Abstract
This study investigated the susceptibility of American singer canaries (Serinus canaria) to West Nile virus (WNV) infection. Adult canaries were inoculated with 105, 102, and 101 plaque forming units (PFU) of WNV. All birds became infected and mortality occurred by 5 days postinoculation. The load of viral RNA as determined by RT-qPCR was dose dependent, and was higher at all doses than the level of viral RNA detected in American crows (Corvus brachyrhynchos) challenged with 105 PFU of WNV. In a subset of birds, viremia was detected by virus isolation; canaries inoculated with 101 PFU of WNV developed viremia exceeding 1010 PFU/mL serum, a log higher than American crows inoculated with 105 PFU of virus. In canaries euthanized at 3 days postinoculation, WNV was isolated at >107 PFU of virus/100 mg of lung, liver, heart, spleen, and kidney tissues. Pallor of the liver and splenomegaly were the most common macroscopic observations and histologic lesions were most severe in liver, spleen, and kidney, particularly in canaries challenged with 102 and 101 PFU. Immunoreactivity to WNV was pronounced in the liver and spleen. IgG antibodies to WNV were detected in serum by enzyme immunoassay in 11 of 21 (52%) challenged canaries and, in 4 of 5 (20%) of these sera, neutralization antibodies were detected at a titer ≥ 1:20. American singer canaries provide a useful model as this bird species is highly susceptible to WNV infection.
The arrival of West Nile virus (WNV) in North America in 1999 in the New York City area was in part detected by morbidity in free-ranging birds, particularly corvids, 13 and captive exotic birds at the Bronx zoo. 16 Early field observations 1,9 and later experimental studies 11,14,18 confirmed the high WNV susceptibility of the corvid family, a member of Passeriformes that includes crows, magpies, and jays. A bird species’ susceptibility to WNV might be measured by the role that species plays in amplification of the virus and transmission of the virus to feeding mosquitoes, thus perpetuating the enzootic cycle of transmission. Alternatively, a bird’s susceptibility might reflect the role the virus plays in the bird’s or the species’ survival and may be considered in the context of conservation. In this context, American crows are acutely affected by WNV, dying within 5–6 days in experimental challenges.
The susceptibility of corvids to WNV has been evaluated through capture and experimental challenge of wild-caught corvids. 11,14,18 The use of wild-caught birds in infectious disease challenges may be compromised by concurrent infection with infectious and parasitic pathogens. In addition, wild-caught birds brought into captivity demonstrate a stress response that diminishes only slowly. 7 The stress response is likely accompanied by an increase in release of corticosterone which, if prolonged, can have a number of negative effects on the innate and acquired immune response. 6,15 As part of a study using domesticated passerines to determine the effect of previous or concurrent infection with Plasmodium relictum on subsequent infection with WNV, our objective was to determine the susceptibility of domesticated canaries challenged with WNV and to compare the clinical and pathological outcome to a group of American crows experimentally infected with WNV at the National Wildlife Health Center.
Materials and Methods
WNV Challenge
Adult canaries (Serinus canaria) were obtained from the Rockefeller University breeding colony and American crows (Corvus brachyrhynchos) were captured by USDA Animal Services staff using rocket propelled nets. On arrival at the USGS National Wildlife Health Center (NWHC) all birds received a brief physical examination, were marked by a unique leg band and were housed in a BSL3 animal room. Canaries were housed in wire cages measuring approximately 60 cm (long) × 40 cm (wide) × 40 cm (height), in groups of 5–7 birds of the same sex and were fed a complete finch diet (Vita Finch®, Vitakraft, Bowling Green, OH) with water supplied ad libitum. Crows were permitted free flight within a BSL3 animal room and were fed moistened dog food supplemented with boiled eggs. All birds were acclimated to the animal room and daily husbandry for at least 14 days prior to experimental challenge. Capture, shipment, husbandry, and experimental protocols were approved by the NWHC Institutional Animal Care and Use Committee. Crows were captured under a US Fish and Wildlife Permit and a WI Department of Natural Resources Scientific Collector’s Permit.
Prior to experimental WNV challenge, canaries were moved to a separate animal room for 3–7 days. A 150 μl blood sample was obtained from the jugular vein of each bird and added to a sufficient quantity of BA1 viral transport medium 10 for a final serum dilution of 1:5, assuming a 50:50 ratio of red blood cells to serum in whole blood samples. The blood was allowed to clot on wet ice, centrifuged at 2000 × g, and stored at –80°C until testing. In the first of 3 canary challenge studies, 10 birds were inoculated subcutaneously with 100 μl of BA1 diluent containing 105 plaque forming units (PFU) of a low passage of a 1999 American crow isolate of WNV (NWHC 16399-3). In the second study, 5 birds in 2 groups were challenged with 102 and 101 PFU, respectively, of the same virus isolate by the same method. A third study was conducted in which 3 birds were challenged with 101 PFU of virus and were euthanized 3 days postinfection (dpi) for collection of tissues for virus quantitation and histopathology. For the first 2 studies, 2 control birds were inoculated subcutaneously with 100 μl of BA1 and were placed on the same sampling schedule. To comply with limitations on the volume of blood that could be collected each day, half of the birds were sampled on days 1, 3, 5 dpi and the remaining birds sampled on days 2, 4. On each sampling day an oropharyngeal swab was obtained from each bird and all birds were weighed daily on an electronic balance. Swabs were placed in 1 ml of BA1, held briefly on wet ice, and stored until viral culture at –80°C.
Following challenge, the birds were observed at least twice daily and birds were euthanized by inhalation of CO2 gas if they were unable to perch or obtain feed or water or were severely dyspneic. All birds were examined at necropsy with collection of skin, lung, liver, heart, spleen, kidney and cerebrum separately in buffered formalin and 1 ml of BA1 medium.
