Abstract
Canine nonepitheliotropic cutaneous T-cell lymphomas (NECTCL) are poorly characterized. In humans, a number of distinct subtypes of NECTCL have been recognized, including subcutaneous panniculitis-like T-cell lymphoma (SPTCL). Five dogs with subcutaneous T-cell lymphomas histologically similar to SPTCL in humans are herein described. The mean age was 8.5 years (5.5 to 12 years). No breed or sex predilection was identified in this small cohort. Two dogs presented with an acute onset of multiple skin masses and 3 dogs had solitary masses with subsequent development of multiple smaller masses within 0.5 to 2 months post-diagnosis without treatment. Locations, when specified, included shoulder, neck, and ventral abdomen. Two dogs were euthanized following diagnosis and one dog treated with chemotherapy (CCNU) survived 7 months post-diagnosis. Histologically, all cases were characterized by proliferations of either small to intermediate or large sized, CD3-positive T cells that infiltrated the subcutis in a lace-like pattern and frequently rimmed adipocytes. No epitheliotropism was observed, neoplastic cells were often karyorrhectic, and there were regions of extensive necrosis. Heavy infiltrates of histiocytes with prominent phagocytosis masked the lymphoid neoplastic cell population in some sections. A clonal T-cell receptor gamma gene rearrangement was found in 4 of the 5 cases. While SPTCLs typically have a less aggressive clinical course in humans, their biological behavior in dogs remains to be determined. In summary, SPTCL may represent a distinct entity in dogs and needs to be accurately diagnosed to better determine clinical behavior.
Primary cutaneous T-cell lymphomas (CTCL) are a diverse group of lymphomas in respect to clinical presentation, morphologic and immunophenotypic characteristics, and disease progression and outcome. CTCLs can be broadly divided into epitheliotropic and nonepitheliotropic forms, of which the former is most common. 17,28 Epitheliotropic forms are often easily identified histologically based on infiltration of the epidermis and/or adnexal structures by neoplastic lymphocytes, and, despite well recognized phenotypic and prognostic differences in dogs in comparison to humans, are subtyped according to the human World Health Organization (WHO) classification. 15,29 These subtypes include mycosis fungoides (MF), pagetoid reticulosis, and Sézary syndrome (SS). While pagetoid reticulosis is limited to the epidermis, MF may be epidermotropic or spread to other sites. In contrast, SS is, by definition, a systemic disease. 15,17,29 On the other hand, the nonepitheliotropic forms of CTCL have been less well characterized in dogs and, from a diagnostic prospective, can prove challenging in some instances due to heavy infiltrations of inflammatory cells. 16
Most nonepitheliotropic cutaneous T-cell lymphomas in humans and dogs are currently classified in the general category of peripheral T-cell lymphomas, not otherwise specified (PTCL-NOS); however, a number of distinct subtypes are being recognized in humans. Although rare, one subtype recognized in humans is the subcutaneous panniculitis-like T-cell lymphoma (SPTCL), which is often indolent. 9,25,31 This subtype is predominately located in the subcutaneous adipose tissue and, in at least the early stages, resembles panniculitis. 7 While SPTCL has been suggested as a potential subtype of canine PTCL-NOS by the application of WHO standards, 27 –29 the entity has not been independently described in dogs with respect to diagnostic criteria and prognostic significance. This study aims to characterize a specific type of nonepitheliotropic cutaneous T-cell lymphoma in a series of dogs similar to SPTCL in humans, and proposes SPTCL as a distinct entity in dogs.
Material and Methods
Case Selection
Five dogs, all of which had been patients at different veterinary clinics within the State of Michigan, were included in this study based on the clinical history of one or more cutaneous/subcutaneous nodules, histomorphologic features of the mass, and immunophenotype. For all cases, the initial diagnosis was nonepitheliotropic lymphoma.
