Abstract
The authors used microscopy and synchrotron-based small-angle X-ray scattering analysis (SAXS) to describe lesions macroscopically typical of tropical keratopathy (“Florida spots”) from 6 cats on St Kitts. Microscopically, there were varying degrees of epithelial hyperplasia and thinning of the cornea (by 4% to 18%) due to loss of corneal stroma associated with dense accumulations of collagen in the superficial stroma. The collagen fibrils in lesions were wider and had more variable diameters (39.5 ± 5.0 nm, mean ± SD) than in normal corneas (25.9 ± 3.6 nm; P < .01). There were occasional vacuoles (<1 μm) in the corneal epithelial basement membrane but no evidence of inflammation, edema, stromal neovascularization, fibrosis, acid-fast organisms, or structures suggestive of a fungal organism. SAXS analysis showed collagen fibril diameters and variation in size were greater in stroma containing the lesions compared to normal corneas (48.8 ± 4.5 nm vs 35.5 ± 2.6; P < .05). The d-spacing of collagen in the stroma of lesions and normal corneas was the same, but the average orientation index of collagen in lesions was greater (0.428 ± 0.08 vs 0.285 ± 0.03; P < .05). A survey revealed Florida spots lesions were static over time and became less obvious in only 1 of 6 affected cats adopted on St Kitts and taken to areas in the US where lesions are not reported. An anterior stromal collagen disorder with various degrees of epithelial hyperplasia is the pathologic hallmark of lesions clinically identical to Florida spots in cats from St Kitts.
Keywords
Corneal lesions in cats and dogs—termed Florida spots, Florida fungus, Florida keratopathy, mycotic keratopathy, acid-fast spots, tropical keratopathy, or atypical keratopathy—are reported to be common in tropical and subtropical areas. 1,10,16,17,22 Clinically, the lesions appear as variably sized (up to 4 mm), round-to-irregular, gray-to-white corneal opacities that are most dense in their centers and can be single or multiple, and unilateral or bilateral. The opacities are inactive, do not stain with fluorescein, and, with slit-lamp examination, appear to be located in the anterior stroma. Their cause is unknown, although there are reports that they are due to infection with acid-fast bacteria 8 or fungi, 20 local factors such as UV light and living outdoors, 22,29 or fire ant exposure. 24,25 The condition does not respond to topical corticosteroids, antibiotics, or antifungals, although keratectomy is curative. 20 There are reports that the lesions resolve when affected animals are moved out of tropical areas. 16,22
Clinically identical lesions to those described above are common in cats on St Kitts. Herein, we describe the gross, microscopic, and synchrotron-based small-angle X-ray scattering (SAXS) abnormalities in 6 cats with typical “Florida spots.”
Materials and Methods
Cats and Samples
In 2016 and 2017, we sampled lesions macroscopically typical of tropical keratopathy (Florida spots) from 6 adult domestic short-haired (DSH) cats (Supplemental Table S1) presented for necropsy at the Ross University School of Veterinary Medicine (RUSVM) Pathology Service after euthanasia for a variety of reasons not associated with the ocular lesions. Typical lesions were identified based on the available descriptions in the literature, 1,10,16,17,20,22 essential criteria being that the lesions were single, multifocal, or coalescing, round to heterogeneous, leukomatous, corneal opacities of varying sizes that were located within the superficial stroma. The opacities had to have a dense, white cotton–like obstructive center with a less dense, respersive, and ill-defined periphery (Figs. 1, 2). Finally, the affected eyes had to have had normal tear production in the Schirmer tear test, negative fluorescein staining, and no clinical evidence of corneal vascularization or edema.
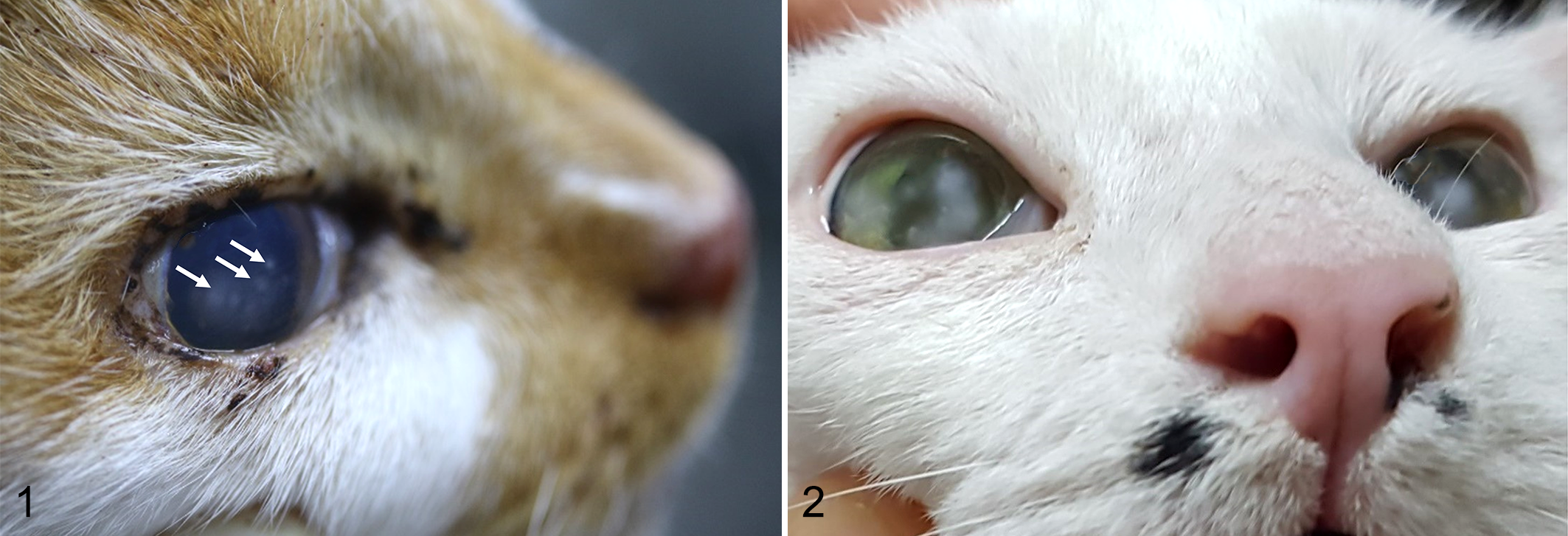
Tropical keratopathy, eye, cat.
