Abstract
Rhabdomyosarcomas are a diverse group of malignant mesenchymal neoplasms exhibiting variable levels of differentiation toward skeletal myocytes. Neoplastic cells may resemble relatively undifferentiated myoblasts, satellite cells, or more differentiated elongated spindle cells and multicellular myotubes. In veterinary medicine, classification into subtypes and variants is based on an outdated system derived from human pathology and is solely based on histologic characteristics. In contrast, classification of human rhabdomyosarcoma is based on histologic, immunohistochemical, and molecular diagnostic techniques, and subclassification has clinical and prognostic relevance. Relevance of tumor subtyping has not been established in veterinary medicine. Recent discoveries of components of the molecular pathogenesis and genomes of human rhabdomyosarcomas have led to new diagnostic techniques and revisions of the human classification system. The current classification system in veterinary medicine is reviewed in light of these changes. Diagnosis of rhabdomyosarcoma using histopathology, electron microscopy, and the clinical aspects of human and canine rhabdomyosarcomas is compared. The clinical features and biologic behavior of canine rhabdomyosarcomas are compared with canine soft tissue sarcomas.
Rhabdomyosarcomas (RMS) are relatively rare neoplasms of domestic animals that have a variety of gross morphologies, histologic variations, and cellular phenotypic variations. Common histologic subclasses include embryonal RMS, alveolar RMS, and botryoid RMS (also called botryoid embryonal RMS). Less common subclasses include pleomorphic RMS. The relative rarity of these tumors makes diagnosis, clinical prognosis, and treatment difficult. In some cases, these neoplasms are lumped into the category of high-grade soft tissue sarcomas 89,158 but are explicitly excluded from the common grading system of canine soft tissue sarcomas. 72 The low prevalence of these neoplasms in the literature may be due to a failure to diagnose them, since their extreme variation in phenotype, age of onset, and cellular morphology makes diagnosis and classification difficult. The most aggressive of these neoplasms arise in juvenile dogs younger than 2 years and include alveolar RMS and embryonal RMS. Histopathological findings of these primitive neoplasms typically consist of sheets or packets of small round cells with a lymphoid appearance, or sheets of round cells with abundant cytoplasm, and may be misdiagnosed as lymphomas or other primitive neoplasms. Other phenotypes arising in adult animals consist of anaplastic cells with large nuclei, abundant cytoplasm, and bizarre mitotic figures. These neoplasms may be misdiagnosed as “undifferentiated sarcoma,” “anaplastic sarcoma,” or even a “poorly differentiated sarcoma” depending on the phenotype of the variant. 33,48,146,158 Diagnostic features of skeletal muscle differentiation may not be evident at the light microscopic level and may only be recognized by immunohistochemistry or electron microscopy. This article reviews the diagnostic features, pathogenesis, and classification of rhabdomyosarcomas in dogs and compares them with their human counterparts to facilitate the recognition of these tumors by veterinary diagnostic pathologists.
Developmental Biology
In the developing embryo, uncommitted mesodermal cells differentiate into myogenic progenitor cells under the control of transcription factors such as PAX3, PAX7, and the Fox family of transcription factors. 2,136 Myogenic precursors then proliferate by asymmetric replication, forming immature myoblasts under the direction of myoblast determination protein 1 (MyoD1) and other transcription factors of the Myf family that includes myogenin (myf4). 165 During development and after injury, these cells proliferate, elongate, and fuse together, forming myotubes. Under the direction of myogenin, the myotubes begin forming actin and myosin filaments organized into sarcomeres characteristic of striated muscle. 58 In both neoplastic and nonneoplastic myogenic cells, MyoD1 is associated with cellular differentiation into myoblast cells with a high proliferative capacity, whereas myogenin is associated with cell cycle arrest, cell fusion into multinucleated myotubular cells, and formation of muscle fibers. 58 Mature skeletal muscle expresses very little of these transcription factors, 19,60,67,121 and as normal skeletal muscle matures, it loses its replicative capacity but retains undifferentiated satellite cells at the periphery of the myotube as stem cells, which are activated when the muscle is damaged. Figure 1 illustrates the major events in skeletal muscle myoblast fusion. More extensive reviews of the subject have been published. 22 –24,32,111