Five crows were challenged subcutaneously with 105 PFU of the same passage of WNV. Sera and oral and cloacal swabs were obtained daily for detection of WNV. All birds that had not been euthanized earlier due to the severity of clinical signs were euthanized on 6 dpi and examined at necropsy. All WNV challenge titers were verified by titration in Vero cells.
Detection of WNV
For all studies, RNA was extracted from 140 µL of diluted serum in BA1 medium and WNV was amplified from 2.5 µL of extracted RNA using the WNV-ENV forward and WNV-ENV reverse primers, as described. 12 Amplified RNA was detected using a FAM-TAMRA probe (Integrated DNA Technologies, Coralville, IA) by an ABI Prism 7000 Real Time PCR Instrument (Applied Biosystems, Foster City, CA), as described. 10 Negative controls (dH2O) were included in each group of samples for RNA extraction and each group of extracts for RT-PCR. Each sample’s cycle threshold (Ct) was referred to a standard curve calculated from a 10-fold dilution series containing 1.2 × 107 – 1.2 × 10-1 genome equivalents (GE) of WNV/ml created from aliquots of RNA frozen at -80°C. These standards were assigned arbitrary units (AU) from 9 to 1. 10 In addition to RT-PCR detection of WNV, canary sera from the third study and crow sera were cultured on Vero cells grown on 6-well plates with plaques quantified using a single overlay method. 3 Plates were examined at 72–108 hours and plaque counts were recorded. Tissue samples in BA1 medium were disrupted with 2.4 mm zirconia beads in a mini bead beater (BioSpec Products Inc, Bartlesville, OK) and, along with oral swabs, were cultured on Vero cells with a single overlay.
Histopathology
Tissue samples in 10% formalin were embedded, sectioned, and stained with hematoxylin and eosin stain using standard methods. Slides were also processed for immunohistochemistry on a Dako autostainer using a peroxidase based polymer system (EnVision-HRP, DAKO, Carpinteria, CA) using standard methods. 4,5 WNV was detected using monoclonal anti-E protein antibody 7H2 (# 81-002, BioReliance, Rockville, MD) and bound antibody was detected with horseradish peroxidase conjugated anti-mouse IgG (EnVision+ System, DAKO) and chromogen (AEC+ Substrate, DAKO). Negative control slides were incubated with anti-WNV E protein and negative control anti-mouse antibody (# NC490, Biocare Medical, Concord, CA) as the primary antibodies. Stained sections were observed at 400x and the images photographed.
Detection of Antibodies to WNV
All serum samples were screened for antibodies to WNV using by the anti-wild bird IgG enzyme immunoassay (WNV IgG EIA), 8 with minor changes, as described. 10 Bound IgY antibodies were detected with horseradish peroxidase labeled polyclonal goat anti-wild bird IgG at a dilution of 1:2000 in wash buffer (# A140-110P, Bethyl Laboratories, Montgomery, TX). Samples in which the ratio of mean test serum optical density (OD) to mean negative control serum OD (P/N), recorded on wells containing WNV antigen, was ≥ 2 were considered positive for anti-WNV antibodies, as described. 8 When sufficient sera were available, samples with a positive result on the screening assay were tested for virus neutralizing antibodies by an enzyme-linked immunosorbent assay (EIA) format microneutralization test, 17 with minor modifications. In the microneutralization test, the virus control was diluted to 5 × 105 PFU/ml in M199 media. The serum and virus samples were incubated for 1 hour at 37°C in 5% CO2 in a total volume of 50 µL. In the EIA, 50 µL/well of West Nile MHIAF polyclonal antibody (FCM30200-06 -1) obtained from the Centers for Disease Control and Prevention was diluted 1:2000 in blocking buffer composed of PBS Tween 20 with 5% nonfat dry milk. Bound antibody was detected by goat anti-mouse IgG HRP conjugate (Bethyl Laboratories, Inc) diluted 1:2000 in blocking buffer, and applied at 50 µL/well. For color development, 50µL/well of substrate (ABTS® 2 component, Kirkegaard and Perry Laboratories, Inc., Gaithersburg, MD) was added, incubated at room temperature for 30 minutes, and stopped with 1% SDS. The optical density was read at 405 nm by a microtiter plate reader. As positive control, a serum pool from chukar partridge (Alectoris chukar), experimentally infected with the same WNV isolate and with neutralization titer established by the plaque reduction neutralization test (PRNT) on 6 well plates coated with Vero cells, 3 was included with dilutions representing neutralizing titers from 1:20 to 1:1280. Negative control serum was also a serum pool from chukar partridge confirmed negative by PRNT. A virus neutralization of ≤ the mean optical density of the virus control wells – 2 SD was considered positive for neutralizing antibodies to WNV.
Statistical Analysis
Survival curves were plotted and were compared using the Log-Rank and the Gehan-Breslow Wilcoxon tests (GraphPad Software, La Jolla, CA). For each RT-PCR run, the linear relationship between the Ct values, as dependent variable, and the standard AU, as predictors, was determined (GraphPad Prism 5.00 software). Using the linear relationship, for each serum sample, the AU was calculated from the sample’s Ct and the mean and SE were plotted using the ggplot2 R package. 19 Means, medians, and 95% confidence intervals for proportions, and the Kruskal-Wallis and Mann-Whitney tests were calculated using GraphPad Software.
