Abstract
Contaminated honey is a common cause of grayanotoxin intoxication in humans. Intoxication of animals, especially cattle, is usually due to ingestion of plants of the Ericaceae family, such as Rhododendron. Here, we report the ingestion of Pieris japonica as the cause of grayanotoxin I intoxication in 2 miniature pigs that were kept as pets. The pigs showed sudden onset of pale oral mucosa, tachycardia, tachypnea, hypersalivation, tremor, and ataxia that progressed to lateral recumbency. The pathological examination of one pig revealed no specific indications for intoxication except for the finding of plant material of Pieris japonica in the intestine. Grayanotoxin I was identified in the ingested plant, gastric content, blood, liver, bile, kidney, urine, lung, and skeletal muscle via HPLC-MS/MS. Grayanotoxin I should be considered as a differential etiological diagnosis in pigs with unspecific signs and discovery of ingested plant material as the only indication in the pathologic examination.
Grayanotoxin (GTX) poisoning is a well-known intoxication in humans, and was documented by the ancient Greek writer Xenophon. 8 The toxins are classified as diterpenes and more than 25 isoforms are known. The principal toxic isomers are GTX I (also known as andromedotoxin, acetylandromedol or rhodotoxin), and GTX III (also known as andromedol). GTX II (also known as andromedenol) is less toxic. 8,11,20,21 The binding to voltage-dependent sodium ion channels is the primary pathomechanism of these neurotoxins. 8 The most common and best-studied source of GTX intoxication in humans is the ingestion of contaminated honey. 8 These cases are therefore also known as mad honey disease. The honey can be contaminated by the toxin when it is produced from nectar derived from Ericaceae plants. 8 Rarely, poisoning unrelated to mad honey has been reported in humans when patients ingested leaves or flowers containing GTX. 8
In contrast to humans, in animals, mainly cattle, the disease is often fatal. GTX intoxication is also reported in sheep, goats, cats, dogs, and donkeys. 8 The ingestion of toxin-containing plants of the Ericaceae family such as Rhododendron and Pieris, 21 which may be accessible on pastures or fed by bystanders are the major source of GTX intoxication in livestock. Most Ericaceae species are native in Asia and North America and a few in Europe, however, the plants are distributed worldwide as ornamental plants in gardens, parks, or houses. 21 Rhododendron catawbiense, Pieris floribunda and Leucothoe fontanesiana are naturally found in North America, Rhododendron ponticum naturally grows in the Black sea region, whereas Rhododendron simsii originates in east Asia and the natural habitat of Pieris japonica is China, Taiwan and Japan. 12,21
Pets such as dogs and cats are usually intoxicated by oral uptake of Azalea, which are used as ornamental plants. 15 Intoxication of pigs by plants is uncommon, probably due to husbandry conditions with limited access to plants and standardized feeding.
Two 2.5-year-old miniature pet pigs (a male-castrated of 30 kg and a female of 32 kg) spontaneously showed tremor and ataxia and were presented to a local veterinarian in lateral recumbency. Both animals were constantly smacking with foam at the snout as a sign of hypersalivation, with pale oral mucosa, tachycardia, tachypnea, and abdominal tenderness and pain. A body temperature of 38.0°C and 39.1°C were measured in the female and male pig, respectively. The female pig received dipyrone as well as 500 ml saline and 500 ml Ringer’s lactate solution intraperitoneally and recovered within a few hours. The male pig was initially treated with amoxicillin, dipyrone, and prednisolone, died during transport to the veterinary clinic, and was submitted for a complete postmortem examination.
The animal was in good nutritional status and moderately cyanotic. Multifocal, acute, petechial hemorrhages were identified in the epicardium around the coronary vessels. A segment of the duodenum and proximal jejunum was severely hyperemic and contained numerous plant materials (Fig. 1). All other parts of the gastrointestinal tract were properly filled with normal content. Additionally, a mild urolithiasis was diagnosed.

Pieris japonica toxicity, miniature pig.