Tissue Processing
One formalin-fixed haired skin biopsy specimen collected from a single cutaneous/subcutaneous nodule from each patient was submitted by the respective veterinary practitioner between the years 1995-2015 to the Michigan State University Veterinary Diagnostic Laboratory (MSU VDL). Tissues were routinely sectioned and embedded in paraffin blocks upon receipt. All paraffin-embedded tissues from the original cases were archived at the MSU VDL and 5 µm in thickness sections of each archived tissue block were routinely stained with hematoxylin and eosin, and were examined histologically by two of the authors (MK, ELN).
Immunohistochemistry
Immunohistochemistry using polyclonal rabbit anti-human CD3 (DAKO, 1:200), polyclonal rabbit anti-human CD20 (Thermo/Neomarkers, 1:200), and monoclonal mouse anti-human CD56 (BC56C04, BioCare, 1:100) was performed on a Bond-Max automated system (Leica Microsystems, Bannockburn, IL) using a biotin-free, polymeric peroxidase-based detection kit with DAB chromagen and hematoxylin counterstain (Bond Polymer Refine Detection kit; Leica Microsystems) as previously described. 4,18,23 Immunohistochemistry for monoclonal mouse anti-human CD30 (Ber-H2, DAKO, 1:50) was performed on a DAKO 48 immunostainer using a biotin-free, polymeric peroxidase-based detection kit with DAB chromagen and hematoxylin counterstain (FLEX Detection kit; DAKO). 19 Antigen retrieval was achieved by incubating slides in low pH retrieval solution (CD30) for 20 minutes on the Dako PT link or in Bond Epitope Retrieval Solution I (CD3 and CD20) or Bond Epitope Retrieval Solution 2 (CD56) for 20 minutes on the Bond-Max automated system. Cells with CD3, CD20, or CD30 membranous or cytoplasmic labeling and cells with CD56 cytoplasmic labeling were evaluated as positive. Positive controls consisted of sections of canine lymph node for CD3 and CD20, canine tonsil for CD30, and canine cerebrum for CD56. Negative controls for all antibodies were made by omitting the primary antibodies.
PARR Testing
PARR testing was performed at two separate institutions, the MSU VDL and the Department of Pathobiology at Ontario Veterinary College. For both institutions, DNA extraction was performed on 5 serial 5-μm-thick sections (25 µm-thickness total) from formalin-fixed, paraffin-embedded tissues. Rearrangements of the TCRγ and immunoglobulin heavy chain gene variable regions was assessed by amplifying the complementary determining region 3 (CDR3) as previously described. 3–4,10,28
Results
Clinical History
Three dogs were females (2 spayed) and 2 were males (1 neutered). The mean age at the time of diagnosis was 8.5 years (range: 5.5-12 years, median: 8 years). No specific breed was overrepresented. Individual signalments are listed in Table 1. Cases 1 and 2 had an acute onset of multiple skin masses. Cases 3-5 had a solitary mass with subsequent development of multiple smaller masses within 0.5 to 2 months later in the untreated animals (cases 3-4) and 7 months later in the treated animal (case 5). Locations, when specified, included the shoulder, neck, and ventral abdomen regions. Such masses were erythematous and/or ulcerated from cases 2 and 4, and there was purple bruising of the skin overlying the mass from case 5. Concurrent provided conditions included diabetes mellitus (case 2) and hip dysplasia (case 4). Dog 4 had status epilepticus and false pregnancy three months prior to biopsy. No additional seizures were observed in this animal and the cutaneous/subcutaneous mass was not detected at that point in time.
Signalment and Clinical Findings in Dogs With Subcutaneous Panniculitis-Like T-Cell Lymphoma.
Treatment and Outcome
Cases 3-5 were euthanized due to disease progression (0.5-7 months post-diagnosis) either without treatment (case 3) or with treatment (cases 4-5) (see Table 1 for details). For Case 5, chemotherapy was stopped after 4 rounds due to elevated serum concentrations of liver enzymes at 3 months post-biopsy. The animal was in clinical remission at that time. Additional clinical information was not available for two dogs (cases 1-2).