Results
Survival
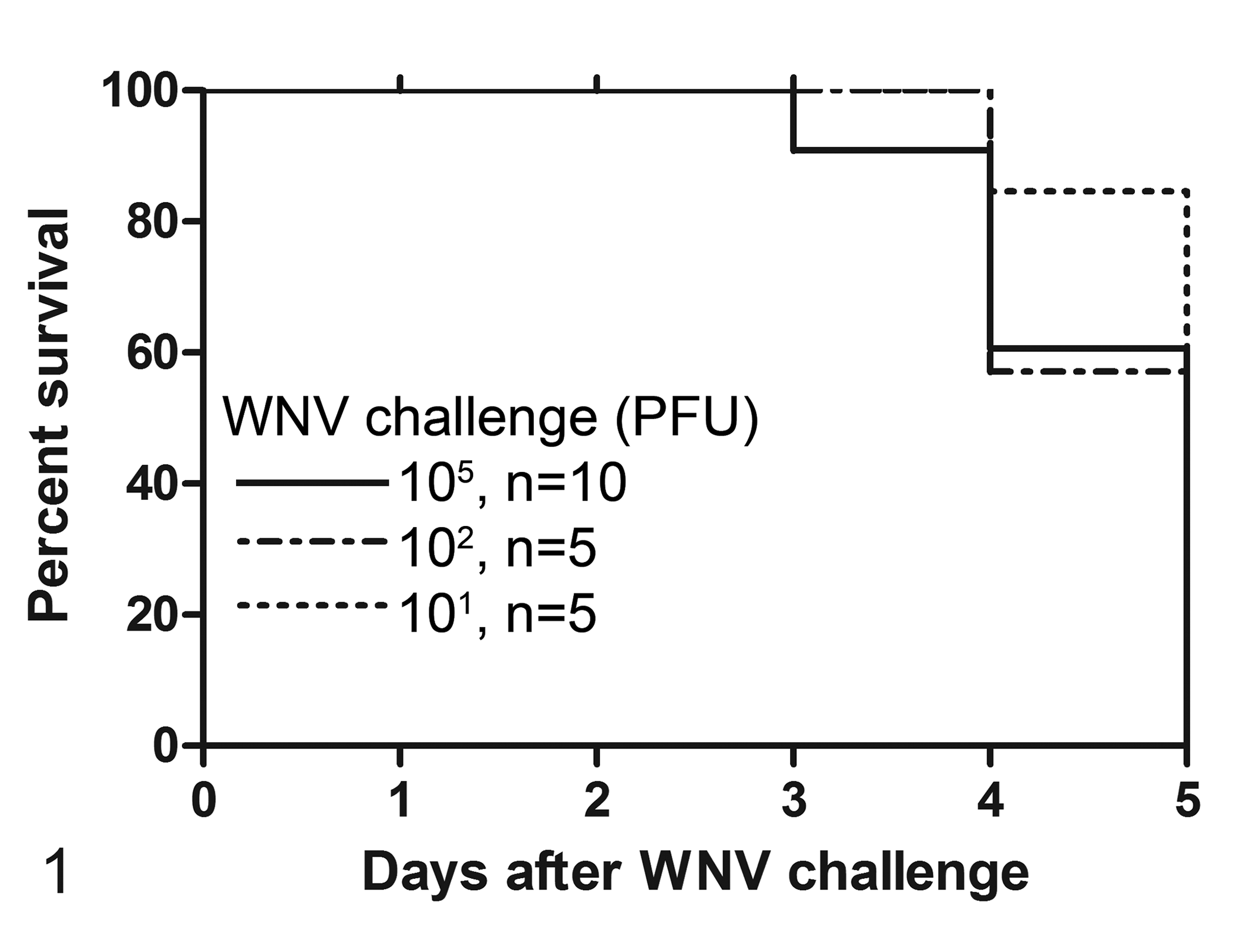
All canaries in the first and second challenge studies developed severe clinical signs including inactivity with fluffed feathers, the inability to stand or perch, and dyspnea and, depending on the severity of signs, were euthanized or were found dead by 5 dpi. Overall, the survival curves (Fig. 1) did not differ statistically; however, survival of canaries challenged with 105 PFU of virus was marginally shorter than birds challenged with 101 PFU of virus (Gehan Breslow Wilcoxon χ2 = 2.98, P = .08). Median survival for all challenge groups was 5 days. In comparison, median survival of canaries challenged with 105 PFU of WNV (5 days) was 1 day shorter than crows and the survival curves were significantly different as assessed by both the Gehan Breslow Wilcoxon χ2 = 8.8, P < .01 and the log rank χ2 = 11.9, P < .01 (Fig. S1).