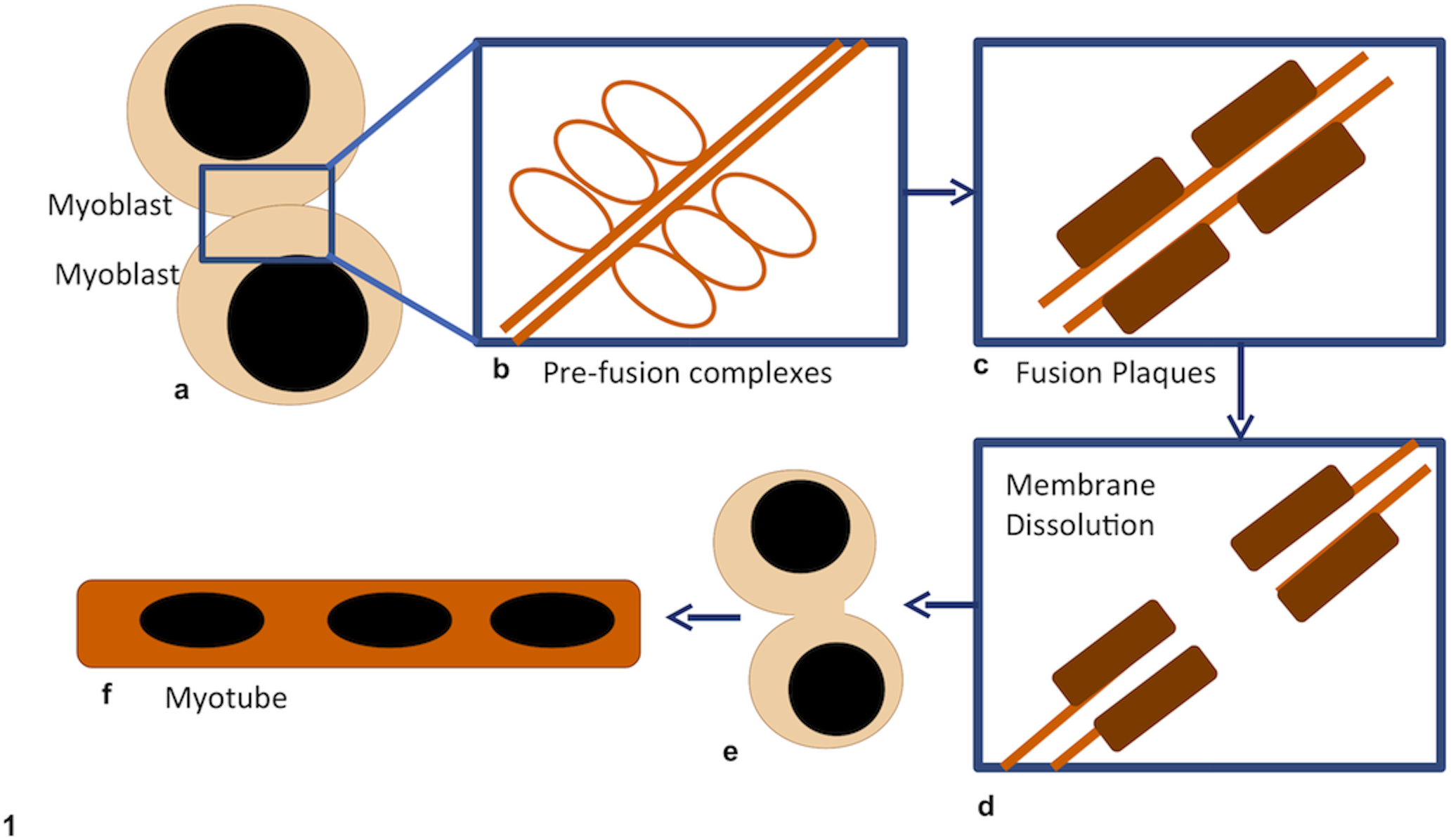
Skeletal muscle development and myoblast fusion. Under the direction of myogenin as well as PAX, myoblasts leave the cell cycle and begin to fuse with each other or with preexisting myotubes. Myotube fusion begins with 2 cells lining up, with their plasma membranes contacting each other (a). Next, paired vesicular structures form along the apposed membrane surfaces (prefusion complexes) (b). These prefusion complexes resolve into fusion plaques (c) that appear similar to adherens junctions and contain M-cadherin. The fusion process requires calcium, which is further evidence that these fusion plaques are members of the adherens junction family of cell-cell attachments. Once fusion plaques form, the plasma membrane adjacent to them dissolves and the cytosols of the 2 cells are in direct contact with each other (d). By some yet unexplained mechanism, the fusion plaques then disappear, and the walls of each adjoining cell are removed, fusing the 2 cells (e). The fused cells then elongate, the nuclei line up in rows to form myotubes, and the cells begin producing contractile filaments and myoglobin (f). Additional myoblasts may fuse with myotubes in a similar fashion. Adapted from Doberstein et al 40 and Paululat et al. 112
Diagnosis and Classification
Histologic Patterns
RMS in humans and dogs can resemble undifferentiated myoblasts or early embryonic myotubes. The term embryonal is a descriptive term for neoplasms exhibiting a range of cellular morphologies that resemble various developmental intermediates and often have a myxomatous stroma as seen in developing muscle. 31,109,127 In the case of RMS, these cells range in morphology from round, immature myoblast cells with variable amounts of eosinophilic cytoplasm to multinucleated myotubular cells that resemble the cellular phenotypes of developing skeletal muscle. Embryonal muscle tumors consist of embryonal RMS, alveolar RMS, and botryoid RMS; however, alveolar RMS is given its own category due to its less favorable prognosis in humans. 19,127 In humans, alveolar RMS and embryonal RMS occur predominantly in children younger than 15 years and is dubbed juvenile or pediatric RMS. 127 In dogs, this same association between age of onset and histologic type has been recognized by several authors 61,65 but has not been universally accepted, as many cases of embryonal RMS occur in dogs older than 2 years. 31 Pleomorphic RMS occurs mostly in adult people, 19,109 and the 2 cases in dogs have been in a 3-year-old and a 14-year-old dog. 8,31
Attempts have been made to segregate neoplasms into different categories for the purpose of making predictions of biologic behavior based on histomorphologic, ultrastructural, or immunohistochemical features. The most recent, and widely used, human classification system is the international classification system developed by Newton et al; 104 however, similar yet differing systems exist and may be encountered periodically in literature searches. 109 According to the international classification system, human RMS is subdivided into embryonal, botryoid, alveolar, and pleomorphic (anaplastic) subcategories. 31,65,109 With regard to human childhood RMS, there are 3 main categories: embryonal, alveolar, and undifferentiated, and these are distinguished from each other by histologic pattern and cytogenetic studies. 109,145 The undifferentiated juvenile RMS has not been recognized in veterinary medicine. Classification of canine RMS closely parallels classification schemes in human medicine (Table 1). Furthermore, embryonal and alveolar RMS can be further subdivided into variants based on the predominant cell morphology. Variants of embryonal RMS include the rhabdomyoblastic, myotubular, and spindyloid variants. Variants of alveolar RMS include the classic alveolar pattern and the solid alveolar pattern, which lacks fibrous septa. 31 Such a precise classification system would potentially allow for subsequent study of clinical course of disease and response to treatment. Detailed descriptions of histologic characteristics of each category are below.