Histologic Lesions
Masses of all cases were characterized by proliferations of neoplastic lymphocytes infiltrating the subcutaneous adipose tissue in a predominately lobular pattern, subgrossly mimicking an inflammatory infiltrate (Figs. 1–2). Within the subcutis, neoplastic cells frequently rimmed adipocytes in a lace-like configuration and were regionally masked by heavy infiltrates of histiocytes with prominent phagocytosis (Figs. 3–4). Neoplastic lymphocytes had small to intermediate-sized nuclei that were densely stippled to hyperchromatic and occasionally had 1-2 prominent nucleoli. In some cases (Cases 1, 3, and 5), there were also regions of neoplastic cells with large, vesiculate nuclei with prominent nucleoli and numerous mitotic figures (Fig. 5). There was frequent karyorrhexis of individualized or clusters of neoplastic cells scattered throughout all masses (Fig. 4) and multifocal large areas of necrosis. In two cases (cases 2-3), neoplastic cells dissected between underlying skeletal muscle bundles.
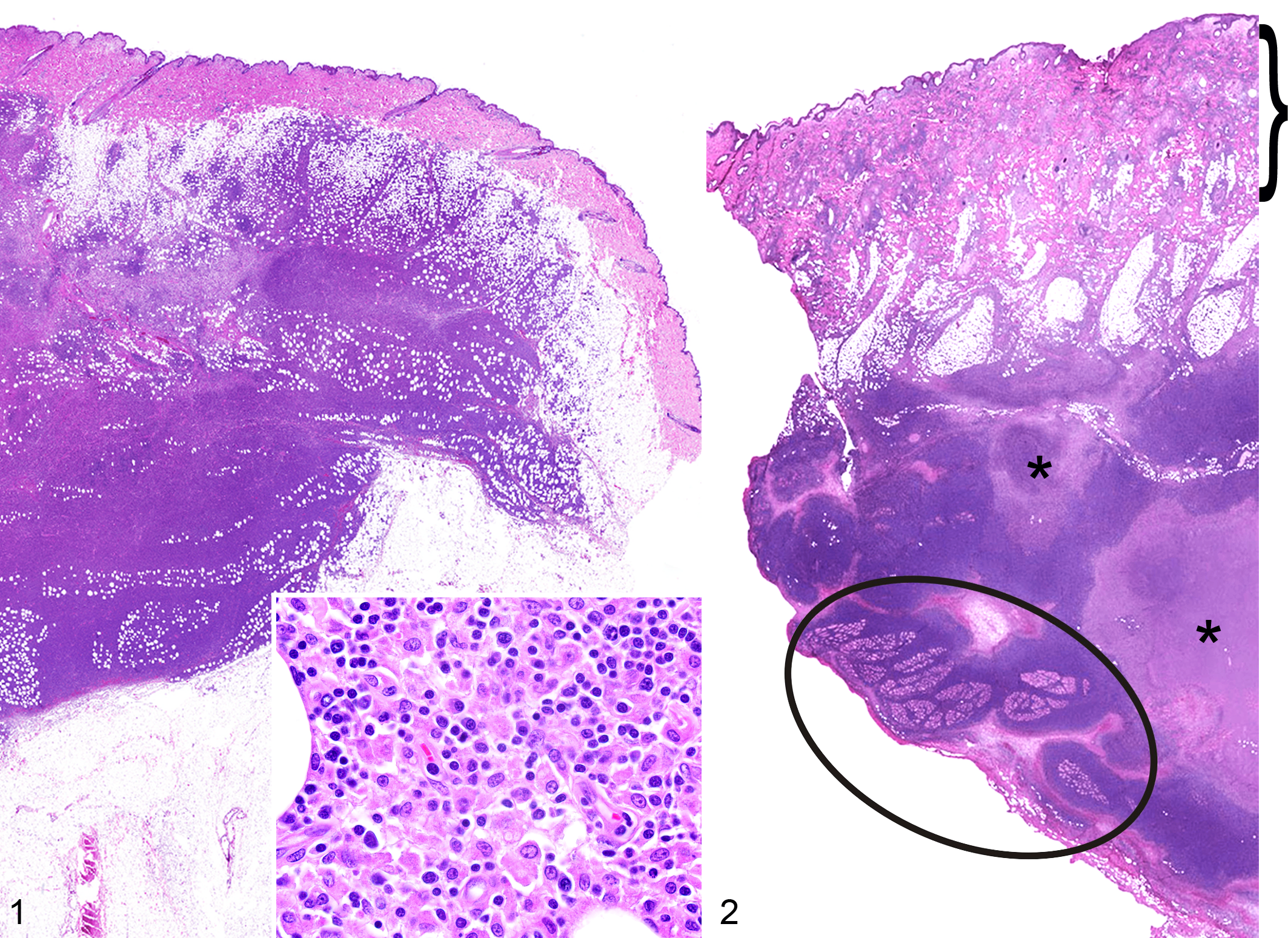
Subcutaneous panniculitis-like T-cell lymphoma, haired skin, dog. Hematoxylin and eosin.