As controls, we used samples from clinically normal areas adjacent to lesions in affected cats, as well as samples from 2 cats with clinically normal corneas: a 6-month-old male DSH cat necropsied at RUSVM and an adult female Siamese necropsied at Massey University School of Veterinary Medicine, New Zealand, where tropical keratopathy is unknown. These cats had also been euthanized for reasons unrelated to eye disease.
Skin biopsy punches (3-mm diameter) were used to obtain samples from the lesions and the normal areas of the corneas. The biopsy samples were divided into quarters, of which 2 were placed in 10% formalin in phosphate buffered saline for light microscopy, 1 in modified Karnovsky’s fixative (2.5% glutaraldehyde in 100 nM phosphate buffer, pH7.0) for transmission electron microscopy (TEM), and 1 in Triton X surfactant (0.25% Triton X-100, 0.25% sodium deoxycholate, 0.02% EDTA, 20 mg/L RNase I and 200 mg/L DNase I) for SAXS.
Histology
Formalin-fixed samples were processed routinely, sectioned at 5 μm, and routinely stained with hematoxylin and eosin (HE). In addition, several special stains were used: periodic acid Schiff (PAS) to highlight and determine the integrity of the epithelial basal lamina 27 and Descemet’s membrane, 19 acid fast stain (modified Ziehl-Neelsen) to detect acid-fast organisms, 8 and Alcian blue stain (pH 2.5) to detect abnormal accumulations of acid polysaccharide in the stroma. 31 Slides were examined using a conventional light microscope (Olympus BX 43) equipped with an Olympus DP 26 digital camera and the cellSens standard image analysis software.
Care was taken during collection of samples to avoid compression due to handling. Tissues in our lab are routinely handled on the back/side edge to avoid sample damage. We paid particular attention to orientation when trimming the biopsies to ensure that perpendicular sections were obtained through the cornea, as close as possible to the centers of the lesion. As an internal quality control, we observed normal cornea on both sides of the lesions.
Transmission Electron Microscopy
To determine the ultrastructure of the lesions, samples fixed in glutaraldehyde were trimmed, washed 3 times in phosphate buffered saline (0.1 M, pH 7.2), postfixed in osmium tetroxide (0.1 M) for 1 hour, dehydrated in graded ethanol washes (from 25% to 100% in increments of 25), and embedded in epoxy resin (EponTM Epoxy Embedding Medium Kit, Merck, USA). Ultra-thin sections of 60 to 70 nm were cut with an ultra-microtome (Leica EM UC7, Leica, Germany) and mounted on a copper grid. After negative staining with ammonium molybdate, the sections were examined by TEM (Tecnai G2 BioTWIN, FEI Company, Czech Republic).
Synchrotron-Based Small-Angle X-Ray Scattering
The SAXS beamline at the Australian Synchrotron was used to examine the collagen structure in the corneal lesions (n = 10 measurements from 2 cases) and normal (n = 10 measurements from 3 cases) (Figs. 3-4; Supplemental Table S2). Each of the punch biopsy quarters preserved in Triton X for SAXS analysis were divided in half and mounted flat on to the X-ray beam to perform surface measurements, where 9 diffraction patterns were recorded using a 3-by-3 grid with 0.25-mm spacing. The diffraction patterns were recorded at the Australian Synchrotron SAXS/WAXS beamline. A high-intensity undulator source was used with an energy resolution of 10-4 from a cryo-cooled Si (111) double-crystal monochrometer. The beam size full width half maximum focused at the sample was 250 × 80 μm with a total photon flux of approximately 2 × 1012 photons s-1. All diffraction patterns were recorded with an X-ray energy of 11 keV using Pilatus 1 M detector with an active area of 170 × 170 mm and a sample detector distance of 3371 mm. The exposure time for diffraction patterns was in the range of 1–5 seconds. Data processing on the 90 scatter patterns created from samples of lesions from 2 cats and samples of normal cornea from 3 cats was carried out using the SAXS151D software. 2 Intensities displayed are all absolute detector counts (1 X-ray detected 1 detector count), except when stated otherwise.
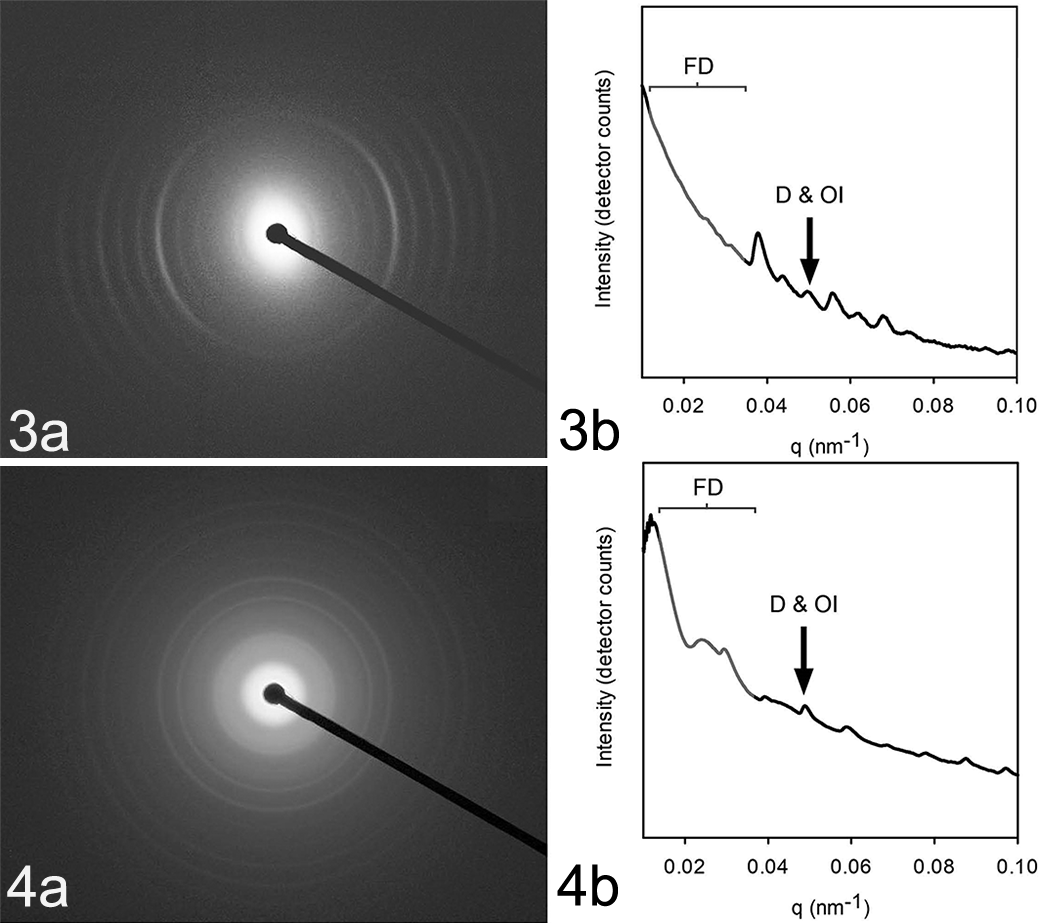
Photoimages of the radial X-ray diffraction patterns produced by affected (Fig. 3a) and normal (Fig. 4a) collagen and graphs of the intensity of the diffraction at different radial distances from the beam (Figs. 3b, 4b). The fibril diameter (FD) is determined by modeling the shape of the curve; D-spacing (D) and orientation index (OI) are determined from the fifth-order peak.
