Abstract
The diagnosis of cutaneous epithelial tumors (CETs) in dogs is based on predominant histological differentiation patterns. However, the expression of a broad panel of antigens has not been comprehensively examined with immunohistochemistry. The present study aims to establish a comprehensive expression profile and identify useful diagnostic markers for each CET type. Cytokeratin (CK), stem cell, and other associated markers were immunohistochemically examined in 110 canine CETs. Among these, CK16 was useful for differentiating between basal and squamous cell carcinomas. Acantholytic squamous cell carcinomas were positive for CK8, CK18, and CK19, suggesting their close association with the apocrine duct. Unlike their benign counterparts, sebaceous carcinomas coexpressed CK5/6 and adipophilin. Smooth muscle actin (SMA) and p63 immunostaining were useful for accurately distinguishing between glandular and ductal differentiation in apocrine tumors. A case of apocrine carcinoma and malignant myoepithelioma was identified using anti-SMA antibodies. Stem cell expression profiles (CK8, CK15, CK19, and CD34) of hair follicle tumors were discrete and indicative of their anatomic origins. The effectiveness of immunohistochemistry for tumor diagnosis was further confirmed by hierarchical cluster analysis, through which selected markers were able to sort CETs into specific groups: CK5/6, CK8, CK14, CK16, CK18, CK19, p63, adipophilin, and SMA sorted tumors of epidermal, apocrine, or sebaceous origin; while CK8, CK14, CK15, CK16, CK19, CD34, and p63 sorted hair follicle tumors in agreement with their histological differentiation. In conclusion, the present study provides comprehensive immunohistochemical information, which could complement histomorphological features for the future classification of canine CETs.
Keywords
Among domestic animals, cutaneous epithelial tumors (CETs) are particularly common in dogs 36 and are classified based on their predominant histological differentiation pattern, which resembles the particular adnexal structures found in skin. 11 Occasionally, some canine cases exhibit multiphenotypic differentiation, 32,37 causing difficulty in establishing an accurate diagnosis based on histological evaluation alone. Despite their neoplastic transformation, most CET cells have well-conserved cytokeratin (CK) expression, 20 and these CKs could serve as valuable immunohistochemical (IHC) markers for precisely identifying the tumor’s origin or differentiation. 7,16,24
Cytokeratins belong to a group of intermediate filaments forming the main cytoskeletal structures of epithelial cells and are classified into several types based on their physiochemical properties. 6 An IHC study of the CK profiles of canine CETs was previously performed, in which useful CK markers for CET diagnosis were identified. 15,36 In recent years, the use of epidermal stem cell markers for the characterization of canine CETs, particularly of hair follicle tumors, has also been investigated. 5 While these studies 5,15,36 provide valuable information about the origin and differentiation of some CET types, the complete IHC profile of canine CETs remained unclear, owing to a lack of comprehensive studies since the publication of the latest edition of the World Health Organization (WHO) International Histological Classification of Skin Tumors of Domestic Animals. 11
In previous studies, cluster of differentiation (CD) 34 9,26 and nestin 18 were found to be useful stem cell markers expressed in the isthmic segment of the canine hair follicle. The expression of p63, a homologue of p53 protein, 38 was observed in the basal cells of the epidermis and its associated adnexa, as well as in the myoepithelial cells of the apocrine gland; thus, its expression in the CETs is of great diagnostic importance. 30,31 Adipophilin reacts with intracytoplasmic lipids and is an important marker for the diagnosis of sebaceous carcinoma in humans. 2,23,25 Vimentin and smooth muscle actin (SMA) were useful in identifying mesenchymal components, as well as myoepithelial cells of the apocrine gland, which are of true epithelial origin. 3,27 The present study used a panel of antibodies against cytokeratins (CKAE1/AE3, CK5/6, CK7, CK8, CK10, CK14, CK15, CK16, CK18, and CK19), CD34, p63, nestin, adipophilin, vimentin, and SMA for the characterization of canine CETs.
Owing to the need for the complete IHC characterization of canine CETs, the present study aimed to perform IHC expression profiling for these tumor entities. The IHC results were then analyzed by hierarchical cluster analysis to objectively delineate an optimal panel of diagnostic markers for the classification of canine CETs.
Materials and Methods
Cases
Biopsies of canine cutaneous epithelial tumors submitted to the Laboratory of Veterinary Pathology, Graduate School of Agricultural and Life Sciences, the University of Tokyo, between January 2010 and March 2017, were retrieved and examined continuously over a period of 2 years (from May 2015 to April 2017). Cases were selected based on the availability of paraffin-embedded tissue specimen for IHC evaluation. Core biopsies and poorly preserved specimens were excluded from the study. Based on these criteria, a total of 110 canine epithelial skin tumor samples were selected and consisted of basal cell carcinomas (n = 2), well-differentiated squamous cell carcinomas (n = 3), acantholytic squamous cell carcinomas (n = 4), infundibular keratinizing acanthomas (n = 11), inferior tricholemmoma (n = 1), isthmic tricholemmomas (n = 2), trichoblastomas (n = 12), trichoepitheliomas (n = 10), malignant trichoepitheliomas (n = 5), pilomatricomas (n = 9), sebaceous adenomas (n = 10), sebaceous epitheliomas (n = 11), sebaceous carcinomas (n = 4), apocrine adenomas (n = 6), complex apocrine adenomas (n = 7), apocrine ductal adenoma (n = 1), apocrine carcinomas (n = 3), complex apocrine carcinomas (n = 6), apocrine carcinoma and malignant myoepithelioma (n = 1), and apocrine ductal carcinomas (n = 2). Normal skin samples were also collected from the neck, dorsal trunk, and hind limbs of 2 dogs submitted for necropsy examination. All of the tissue samples were fixed in 10% phosphate-buffered formalin and embedded in paraffin wax for histological and immunohistochemical evaluations.
Histological Analysis
Formalin-fixed, paraffin-embedded tissues were cut into 4-μm-thick sections and stained with hematoxylin and eosin (HE) and periodic acid–Schiff (PAS). The tumors were classified according to the WHO International Histological Classification of Skin Tumors of Domestic Animals 11 and its recent update. 12 An exception was made for the apocrine carcinoma and malignant myoepithelioma, in which the nomenclature was adopted from the recent update on the classification of canine mammary tumors. 10 The sections were evaluated independently by the first author (M.K.K.) and the 2 coauthors, who are board-certified pathologists (K.U. and J.K.C.) from the Japanese College of Veterinary Pathologists (JCVP). A consensus histological diagnosis was reached for all cases included in the study. The histological features of CETs have been summarized in Supplemental Table S1 and Supplemental Figs. S1 to S19.
Immunohistochemistry
Deparaffinized sections (4 μm thick) were subjected to antigen retrieval. The antigen retrieval for nestin was performed according to the method described by Gerhards et al. 9 Nonspecific reactions were blocked by immersing the sections in 3% hydrogen peroxide in methanol at room temperature for 5 minutes, followed by incubation in 8% skimmed milk at 37°C for 40 minutes. The sections were then incubated overnight with one of the primary antibodies (Table 1) at 4°C. Next, the sections were washed 3 times with Tris-buffered saline (TBS) and incubated with Dako EnVision+ System horseradish peroxidase (HRP)–labeled anti-mouse or anti-rabbit secondary antibodies (Dako, Tokyo, Japan) at 37°C for 40 minutes. For the detection of CD34, tissue sections were incubated with biotinylated anti-goat immunoglobulin G (1:400; KPL, Gaithersburg, MD) at 37°C for 40 minutes and then with HRP-conjugated streptavidin (1:200; Dako) at room temperature for 40 minutes. To visualize the products of the reaction, sections were subjected to chromogen treatment with 0.05% 3.3′-diaminobenzidine and 0.03% hydrogen peroxide in Tris–hydrochloric buffer (DAB) and counterstained with Mayer’s hematoxylin. The surrounding normal tissues in each tumor section were used as an internal positive control. For nestin immunostaining, a peripheral nerve sheath tumor sample was used as positive control. For negative control, the primary antibodies were replaced with TBS, while the panel of antibodies was used as the reagent control for verifying the specificity of each antibody. 29
Primary Antibodies and Protocols Used for Immunohistochemistry.
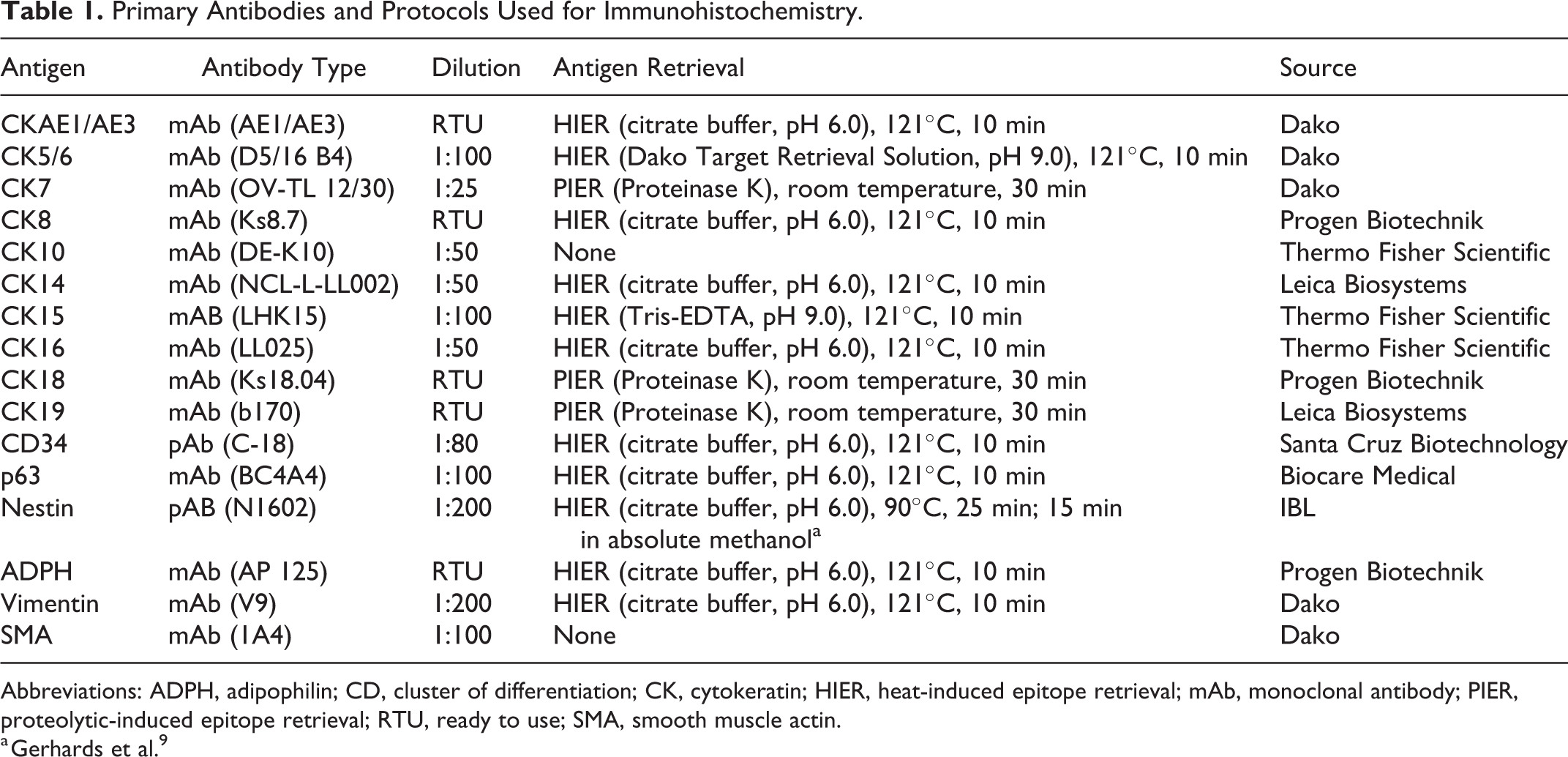
Abbreviations: ADPH, adipophilin; CD, cluster of differentiation; CK, cytokeratin; HIER, heat-induced epitope retrieval; mAb, monoclonal antibody; PIER, proteolytic-induced epitope retrieval; RTU, ready to use; SMA, smooth muscle actin.