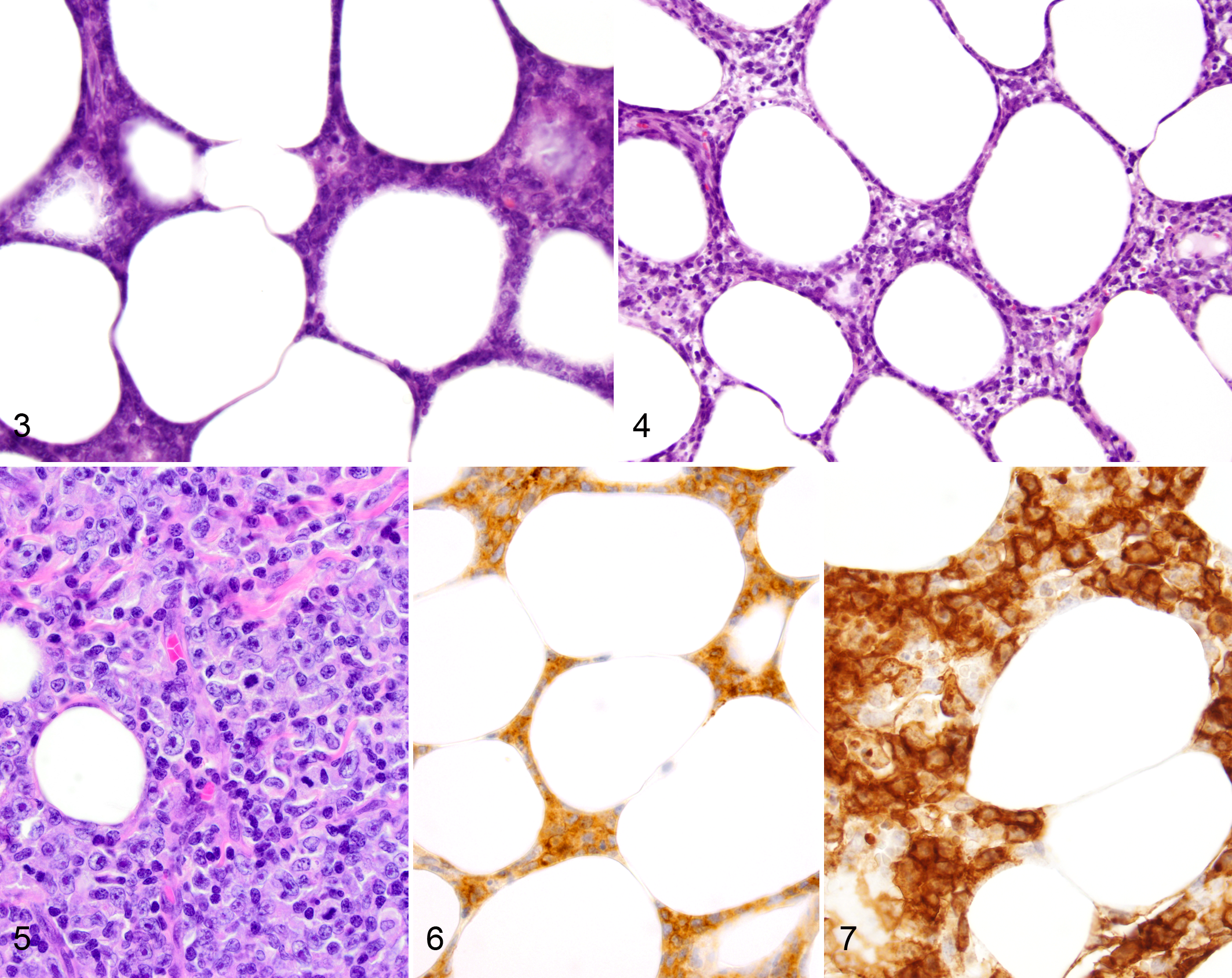
Subcutaneous panniculitis-like T-cell lymphoma, haired skin, dog.