Virus Detection
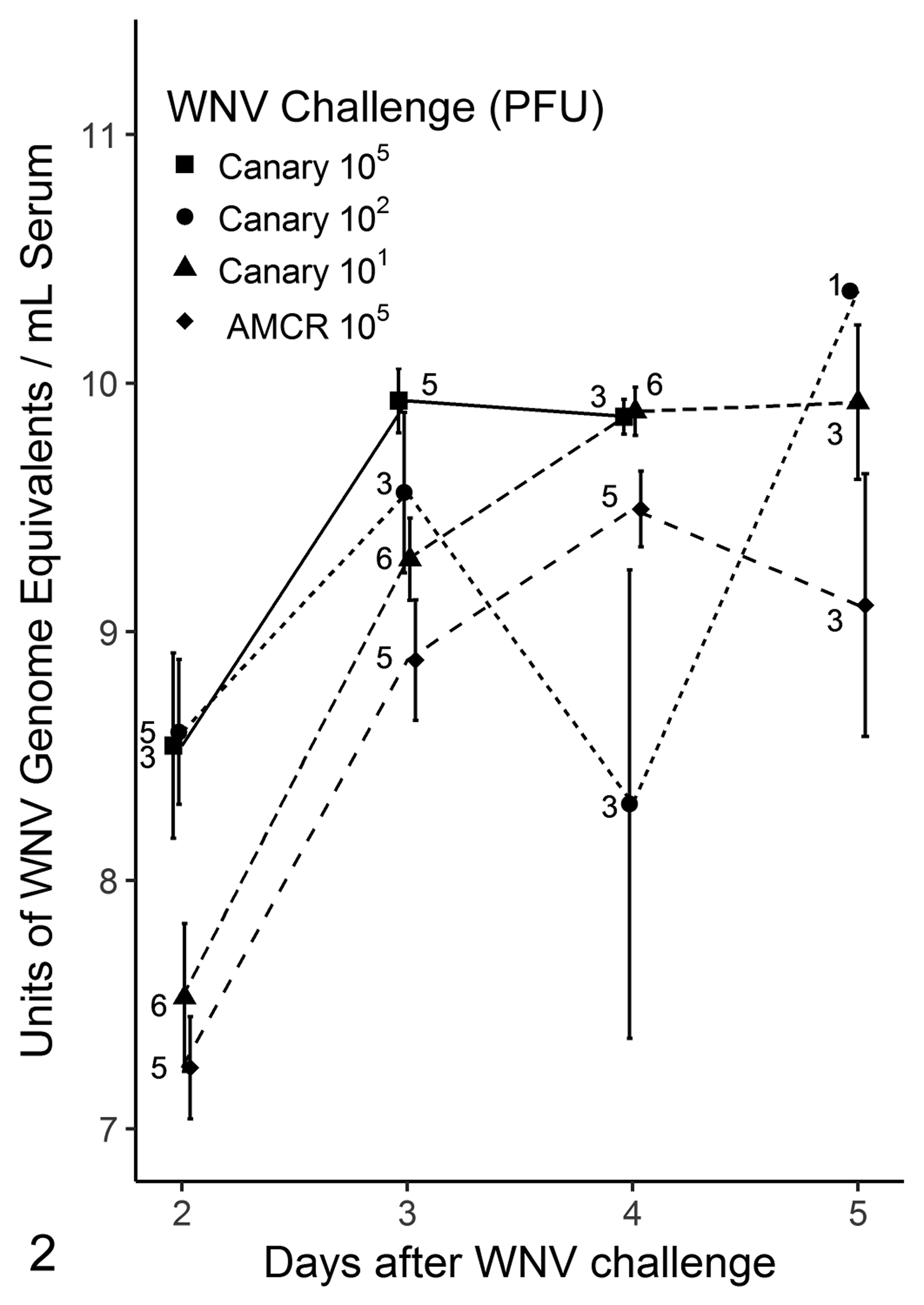
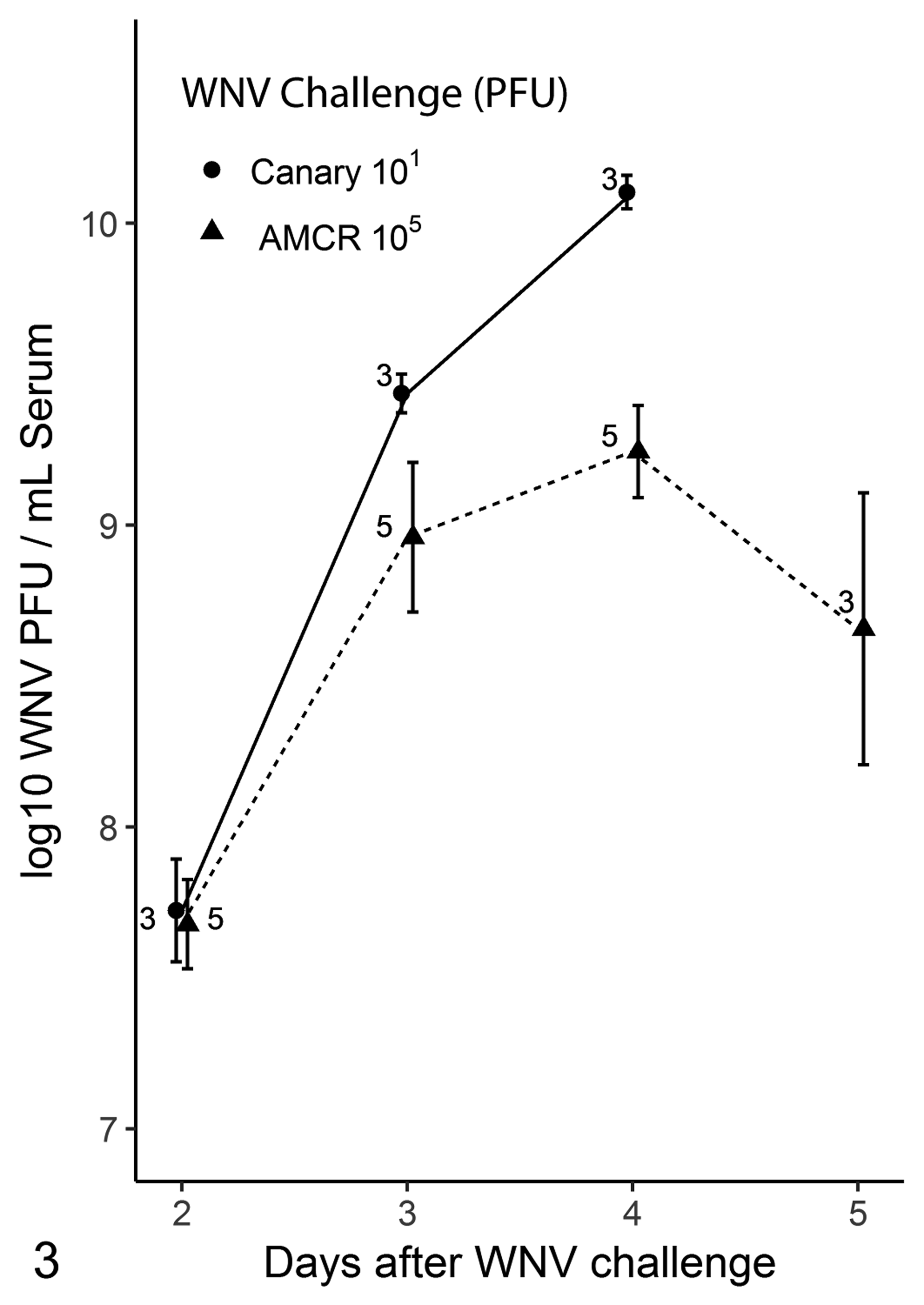
By RT-PCR, WNV was detected in all samples collected from all challenged canaries and crows. On day 2 and 3 dpi the AU of WNV RNA detection generally followed a dose dependency and were all higher in canaries than in crows (Fig. 2). Following 3 dpi the number of birds sampled at each time point was smaller due to birds dying or being euthanized and the mean AU had a larger variability. On days 3 and 4 dpi of the third trial, the quantity of virus isolated from serum from canaries (n = 3) was higher than that from crows (n = 5) (Fig. 3). Overall, comparing log10 PFU/ml, canaries had a significantly higher virus burden than crows (P = .002, Kruskal-Wallis test). However, the log10 PFU/ml virus burden was significantly higher in canaries only on 4 dpi (P = .03, Mann-Whitney test).