We were not able to perform focal SAXS measurements of just the lesion without the surrounding normal collagen. Instead, we obtained total measurements across the entire anterior-to-posterior thickness of the cornea, which contained collagen of both the lesion and adjacent normal cornea. We therefore selected biopsies from more severe lesions for the SAXS studies as these contained the highest ratios of abnormal to normal collagen, which would enable us to more readily characterize the collagen in the lesion.
Fibril diameter and size distribution
The Irena software package running with Igor Pro 11 was used to calculate the fibril diameters from the SAXS data. The range of 0.01–0.04 Å-1 was used to fit the wave vector, Q, and at an azimuthal angle of 90° over a 5° segment to the long axis of most of the collagen fibrils. This azimuthal angle of the long axis was determined as the position for the maximum intensity when referring to the d-spacing diffraction peaks. The “cylinderAR” shape model (with an arbitrary aspect ratio of 100) was used for all fittings. The aspect ratio was not optimized for each individual fitting, and it was noted as being possible for the collagen fibrils unbranched lengths to exceed the aspect ratio of 100. Fibril diameters were also determined from TEM images showing end-on collagen fibrils using ImageJ (National Institute of Health, Bethesda, MD) with the particle analysis function having a circulatory threshold of 0.7–1.0. We measured fibrils from 7 samples (4 normal corneas and 3 lesions) over a total area of 5.1 μm2 (2.4 μm2 for the normal tissue and 2.7 μm2 for the lesions).
Orientation index
The collagen orientation was determined from the azimuthal angle for the maximum intensity of the d-spacing diffraction peaks. The orientation index (OI) was the primary measure used, where OI is defined as (90° – OA)/90°, where OA is the minimal azimuthal angle range, centered at 180°, that contains 50% of the micro fibrils. 23 The OI range is from perfect alignment, represented by an OI of one, through to isotropy where the OI is zero. The OI was calculated from the spread in the azimuthal angle of the d-spacing peak at 0.059–0.060 Å. Each OI value represents an average of 9–18 measurements of a single sample.
D-spacing
The central position of Gaussian approximations, fitted to the fifth-order diffraction peak from the integrated intensity at the azimuthal angle range of 45°–135°, was used to calculate the d-spacing according to Bragg’s law.
Statistical Analysis
Analyses were performed using Microsoft Excel for ANOVA and Minitab 17 Statistical Software (2010) (State College, PA: Minitab, Inc,; www.minitab.com) for the 2-sample t-test for unequal variance. A P < .05 was regarded as significant.
Alumni Survey
Previous students at RUSVM were approached through the RUSVM Alumni Association to anonymously complete an online Qualtrics survey (Qualtrics software, Version 122014, Provo, UT, http://www.qualtrics.com), which requested details on the outcome of Florida spots in cats adopted on St Kitts and returned to the USA. The survey was approved by the Institutional Review Board of RUSVM.
Results
Cats
The cats used in the study (Supplemental Table S1) had multiple lesions, 0.5 to 4 mm maximum dimension, multifocal to coalescing, in one or both eyes (Figs. 1, 2).