While the bulk of each mass was within the subcutis, in the majority of cases (cases 2-5) lesser numbers of neoplastic cells extended into the dermis (Fig. 2). Dermal extension included the periadnexal dermis and multifocal variably sized aggregates around vessels of the deep and mid-dermal plexuses (cases 2-4), and in some cases included few perivascular to interstitial aggregates within the superficial dermis (cases 3-4). For case 5, few mid-dermal vessels contained clusters of intraluminal neoplastic cells. While neoplastic cells occasionally tightly surrounded dermal adnexa or encroached on the epidermis, discrete aggregates or nests of neoplastic cells within the follicular/adnexal epithelium or epidermis (epitheliotropism) were not observed. In addition, the superficial dermal stroma in all cases was moderately expanded by edema and variable numbers of superficial perivascular to scattered interstitial mast cells, lymphocytes, and plasma cells. Rare neutrophilic vasculitis of the superficial and mid-dermal plexi was noted in case 2.
Immunohistochemical Findings
In all cases, the neoplastic cells had membranous or cytoplasmic immunoreactivity for CD3 (Fig. 6); however, for some dogs (cases 2, 4, and 5), CD3 immunoreactivity was weak either diffusely throughout or within regions of the mass. Throughout the lesion in all cases, there were small numbers of scattered individualized small lymphocytes or multifocal moderate-sized clusters of small lymphocytes that had strong membranous or cytoplasmic immunoreactivity for CD20 (cases 1 and 4), consistent with inflammatory infiltrates. The neoplastic cells were not immunoreactive for CD20. Neoplastic lymphocytes diffusely had strong membranous or cytoplasmic immunoreactivity for CD30 in cases 1 and 3 (Fig. 7), and had weak membranous or cytoplasmic CD30 immunoreactivity within patchy regions of the mass in cases 2 and 5. Neoplastic lymphoid cells in case 4 were not immunoreactive for CD30. In all cases, the neoplastic cells were not immunoreactive for CD56.
PARR Testing
PARR testing was performed by two separate laboratories for all 5 cases. For three dogs (cases 1, 2, and 5), both laboratories reported a monoclonal T-cell receptor gamma gene rearrangement. In case 3, one laboratory (MSU VDL) reported a monoclonal T-cell receptor gamma gene rearrangement, while no amplification of the DNA from paraffin shavings of the tissue block in this case was reported by the other laboratory (Ontario Veterinary College). In case 4, a polyclonal result was reported by one laboratory (MSU VDL), while at the other laboratory (Ontario Veterinary College) the sample failed to amplify for either the T- or the B-cell primer sets.