WNV was isolated from all oral swabs from canaries on 2-4 dpi at the 105 PFU challenge level, with ranging from 102.5 to 106.6 PFU per swab. In the 101 PFU challenge of canaries, virus was isolated from swab samples obtained from 1/3 birds at 1 dpi and thereafter in all cultured swabs at 103 to 105.4 PFU per swab. Similar levels of virus were isolated from birds inoculated with 102 PFU of WNV. Among crows, WNV was isolated from the oral swabs of 3 of 5 birds on 2 dpi with 101.7 to 102.1 PFU per swab, with increased amount of virus isolated each day; by 5 dpi, a range of 105.1 to 106.3 PFU per swab was detected in 4 crows.
In the third trial, in which canaries were challenged with 101 PFU of virus, WNV was cultured from all tissues from 3 birds sacrificed at 3 dpi (Table 1). The mean quantity of virus cultured from all tissues was uniformly > 106 PFU per 100 mg of tissue and did not differ significantly among tissues (Kruskal-Wallis statistic 4.55, P = .60). Because these birds received the lowest WNV challenge, the WNV tissue burden in birds of the other challenge groups may have been higher. Tissues were not available from crows for viral culture.
Isolation of West Nile Virus From Tissues of Canaries at 3 Days After Experimental Infection With 101 PFU of Virus.
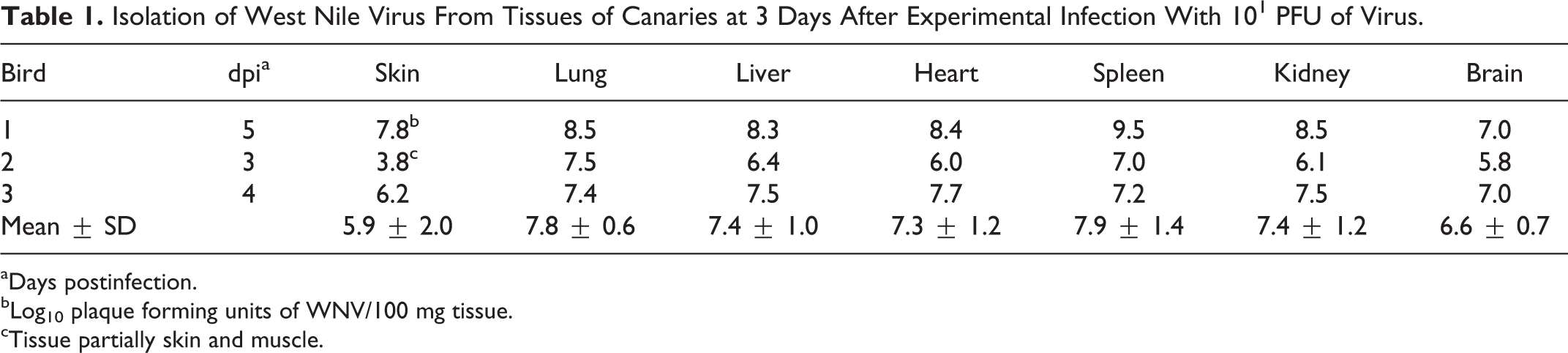
aDays postinfection.
bLog10 plaque forming units of WNV/100 mg tissue.
cTissue partially skin and muscle.
Anti-WNV Antibody Detection
Using the IgG EIA for antibodies to WNV, a P/N ratio of >2 was detected in 11/21 birds with sufficient sera for testing (52%, CI: 32–72%). P/N ratios ranged from 2.2 to 9.6 with a median of 3.4 and were detected as early as 2 dpi. Sufficient sera remained to perform the microneutralization test on 5 samples with P/N ≥ 2 on the screening EIA. Four of these samples were positive for neutralizing antibodies with titers ≥ 1:20. The microneutralization EIA detected neutralizing antibodies in the positive control sera with endpoint titer agreeing with the PRNT result.
