. 26,38 The finding of severe myopathy in 2 pups that were in excellent body condition with no other lesions suggests that MNM may be fatal.
Additional Cases From TMMC
An additional 21 northern elephant seal pups from TMMC were examined. All were extremely emaciated, and the cause of death was attributed to malnutrition/starvation. The rookeries of origin of these pups were not known and could have included any of those from Baja California, Mexico, to central California. Gross lesions in the skeletal muscles were not observed in any of these pups. Histological evidence of MNM was identified in 0 of 10 pups from 2010 to 2012, 1 of 3 pups in 2014, 5 of 5 pups from 2015, and 1 of 3 pups from 2016 (Table 1). The case from March 2014 had mild multifocal necrotizing myopathy. Four cases from 2015 had small lesions of chronic-active necrotizing myopathy, and 1 had small regions of skeletal muscle fibrosis suggestive of healed areas of myopathy. The case from March 2016 had a few small areas of fibrosis within the abdominal muscles. We speculate that pups with mild MNM might have had difficulty in swimming and diving that interfered with feeding, which may have been the reason for stranding of some pups examined from TMMC.
Discussion
The myopathy described in this field study of northern elephant seal pups had gross and histological lesions similar to white muscle disease or nutritional myopathy caused by a dietary deficiency of selenium and vitamin E in young calves, lambs, foals, and swine. 36 However, other possible causes for this condition should be considered and include marine biotoxins, protozoa, bacteria, or viruses.
Vitamin E and selenium liver concentrations in the pups with MNM were not significantly different from pups that did not have MNM. The selenium levels in these pups were similar to levels reported in other pinnipeds. 4,24 It has been established that vitamins are transferred to newborn pups via the milk. 12,13 The vitamin E and selenium levels in the stillborn northern elephant seal examined in this study were lower than in the older pups but do confirm that vitamin E and selenium are transferred to the fetus to some degree. Evidence for a nutritional cause of MNM was not found in this investigation.
Mercury concentrations were not significantly different in pups with and without MNM (Table 2). Similarly, the mercury levels in these northern elephant seal pups were not significantly different from levels reported in other pinnipeds. 4,24
A suspected nutritional myopathy has been reported in a yearling captive-born California sea lion. 1,9 This sea lion had gross lesions similar to the elephant seal pups but in a somewhat different distribution: the forelimb muscles and most intercostal muscles were heavily affected, but the inner abdominal wall was not. The suspected cause was vitamin E deficiency because the diet had not been supplemented with vitamin E. However, a definitive diagnosis could not be made. 1,9
A diffuse muscular degeneration has been reported in 3 captive harbor seals. 34 The histological lesions were similar to the lesions in the elephant seal pups, but there were some differences. First, the significant gross muscular lesions were in the temporal muscles; tongue; psoas; pectoral, intercostal, and thigh muscles; and ventricular walls of the heart, whereas the gross distribution of the lesions in elephant seal pups was the inner abdominal wall and the small ventral intercostal muscles. Lesions were not found in the tongue in any of the affected northern elephant seal pups. In addition, the harbor seals were yearlings that had been in captivity for 2 months, not free-ranging pups less than several weeks old. The cause of this myopathy in harbor seals was suspected to be a vitamin E–deficient diet but was not verified. 34
A similar myopathy characterized by multifocal necrosis of skeletal muscle was reported in northern fur seal pups on the Pribilof Islands, Alaska, in 1990. This myopathy resulted in extremely high preweaned pup mortality on specific rookeries. 28 It was suggested that the lesions could have been caused by a biotoxin reaching the pups transplacentally, through the mother’s milk, or perhaps absorbed through their flippers. The gross and histological lesions in this condition in northern fur seals were similar to those in these northern elephant seals, but their distribution was different: those in the northern fur seal pups were most severe in the pectoral muscles, neck muscles, diaphragm, intercostal muscles, and the heart. The cause of the myopathy in these northern fur seals pups was not determined.
A condition in adult California sea lions with similar gross and histological lesions has been described and reported to be a suspected autoimmune condition associated with Sarcocystis neurona. 7,8 The MNM in northern elephant seal pups is not thought to be associated with a Sarcocystis sp. for 2 reasons. First, the Sarcocystis infection in California sea lions was reported in adult animals, not in pups on rookeries. Second, Sarcocystis was not found in nearly 300 histological sections, including electron microscopy and special stains of skeletal muscle.
A gammaherpesvirus has been reported in 3- to 4-month-old silver-coated northern elephant seal pups from TMMC. This condition was reported to be nonfatal but was associated with grossly visible ulcerations of the mouth, tongue, hard palate, and tonsil. 15 These lesions were not observed in any of the northern elephant seal pups with MNM from San Miguel Island. A novel gammaherpesvirus has been isolated from the vagina in apparently healthy northern fur seals and California sea lions living sympatric with the northern elephant seal pups on San Miguel Island examined in this study. 11 This novel gammaherpesvirus (named otariine herpesvirus 4) is reported to be closely related to the herpesvirus (otariine herpesvirus 1) associated with the urogenital carcinomas of California sea lions. 11 Using an immunohistochemical staining protocol for Epstein-Barr virus, which was reported to have shown positive staining for the gammaherpesvirus in northern elephant seals from TMMC, 15 showed positive staining in elephant seal pups seals with and without MNM from San Miguel Island and from elephant seal pups from Año Nuevo, where evidence of MNM was not found. Thus, we consider these results to suggest the presence of a gammaherpesvirus that is not associated with MNM and is presumably not the cause of this condition in northern elephant seal pups; alternately, this could be an artifact with this particular IHC protocol in elephant seal tissues.