Discussion
This case series characterizes a unique subtype of canine cutaneous nonepitheliotropic T-cell lymphoma in dogs that has not been previously independently described. While the histomorphology of SPTCL in dogs was similar to that described in humans, the biologic behavior was more aggressive in dogs than has typically been reported in humans based on limited follow-up information. In dogs, animals that were not treated with chemotherapy were euthanized due to progressive disease at 16 or 17 days post-diagnosis and one dog treated with chemotherapy was euthanized 7 months post-diagnosis. In contrast, SPTCLs in humans are often indolent and, in many cases, respond well to high doses of systemic corticosteroids as a first line of defense, although hemophagocytic syndrome may develop in some human cases and be associated with poor prognosis. 14,20,26 In cases with recurrence or resistance to immunosuppressive therapy, combination chemotherapy with cyclophosphamide, doxorubicin, vincristine, and prednisolone (CHOP) or CHOP-like chemotherapy and/or stem cell transplants may be employed. 6,26,31 For localized disease, long-term remission has been reported with radiation therapy. 31 Differences in biologic behavior/phenotype have been previously reported between morphologically similar canine and human lymphomas. For example, canine cutaneous epitheliotropic T-cell lymphoma (MF subtype) shares histomorphologic similarities to lesions in human MF, but in contrast to the human entity carries a poor prognosis in dogs. Furthermore, in the majority of canine cases, the neoplastic infiltrate is composed of CD8+ lymphocytes while most human MFs consist of CD4+ lymphocytes. 11,15,22
However, aggressive forms of SPTCL do occur in humans. Aggressive behavior is typically associated with a γδ+ T-cell receptor phenotype as opposed to the αβ+ phenotype that is more often seen with an indolent course. 24 Histologically, these phenotypic subtypes are cytologically indistinguishable on routine microscopic evaluation, but extension of neoplastic cells into the dermis and epidermal ulceration have been more commonly reported with the γδ+ phenotype in human SPTCLs. 31 Due to the difference in clinical outcome and thus initial treatment approach, those nonepitheliotropic T-cell lymphomas with a γδ+ phenotype have been reclassified according to the WHO as a separate entity known as primary cutaneous γδ T-cell lymphoma, reserving the designation of SPTCL for those cases with a αβ+ phenotype. 20 Notably, despite this reclassification, there are cases of aggressive or refractory αβ-SPTCLs in 15-20% of affected humans, typically associated with the aforementioned hemophagocytic syndrome. 6,20 At the same time, indolent forms of primary cutaneous γδ T-cell lymphoma in humans have at least been suggested. 13 In addition, there have been reports of transformation of cutaneous γδ T-cell lymphoma to a more aggressive phenotype years after an initial indolent onset and also potentially arising within long-standing panniculitis. 1,8 Typing of neoplastic T cells as αβ or γδ is not routinely performed on histological specimens in dogs due to the lack of anti-αβ and anti-γδ antibodies that work in formalin-fixed, paraffin-embedded tissues. As such, the use of SPTCL as an umbrella term potentially containing both phenotypic subtypes is recommended for dogs until such time that definitive subtypes with different biologic behaviors can be routinely identified.
While it is impossible to differentiate αβ-SPTCL and γδ–SPTCL in formalin-fixed tissues in dogs due to the lack of T-cell receptor antibodies, some other antibodies may be useful in predicting the biological behavior of these neoplasms. In humans, SPTCLs are typically CD3+, CD4-, CD8+, βF1+, CD56-, CD30-, and positive for cytotoxic proteins (TIA-1, granzyme B, or perforin). 20,31 This is in contrast to the typical profile of primary cutaneous γδ T-cell lymphomas, which are CD3+, CD4-, CD8-, βF1-, CD56+, CD30-, and positive for cytotoxic proteins. 20,31 Approximately 10-20% of cases of human cutaneous γδ T-cell lymphomas are positive for CD8, and CD30+ cutaneous γδ T-cell lymphomas have at least been reported. 6,20,21,30 There are commercially available antibodies for CD56 and CD30 that cross-react with the canine formalin-fixed antigens. 18–19,23 Both NK cells and NK-like T cells express CD56, the latter of which also express T-cell markers such as CD3. CD30 is a cell surface receptor that is highly expressed on recently activated γδ T cells in humans, as well as on normal activated T and B lymphocytes. 2 In our series of dogs, all cases were CD56-, which is more typical of αβ-SPTCL and would suggest a more favorable prognosis in most human cases. On the other hand, 4/5 cases had CD30 immunoreactivity, albeit variable, which has been reported in some γδ-SPTCL in humans. In addition, localization of neoplastic cells around blood vessels or scattered throughout the interstitium of the dermis, angioinvasiveness, and angiodestruction, and dermal edema have all been associated with aggressive behavior in humans. 31 In four of the dogs (cases 2-4), dermal perivascular aggregates and associated edema were noted and, at least one of the dogs (case 5), had neoplastic cells within the lumens of vessels in the dermis. In limited follow-up data, there were no reported clinical signs to suggest an associated hemophagocytic syndrome.