Classification of Rhabdomyosarcomas, With Variants and Relevant Clinical Aspects
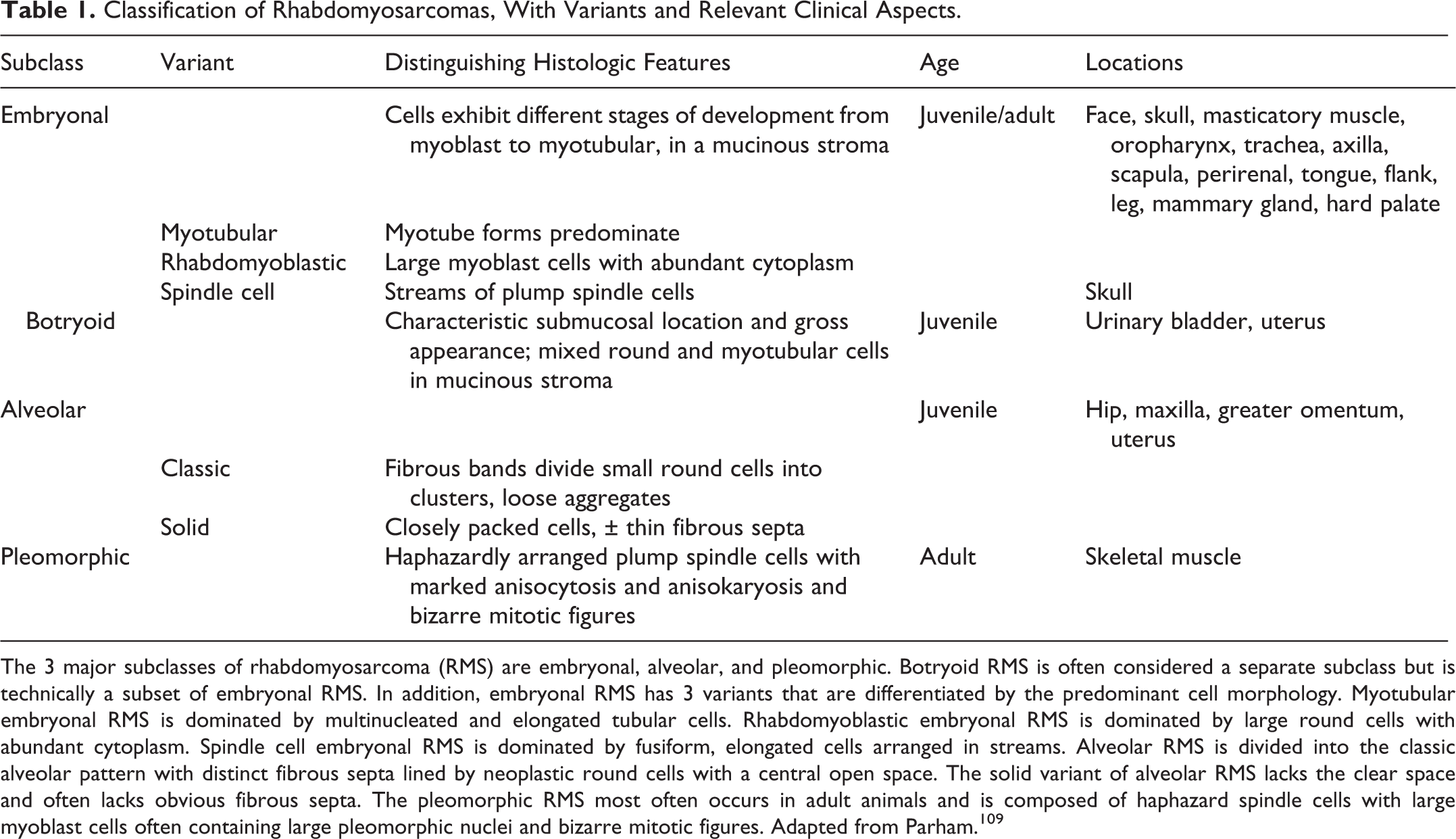
The 3 major subclasses of rhabdomyosarcoma (RMS) are embryonal, alveolar, and pleomorphic. Botryoid RMS is often considered a separate subclass but is technically a subset of embryonal RMS. In addition, embryonal RMS has 3 variants that are differentiated by the predominant cell morphology. Myotubular embryonal RMS is dominated by multinucleated and elongated tubular cells. Rhabdomyoblastic embryonal RMS is dominated by large round cells with abundant cytoplasm. Spindle cell embryonal RMS is dominated by fusiform, elongated cells arranged in streams. Alveolar RMS is divided into the classic alveolar pattern with distinct fibrous septa lined by neoplastic round cells with a central open space. The solid variant of alveolar RMS lacks the clear space and often lacks obvious fibrous septa. The pleomorphic RMS most often occurs in adult animals and is composed of haphazard spindle cells with large myoblast cells often containing large pleomorphic nuclei and bizarre mitotic figures. Adapted from Parham. 109
Embryonal Rhabdomyosarcoma
Embryonal RMS includes 3 variants that are named because of their predominant cellular morphology. 31,35,36,109 The myotubular variant of embryonal RMS is dominated by multinucleated “strap” cells forming myotubes with frequent cross-striations (often only identified by histochemical staining with phosphotungstic acid hematoxylin [PTAH] stain, which highlights striations as dark blue/purple lines among paler blue/purple myofibers), whereas the rhabdomyoblastic variant is dominated by round to polygonal cells with abundant eosinophilic cytoplasm (Fig. 2), with only rare PTAH positive cross-striations. 36,109 The rhabdomyoblastic variant is included along with alveolar RMS and undifferentiated RMS in the general category of “small round cell” neoplasms of childhood sarcomas. 5,149 These 3 categories are more commonly diagnosed with the aid of molecular diagnostic techniques that are unavailable in veterinary medicine. 145 There is no evidence that classification into the myotubular or rhabdomyoblastic variants has any bearing on prognosis or clinical course in human or canine medicine. 36,109
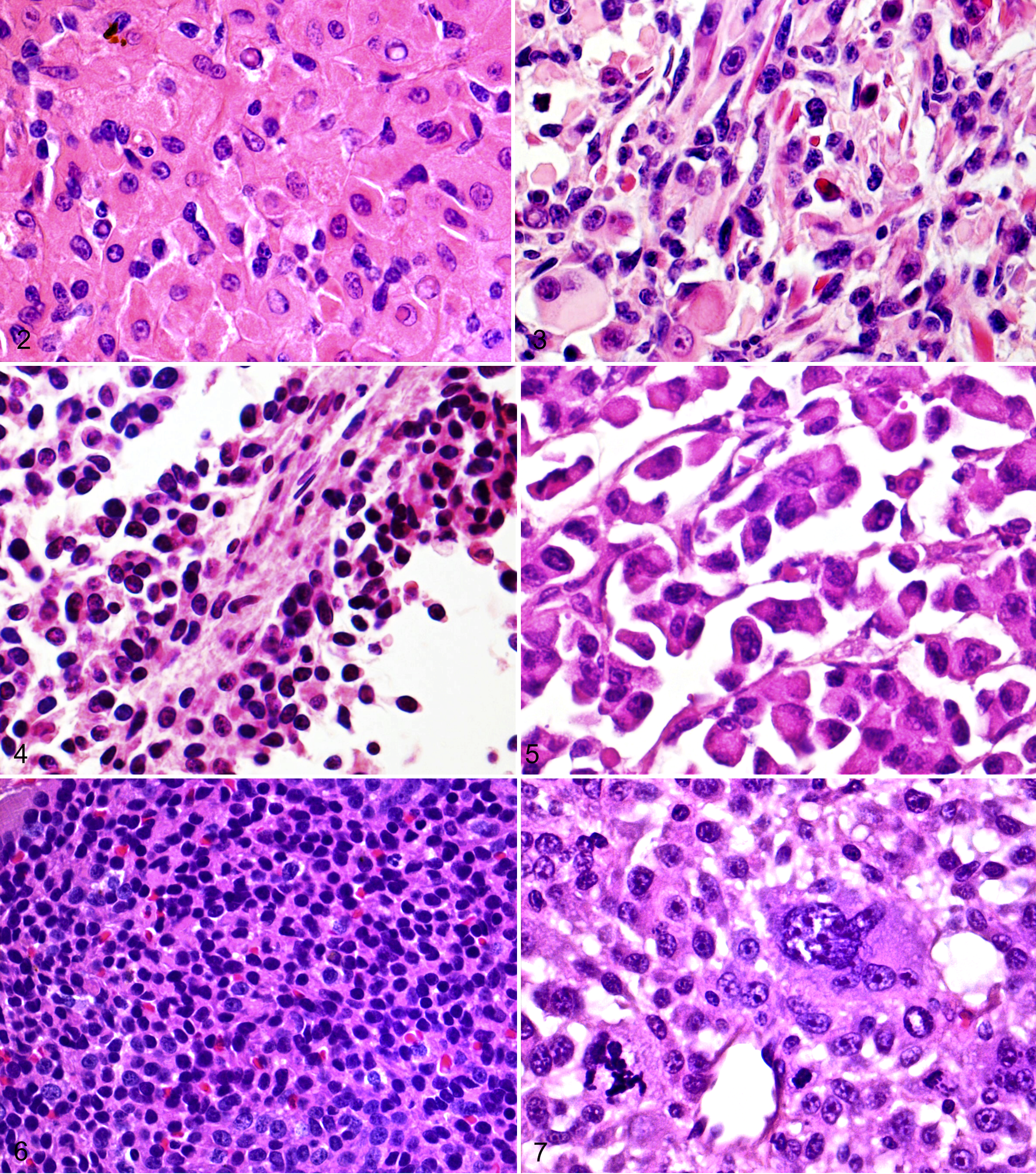
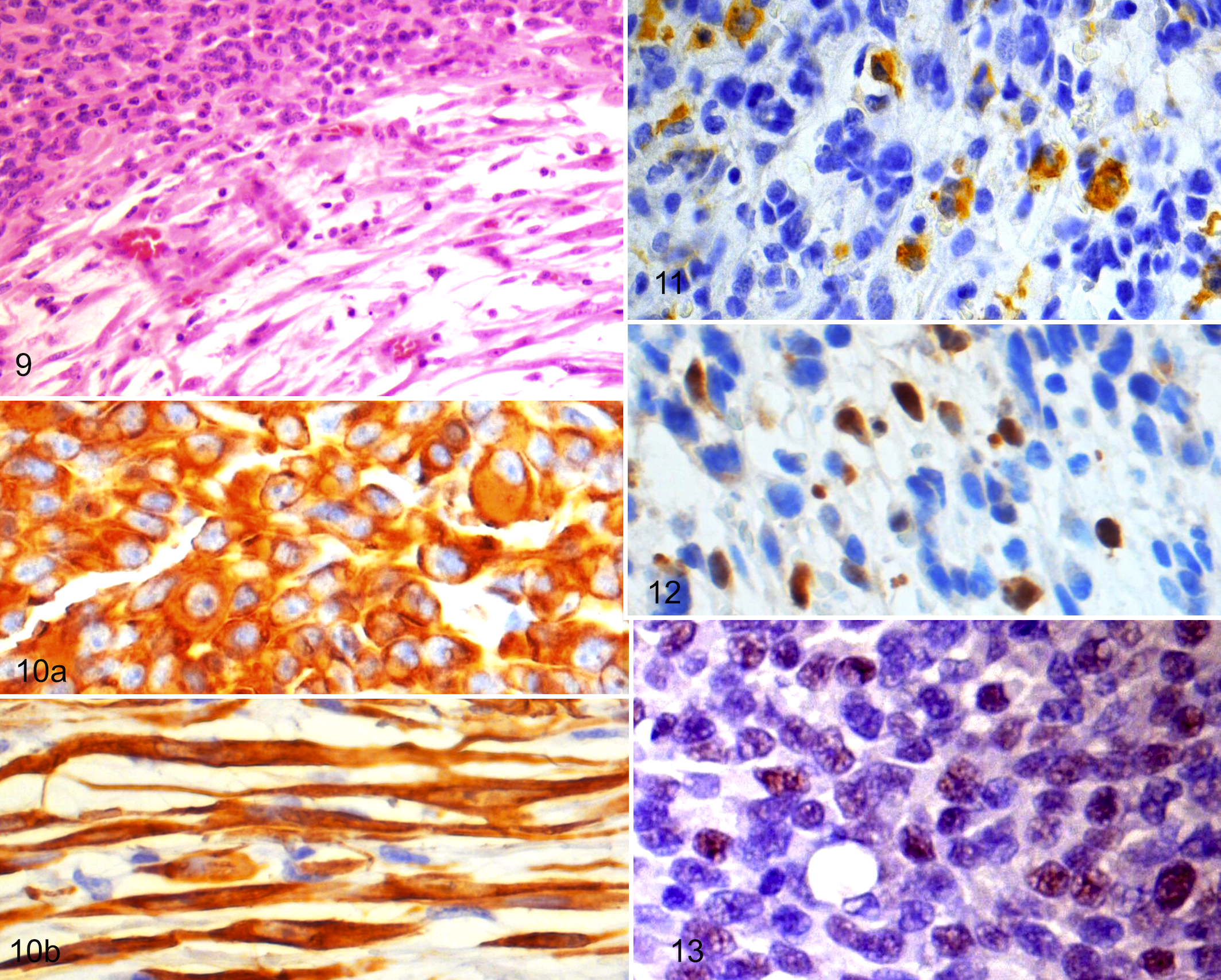
Dog, embryonal rhabdomyosarcoma (RMS), rhabdomyoblastic variant: neoplastic cells are uniform in size with abundant eosinophilic cytoplasm with variable amounts of cytoplasmic glycogen or lipid vacuoles. Hematoxylin and eosin (HE).
Botryoid Embryonal RMS