a Gerhards et al. 9
To detect the ductal component of the apocrine gland, double immunostaining with p63 and SMA antibodies was performed on normal cutaneous tissue sections. After the visualization of p63 immunostaining with Dako EnVision+ System HRP-labeled anti-mouse secondary antibody and DAB chromogen treatment, the sections were incubated with 8% skimmed milk at 37°C for 40 minutes. The sections were then incubated with anti-SMA antibody at 4°C overnight, followed by treatment with N-Histofine Simple Stain AP (M) (Nichirei Biosciences, Tokyo, Japan), which is an alkaline phosphatase–conjugated anti-mouse secondary antibody. The immunoreaction was visualized using the N-Histofine New Fuschin substrate (Nichirei Biosciences).
Immunohistochemical Scoring
IHC expression in normal skin tissues was graded as –, negative; +/–, some cells were positive; or +, diffusely positive. The number of immunoreactive tumor cells was counted in 10 randomly selected high-power fields (at 400× magnification) in the tumor fields. The IHC score was presented as the mean percentage (± standard deviation) of positive tumor cells for each CET type. Only tumor cells that showed a membranous vesicular staining pattern were considered positive for adipophilin. 23,25
Cluster Analysis
To objectively classify the CETs into specific tumors of epidermal or adnexal origin based on the IHC profile, a multivariate method of cluster analysis, the Ward method, was applied using SPSS 15.0 software (SPSS, Inc., an IBM Company, Chicago, IL). A dendrogram was constructed by assessing the squared Euclidian distances between the clusters. Individual components of sebaceous and apocrine gland tumors were grouped together for carrying out the analysis. If 2 tumor components expressed the same marker (eg, CK8, expressed in luminal and basal cells in apocrine ductal adenoma), the higher value of the marker was used for analysis.
Results
Immunohistochemistry of Normal Skin
Immunohistochemical Features of the Normal Skin in Dogs.
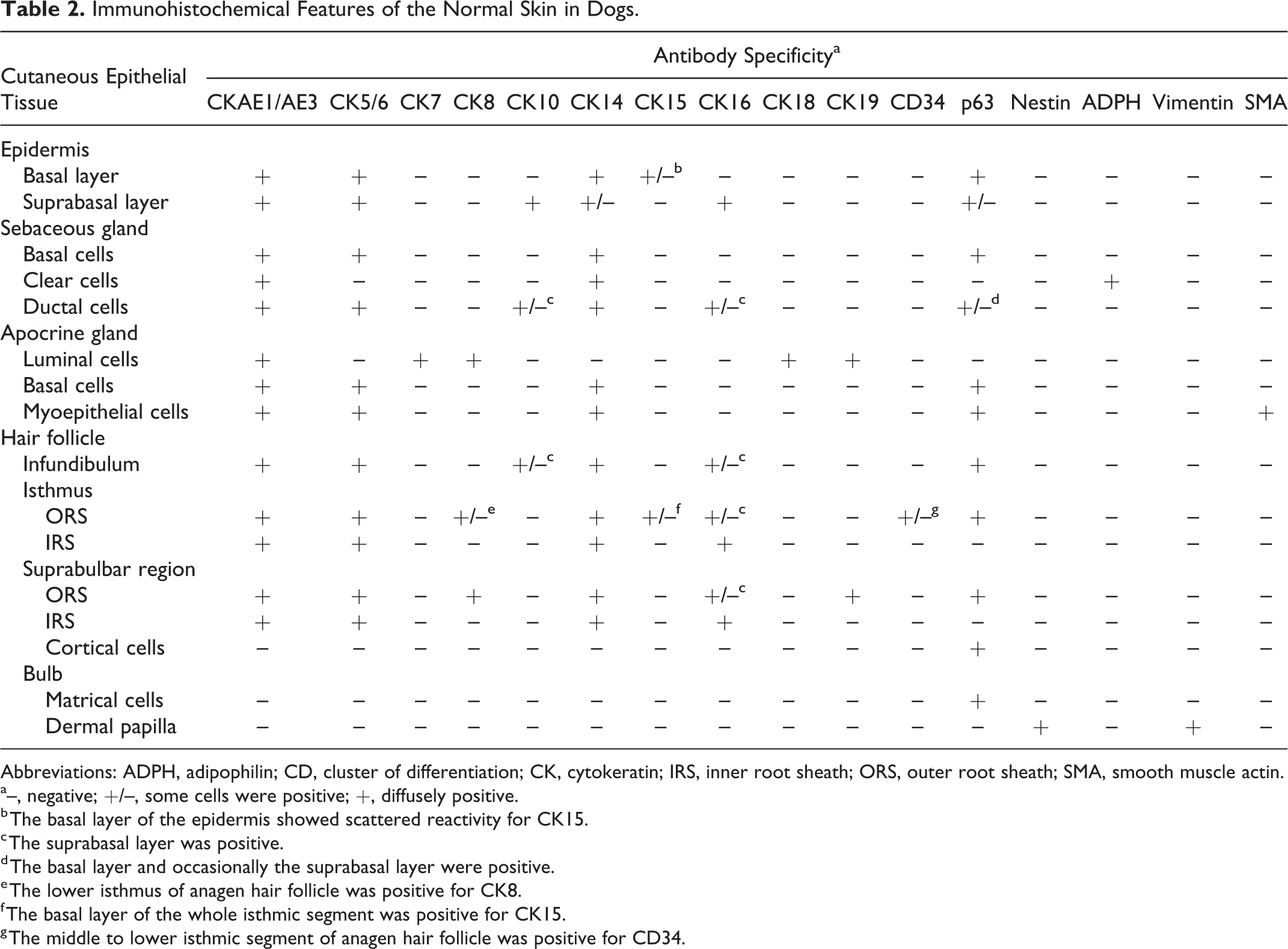
Abbreviations: ADPH, adipophilin; CD, cluster of differentiation; CK, cytokeratin; IRS, inner root sheath; ORS, outer root sheath; SMA, smooth muscle actin.
a–, negative; +/–, some cells were positive; +, diffusely positive.
b The basal layer of the epidermis showed scattered reactivity for CK15.
c The suprabasal layer was positive.
d The basal layer and occasionally the suprabasal layer were positive.
e The lower isthmus of anagen hair follicle was positive for CK8.
f The basal layer of the whole isthmic segment was positive for CK15.
g The middle to lower isthmic segment of anagen hair follicle was positive for CD34.
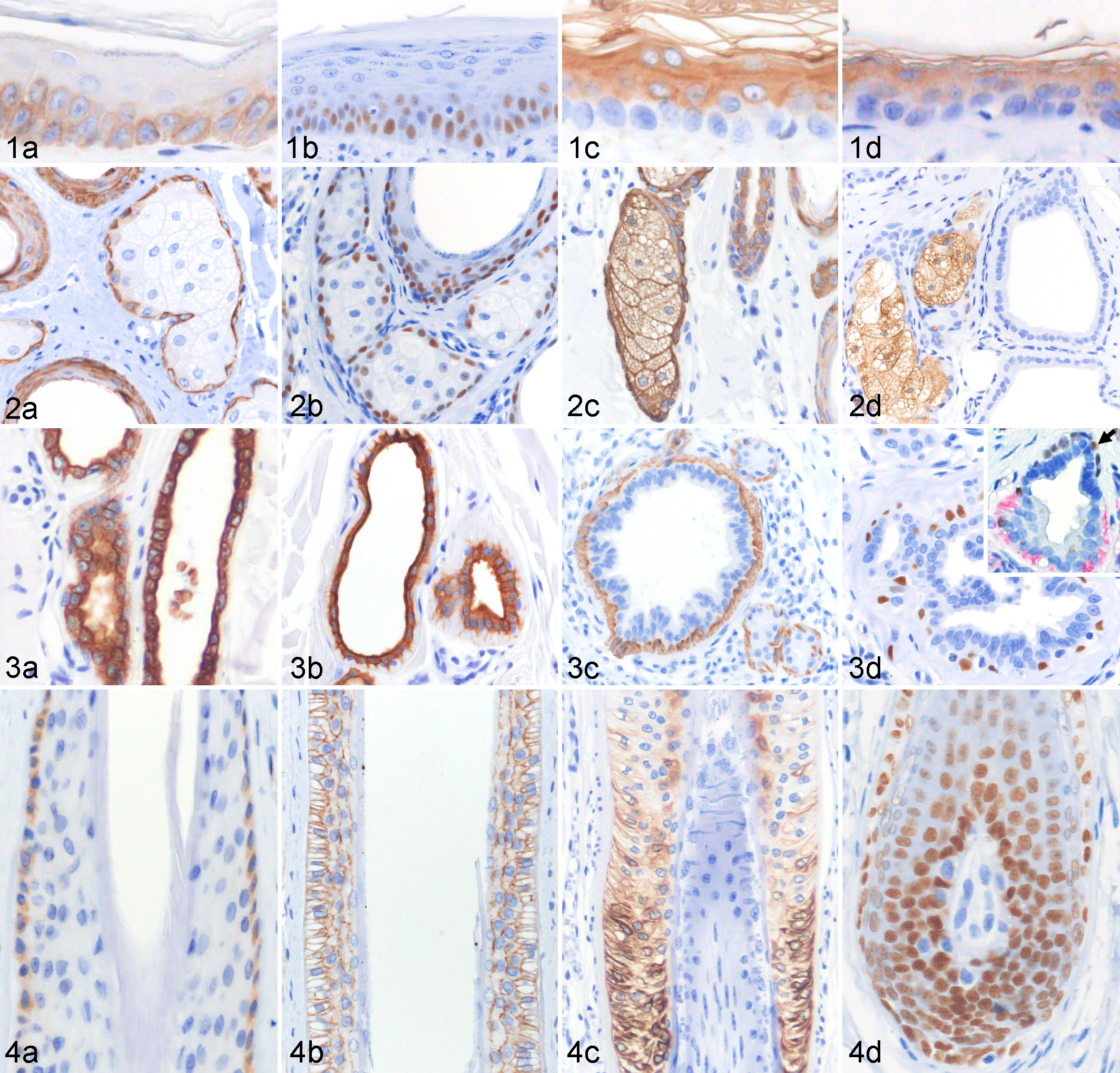
Normal skin, dog. Immunohistochemical features of the epidermis and adnexa.