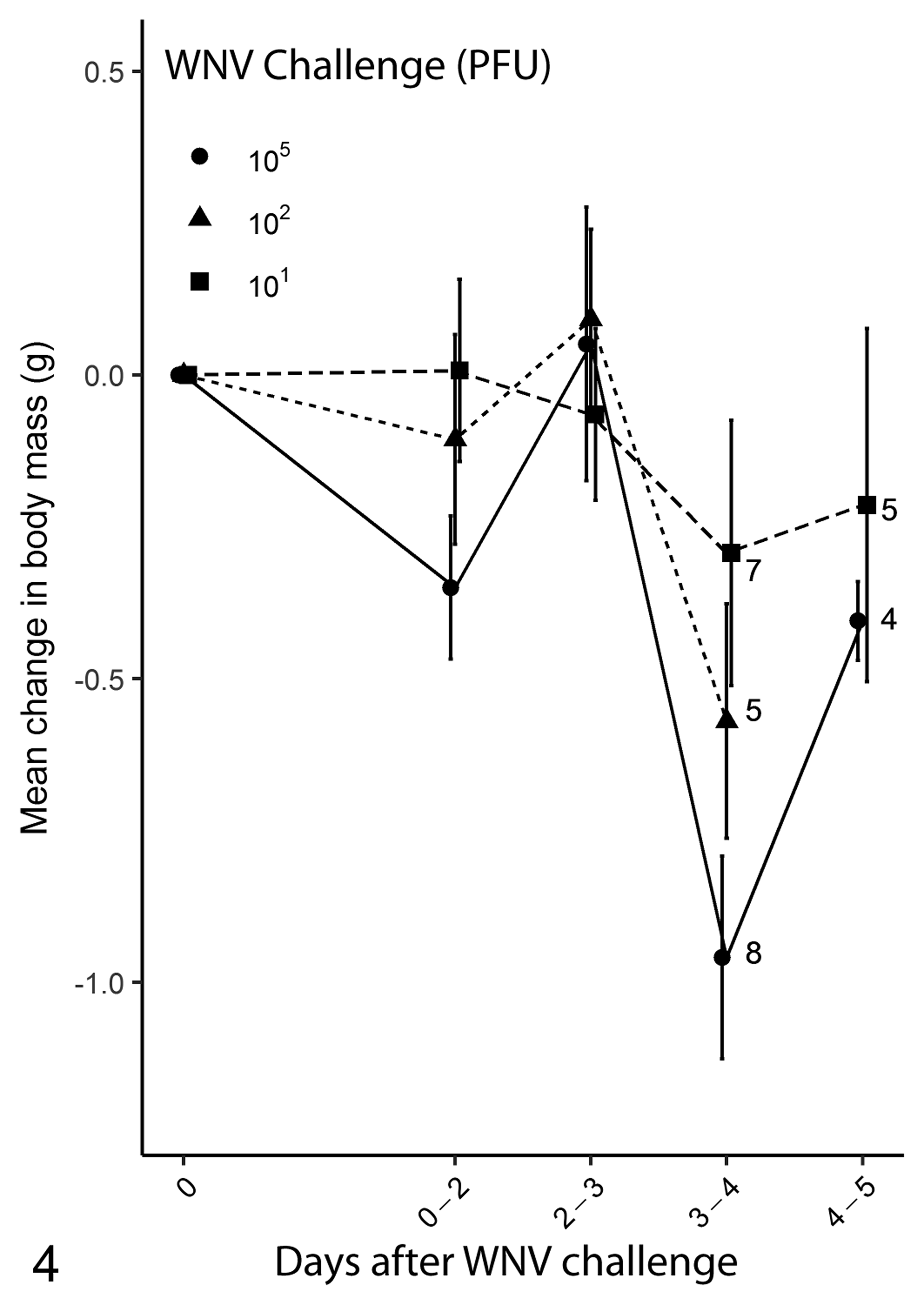
Body Mass Changes
In the interval between 0 and 2 dpi, canaries inoculated with 105 or 102 PFU of virus lost body mass in a dose dependent manner (Fig. 4). However, these birds recovered their prechallenge body mass by 3 dpi before a second decline in body mass between 2 and 4 dpi. For birds inoculated with 105, 102, 101 PFU of virus, the mean loss of body mass at death was 8%, 2%, and 4%, respectively (refer to doi:10.5066/F7F18XWV). Crows inoculated with WNV lost on average 8% of body mass (refer to doi:10.5066/F7F18XWV).
Macroscopic Lesions
At necropsy, most canaries had splenomegaly; pallor, though not enlargement, of the liver; and, in a smaller number of birds, slight injection of vessels on the surface of the brain. Gross lesions were not observed in control birds.
Histopathology
Organs most affected by WNV challenge in canaries were the liver and the spleen. Pathologic changes present were influenced by dose of WNV inoculum, as follows.
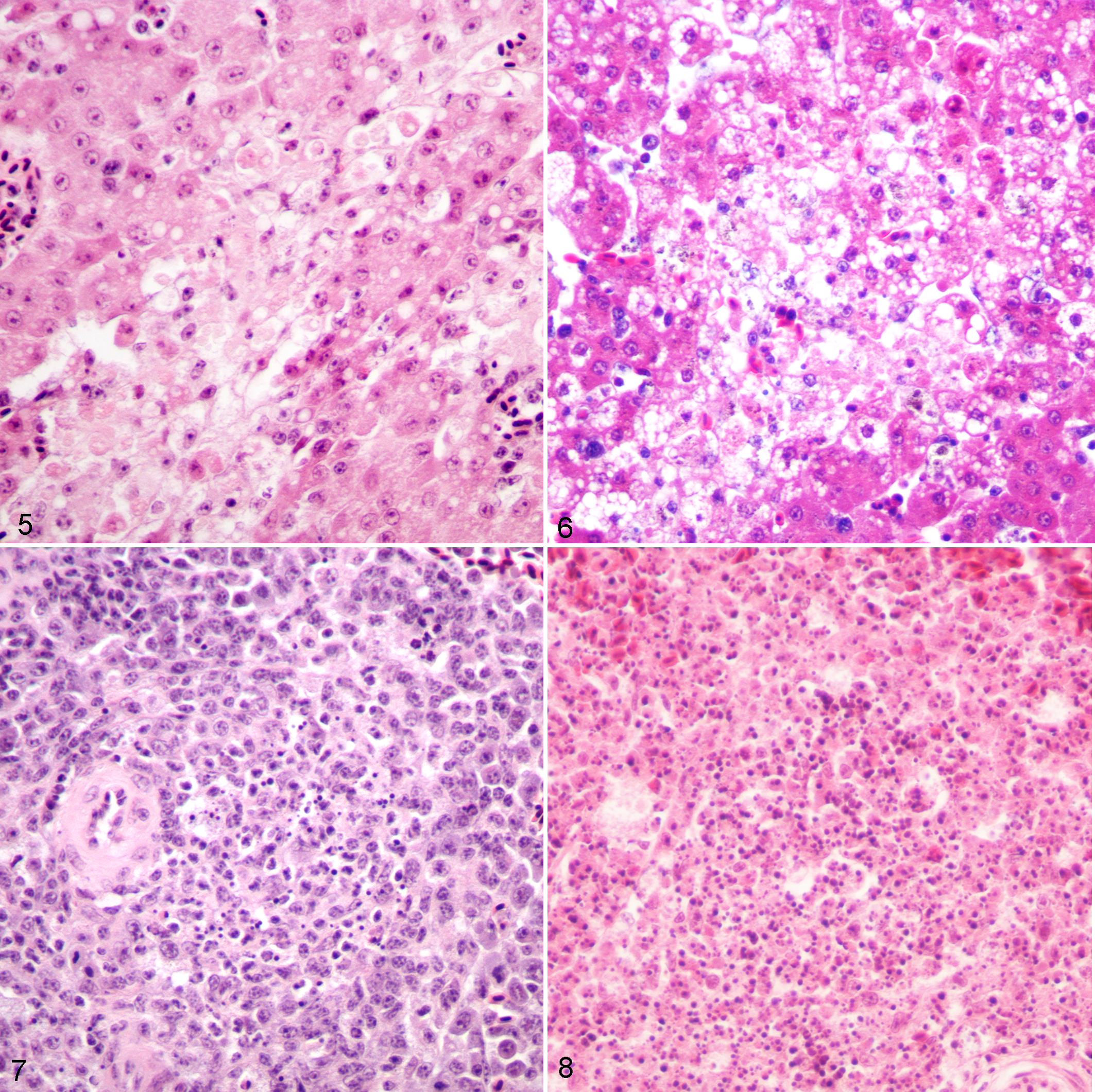
Canaries inoculated with 105 PFU of virus had either no significant lesions in the liver or rare multifocal lymphohistiocytic nodules associated with mild acute hepatocellular necrosis. In the spleen, there were moderately increased numbers of macrophages in the red pulp, with erythrophagocytosis, and rarely, mild, multifocal fibrinous necrosis.