Botryoid rhabdomyosarcomas are tumors that have a characteristic gross appearance and anatomic location. The term botryoid refers to the grape-like masses protruding from the mucosa of the urinary bladder, which is the most common location. Like embryonal RMS, these tumors also recapitulate skeletal muscle embryogenesis and therefore contain undifferentiated myoblast cells as well as multinucleated myotube cells within a myxomatous stroma (Fig. 3). 31,127 Histologically, botryoid RMS of the urinary bladder is located in the submucosa, and neoplastic cells are separated from the mucosal epithelium by a “cambium” (ie, transitional) layer of connective tissue. Because of their distinct anatomic location and histologic characteristics, these neoplasms have been placed into a distinct subcategory of embryonal RMS in human and veterinary medicine. 109
Spindyloid Embryonal RMS
The spindyloid variant of embryonal RMS is a relatively new category. This type of tumor has been reported only once in veterinary medicine as a mass arising from the skull of an 11-month-old Boxer dog. This mass was composed of interlacing fascicles of plump spindle cells. 35 The spindyloid embryonal RMS, as the name implies, is composed of thin spindyloid myoblast cells forming bundles within a myxoid stroma. Although limited case reports exist, these neoplasms in humans typically have a better prognosis than any other variant of RMS. 94
Alveolar RMS
Alveolar RMS is the second RMS in the small round cell category of childhood RMS. It is characterized by small, poorly differentiated, round cells with scant cytoplasm, arranged into packets of cells separated from each other by fibrous connective tissue, forming alveolar-like patterns (Figs. 4, 5). 149 Alveolar RMS is further subdivided into the classic alveolar pattern, which is characterized by extensive sloughing of neoplastic cells into a central open space with additional neoplastic cells lining the fibrous septa. The second subtype of alveolar RMS is the solid variant, which is characterized by sheets of small round cells closely packed together (Fig. 6). 5,149 This variant in dogs may have thin fibrous septa dividing nests of round cells similar to the classic “neuroendocrine pattern” 31 but is most often lacking this histologic distinction, making differentiation between solid alveolar RMS and rhabdomyoblastic embryonal RMS very difficult. 36,109 Differentiation between alveolar RMS and embryonal RMS is important in human medicine because alveolar RMS of any variant is more locally aggressive and has a higher metastatic rate, carrying a poorer prognosis than embryonal RMS. 36,60,116,127 Currently, there are insufficient data to draw the same prognostic conclusion in veterinary medicine.