Basal cells of the sebaceous gland expressed CK5/6 (Fig. 2a) and p63 (Fig. 2b), while both basal and differentiated clear cells (Fig. 2c) expressed CK14. Adipophilin was expressed in the clear cells with a membranous labeling of the intracytoplasmic lipid droplets (Fig. 2d).
The secretory and ductal components of the apocrine gland displayed a similar CK and p63 expression. The luminal cells expressed CK7 (Fig. 3a), CK8, CK18 (Fig. 3b), and CK19. The myoepithelial cells and basal cells expressed CK5/6 (Fig. 3c), CK14, and p63 (Fig. 3d, inset). Immunoreactivity to SMA was observed in the myoepithelial cells of the secretory component but not in the basal cells of the apocrine duct (Fig. 3d, inset).
In the anagen hair follicle, CK15 (Fig. 4a) was expressed in the basal cells of the isthmic outer root sheath (ORS). The middle to lower isthmic ORS expressed CD34 (Fig. 4b). CK8 was expressed in the ORS of the lower isthmus to suprabulbar region. CK19 was expressed in the suprabulbar ORS, and its expression gradually decreased toward the lower isthmus (Fig. 4c). CK16 was expressed in the suprabasal layer of the ORS and the inner root sheath of the isthmic and suprabulbar regions. Matrical cells were negative for all CKs and expressed only p63 (Fig. 4d). The dermal papilla expressed nestin and vimentin.
Immunohistochemistry of Cutaneous Epithelial Tumors
Information regarding the IHC profiles of epidermal tumors is summarized in Table 3. Tumor cells of basal cell carcinomas displayed diffuse cytoplasmic expression of CK14 (87% ± 19%; percentage of positive tumor cells, mean ± SD) (Fig. 5) and nuclear expression of p63 (90% ± 10%), while a small number of tumor cells expressed CK16 (4% ± 1%). Diffuse immunoreactivities to CK14 (95% ± 8%) and p63 (85% ± 14%) were also observed in squamous cell carcinoma cells. CK10 was weakly expressed (2% ± 3%), while CK16 expression (30% ± 17%) was observed at the center of the tumor cords or islands (Fig. 6). Nonspecific granular staining of adipophilin, similar to that of normal epidermis, was detected in some tumor cells of squamous cell carcinomas (as well as squamous epithelium of infundibular keratinizing acanthomas and trichoepitheliomas). In acantholytic squamous cell carcinomas, tumor cells lining the pseudoglandular structure occasionally expressed CK8 (3% ± 4%) and CK18 (3% ± 6%) (Fig. 7a), while a moderate to high number of tumor cells were positive for CK19 (54% ± 22%) (Fig. 7b).
Immunohistochemical Features of Epidermal Tumors in Dogs.
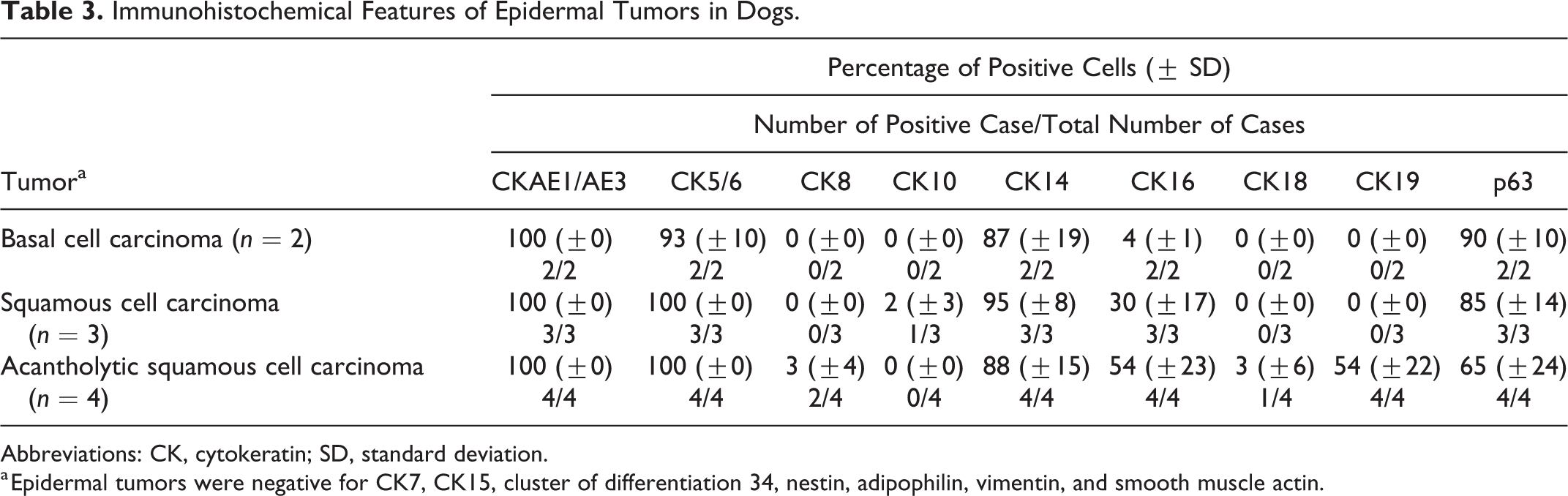
Abbreviations: CK, cytokeratin; SD, standard deviation.
a Epidermal tumors were negative for CK7, CK15, cluster of differentiation 34, nestin, adipophilin, vimentin, and smooth muscle actin.
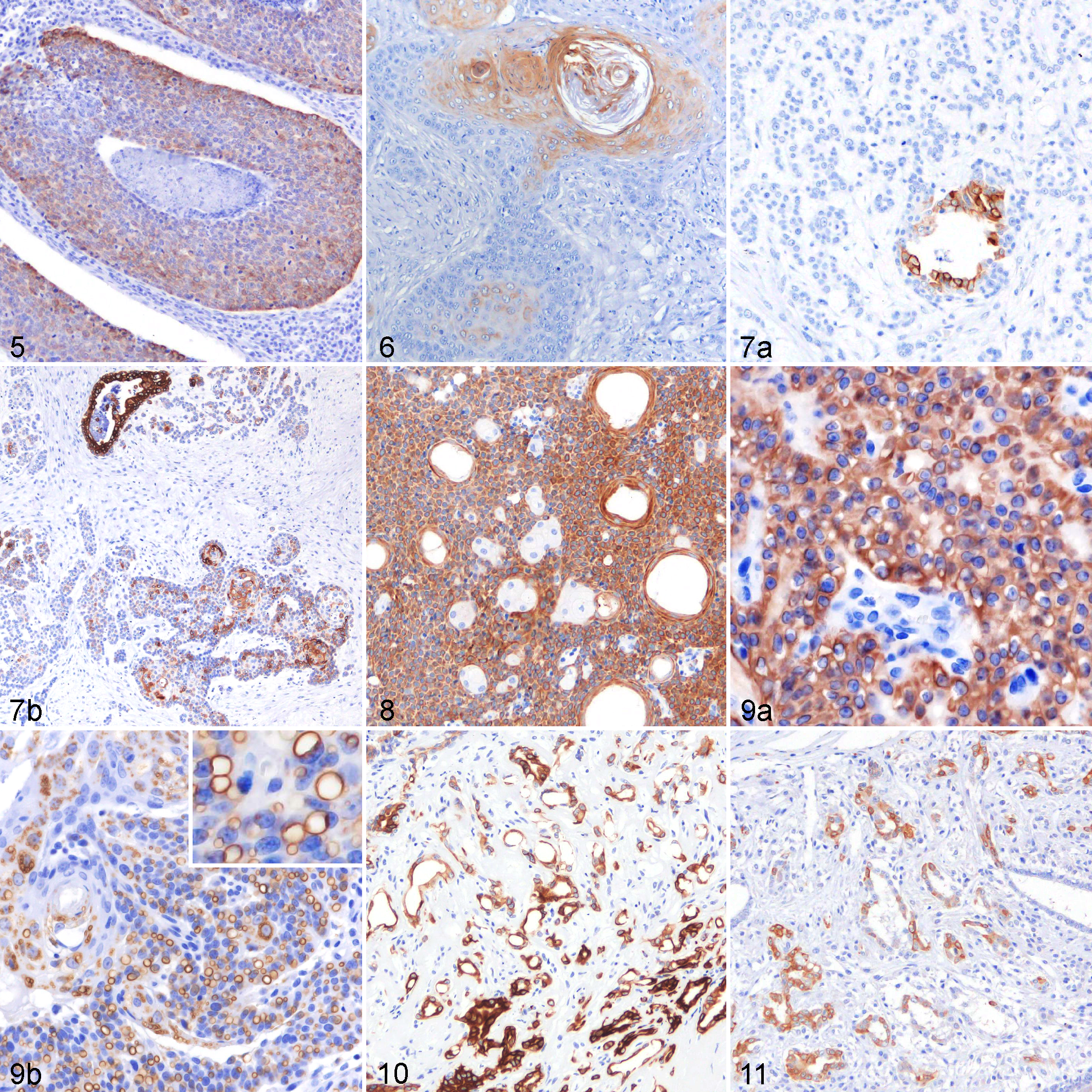
Cutaneous epithelial tumors, dog.
Immunohistochemical Features of Sebaceous Tumors in Dogs.