Histologic examination revealed unspecific signs such as a moderate, acute, segmental, submucosal edema, deposition of fibrin, and hemorrhages in the hyperemic segment of the small intestine. There were mild multifocal acute hemorrhages in the epicardium and kidneys as well as edema with fibrin in the epicardium and spleen. In addition, there were mild chronic multifocal eosinophilic enteritis and moderate multifocal acute adrenocortical necrosis with mild neutrophilic infiltration. Histopathological analysis of the brain revealed no specific findings.
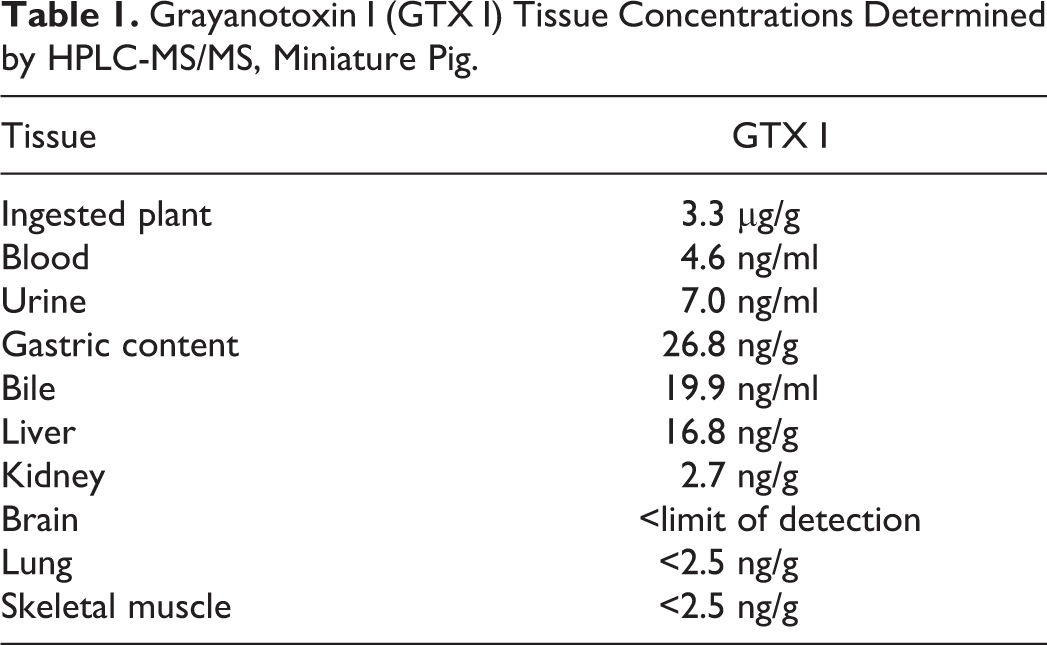
The plant parts found in the small intestine (Fig. 2) were classified as Pieris japonica due to the following characteristics: specific leaf venation, leaf margin with shallow serration, which smoothened towards the leaf node, alternate leaf arrangement, and red coloring with zigzag form of the stem. Pieris japonica is a GTX-containing plant of the Ericaceae family. Tissues were subsequently analyzed via HPLC-MS/MS 3,13,23 and GTX I was detected in the ingested plant material, in different tissues, and body fluids (Table 1). A detailed description of the materials and methods used is provided in the Supplemental Material.
Grayanotoxin I (GTX I) Tissue Concentrations Determined by HPLC-MS/MS, Miniature Pig.
A polymerase chain reaction and an ELISA failed to detect nucleic acid of classical or African swine fever viruses in samples of liver, kidney, and lung, or antibodies in blood against these viruses (data not shown).