Histology
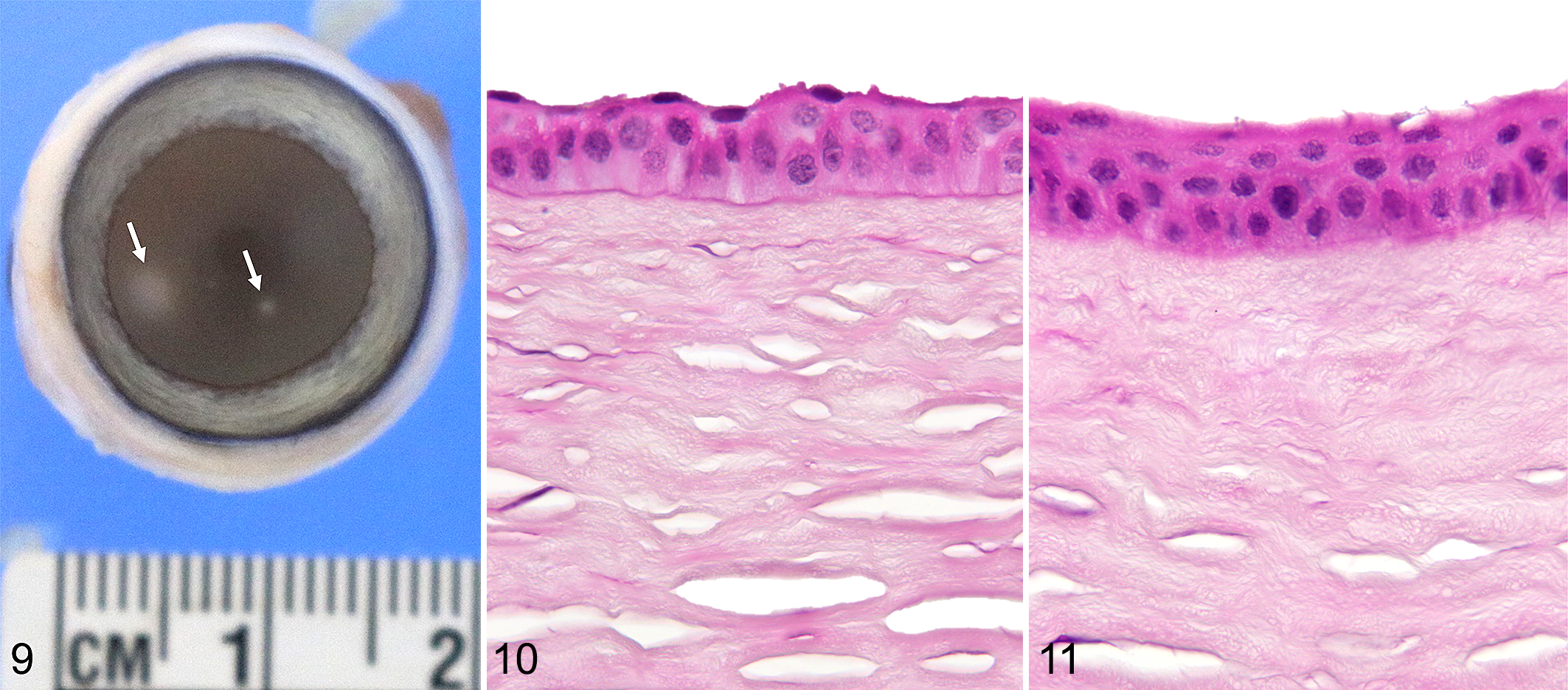
All histological lesions that we studied were essentially the same, although their extent and severity varied between cats. The major change in the epithelium (Fig. 5) was progressive epithelial hyperplasia toward the center of the lesion with up to 13–15 cell layers (Figs. 6, 7; Supplemental Table S3; Supplemental Figs. S1, S2) compared to only 6–9 layers in the normal controls (Supplemental Figs. S3, S4) or in the unaffected areas adjacent to the lesions (Fig. 8) (P = .009). No mitotic figures were observed in the epithelial lesions examined. The major change in the stroma was in the subepithelial and superficial regions, where the corneal stromal lamellae appeared more dense as evidenced by loss of artifactual stromal clefts that appeared during normal histological processing. 28 The thickness of the irregular collagen also increased progressively toward the centers of the lesions. Measurements showed the overall thickness of the cornea in the center of lesions was 4% to 18% less than that of the normal cornea on the edge of the lesions (not statistically significant). This reduction in corneal thickness was due to a decrease in the thickness of the corneal stroma. In some cases (Fig. 9), the lesion was much more subtle, with mild changes in the superficial stroma and minimal changes in the overlying epithelium compared with a nearby nonaffected area (Fig. 10). Still, this change or this stage of the lesion (Fig. 11) could be observed on light microscopy, with patience and a slide of good quality.
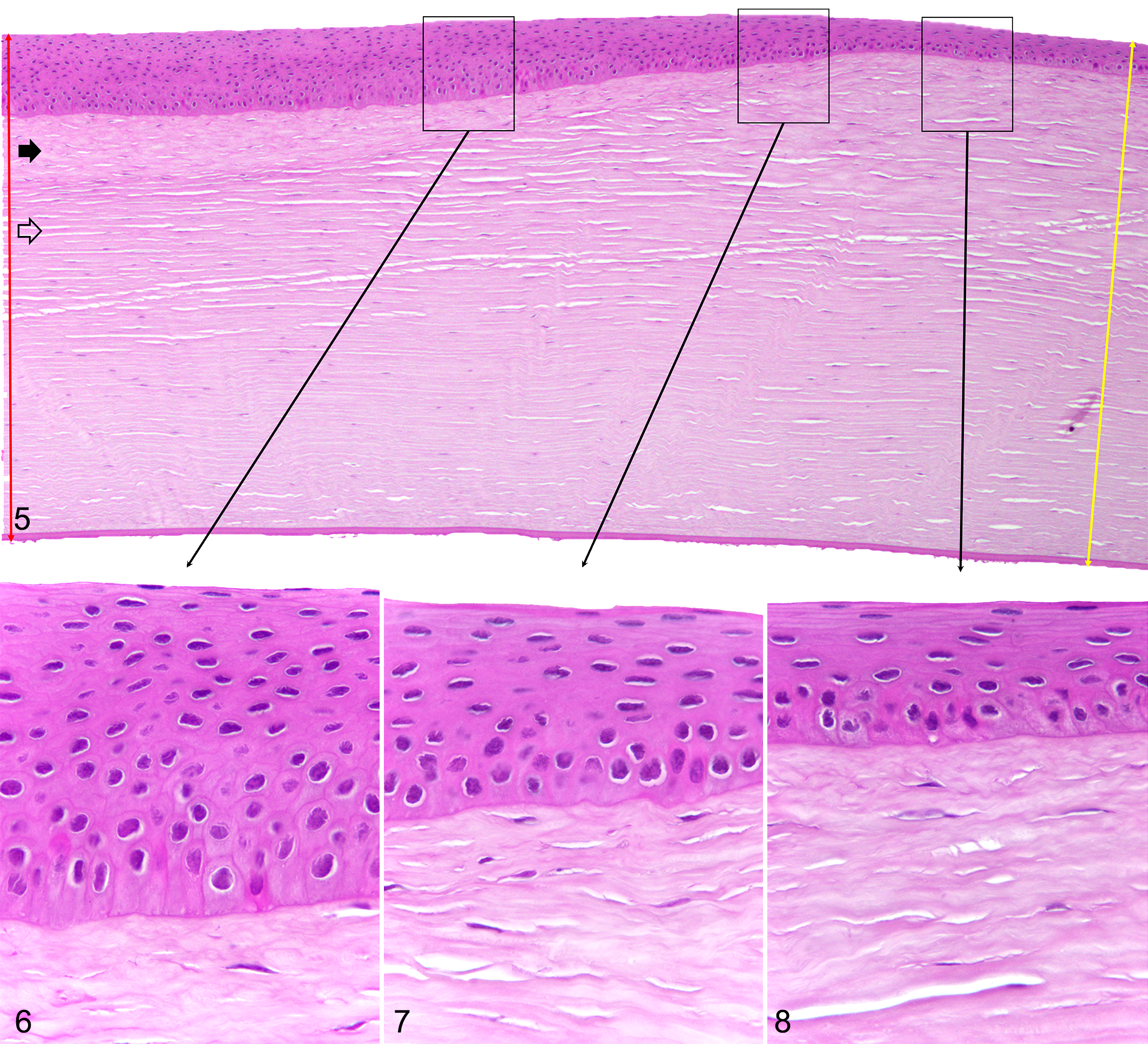
Tropical keratopathy, cornea (left eye), cat, case 2.