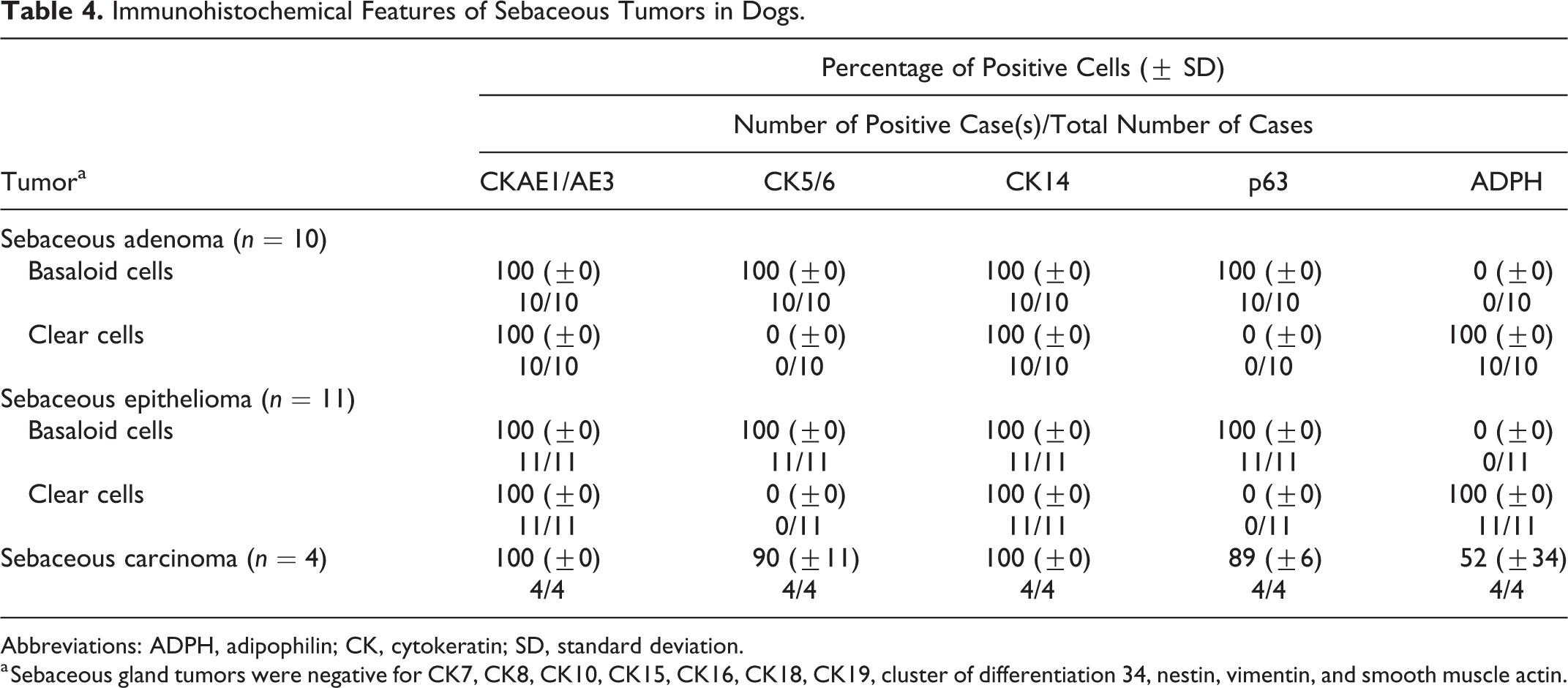
Abbreviations: ADPH, adipophilin; CK, cytokeratin; SD, standard deviation.
a Sebaceous gland tumors were negative for CK7, CK8, CK10, CK15, CK16, CK18, CK19, cluster of differentiation 34, nestin, vimentin, and smooth muscle actin.
Table 5 summarizes the IHC features of apocrine gland tumors. In apocrine adenomas, complex apocrine adenomas, and carcinomas, the luminal components expressed CK7 (36% ± 48%, 40% ± 29%, and 47% ± 15%, respectively) (Fig. 10), CK8 (100% ± 0%, all 3 tumor types), CK18 (82% ± 11%, 91% ± 17%, and 77% ± 28%, respectively), and CK19 (84% ± 17%, 93% ± 19%, and 100% ± 0%, respectively). Scattered immunoreactivity to CK15 (17% ± 6%) was observed in the luminal cells of complex apocrine carcinomas (Fig. 11). The myoepithelial cells of apocrine adenoma, which were inconspicuous on HE-stained sections, were highlighted by several IHC markers and were seen as a discontinuous layer of flattened cells lining the luminal component (Fig. 12). These myoepithelial cells, along with those of complex apocrine adenomas and carcinomas, were diffusely positive (100% ± 0%) for CK5/6, CK14, p63, and SMA. A low number of myoepithelial cells in apocrine adenomas and complex apocrine adenomas were positive for vimentin (1% ± 2% and 2% ± 6%, respectively), although a higher expression was observed in the malignant counterpart (complex apocrine carcinoma) (61% ± 47%) (Fig. 13). In an apocrine carcinoma and malignant myoepithelioma, the luminal cells expressed CK5/6 (91%), CK7 (5%), CK8 (100%), CK16 (6%), CK18 (100%) (Fig. 14a), CK19 (100%), and vimentin (20%), while the pleomorphic myoepithelial cells were positive for CK5/6 (100%), CK14 (78%), p63 (71%), vimentin (86%), and SMA (100%) (Fig. 14b). The luminal cells of apocrine carcinomas displayed immunoreactivity to CK5/6 (37% ± 49%), CK7 (42% ± 35%), CK8 (96% ± 4%), CK14 (52% ± 48%) (Fig. 15), CK18 (62% ± 45%), CK19 (70% ± 42%), p63 (2% ± 3%), and vimentin (7% ± 12%). In apocrine ductal adenomas, the luminal cells expressed CK5/6 (60%), CK8 (62%), CK14 (60%), CK18 (52%), CK19 (60%), and p63 (56%). Some luminal cells displayed squamous metaplasia and were positive for CK10 (5%) and CK16 (6%). The basal cells expressed CK5/6 (88%), CK14 (77%), and p63 (44%), and some cells were positive for CK8 (20%). In apocrine ductal carcinomas, luminal cells displayed a lower expression of CK5/6 (27% ± 19%) and CK14 (10% ± 14%), as well as variable reactivity to CK18 (56% ± 61%) and CK19 (58% ± 60%). Some luminal cells also expressed CK7 (11% ± 4%), while p63 was specifically expressed in the basal cells (100% ± 0%) (Fig. 16). There was no reactivity to SMA detected in the lesions of apocrine ductal adenomas and carcinomas.
Immunohistochemical Features of Apocrine Gland Tumors in Dogs.
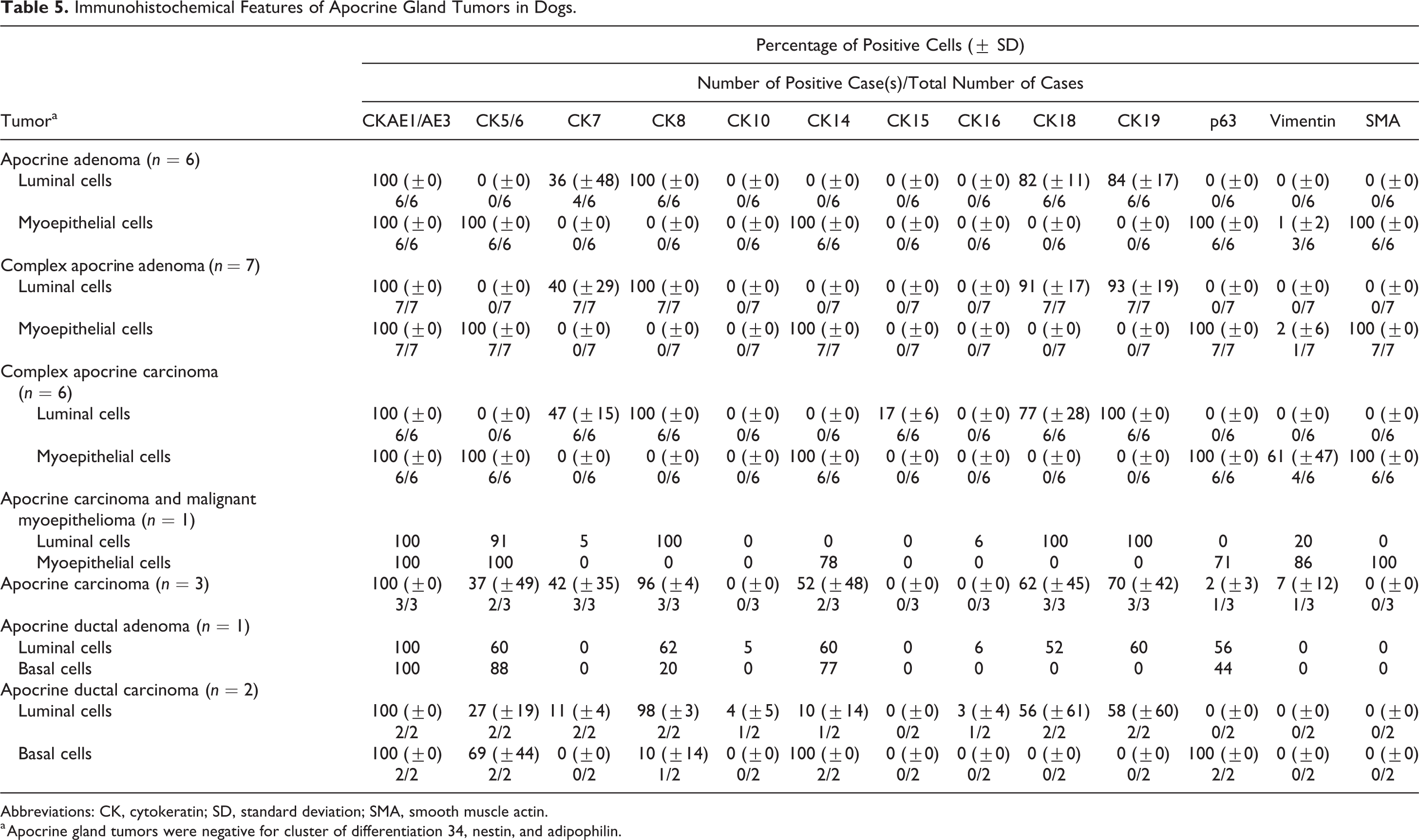
Abbreviations: CK, cytokeratin; SD, standard deviation; SMA, smooth muscle actin.
a Apocrine gland tumors were negative for cluster of differentiation 34, nestin, and adipophilin.
Cutaneous epithelial tumors, dog.