Pleomorphic Rhabdomyosarcoma
Pleomorphic RMS occurs less frequently than alveolar and embryonal RMS in humans. It is found more often in adult humans and animals and arises almost exclusively within skeletal muscle. Histologically, it is composed exclusively of haphazardly arranged spindle cells with rare multinucleated cells present and is recognized by the appearance of bizarre or multipolar mitotic figures and a high degree of cellular and nuclear pleomorphism (Fig. 7). 35,109 This diagnosis should be used only when there are no areas of embryonal or alveolar morphology within the tumor. 31 This tumor undoubtedly causes confusion for pathologists, as histologic evidence of skeletal muscle differentiation can be rare. Without the aid of immunohistochemistry, some of these neoplasms may be diagnosed as anaplastic sarcomas.
Ultrastructural Diagnosis of Rhabdomyosarcoma
Transmission electron microscopy is the gold-standard diagnostic test for rhabdomyosarcomas in humans and dogs, but attempts to use ultrastructural characteristics to aid in classification have been relatively unrewarding in human medicine, with both alveolar and embryonal RMS exhibiting a variety of subcellular structures. 133 Common to all human RMS are Z-lines, large numbers of mitochondria, myofilament tangles, and myosin-ribosome complexes (Fig. 8). Several studies have indicated the presence of basement membranes in both alveolar and embryonal tumors in human RMS. 95,142 Desmosome-like structures may be a unique feature of alveolar RMS, 27 but since publication of this study, there has not been further investigation into these unique structures.
Cytoskeletal Proteins
In the absence of myotubes, strap cells, and cross-striations, small round cell neoplasms must be differentiated from lymphomas and primitive neuroectodermal tumors or other embryonal neoplasms. Early studies of human RMS identified many ultrastructural features unique to skeletal muscle that can aid diagnosis of rhabdomyosarcomas. Identification of sarcomeric structures such as unambiguous Z-lines within I-bands, A-bands with H- and M-bands, and other structures such as free ribosomes closely associated with myofilaments, abundant glycogen granules, external lamina, prominent nucleoli, and pleomorphic mitochondria can all be used to support a diagnosis of rhabdomyosarcoma. 17 Electron microscopy studies of both alveolar and embryonal RMS in humans have consistently identified actin and myosin filaments, Z-lines, and A- and I-bands in both alveolar and embryonal tumors. 129,133,139 In some cases of embryonal RMS, myofilament tangles are observed, indicating poor formation and disorganization. 133
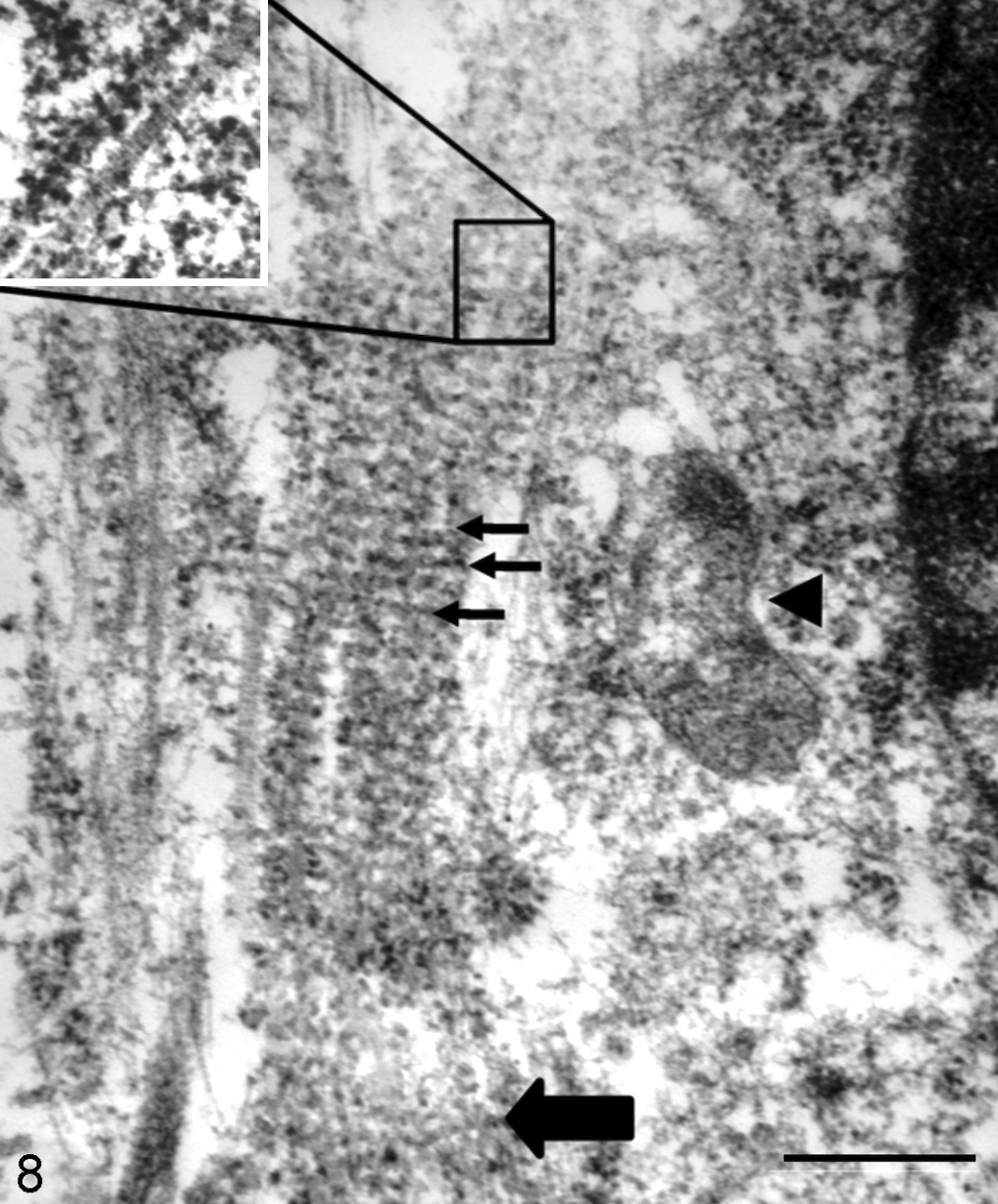
Dog, skeletal muscle, rhabdomyosarcoma (RMS). Transmission electron micrograph showing a small crescent of the nucleus on the right, a mitochondrion (arrowhead), and a developing muscle fiber consisting of numerous polyribosomes with myosin thick filaments (short arrows) and an M-line (block arrow). Z-lines were poorly formed and scattered haphazardly (30 k; scale bar = 500 nm). Inset (upper left corner) shows a closeup of the polyribosomes and myosin filaments (40 k).