The rimming of fat spaces by round cells is not exclusive to SPTCL. Rimming of adipocytes by round cells may be seen in benign lobular panniculitis as well as in other primary or secondary cutaneous lymphomas. 12 CD30 immunoreactivity may be seen in other peripheral T-cell lymphomas. In particular, primary cutaneous anaplastic T-cell lymphoma in humans is characterized by diffuse CD30 positivity, but given the cellular morphology of the neoplastic cells in this case series, a SPTCL with CD30 positivity was favored. 5 Because of the overlap in histologic appearances between SPTCL, other subtypes of lymphoma, and panniculitis, a diagnosis of SPTCL must be done with consideration of the histopathologic findings, immunophenotyping, clinical history, and a thorough clinical evaluation. At a minimum, immunophenotyping using markers such as CD3 and CD79a/CD20/Pax5 is necessary to further define the cell populations present and to illustrate that the suspected neoplastic population is most likely of T cell origin. PARR testing to demonstrate clonality may be necessary in cases where neoplastic T lymphocytes have lost CD3 expression or when the neoplastic population is not readily recognized. While a clonal TRG rearrangement does not prove T cell lineage, it may help substantiate a lymphoid origin or confirm a neoplastic etiology when panniculitis is a prime differential.
While PARR testing was not necessary to diagnose lymphoma in this series of cases, as this was confidently achieved by histomorphologic features and immunophenotyping alone, PARR testing may be required in cases with marked inflammation that masks the neoplastic cell population. However, in cases with a heavy mixed inflammatory infiltrate, reactive lymphocytes might outcompete neoplastic lymphocytes resulting in a polyclonal, that is, false negative, PARR result. 16 This may explain the polyclonal result in case 4. If suspicious, close monitoring and biopsy of recurrent or new lesions may be required to reach an accurate diagnosis.
Due to lack of necropsies in all cases, it is unclear if and to what extent the SPTCLs had spread to extra-cutaneous sites at the time of euthanasia. However, the rapid deterioration of untreated animals suggests metastatic spread or systemic involvement. In case 4, it is unclear if the seizures occurring around the time of eruption of the cutaneous masses were a manifestation of systemic disease or a concurrent form of epilepsy. Further investigation, including prospective studies, additional immunophenotyping on frozen tissue samples, and genetic analysis is warranted. This case series serves to raise awareness of this entity in dogs and provides a basis to collect cases moving forward in order to better understand the biology of this type of lymphoma and to better guide prognosis.
Footnotes
Acknowledgements
We would like to thank Madison Operacz for performing clonality testing at the Michigan State University Veterinary Diagnostic Laboratory (MSU VDL), and the histology department at MSU VDL, particularly Tom Wood, for immunohistochemistry. Gratitude to the following clinics/clinicians for providing follow-up information: Kaspari Veterinary Clinic, Battle Creek, MI; Rakestraw Animal Hospital, Detroit, MI; Animal Medical Center of Traverse City, Traverse City, MI; and Christine Swanson, DVM, DACVIM, Blue Pearl, Grand Rapids, MI.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