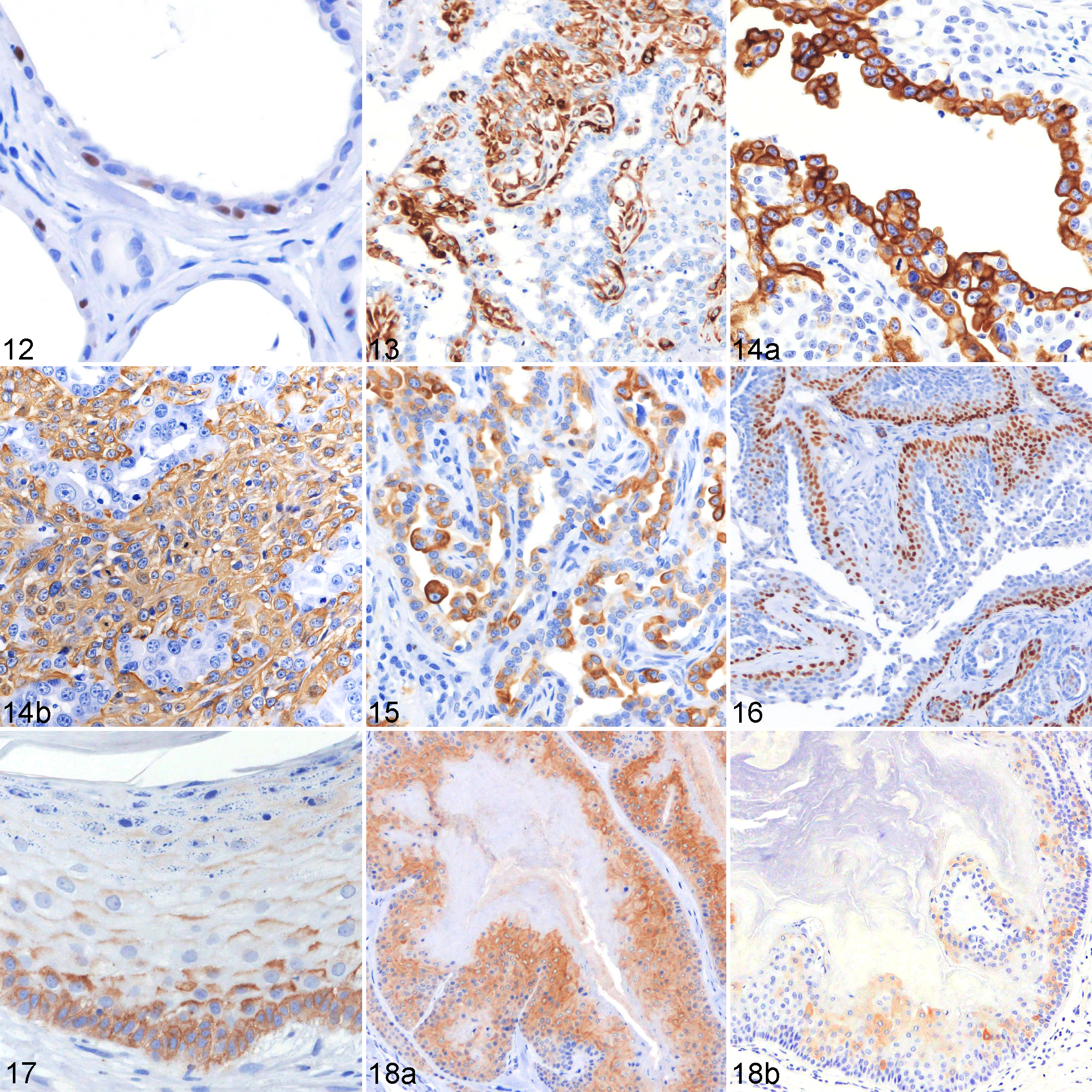
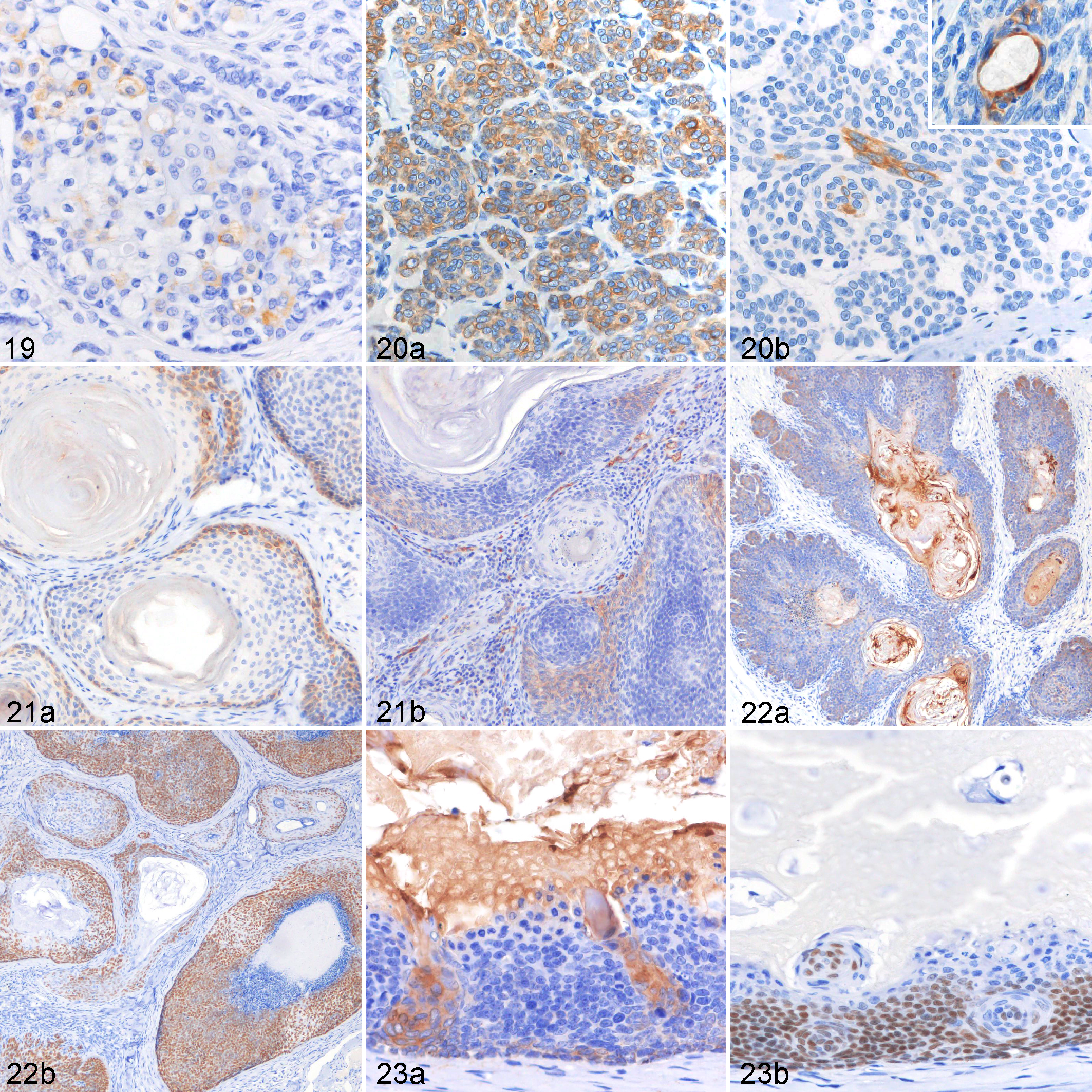
Table 6 summarizes the immunohistochemical profile of hair follicle tumors. All types of tumors except for pilomatricoma could be completely characterized and differentiated based on their immunoreactivity toward the isthmic and inferior stem cell markers (CK8, CK15, CK19, and CD34). In infundibular keratinizing acanthomas, basal cells and cells of the suprabasal layers displayed a membranous to cytoplasmic expression of CD34 (33% ± 23%) (Fig. 17). Isthmic tricholemmoma showed a moderate to strong immunoreactivity toward CK15 (58% ± 30%) (Fig. 18a) and a weak to moderate immunoreactivity toward CK19 (Fig. 18b) and CD34 (11% ± 16% and 8% ± 11%, respectively). Approximately 28% of tumor cells in inferior tricholemmomas expressed CD34. A small number of tumor cells also expressed CK15, CK19, and nestin (mean number of cells: 2%, 6%, and 2%, respectively). In the present study, nestin was detected by immunostaining only in the inferior tricholemmoma (Fig. 19) and was not seen in normal cutaneous epithelial tissues and other types of tumors. Tumor cells of trichoblastomas were positive for CK8 (72% ± 34%) (Fig. 20a), CK15 (33% ± 30%), CK19 (12% ± 12%), and CD34 (73% ± 29%). Spindle-shaped inner cells and cells lining the cystic luminal structure in trichoblastomas expressed CK16 (5% ± 8%) (Fig. 20b). In trichoepitheliomas, a low CK8 (4% ± 8%) and CK15 (10% ± 13%) (Fig. 21a) expression was detected in the basal cells, which displayed a higher immunoreactivity toward CD34 (24% ± 19%) (Fig. 21b). In malignant trichoepitheliomas, the immunoreactivity to CKAE1/AE3 (67% ± 11%) (Fig. 22a) was gradually lost toward the center of the tumor lobules; the tumor cells displayed a diffuse, strong nuclear reactivity to p63 (90% ± 9%) (Fig. 22b). Malignant trichoepithelioma displayed a weak immunoreactivity to all of the stem cell markers (CK8, CK15, CK19, and CD34). In pilomatricoma, only a few areas with transitioning cells displayed immunoreactivity to CKAE1/AE3 (13% ± 14%) (Fig. 23a), CK5/6 (11% ± 4%), CK14 (6% ± 5%), CK16 (5% ± 4%), and CK19 (4% ± 6%), while most tumor cells with matrical differentiation expressed p63 (86% ± 7%) (Fig. 23b).
Immunohistochemical Features of Hair Follicle Tumors in Dogs.
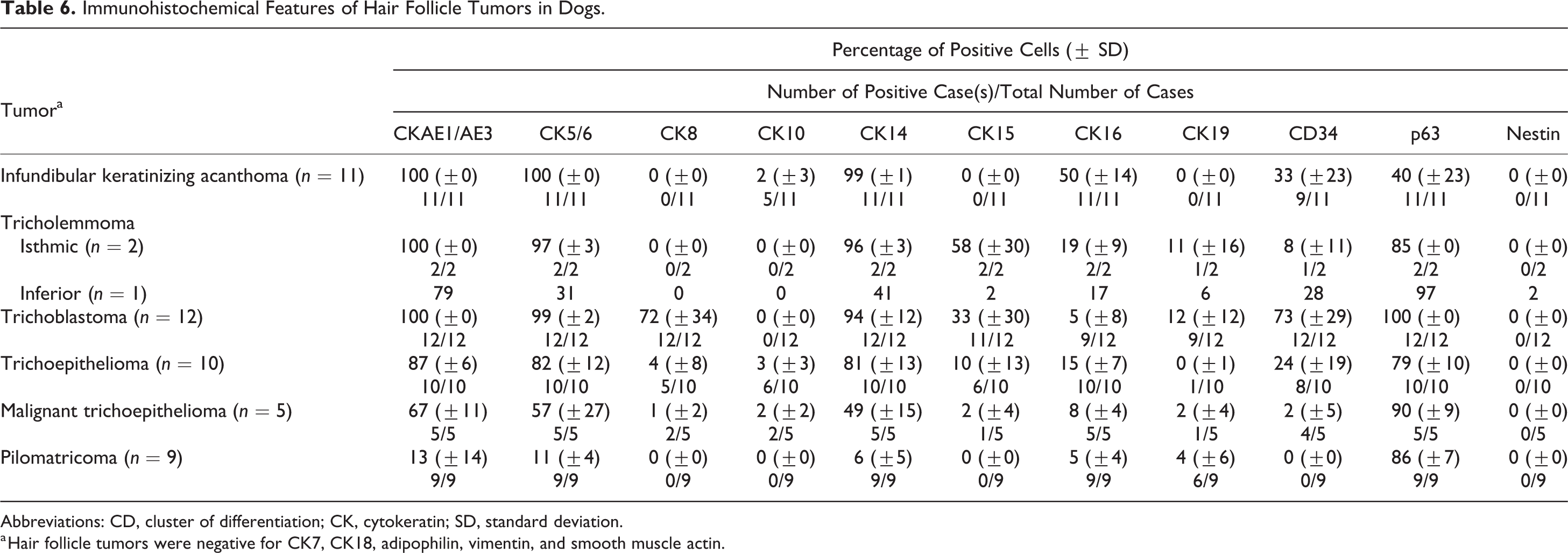
Abbreviations: CD, cluster of differentiation; CK, cytokeratin; SD, standard deviation.
a Hair follicle tumors were negative for CK7, CK18, adipophilin, vimentin, and smooth muscle actin.