Tropical keratopathy, cornea (left eye), cat, case 6.
In routine HE-stained sections, the other corneal structures (deep stroma, Descemet’s membrane, and posterior epithelium/corneal endothelium) appeared normal, and there was no evidence of inflammation, edema, ulceration, stromal neovascularization, or fibrosis associated with the lesions. Similarly, the PAS stain did not reveal any changes in the corneal epithelial basal lamina or in Descemet’s membrane (Supplemental Fig. S5), no organisms were visualized with the acid-fast stain (Supplemental Fig. S6), no accumulations of acid polysaccharides were observed with Alcian blue stain (Supplemental Fig. S7), and there were no structures suggestive of fungal organisms.
Transmission Electron Microscopy
The basal, wing, and squamous cell layers of the epithelium at the site of the lesion were essentially identical to those in the normal corneas (Figs. 12, 15), and no inflammatory cells were seen. Increases in the numbers of polygonal basal cells were responsible for the epithelial hyperplasia (Fig. 13). The numbers of mitotic figures in the hyperplastic epithelium were not increased, and all the cells in this area had normal morphology, containing few organelles and being closely attached by numerous desmosomes. The basal surface of the epithelium appeared firmly attached to the corneal epithelium basement membrane with plentiful hemidesmosomes. In one of the lesions, small vacuoles (< 1 μm) with no limiting membrane and sometimes bounded with osmium precipitates (probably lipids) were seen in the basement membrane and adjacent lamellae (Fig. 14).
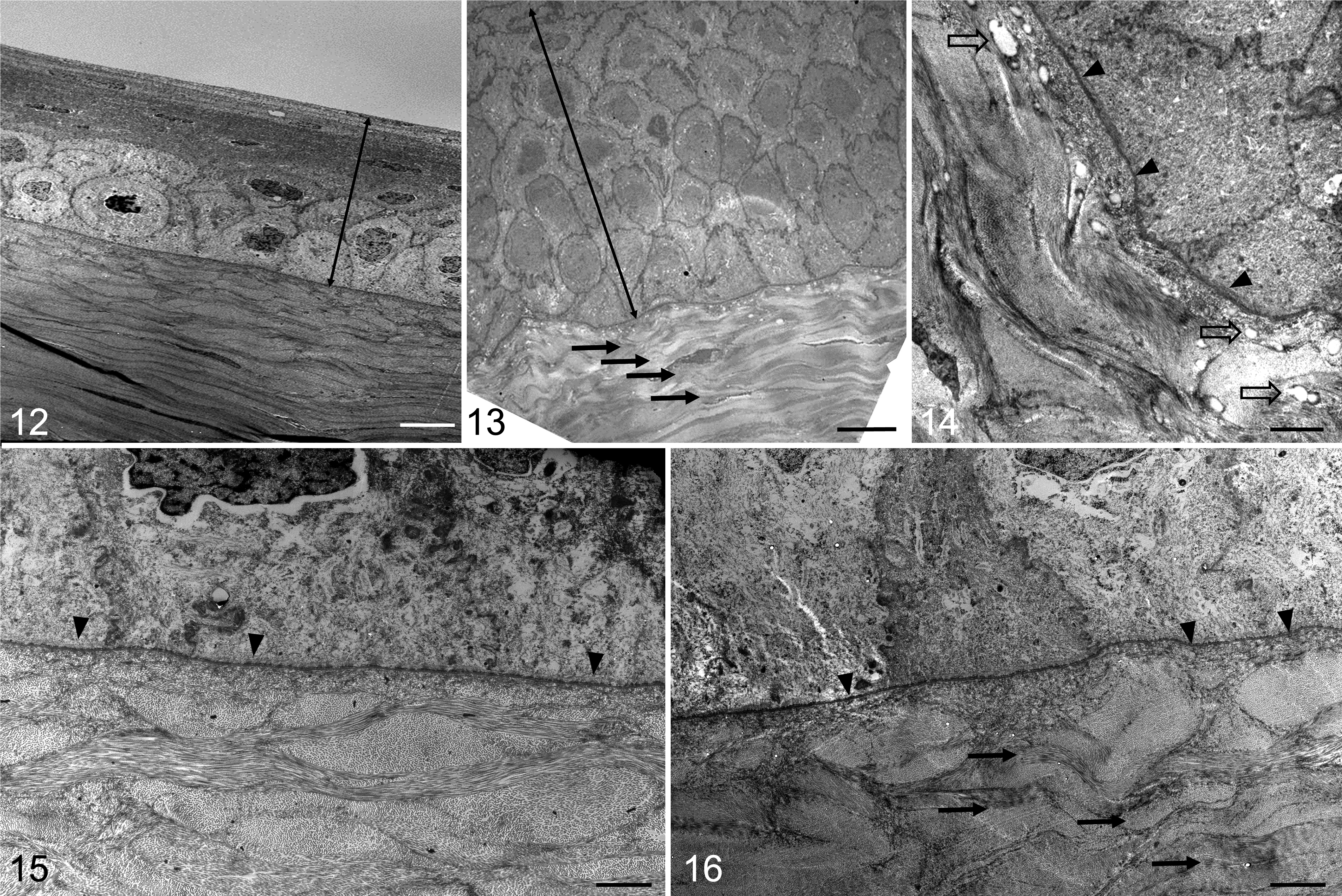
Cornea, cat. Transmission electron microscopy.
Collagen below the hyperplastic epithelium had a more undulating arrangement (Figs. 13, 14, and 16) than the collagen below the lesion, which appeared normal, 3 with regularly arranged, tightly packed, interbranching collagen fibrils oriented in different planes. No neovascularization was seen in the stroma. The keratocytes between the collagen fibrils in both the lesion and normal areas of the stroma below the lesion were similar in number and morphology, and Descemet’s membrane was intact (data not shown on TEM but visible in histology images).