Adhesion Structures in Human RMS
Some alveolar RMS contains “desmosome-like” structures between adjacent neoplastic cells lining the fibrous septa. 27 Churg and Ringus 27 described these structures and hypothesized that they are remnants from the embryological ectoderm before the epithelial to mesenchymal transition. To complicate matters, these structures resemble skeletal muscle fusion plaques found in Drosophila and mouse myoblast cultures. 22,23,40 Fusion plaques appear similar to desmosomes and adherens junctions by electron microscopy. They are transient structures, like adherens junctions, and contain M-cadherin rather than desmosomal cadherins or desmoglein. 10,22,46,53 Myoblasts of Drosophila spp that lack the gene Rolling stone (Rost) fail to complete myoblast fusion and accumulate fusion plaques resembling the junctional complexes described in human alveolar RMS. 27,32,40 It is unknown whether these junctional complexes in human RMS are arrested fusion plaques since they have not been studied extensively. It is unknown whether human embryonal RMS has similar “desmosome-like” structures, and it is unclear whether these junctional complexes are related to the pathogenesis of RMS in humans. RMS cells of C2C12 mouse myoblast cultures have reduced M-cadherin expression and elevated R-cadherin expression, both of which can activate Rac1 GTPase, 22,70 which has been implicated in promoting myoblast proliferation and myoblast fusion, and yet later acts as an inhibitor of myogenesis induction. 22 Although not proven, it is possible that adhesion structures are involved in the failure of many RMS types to fuse into myotubes and therefore fail to histologically resemble skeletal muscle.
Canine RMS
There are few electron microscopic studies of canine RMS and no descriptions of differences between histologic variants. Actin and myosin filaments, polyribosomes, Z-bands, and mitochondria (Fig. 8) are prominent in botryoid RMS, laryngeal muscle tumors (both rhabdomyomas and RMS), and RMS that is not further classified (RMS not otherwise specified [NOS]). 3,76,93,113
Immunohistochemical Diagnosis of RMS
Immunohistochemistry (IHC) is useful in differentiating rhabdomyosarcomas from other mesenchymal tumors or embryonal tumors. A potential diagnostic pitfall is the fact that all myogenic markers used to diagnose RMS will also be present in developing and regenerating skeletal muscle, 19,168 so it is the job of the diagnostic pathologist to first differentiate satellite cell proliferation and differentiation from a neoplastic proliferation. Diagnosis of RMS in humans and dogs is based on detection of 1 or more muscle-specific markers and absence of smooth muscle markers. The presence of these markers depends to some extent on the degree of differentiation of the neoplastic cells but is also subject to variation due to altered expression of myogenic regulating factors. 26,34,97,133
Vimentin and Desmin
Identification of skeletal muscle fibers is largely based on specific expression of cytoskeletal proteins such as intermediate filaments or contractile proteins. Skeletal muscle cells are characterized by expression of different cytoskeletal proteins at progressive stages of development. 31 Like all mesenchymal cells, myoblast cells first express the intermediate filament vimentin. Later, during differentiation, they express another intermediate filament—desmin, which is common to skeletal muscle, cardiac muscle, smooth muscle, and myofibroblasts. 9,97 Desmin has been proven a useful marker even in relatively undifferentiated RMS in both humans and dogs 95,129,166,167 (Figs. 9, 10), but staining can be patchy and sparse, and other neoplastic and normal tissue can express desmin. 71,121 Despite the variability in staining pattern and intensity, it is a useful marker of myogenic differentiation in poorly differentiated RMS such as alveolar and embryonal RMS. Because of desmin’s low specificity and high degree of variability of staining, newer methods of identifying undifferentiated myoblast cells have been developed, including using MyoD1 and myogenin, 2 nuclear transcription factors.
Actin, Myosin, Sarcomeric Actin, and Myoglobin
Muscle actin (ms actin or HHF35) is an antibody that can identify α-actin isoforms in both human and canine skeletal, cardiac and smooth muscle, and myofibroblasts, myointimal cells, and reactive mesothelial cells 49,150 with an equivalent specificity to desmin. 126 Identification of desmin or ms actin in myogenic sarcomas cannot confirm a diagnosis of RMS by themselves, but neoplasms with negative immunostaining for smooth muscle actin can help rule out leiomyosarcomas and malignant myofibroblastic tumors and make a reasonably certain diagnosis of RMS. 51,133
More specific to skeletal muscle is expression of proteins involved in sarcomere construction such as the α-actin isoform found in sarcomeres, identified by the antibody sarcomeric actin (src actin). 31 As skeletal myocytes differentiate further, they begin to accumulate sarcomeric actin, followed by myosin or fast myosin, and finally myoglobin, which is first seen in large amounts following myoblast fusion to form myotubes or strap cells.9,31,36,60,83,101,121,166,167 Mature skeletal muscle typically has little vimentin expression, whereas immature skeletal muscle and undifferentiated rhabdomyosarcomas typically have rare expression of myoglobin and myosin. 36 See Table 2 for a brief summary of common IHC markers used in canine rhabdomyosarcomas.