Hair follicle tumors, dog.
Cluster Analyses
Figures 24 and 25 represent the dendrograms constructed through a hierarchical clustering approach. Sebaceous gland tumors (cluster A), epidermal tumors (cluster B), and apocrine tumors (cluster C) could be clearly separated into 3 distinct clusters by immunostaining for CK5/6, CK8, CK14, CK16, CK18, CK19, p63, adipophilin, and SMA (Fig. 24). Cluster A consisted of all 3 types of sebaceous tumors. In cluster B, consisting of epidermal tumors, basal cell carcinoma and squamous cell carcinoma were found to be more closely related than acantholytic squamous cell carcinoma. In cluster C, apocrine adenoma was clustered with complex apocrine adenoma, complex apocrine carcinoma, and apocrine carcinoma and malignant myoepithelioma. On the other hand, apocrine carcinoma was closely related to apocrine ductal tumors. Hair follicle tumors were classified into 3 different clusters by using markers such as CK8, CK14, CK15, CK16, CK19, CD34, and p63 (Fig. 25). Inferior tricholemmoma, malignant trichoepithelioma, trichoepithelioma, and pilomatricoma were sorted into cluster D, while cluster E consisted of infundibular keratinizing acanthoma and isthmic tricholemmoma. Trichoblastoma was not sorted with other hair follicular tumor types (cluster F).
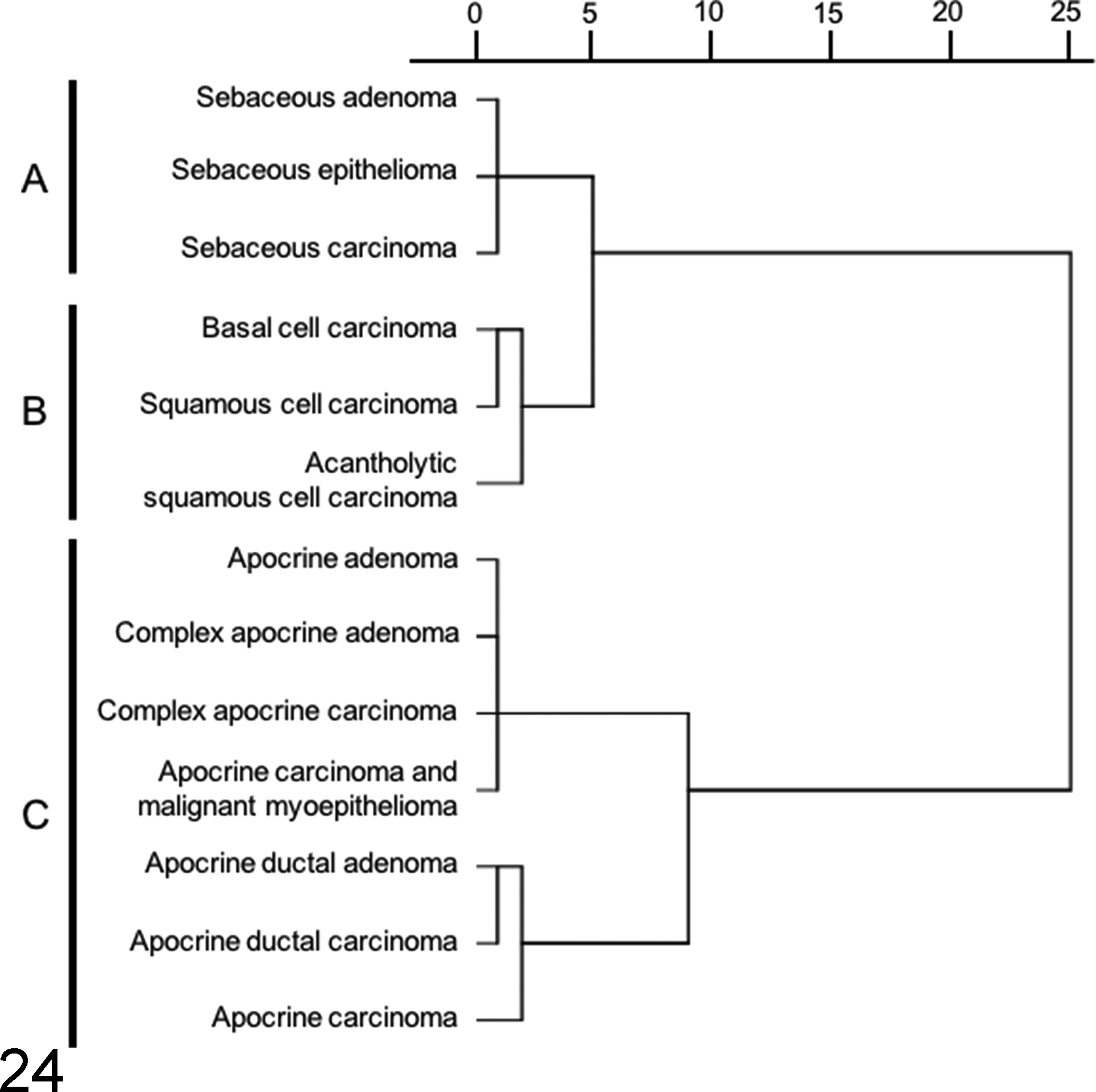
Cluster analysis of cutaneous epithelial tumors (except for hair follicle tumors) based on the results of cytokeratin (CK) 5/6, CK8, CK14, CK16, CK18, CK19, p63, adipophilin, and smooth muscle actin (SMA) immunohistochemistry. A dendrogram constructed using the Ward method demonstrates specific clusters, which are in agreement with their histological differentiation.
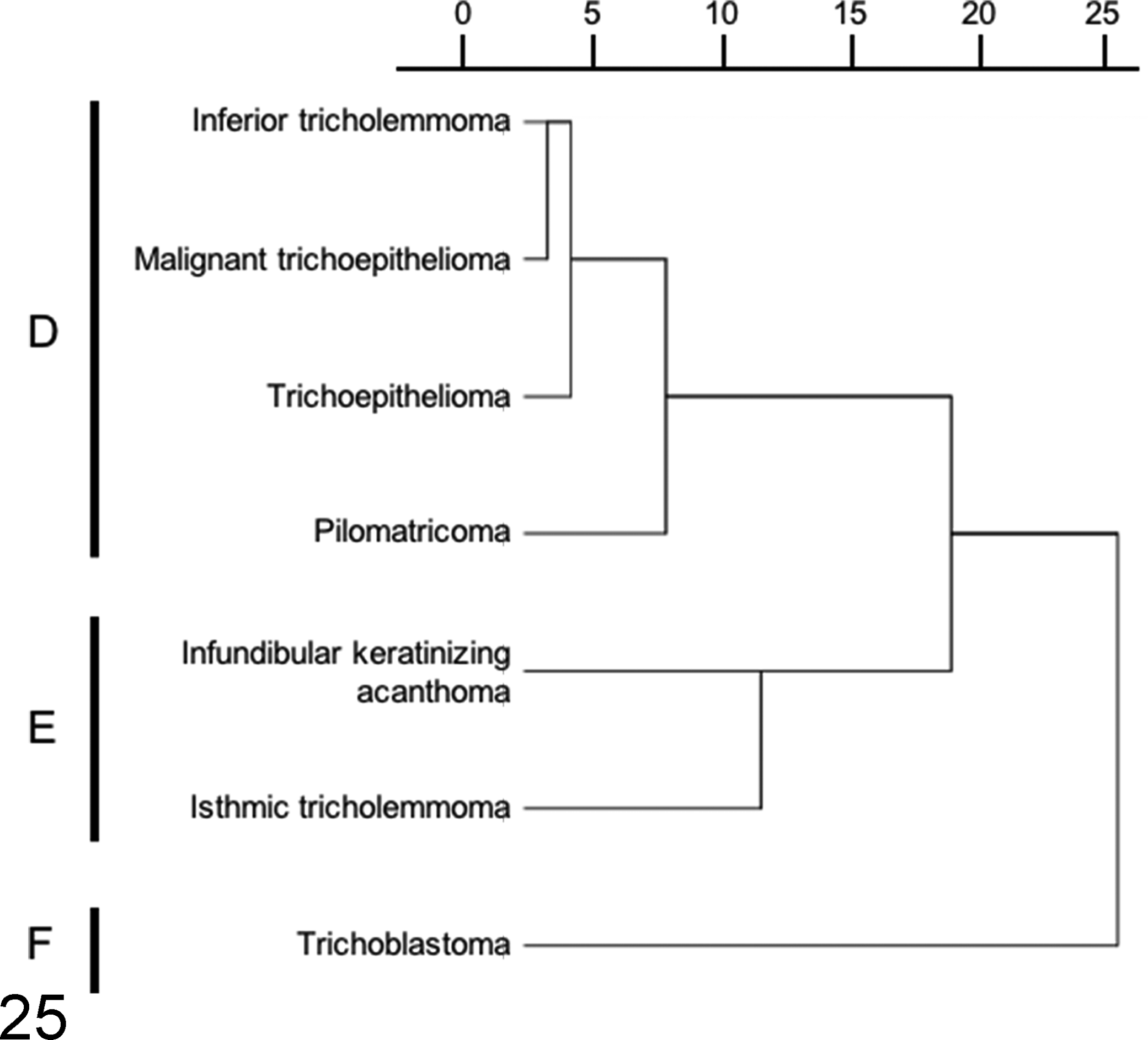
Cluster analysis of hair follicle tumors based on the results of cytokeratin (CK) 8, CK14, CK15, CK16, CK19, CD34, and p63 immunohistochemistry. A dendrogram constructed using the Ward method demonstrates the relationship among hair follicle tumors examined in the present study. Trichoblastoma forms its own cluster.