An otariine herpesvirus-3 has been isolated from stranded California sea lions that also are sympatric with the affected elephant seal pups on San Miguel Island, but the histological findings associated with this viral infection were reported to be lymphomas and esophageal ulcers. 37 These lesions were not found in the northern elephant seal pups in this study.
It has been reported that domoic acid toxicity can cause skeletal muscle lesions in brown pelicans (Pelecanus occidentalis) and Brant’s cormorants (Phalacrocorax penicillatus), but the lesions described in these birds were characterized by acute necrosis with hemorrhage and not a necrotizing granulomatous myopathy as seen in the elephant seals. 35 Domoic acid toxicity has been shown to cross the placenta and cause premature births in California sea lions; therefore, it might be considered a cause of this myopathy in elephant seals. However, the lesions described in premature California sea lion pups included cerebral edema, systemic and localized inflammation caused by bacteria of amniotic origin, and placental abruption. 16 The 2 fetuses examined in this study did not have these lesions, including the brain edema. A marine biotoxin is a possible cause of this myopathy but probably not domoic acid because the feeding habits of northern elephant seals differ from those of California sea lions. Elephant seal adult females feed on mesopelagic prey far offshore in extremely deep waters. 2 In contrast, domoic acid toxicosis is observed annually in California sea lions that feed on epipelagic forage fishes in the coastal portions of the California Current Ecosystem where the phytoplankton (Pseudo-nitzschia sp.) blooms that produce domoic acid occur. 6 Evidence of convulsions in subadult and adult animals in either California sea lions or northern elephant seals was not observed during this study, nor were there an abnormal number of elephant seal abortions or premature births.
The cause of MNM is not known. We speculate that this novel myopathy may be caused by ingestion of a biotoxin acquired through the milk, through the placenta, or both. An infectious agent was not identified in this study but remains possible.
The epidemiology and significance of MNM in the northern elephant seal population are not known. Finding 8 of 18 pups (44%) with this condition on San Miguel Island during 1 week of investigation does suggest that this disease was widespread on the rookeries of San Miguel Island in 2015. This condition could possibly have an impact on population dynamics. Northern elephant seal pups on San Miguel Island have not been previously examined at necropsy; thus, historical data describing this condition are not available. Approximately 600 to 700 California sea lion pups have been necropsied on San Miguel Island over the past 25 years, but evidence of a similar condition has not been observed in these pups (DeLong and Spraker, unpublished data). Necropsy of 21 northern elephant seal pups from the Año Nuevo State Reserve, California, in February 2012 also did not demonstrate any evidence of this condition. 29 Twenty-one northern elephant seal pups were necropsied on San Benitos Island, Baja California, in 2006, and lesions similar to MNM were not reported. 25 In addition, review of 21 necropsy cases of northern elephant seal pups from TMMC did not reveal any evidence of MNM in 10 cases examined histologically from 2010 to 2012, but mild active lesions were present in 1 of 3 pups in 2014, 4 of 5 pups in 2015, and none in 2016. Approximately 4000 northern elephant seals have been rescued by TMMC in the past 25 or so years, and many of the pups that died were examined grossly and histologically; however, MNM has not been reported in these cases. 10 Multifocal necrotizing myopathy has not been described in literature addressing causes of mortality in northern elephant seals of any ages. 5,10,22,33 This suggests that MNM could be an emerging disease that might negatively affect the elephant seal population. Thus, further studies addressing the etiology and epidemiology of MNM between different northern elephant seal populations are necessary for clarification of the significance of this disease.
Footnotes
Acknowledgements
This project was conducted under the National Marine Mammal Laboratory permit, MMPA Research Permit 16087-02. We thank Todd Bass, Ana Mario Benavides, and Joe McDowell for the histological sections and special staining of formalin fixed tissues. We appreciate the technical assistance done by Jennifer Palmer and Carol Moeller for the transmission electron microscopy photographs. We also thank Megan Dillon for the negative staining electron microscopy examination. We appreciate the time Dr. Norman F. Cheville took to examine the electron microphotographs for the possible presence of viral agents. We are thankful for the advice given to us in regard to the histological lesions by Dr. Tawfik Aboellail. We thank Barbie Halaska for helping with the necropsies of the northern elephant seal pups in 2016 at TMMC, as well as Iskra Majewski for photographic formatting and labeling. We also thank the reviewers and editors with Veterinary Pathology for their many helpful comments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received private financial support for the research. Publication of this article was supported by the Colorado State University Diagnostic Medical Center, College of Veterinary Medicine and Biomedical Sciences, Fort Collins.