Computer measurement and analysis of the diameters of collagen fibrils seen end-on in electron microscope images revealed the average diameter and variation in size of the superficial stromal collagen of lesions (39.5 ± 5.0 nm, mean ± SD) (Fig. 17) were greater than that of the underlying normal collagen (25.9 ± 3.6 nm; P < .01) (Fig. 18) (Supplemental Table S4).
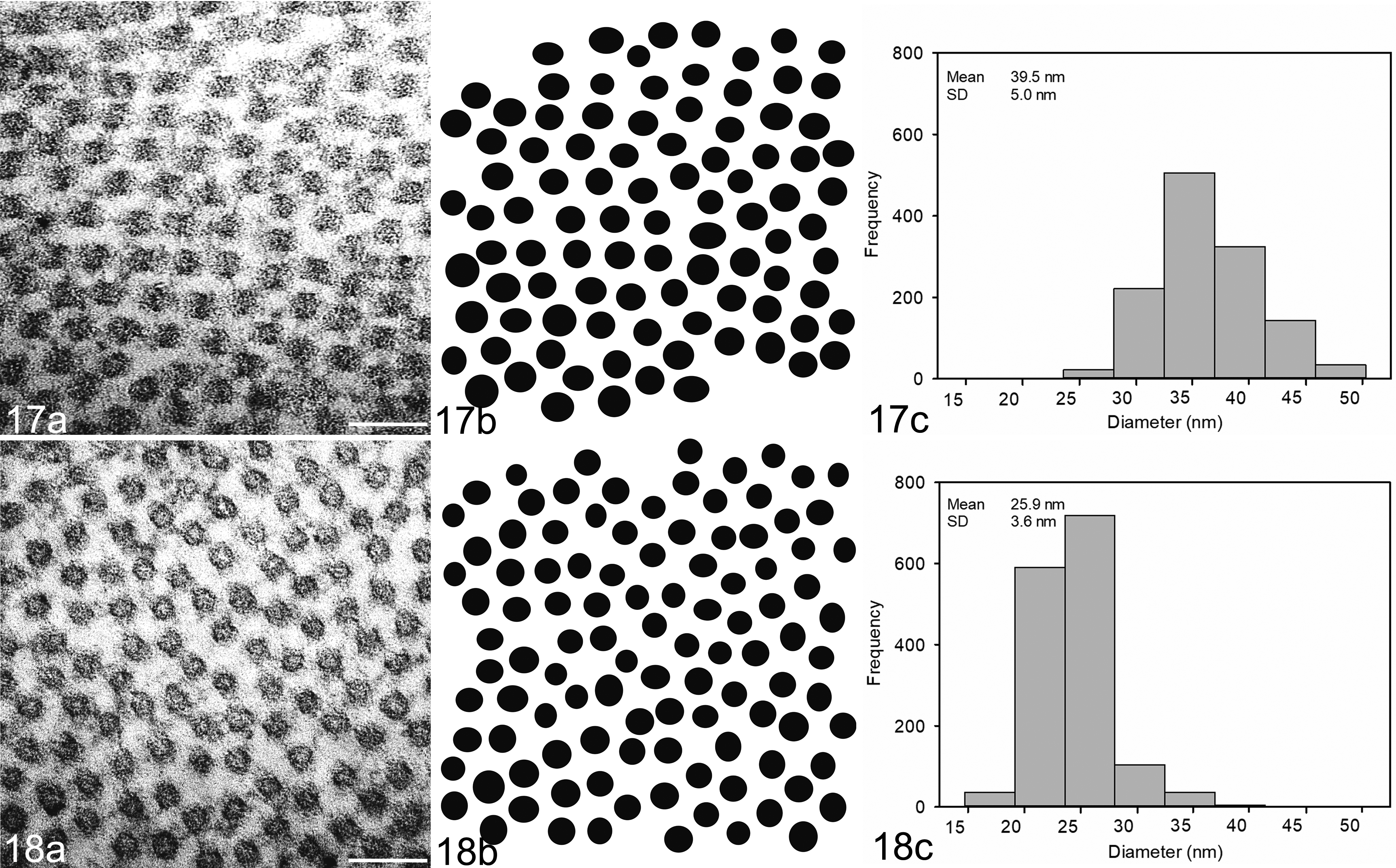
Collagen fibril size distribution in normal cornea (Fig. 17) and in tropical keratopathy lesion (Fig. 18; case 3). (a) Cornea, cat. Typical transmission electron micrograph of the stroma (scale bar is 100 nm). (b) The resulting binary image obtained using ImageJ software. (c) Frequency of different collagen fibril diameters in appropriately oriented transmission electron micrographs of the corneas studied. Collagen fibrils had significantly greater diameter in lesions of tropical keratopathy than in normal cornea (P < .01). SD, standard deviation.
Small-Angle X-Ray Scattering
The average d-spacing in the lesion samples (63.6 ± 1.0; mean ± SD; range, 62.7 to 65.1) was not significantly different from that of the normal corneas (64.9 ± 0.04; range, 63.7 to 65.8; P = .18) (Supplemental Table. S2; Fig. 19). The collagen fibril diameters and fibril diameter distributions were significantly larger in the lesion samples as compared to the samples of normal corneal tissue (35.5 ± 2.6 nm vs 48.8 nm ± 4.5 nm; P < .01) (Supplemental Table S2; Fig. 20). Similarly, there was an increase in the average OI of the collagen in the lesions as compared to that in normal areas (0.428 ± 0.06 vs 0.285 ± 0.03; P < .01) (Supplemental Table S2; Fig. 21). This indicated a greater relative alignment of collagen fibrils in the lesions as compared with that in normal cornea.