Discussion
Canine CETs are classified based on their histomorphological differentiation, 11,12 and there is limited complementary IHC information available for justifying the histogenesis of each tumor type. 36 The purpose of the present study was to establish a complete IHC expression profile for canine CETs. The hierarchical clustering results show that by employing suitable markers, immunohistochemistry is useful for the characterization of CETs. In general, tumors in the same cluster share similar IHC profiles and can be differentiated by specific markers. A simplified illustration (Fig. 26) was prepared based on the results of cluster analysis and the IHC expression profiles of the normal skin tissue and CETs, to show the possible tumor origins and markers useful for distinguishing each tumor type. An IHC algorithm for diagnosis of CETs is shown in Fig. 27.
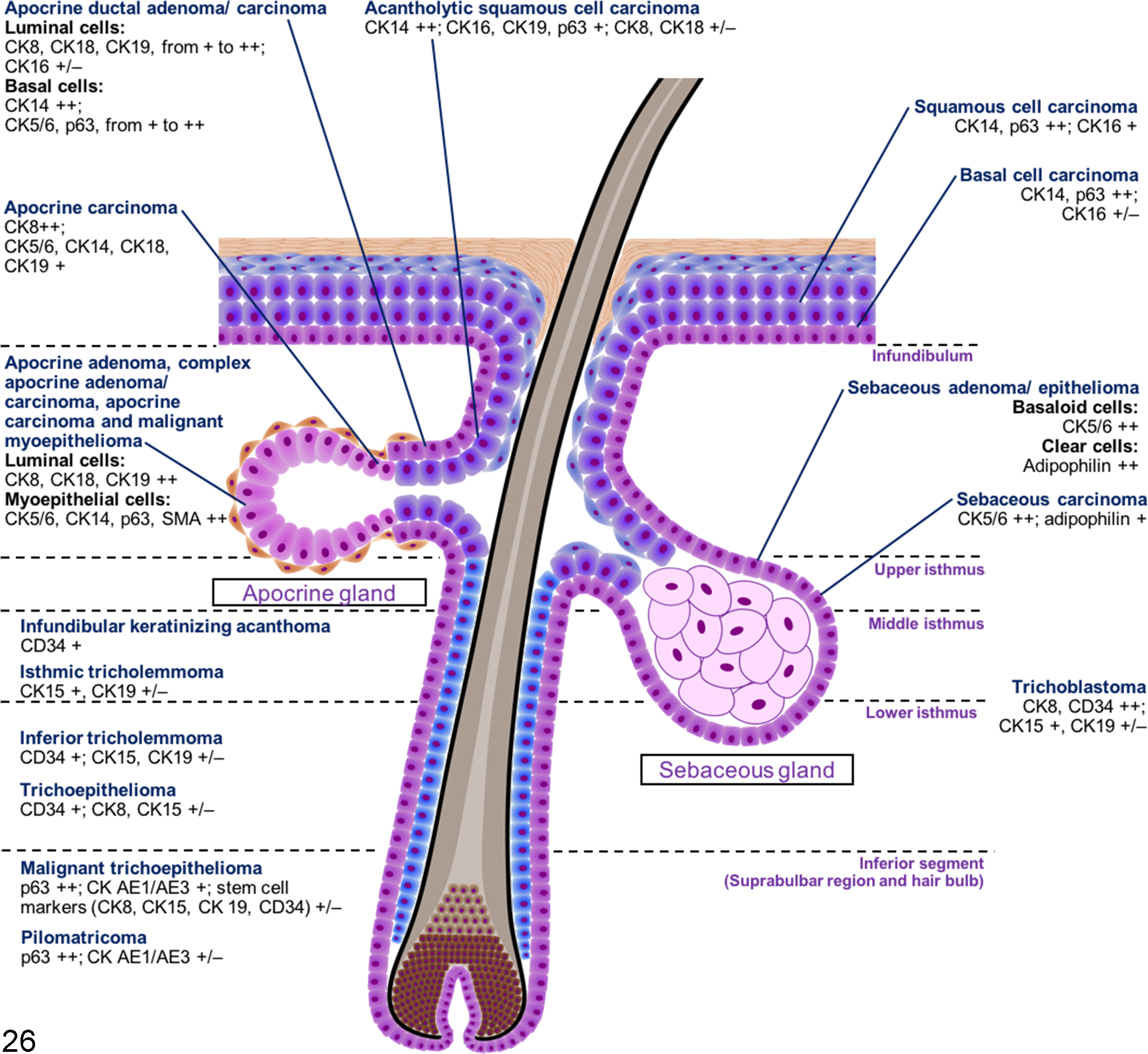
A simplified illustration summarizing the suggested origin and the important immunohistochemical (IHC) markers of each cutaneous epithelial tumor. The hierarchical cluster analysis and a comparison with the immunohistochemical findings of the normal skin tissues suggest that infundibular keratinizing acanthoma and isthmic tricholemmoma potentially originate from the middle isthmus, inferior tricholemmoma and trichoepithelioma from the lower isthmus, and malignant trichoepithelioma and pilomatricoma from the inferior segment of the hair follicle. Trichoblastoma is not grouped into any specific origin. The IHC expression levels are defined as follows: 0% positive cells, negative; 1% to 15% positive cells, weakly positive (+/–); 16% to 70% positive cells, moderately positive (+); and 71% to 100% positive cells, strongly positive (++). CD, cluster of differentiation; CK, cytokeratin.
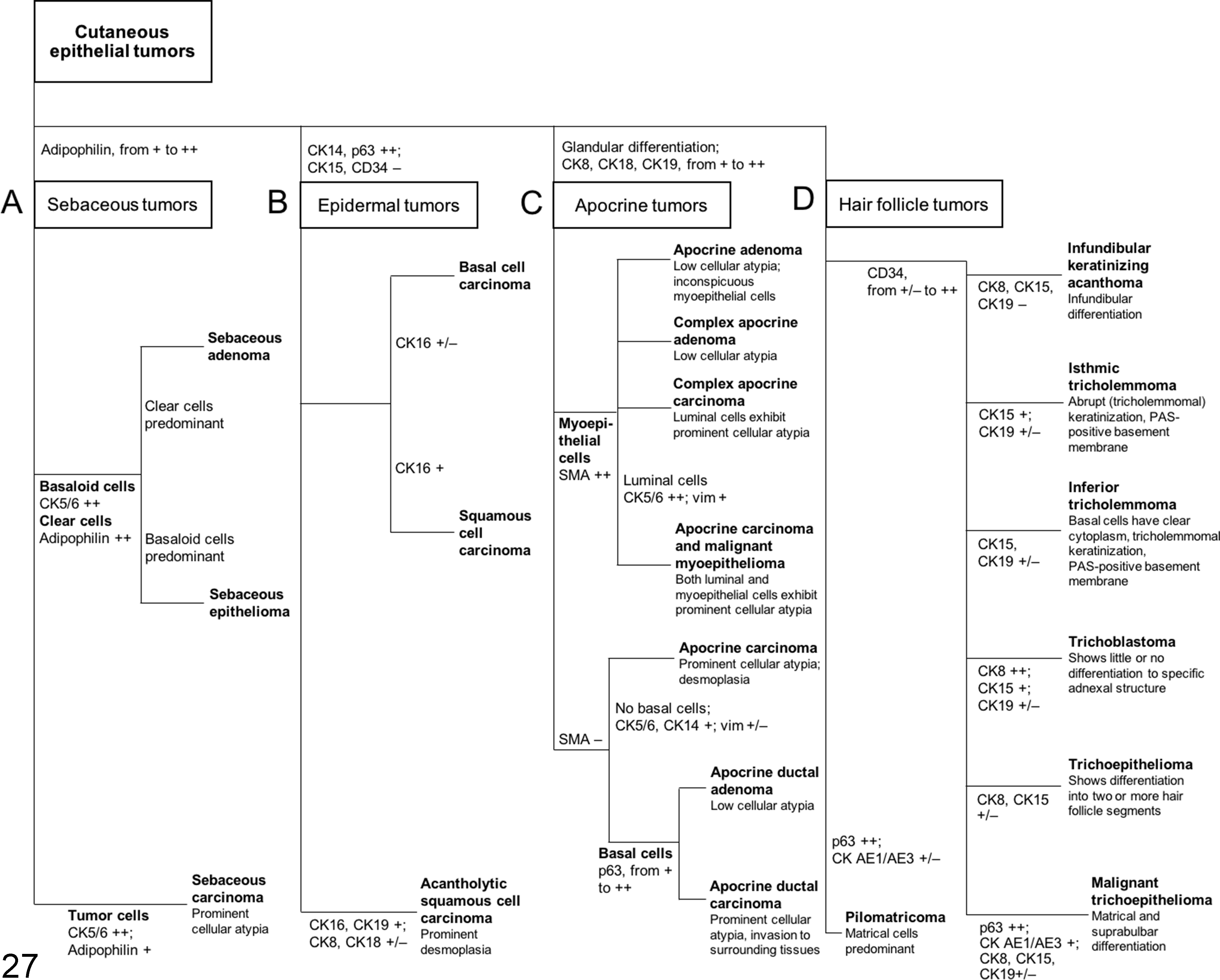
An immunohistochemical (IHC) algorithm to facilitate diagnosis of cutaneous epithelial tumors. Tumors could be generally categorized into 4 groups: sebaceous tumors (group A), epidermal tumors (group B), apocrine tumors (group C), and hair follicle tumors (group D) based on specific markers as shown in the figure. Sebaceous tumors (group A) express adipophilin. Epidermal tumors (group B) are strongly positive for CK14 and p63 but negative for CK15 and CD34. Apocrine tumors (group C) show glandular differentiation and are positive for CK8, CK18, and CK19. Hair follicle tumors (group D), except for pilomatricoma, are positive for CD34. The IHC expression levels are defined as follows: 0% positive cells, negative; 1% to 15% positive cells, weakly positive (+/–); 16% to 70% positive cells, moderately positive (+); and 71% to 100% positive cells, strongly positive (++). CD, cluster of differentiation; CK, cytokeratin; PAS, periodic acid–Schiff; SMA, smooth muscle actin; vim, vimentin.