Lesions were much more prominent in canaries inoculated with 101 or 102 PFU WNV, and tended to be most severe when the bird had survived to day 5 of the study. In these birds, pathologic changes in the liver and spleen were strikingly similar to those in crows that were inoculated with 105 PFU WNV and survived until day 6 postinoculation. Changes in the liver included multifocal, moderate, randomly distributed granulomatous and lymphohistiocytic hepatitis, with mild to moderate, multifocal to focally extensive coagulative hepatocellular necrosis (Figs. 5 and 6). Areas of necrosis were only mildly infiltrated by heterophils. In the spleen, there was multifocal lymphocytic necrosis characterized by the presence of karyorrhectic nuclear debris (Figs. 7 and 8). In addition, there was moderate diffuse plasmacytosis and histiocytosis in the red pulp. The only other significant histopathologic abnormality present in this group of canaries was in the kidney, where mild multifocal proximal tubular necrosis was consistently present. No significant histopathologic abnormalities were present in the skin, heart, brain, lung, skeletal muscle, proventriculus, gizzard, intestine, or pancreas of any of the canaries examined. Microscopic lesions were not observed in the tissues of control birds.
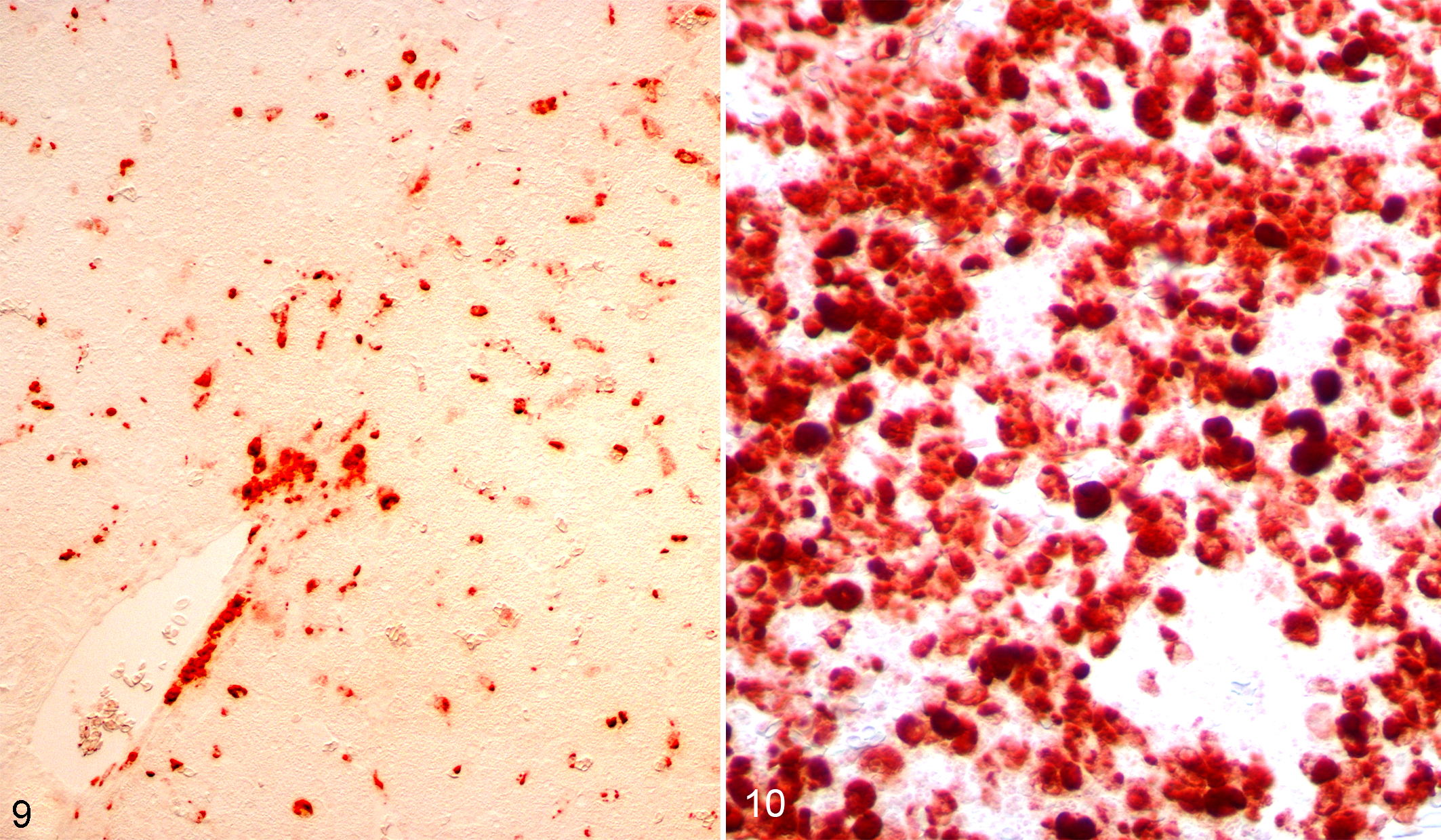
Immunohistochemistry for WNV antigen was strongly positive in both the liver and spleen in inoculated canaries (Figs. 9 and 10). In the liver, scattered hepatocytes and Kupffer cells were IHC-positive; in the spleen, lymphocytes were uniformly strongly positive. The brain, lung, heart, kidney, adrenal glands, and gonads were negative for WNV antigen.