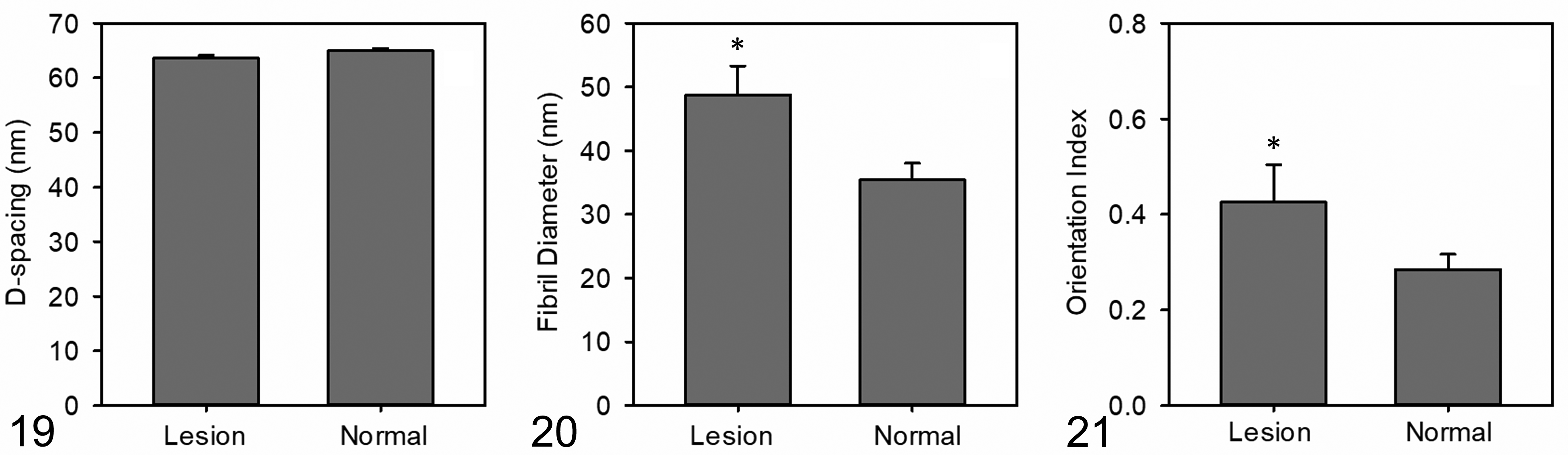
Collagen parameters measured by small-angle X-ray scattering (SAXS) in normal corneas and those with Florida spots.
Alumni Survey
Of the approximately 250 alumni estimated to have opened the email questionnaire, 6 responded and reported they had adopted cats with Florida spots while on St Kitts and subsequently returned them to the USA from 2 to 25 years previously (mean, 11.5 years). The lesions in 1 of the 6 cats were reported to have become less obvious, but none totally regressed or became worse. The 5 cats with static lesions returned to, and lived in, a number of states in the USA, including Minnesota (1 year), Maryland (5 years), Maine (1 year), New Jersey (7 years), Pennsylvania (1 year), New Hampshire (1 year), New York (14 years), Wisconsin (1 year), Oklahoma (1 year), Colorado (2 years), and Texas (1 year). These states are not in the southeastern USA, the region where Florida spots are most frequently reported. 8,17,20
Discussion
Although corneal opacities consistent with Florida spots and its aliases are reported to be common in tropical and subtropical areas, 10,22 there have been few reports on their underlying pathology. An early study on radially symmetric light-scattering opacities that occur in 10% of South Florida cats described innumerable granules, probably lipid, in the basement membrane of affected corneas. 29 These lesions seem grossly similar to those described in people with punctate keratopathy (Rice’s keratopathy), 9 which is reported from the West Indies and Central and South America. 14,26,30 Clinically, the human lesions are also nonactive and asymptomatic but are very much smaller (under 1 mm in diameter) 9 than those we observed in cats (up to 4 mm). In one of the cases we examined with TEM, there were simple vacuoles in the epithelial basement membrane and immediately adjacent lamellae. They did not have a lining membrane (Fig. 12) and were similar in location and morphology to some of the vacuoles described by Rice et al 21 in West Indians with punctate keratopathy. It was suspected these empty simple vacuoles originally contained fat that was likely dissolved in the processing steps for electron microscopy. Although not observed in the cats in our study, Rice et al found several other types of vacuoles, including those with a lining membrane that formed villi, others in which the villi had become electron dense, and finally vacuoles almost filled with electron-dense villi. It was suggested that these vacuoles, located under regions of epithelial detachment from Bowman’s membrane due to epitheliolysis of basal cells, most likely contained lipid and calcium deposits. 21
Numerous round-to-oval vacuoles (30–200 μm) between normal collagen fibrils were seen in another early light microscopy study of keratectomy specimens from 2 cats and 3 dogs with Florida spots. These were suggested to contain organisms similar to Rhinosporidium, 20 and there were no signs of inflammation or irritation. In 1987, Fischer and Peiffer 8 described a dog with acid-fast organisms within the stroma in a Florida spot lesion. Transmission electron microscopy revealed a most likely lipoidal light-staining material adjacent to keratocytes and 0.5 to 1 μm vacuoles scattered throughout the stroma. There were no alterations in the overlying corneal epithelium or stroma. In a study of 16 cats from Israel with Florida keratopathy, 1 case was examined histopathologically and no changes were found in the cornea. 24
Although the keratopathy we found in cats from St Kitts is clinically the same as the corneal opacities described in the veterinary reports above, its microscopic features are distinct. Previous reports and histopathologic examination suggested that the condition was not associated with any microscopic changes. 6 We found that the lesions can be small, so careful trimming of biopsies and proper placement in embedding media are needed to enable microscopic detection. Also, the extent of the epithelial hyperplasia and increase in density of the subepithelial/superficial collagen varied in severity between lesions. In some cases, we struggled to identify subtle lesions, and it required careful examination of multiple serial sections. It is also possible there might be a number of underlying pathologies for the corneal opacities that are common in cats in the region. Additional studies are needed on cats in other areas to provide a better understanding of the lesions and enable a more appropriate name for the condition.