Common Immunohistochemistry Antibodies in Veterinary Medicine
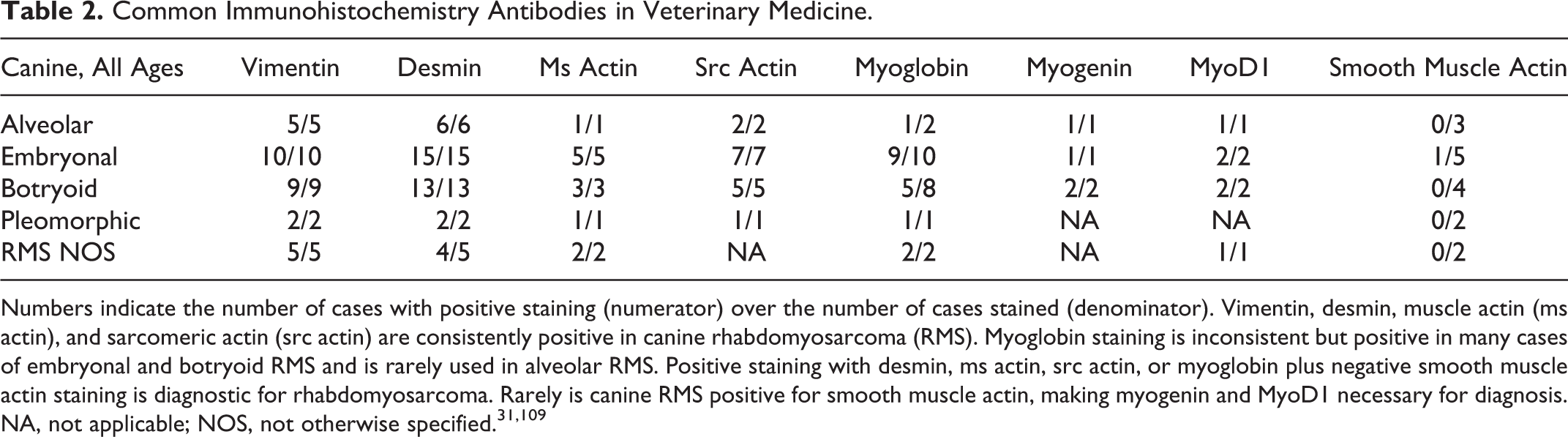
Numbers indicate the number of cases with positive staining (numerator) over the number of cases stained (denominator). Vimentin, desmin, muscle actin (ms actin), and sarcomeric actin (src actin) are consistently positive in canine rhabdomyosarcoma (RMS). Myoglobin staining is inconsistent but positive in many cases of embryonal and botryoid RMS and is rarely used in alveolar RMS. Positive staining with desmin, ms actin, src actin, or myoglobin plus negative smooth muscle actin staining is diagnostic for rhabdomyosarcoma. Rarely is canine RMS positive for smooth muscle actin, making myogenin and MyoD1 necessary for diagnosis. NA, not applicable; NOS, not otherwise specified. 31,109
In humans and dogs, rhabdomyosarcomas are consistently immunohistochemically positive using antibodies for vimentin, desmin (Fig. 10), muscle actin, and src actin (Fig. 11), 133 but because of their lack of cellular differentiation, myoglobin staining is variable. 3,31,61,65,109,167 In dogs, embryonal and botryoid RMS are frequently positive for myoglobin compared with alveolar RMS (Table 2). 67
Myo D1 and Myogenin
MyoD1 and myogenin are early embryological transcription factors involved in the differentiation of mesoderm cells into myoblast cells, myoblast cell proliferation, and myoblast differentiation into multinucleated myotubes. MyoD1 expression is associated with immature rhabdomyoblasts that have a higher proliferative capacity, whereas myogenin is expressed during myoblast fusion and is associated with exit from the cell cycle. Rhabdomyosarcomas that are composed of relatively undifferentiated cells should be expected to express less desmin, actin, myosin, and myoglobin and more MyoD1 and myogenin. Several studies have confirmed the specificity of myogenin and MyoD1 as specific and sensitive markers of undifferentiated RMS in humans. 19,60,101,121,127,165 Furthermore, several studies have concluded that differential expression of myogenin can be used to subclassify human RMS into alveolar or embryonal RMS (Table 3). 19,60,109,165 Human alveolar RMS has been found by several investigators to diffusely express myogenin in the majority of nuclei of neoplastic cells by immunohistochemistry, whereas nuclear MyoD1 expression, although consistently present, is patchy and less intense. Conversely, human embryonal RMS has few cells expressing either myogenin or MyoD1. 19,39,60,109 The different IHC staining patterns between myogenin and MyoD1 were used to differentiate human alveolar RMS from embryonal RMS. 127,146 Although immunohistochemical identification of myogenin and MyoD1 is a useful tool in human medicine, more specific tools have been developed that are in the process of supplanting these methods.
Dog, embryonal rhabdomyosarcoma (RMS). This is a biphasic embryonal RMS with round myoblast cells in the upper left corner and a more spindyloid population in the lower right with a very myxomatous stroma. Hematoxylin and eosin (HE).
Comparison of Myogenin and MyoD1 Staining in Different Categories of RMS Between Human and Canine Neoplasms

Alveolar rhabdomyosarcoma (RMS) tends to have uniform and more abundant myogenin staining compared with embryonal RMS. MyoD1 staining is present is both but sparse. In dogs, both alveolar and embryonal RMS stains with myogenin and MyoD1, but staining can be sparse, and there is no proven difference between the two. In canine botryoid RMS, myogenin stained the myotube cells preferentially, and MyoD1 stained the round cells preferentially. 60,68,121,127 NA, not applicable.
Precise correlations cannot yet be made between human and canine RMS using myogenin or MyoD1 staining. One study of canine botryoid RMS found myogenin IHC stained elongated strap cells preferentially, whereas immunohistochemical detection of MyoD1 was limited to the nuclei of small, round myoblast cells. 67 In published case reports, both myogenin and MyoD1 have been used to diagnose canine RMS, but there have been no differences in the staining pattern or intensity described.21,25,35,62,67,100,140 Myogenin staining can also be seen in large round rhabdomyoblastic cells of embryonal RMS in dogs (Fig. 12). These cells often contain large amounts of eosinophilic cytoplasm but may be mixed with smaller cells that resemble undifferentiated myoblast cells. In dogs, MyoD1 immunostaining of embryonal RMS is rare and sometimes nonexistent (B. G. Caserto, unpublished data), but MyoD1 staining can be observed in alveolar RMS more frequently (Fig. 13). At present, there is insufficient information to conclude whether myogenin or MyoD1 is useful in subclassifying RMS into alveolar or embryonal categories.