Discussion
In this study, we describe the clinical outcome and the lesions in canaries challenged with WNV. Through a series of experimental challenges with a decreasing level of viral challenge we show that even when challenged with as few as 10 PFU of WNV, severe clinical disease including mortality occurred in canaries. Clinical disease was accompanied by a high level of viremia, oral shedding of virus, and a high level of WNV in all tissues collected at death. The liver and spleen, as we and others 14,18 have observed in crows, were highly affected with gross and microscopic lesions. In addition, like experimentally and naturally infected crows, the brain of canaries was not highly affected by the virus and, even though flight and posture were affected, the birds did not die from neurological disease.
The acute clinical course of disease in canaries in this study paralleled those observed in crows, and as reported by other investigators. 11,14,18 Viremia, oral shedding of virus, and the tissue burden of virus all equaled or exceeded the levels we and others have reported in crows and other corvids. In addition, the loss of body mass along with apparent weakness or ataxia in canaries is similar to our and others’ observations following experimental challenges of corvids. Previously, WNV neutralizing antibody has been reported in experimentally infected crows that died or were euthanized because of severe clinical signs of disease. 14 We also detected WNV neutralizing antibody in a small number of samples remaining that were positive on the WNV screening assay.
The microscopic lesions observed in canaries in this study are in agreement with those observed in crows that were experimentally infected with WNV at NWHC and reported by others. 14,18 Hepatitis and splenitis were most commonly observed in both canaries and crows in this study, while lesions in the brain were either uncommon and mild or nonexistent. Myocarditis has been observed in crows, but was not present in canaries examined in this study. Other studies in crows have also included examination of other tissues, 14,18 and hematological, serum chemistry, and behavioral observations. 14
Corvids are recognized to be among the most susceptible avian species to WNV in North America and have been used in a number of experimental studies with the virus. Major deterrents to conducting studies with wild-caught corvids are the relative difficulty in capturing the birds and acclimating captured birds to biosafety level 3 containment. Another drawback in the use of corvids, or any wild caught bird, is the inability to effectively control confounding infection or parasitism and the stress of captivity which may alter the outcome of a WNV challenge. The use of a domesticated passerine species is considerably easier and decreases the effect of stress and coinfection on the outcome of the challenge study.
A limitation in using small domestic passerines such as zebra finches 10 or canaries is the limited volume of blood that can be obtained at each sample collection. Generally, blood collection is limited by US Public Health Service policy to 10% of circulating blood volume. When sampled more than once weekly the limitation is further reduced to 5% per sample. In addition, while larger tissues are easily obtained at necropsy, smaller tissues are more difficult to obtain precisely and process for histopathology. Domestication of an avian species also may result in the loss of allelic variation of receptors of the immune system as has been described for MHC I in domesticated Australian and Timor zebra finches compared to wild-type zebra finches. 2 The loss of genetic diversity through domestication may affect the response compared to challenge of wild-type birds.
Despite these limitations, small passerines can be easily maintained in a colony which allows some control over the role of stress in a challenge study. Canaries could be used to examine the pathogenicity of emergent strains of WNV that are not maintained subsequently in enzootic cycles. Zebra finches with moderate susceptibility to WNV with little mortality 10 and canaries with high susceptibility and mortality might be used to quantify the relative transmission of WNV to feeding mosquitoes from birds of varying susceptibility. Our discovery of the high susceptibility of canaries may lead to their use as a laboratory model for not only studies of WNV in passerines, but potentially other flaviviruses.
Supplemental Material
Supplementary Material, DS1_VET_10.1177_0300985818760377 - West Nile Virus Infection in American Singer Canaries: An Experimental Model in a Highly Susceptible Avian Species
Supplementary Material, DS1_VET_10.1177_0300985818760377 for West Nile Virus Infection in American Singer Canaries: An Experimental Model in a Highly Susceptible Avian Species by Erik K. Hofmeister, Melissa Lund, and Valerie Shearn Bochsler in Veterinary Pathology
Footnotes
Supplementary material for this article is available online.
Acknowledgements
The authors wish to thank L. Karwal for laboratory support, one anonymous reviewer for comments on a previous version of this manuscript, and the Rockefeller University’s Field Research Center for the generous donation of canaries. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the US government. The data supporting this manuscript are available at doi:10.5066/F7F18XWV.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was conducted with USGS Ecosystems Mission Area funds.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