The most prominent feature of the condition in the cats on St Kitts is the abnormality in the collagen fibrils of the superficial stroma, which is the site of the opacity seen with slit-lamp examination. Transparency of the cornea depends on the relatively regular parallel arrangement of collagen fibrils that are almost uniform in diameter and equally spaced (short-range ordering), under 1/2 the wavelength of visible light (400–700 nm). 5,7,15 The large range in fibril diameters we found and the variability in spacing of the collagen fibrils in the abnormal stroma most likely account for the opacities seen clinically in the corneas we studied. As the collagen in the stroma is generally aligned to counteract sheer stresses and thereby maintain corneal shape, it seems likely that the greater alignment we observed in the lesions was induced by altered forces in the cornea. It remains to be elucidated at this stage whether or not the initial trigger for the lesion first affected the corneal epithelium or whether the hyperplasia resulted from the stromal thinning we observed causing depressions in the cornea, which became filled with epithelial cells. This basal cell proliferation and migration of epithelial cells from the limbus would have continued until the resulting hyperplastic epithelium became level with the adjacent corneal surface and therefore exposed to the palpebral “snow plough” effect that maintains the smooth and uniform epithelial surface. 4,13
It is noteworthy that although epithelial hyperplasia can be marked, it does not contribute to corneal opacity, and the epithelial layer was normal in slit-lamp examinations of affected eyes. In our study, the individual cells in the hyperplastic epithelia appeared normal by both light and electron microscopy and thus provide the relatively small fluctuations between the refractive indices of the cell cytoplasm and organelles (short-range order) needed for transparency. 12 Also, the cells appeared to be functioning normally and providing a diffusion barrier to prevent fluid (tears) flow into the hydrophilic stroma: The cells were tightly attached to one another with large numbers of desmosomes, there were no obvious intracellular spaces, and there was no stromal edema.
Although previous studies in cats and dogs also described vacuoles in lesions, the ones we report in our case were not related to acid-fast organisms 8 and were far smaller than the vacuoles described that possibly contained Rhinosporidium. 20 Also, they were not visible as light-scattering granules with HE staining 29 and were localized to the basement membrane or the area immediately beneath it, unlike the above vacuoles that were widely distributed in the stroma. The vacuoles described in human patients with tropical keratopathy were also mainly in the basement membrane but were considerably more numerous, forming dense aggregates. They also appeared in a variety of forms not seen in our study and were regarded as being evidence of fatty degeneration and calcification possibly induced by trauma. 21,26
Our study did not show the cause of the lesions, but we found no evidence they were caused by acid-fast organisms, as reported for Florida spots, 8 or by fungi as reported with mycotic keratopathy. 20 Our results do show, however, that the main problem is in the superficial stroma where collagen fibrils with a wide range of diameters were seen microscopically and confirmed by SAXS measurements. In the cases we examined, a corneal wound can be ruled out by the absence of classical histological features, such as edema, stromal myofibroblastic metaplasia of keratocytes, or angiogenesis. 18 Further studies are needed to determine if these changes are due to the other suggested causes of the lesions, mainly exposure to UV light, 29 fire ant (Wasmannia auropunctata) venom, 24,25 or microtrauma as a result of dust or other air-borne pollutants. 14,21,22 Based on local experience, we doubt that fire ants are a significant cause of the lesions. Although these ants are present on St Kitts, we very rarely see fire ant–related skin lesions in cats, let alone the severe acute keratitis associated with fire ants. 25
There is one report that lesions in 3 cats from Martinique resolved when the cats returned to France and that they recurred when the cats returned to the island. 22 In our survey, the lesions in only 1 of the cats was reported to improve but not resolve. The lesions in the other 5 cats, however, did not change even though most of the cats went to areas in the USA not associated with Florida spots and therefore presumably without the agents causing the lesions. This lack of resolution seems more consistent with the seemingly irreversible lesions we observed in the study.
It is of note that other species on St Kitts with lesions clinically identical to the feline “Florida spots” we describe have very similar histological changes. These include dogs, which have been previously reported to have “Florida spots,” 9,21,23 Indian mongooses (Herpestes javanicus), and vervet monkeys (Chlorocebus aethiops sabaeus) (data not shown).
Conclusion
In conclusion, our study represents the first detailed description of the corneal opacities seen commonly in cats on the Caribbean island of St Kitts. The major abnormality is the thinning of the corneal stroma and more-aligned collagen fibrils in the superficial region that have a wide variety of fibril sizes. The other changes (various degrees of overlying epithelial hyperplasia and occasional vacuoles in the basement membrane) do not appear to contribute to the opacification of the cornea. The lesions appear not to be associated with active infectious or inflammatory processes. Further studies are needed to determine the cause of the lesions and their occurrence in cats from other regions.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818789483l - Tropical Keratopathy (Florida Spots) in Cats
Supplemental Material, DS1_VET_10.1177_0300985818789483l for Tropical Keratopathy (Florida Spots) in Cats by Pompei Bolfa, Susyn J. Kelly, Hannah C. Wells, Katie H. Sizeland, Erin M. Scott, Nigel Kirby, Stephen Mudie, Anibal G. Armien, Richard G. Haverkamp, and Patrick J. Kelly in Veterinary Pathology
Supplemental Material
Supplemental Material, DS2_VET_10.1177_0300985818789483l - Tropical Keratopathy (Florida Spots) in Cats
Supplemental Material, DS2_VET_10.1177_0300985818789483l for Tropical Keratopathy (Florida Spots) in Cats by Pompei Bolfa, Susyn J. Kelly, Hannah C. Wells, Katie H. Sizeland, Erin M. Scott, Nigel Kirby, Stephen Mudie, Anibal G. Armien, Richard G. Haverkamp, and Patrick J. Kelly in Veterinary Pathology
Supplemental Material
Supplemental Material, DS3_VET_10.1177_0300985818789483l - Tropical Keratopathy (Florida Spots) in Cats
Supplemental Material, DS3_VET_10.1177_0300985818789483l for Tropical Keratopathy (Florida Spots) in Cats by Pompei Bolfa, Susyn J. Kelly, Hannah C. Wells, Katie H. Sizeland, Erin M. Scott, Nigel Kirby, Stephen Mudie, Anibal G. Armien, Richard G. Haverkamp, and Patrick J. Kelly in Veterinary Pathology
Footnotes
Acknowledgements
Part of this research was undertaken on the SAXS/WAXS beamline at the Australian Synchrotron, part of the Australian Nuclear Science and Technology Organization (ANSTO). We thank Jordan Taylor of the Manawatu Microscopy Centre. We are also grateful to Dr Fernanda Castillo Alcala from the Institute of Veterinary, Animal and Biomedical Science at Massey University and to Dr Teri Weronko and Dr Andrea Peda from Ross University Veterinary Clinic; and David Hilchie, histology lab manager, Ross University School of Veterinary Medicine, for technical support and assistance with sourcing samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: The Australian Synchrotron paid for travel and accommodation for the SAXS measurements.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