Differential etiological diagnoses, which also cause clinical signs of neurotoxicity with unspecific pathological manifestation include intoxication with organophospates such as parathion, carbamates, or metaldehyde. 14 The neurotoxins veratridine and aconitine produced by Aconitum or Veratrum plants, respectively, and the batrachotoxin found for example in poison dart frogs also target voltage-gated sodium channels identically to GTX causing a similar disease pattern. 24
The miniature pigs clinically showed unspecific clinical signs with sudden onset such as shivering, ataxia, hypersalivation, lateral recumbency, and vomiting, which have also been reported in GTX I intoxication of other species including humans. 8,22 These clinical signs vanished suddenly in the female pig after several hours. In contrast the male pig died, which is a typical disease outcome in animals poisoned with GTX I. 8 The pathological examination showed no specific lesions but identified plant material in the intestinal tract as the exclusive specific indicator of a GTX intoxication. Specific lesions of the nervous system after GTX intoxication have never been reported. 2,17,22 Only intestinal hemorrhages and aspiration pneumonia due to vomitus had been observed. 5,17,22 The alpha subunit of activated voltage-dependent sodium ion channels on cell membranes of neurons is the primary target of GTX. Its binding to the channel results in a permanent depolarization. Secondary, calcium ion influx through synaptic voltage-dependent calcium ion channels on ventromedial hypothalamic neurons leads to increased gamma-aminobutyric acid (GABA) and glutamate release. This culminates in autonomic effects like hypotension and bradycardia. 1,6,9 M2-subtype muscarinic acetylcholine receptors may also be involved in GTX-induced cardiotoxicity. 16 Contact of GTX with mucosa leads to local irritation. 7 Knight stated that vomition is induced by peripheral stimulation of the vomiting center via the nervus vagus. 10
Clinical signs in humans with mad honey disease belong to the cholinergic toxidrome and can be treated with atropine. 4 Bradycardia and hypotension occur in most cases; dizziness, respiration and an altered mental status are common; syncope, blurred vision are found in some cases; and salivation was less frequent. 7,25,26 Similar clinical signs in animals are reported including vomiting, depression, abdominal pain, salivation, bloat, muscle tremor, convulsions, cardiac arrhythmias (bradycardia, tachycardia and others), blindness, weakness, dyspnea, ataxia, recumbency and ophisthotonos and only rarely diarrhea. 2,22 These cholinergic type signs begin 3 to 14 hours after ingestion of toxic plants and last approximately 24 hours. 22
The detection of the toxin in several tissues and the failure to identify other diseases or causes points towards the ingestion of GTX-containing Pieris japonica as causative for the clinical signs in these pigs. The concentrations of GTX I which were measured in porcine tissues were lower compared to reports in other species. In a human case with mad honey disease, GTX I concentrations of 30.62 ng/mL and of 0.447 mg/mL were determined in the blood and the urine, respectively. 1 In poisoned goats, urine contained 50-300 ng/g GTX I. 19 Species differences in terms of their sensitivity to intoxication from plants are known, 18 and it is tempting to speculate that pigs are more sensitive than other species. Underlying diseases such as heart or liver insufficiency have an impact on the severity and outcome of intoxications. Although the anamneses and pathological examination of this miniature pig did not identify such conditions, we cannot exclude that further factors exceeding the intoxication contributed to the clinical signs and death.
Pieris japonica also known as Japanese pieris, Japanese Andromeda or Lily-of-the-Valley Shrub is popular as an ornamental plant and was also planted in the garden that was accessible to the pigs of this case report (Fig. 3). However, the toxicity of this and other plants is widely unknown to pet owners and there is increased popularity of pigs, especially miniature pigs, as pets. Plant intoxications always have to be considered as a putative cause of a disease, also in pigs, and the discovery of ingested plant material is often the only indication for intoxication.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818789482 - Grayanotoxin I Intoxication in Pet Pigs
Supplemental Material, DS1_VET_10.1177_0300985818789482 for Grayanotoxin I Intoxication in Pet Pigs by Hannah Pischon, Anne Petrick, Matthias Müller, Nils Köster, Jörg Pietsch, and Lars Mundhenk in Veterinary Pathology
Footnotes
Acknowledgements
The authors thank Prof A. Hensel, Dr M. Lechtenberg, and J. Wang from the Institute of Pharmaceutical Biology and Phytochemistry, University of Münster, for providing the GTX I standard and S. Binder from the Landeslabor Berlin-Brandenburg for the investigation for classical and African swine fever.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