The CK expression profile for sebaceous gland tumors was not different from that of epidermal tumors (Tables 3 and 4). The complementary use of adipophilin, an intracellular lipid-specific marker, 23,25 allowed this tumor group to be characterized more effectively, as shown in cluster A (Fig. 24). Adipophilin is expressed in a wide range of tissues that contain intracytoplasmic lipid droplets. 13 It is a surface protein found in the lipid droplet and displays a membranous vesicular staining pattern. 23,25 However, a careful evaluation is required as cross-reactivity (granular staining pattern) was often detected with other intracytoplasmic antigens, mainly in the keratohyalin granules of granular cells in the epidermis, squamous cell carcinomas, infundibular keratinizing acanthomas, and trichoepitheliomas. Previous studies with humans had concluded that these were a result of nonspecific staining. 23,25 Adipophilin only helps to reveal the presence of intracellular lipids and is not indicative of differentiation to any cell type. 25 Hence, adipophilin should be used together with CK5/6 (expressed in basaloid cells but not in clear cells of the sebaceous tissue) to confirm sebaceous differentiation. The coexpression of CK5/6 and adipophilin is useful for confirming a diagnosis of sebaceous carcinoma, as some of its tumor cells exhibit an intermediate stage of sebocytic differentiation.
The epidermal tumors included in cluster B (Fig. 24) consisted of basal cell carcinoma, squamous cell carcinoma, and acantholytic squamous cell carcinoma. Hierarchical clustering shows that basal cell carcinoma and squamous cell carcinoma are closely related neoplasms. These tumors differed in the degree of differentiation and predominant cell types, but both displayed diffuse reactivity to the basal cell markers CK14 and p63. The upregulation of CK14 and p63 and the downregulation of CK10, a terminal differentiation marker, 6 indicate an active proliferative state in squamous cell carcinomas. CK16, an alternate suprabasal marker, which was consistently expressed in squamous cell carcinomas, represents the most suitable marker for differentiation in epidermal tumors. Acantholytic squamous cell carcinoma was relatively different from the other 2 tumors (basal cell carcinoma and squamous cell carcinoma) in the same group, and this difference was attributable to the weak expression of CK8 and CK18 and moderate expression of CK19 within the tumor lesion. A previous study had confirmed the coexpression of CK8 and CK18 as a specific feature of the apocrine gland and its associated tumors. 17 Acantholytic squamous cell carcinomas consist of proliferating squamous epithelial cells in a pseudoglandular arrangement, which, in addition to IHC findings (CK8, CK18, and CK19 reactivity) for these carcinomas, suggest that a subset of the acantholytic squamous cell carcinomas, if not all, were of apocrine ductal origin.
Cluster C (Fig. 24) consisted of apocrine tumors. The apocrine adenoma cases were diagnosed based on the single-layered lining of tubular or papillary structure by luminal epithelial cells. CK5/6, CK14, p63, and SMA were positively expressed in some areas of the apocrine adenomas, indicating the presence of myoepithelial cells. However, they were not reclassified as complex apocrine adenoma, as the focal aggregation of myoepithelial cells, which serves as an important histological criterion for complex apocrine tumor, was absent in all of the cases. 11,12 According to the WHO International Histological Classification of Skin Tumors of Domestic Animals, 11 malignant tumors of the apocrine gland were composed of apocrine carcinoma, complex apocrine carcinoma, mixed apocrine carcinomas, and apocrine ductal carcinoma. In both complex and mixed apocrine carcinomas, the myoepithelial component was nonmalignant in nature. In the present study, we described a new CET type, apocrine carcinoma and malignant myoepithelioma, the nomenclature for which was adopted from the recent study on the classification of canine mammary tumors. 10 The diagnosis was based on the presence of a malignant myoepithelial component in the complex apocrine carcinoma, as confirmed by SMA immunoreactivity. To our knowledge, the same tumor entity had also been described in a case report. 33 Hierarchical cluster analysis showed that apocrine carcinoma and malignant myoepithelioma were closely sorted with apocrine adenoma, complex apocrine adenoma, and complex apocrine carcinoma, mainly due to the presence of the myoepithelial component in these tumor types. The aberrant expression of CK15 in luminal cells was observed in complex apocrine carcinomas. In humans, CK15 reactivity was reported in apocrine mixed tumors, which sometimes exhibit follicular (hair follicle) or sebaceous differentiation. 19 Misago and Narisawa 19 suggest that the human apocrine mixed tumor might originate from multipotent stem cells in the hair follicular bulge. However, the implication of CK15 reactivity in canine complex apocrine carcinoma is still unknown. Previous immunohistochemical studies have shown conflicting results on vimentin expression in the myoepithelial component of the normal apocrine gland. 8,27 Similar to the study by Ferrer et al, 8 we did not observe any positivity for vimentin in the myoepithelial cells. On the other hand, vimentin was found to be expressed in the myoepithelial component of apocrine adenoma, complex apocrine adenoma, complex apocrine carcinoma, and apocrine carcinoma and malignant myoepithelioma. These findings may be attributed to the acquisition of vimentin intermediate filament, which is closely related to the myofilaments, in active proliferating myoepithelial cells. 3,8,28 Some luminal cells of apocrine carcinoma and malignant myoepithelioma and apocrine carcinoma were vimentin positive. These cells exhibited a high degree of cellular atypia and invaded the surrounding stroma, suggesting that the increased malignancy occurred as a result of epithelial-mesenchymal transitions. 14 The apocrine carcinoma was more closely related to apocrine ductal tumors, not only because of the absence of the myoepithelial component but also due to aberrant CK5/6 and CK14 expression, observed in the luminal cells. The expression of these markers was not seen in the luminal cells of complex apocrine carcinomas, with the exception of apocrine carcinoma and malignant myoepithelioma, which expressed CK5/6. A portion of the luminal component of apocrine ductal tumors expressed CK5/6 and CK14, which was also seen in the junction between the apocrine duct and the hair follicle infundibulum. Immunoreactivity for p63, as observed in some luminal cells of apocrine ductal adenoma, was attributed to the gradual squamous differentiation of the ductal epithelium. In the apocrine tumor group, the concurrent use of p63 and SMA is useful for distinguishing between myoepithelial cells (p63 and SMA positive) and basal cells (p63 positive and SMA negative).
In a separate hierarchical clustering (Fig. 25) event, hair follicle tumors were sorted into 3 different clusters. The IHC expression profiles of the normal hair follicle and their tumorous counterparts indicate that hair follicle tumors were sorted according to their origin. Cluster D (inferior tricholemmoma, malignant trichoepithelioma, trichoepithelioma, and pilomatricoma) consists of a group of tumors potentially originating from the inferior (bulb and suprabulbar region) or lower isthmic segment of the hair follicle. In this cluster, inferior tricholemmoma (CD34 positive, CK15 and CK19 weakly positive) and trichoepithelioma (CD34 positive, CK8 and CK15 weakly positive) may have arisen from the lower isthmus (CK8, CK15, and CD34 positive) of the hair follicle. On the other hand, malignant trichoepitheliomas and pilomatricomas exhibited low CK and high p63 expression levels and were associated with the inferior segment (suprabulbar and hair bulb regions). Cluster E (infundibular keratinizing acanthoma and isthmic tricholemmoma) consists of a group of tumors potentially originating from the middle isthmic segment. The expression of CK15 and CD34 has been observed in the normal middle portion of the isthmus. Infundibular keratinizing acanthomas expressed CD34, while isthmic tricholemmomas mainly expressed CK15. Both tumor types had similar histological structures and showed a progressive differentiation toward the upper hair follicle segment. Their differences, apart from histomorphology, were attributable to the presence of a PAS-positive basement membrane (as seen in isthmic tricholemmoma) and the expression profiles of CK15 and CD34. Trichoblastomas were not sorted into any of the 2 groups, and they formed cluster F. These tumors, which are predominantly composed of undifferentiated, germinative epithelial cells, expressed CK8, CK15, CK19, and CD34. The expression of significant percentages of these stem cell markers in comparison to their expression in other hair follicle tumors prevented the inclusion of trichoblastomas in any of the 2 clusters. The inner, spindle-shaped cells of trichoblastomas and the cells lining the cystic structures expressed CK16. This shows that despite a predominant germinative cell population, a portion of the tumor cells was differentiating into the inner structure of the hair follicle in a restricted pattern. It has been suggested that CK8, CK15, and CD34 are useful hair follicle stem cell markers in canine species. 9,17 In addition to these markers, the present study showed that CK19 was also useful in identifying stem cells located in the suprabulbar region of the anagen hair follicle. The difference in the stem cell expression profiles of hair follicle tumors suggests that their terminal differentiation is dependent on the type of stem cells involved in tumorigenesis. Previous studies had demonstrated that nestin was expressed in the isthmus segment of the hair follicle 18 and some types of hair follicle tumors, 5 while another study showed the absence of nestin reactivity in cutaneous epithelial tissues. 9 The present study is in agreement with the latter, 9 as only inferior tricholemmoma showed reactivity to nestin among all the epithelial tissues examined.
In the present study, canine CETs could be classified based on their IHC expression profile. Due to the low incidence, some of the tumor types (eg, basal cell carcinoma, apocrine ductal adenoma, inferior tricholemmoma, and isthmic tricholemmoma) are underrepresented, and further studies with larger sample sizes are required to reaffirm the results of the hierarchical cluster analysis. In tumors that clearly exhibited different components (eg, benign sebaceous gland tumors and some apocrine gland tumors), the IHC expression of each cellular phenotype was examined. The present results may be used in future studies to immunohistochemically evaluate the proportion of each cellular component in certain CETs. In particular, we believe that examining the proportion of the myoepithelial cells in complex apocrine tumors is prognostically important, as they have been known to play a role in the suppression of tumor invasion and metastasis. 1,35
Canine CETs, particularly those of hair follicular origin, show a wide range of histomorphological differentiation. The diverse histological types may be attributed to the presence of a heterogenous stem cell population in the skin, which constantly undergoes self-renewal to maintain the integrity of the cutaneous adnexa. 4,34 Previous murine studies had shown that cutaneous epithelial stem cells were the primary targets of cutaneous carcinogens and are responsible for CET formation. 21,22 In dogs, we postulate that tumorigenesis of sporadic CETs may occur, at least partially if not wholly, as a result of an impairment in the process of regeneration of epithelial stem cells. Further investigations are needed to support this hypothesis.
Conclusion
The panel of IHC markers used in the present study allowed a better characterization of canine CETs and helped in understanding the origin and differentiation of each tumor type. The IHC expression profiles of both normal skin and tumor cells may serve as reliable references for CET classification and research in the future.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818785680 - Hierarchical Cluster Analysis of Cytokeratins and Stem Cell Expression Profiles of Canine Cutaneous Epithelial Tumors
Supplemental Material, DS1_VET_10.1177_0300985818785680 for Hierarchical Cluster Analysis of Cytokeratins and Stem Cell Expression Profiles of Canine Cutaneous Epithelial Tumors by Mun Keong Kok, James K. Chambers, Siew Mei Ong, Hiroyuki Nakayama, and Kazuyuki Uchida in Veterinary Pathology
Footnotes
Acknowledgement
The authors are grateful to Ms. S. Kato for her invaluable technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