Histogenesis and Cytogenetics
Many different hypotheses have been proposed regarding the histogenesis of RMS in all species. RMS of any species may develop from pluripotent stem cells from the primitive urogenital ridge remnants (in the case of botryoid RMS) or from mesenchymal progenitor cells invading the Müllerian and Wolffian ducts, either locally or through the circulation originating from the bone marrow stroma. 58,91 The presence of mesenchymal stem cells capable of myogenic differentiation in the bone marrow and elsewhere in the body may explain why RMS can be found in tissue with no skeletal muscle. 58 Alternatively, RMS can develop from early myogenic progenitor cells remaining from embryonic development or from myoblast cells within the pool of satellite cells. Using a mouse model, Nitzki et al 105 suggest that early mesenchymal stem cells (embryonic cells uncommitted to myogenesis) are the major source of embryonal RMS. This view is contradicted by others who have suggested that embryonal RMS develops from muscle progenitor cells, or skeletal muscle stem cells present within the pool of satellite cells, because embryonal RMS in mice contains a high number of muscle stem cells. 58 It has been suggested that the pleomorphic RMS subclass is derived from adult skeletal muscle, but whether they are derived from satellite cells or other cell populations in the adult muscle was not mentioned. 125
Cytogenetics of RMS in Domestic Animals
Very little is known about the cytogenetics of RMS in dogs and other domestic animals when compared with knowledge of human RMS. Although a tumor cell line was established from a pleomorphic RMS from a 14-year-old mixed breed dog, no cytogenetic abnormalities were reported. 8 One report described single or multiple RMS developing in 25 of 100 piglets from multiple litters over a 6-month period in the Netherlands, suggesting an inherited component. 157 van der Loop and colleagues established a cell culture from a pleomorphic RMS in a 3.5-week-old female piglet and described a deletion of the long arm of the X chromosome (Xq24-qter) in these cells. 154
Cytogenetics of Human RMS
Human embryonal RMS has been associated with abnormalities of chromosome 11p15, leading to loss of maternal information and duplication of paternal genetic information (loss of heterozygosity or LOH). The IGF-II gene is normally expressed by the paternal allele only, so the loss of the maternal allele in these tumors with a duplication of the paternal allele leads to a 2-fold increase of IGF-II, leading to uncontrolled cell cycle progression and cell proliferation. 36 Embryonal RMS can also have DNA contents ranging between diploid and hyperdiploid, with diploid tumors having a worse prognosis than hyperdiploid tumors. 36
Human alveolar RMS has a characteristic translocation between the long arm of chromosome 2 and the long arm of chromosome 13: [t(2;13)(q35;q14)]. This mutation fuses the PAX3 gene (Paired box family) with the FKHR gene (Forkhead box family). A variant of the mutation, t(1;13)(p36;q14), fuses the PAX7 gene from chromosome 1 with the FKHR gene and leads to more aggressive neoplasms in younger children. These mutations are detectable in 70% to 90% of histologically diagnosed alveolar RMS. 2,36,60,127,136
Polymerase chain reaction (PCR) identification of mRNA transcripts of fused genes (PAX3 or PAX7-FKHR) can identify and distinguish human alveolar RMS from embryonal RMS. 136,160 This is a rapid diagnostic test that can detect the mutation in cases identified by histology as alveolar RMS. In addition, studies of alveolar RMS lacking this mutation (fusion-negative alveolar RMS) found that the biologic behavior was similar to embryonal RMS and, like embryonal RMS, showed high levels of expression of genes from chromosome 8. The biological behavior of fusion-negative alveolar RMS is distinct from fusion-positive alveolar RMS, indicating that histologic pattern is not as specific a predictor of biologic behavior as cytogenetic studies. 160 It was concluded that the presence of the PAX-FKHR mutation is a significant factor in the aggressive phenotype of human alveolar RMS. Using this method to help classify human RMS is especially helpful in cases where there is lack of distinct histologic features. 160 It is becoming more common in human medicine to test for these mutations using reverse transcription PCR (RT-PCR) and making the diagnosis in conjunction with a biopsy. 145,160
Proposed Molecular Pathogenesis of RMS
Human alveolar RMS and embryonal RMS are thought to arise by a multistep process leading to loss of tumor suppressor genes and genes affecting the regulation of apoptosis and cellular senescence. 79 Recent molecular studies have suggested that alveolar RMS represents an arrested stage of development in undifferentiated myoblast cells. 165 The cause of the arrested stage of development could be related to the PAX-FKHR mutation; under certain circumstances, increased expression of PAX inhibits myogenic differentiation in mesoderm cells. 43 The fusion of the PAX and FKHR genes leads to uncontrolled cell growth signals, cell cycle progression, failure of tumor suppressor gene function, and myogenic differentiation. Fusion of PAX-FKHR causes increased levels of MyoD1, resulting in cell proliferation and also increased levels of myogenin, leading to myogenic differentiation. Increased levels of IGF-II also result in cell proliferation. 163 Human alveolar RMS has been associated with increased expression of c-Met receptor and CXCR4, both of which enhance cell growth and cell cycle progression. 87
Studies of the mRNA transcripts and in vitro studies of transfected cultured myoblasts have discovered increased activity of MDM2, CDK4, and cyclin D. 163,169 A recent study of in vitro mouse mesenchymal stem cells demonstrated the ability of the PAX-FKHR translocation to produce alveolar RMS when coupled with loss of RB and p53 tumor suppressor gene function and constitutive H-RAS activation. 120 In normal cells, CDK4 and cyclin D form a complex that begins a cell’s progression through the G1 phase of the cell cycle. Uncontrolled accumulation of CDK4 and cyclin D lead to uncontrolled cell cycle progression through phosphorylation of RB. RB is a tumor suppressor protein normally present in the nucleus bound to E2F, thereby preventing transcription of mRNA needed for cyclin production and cell cycle advancement beyond the G1 phase. When RB is phosphorylated, E2F is freed, leading to transcription of cyclins needed for cell cycle progression. If H-RAS is constitutively active as in this mouse model, 120 RAS continually sends positive signals for cell cycle progression. These continuous growth signals, coupled with the inability to halt the cell cycle or undergo apoptosis, leads to neoplastic growth. 169,170
P53 is an important regulator of cell senescence and apoptosis. It senses DNA damage and can halt cell cycle progression, leading to either senescence or apoptosis. MDM2 is responsible for inactivating p53, thereby possibly preventing apoptosis in these cells. 163,170 Inactivation of P53 coupled with uncontrolled cell cycle advancement results in neoplastic growth. There are more alterations in cell senescence and growth found in human and mouse models of RMS. Mirk expression is elevated in human RMS. Normal functions of Mirk range from cell senescence in the G0 phase to responses to cellular injury and reduction in apoptosis. Increased expression of Mirk in RMS is thought to result in reduced apoptosis and increased survival of all RMS variants. 90 Alterations in other tumor suppressors have also been found. Micro-RNAs act as tumor suppressors and are downregulated in human RMS. 124
Furthermore, other studies of transfected cultured pediatric RMS and mouse mesenchymal stem cells have demonstrated that the PAX-FKHR mutation is necessary for myogenic differentiation in alveolar RMS but in itself is not sufficient to produce a neoplasm without additional mutations. Consequently, multiple mutations over several generations are needed to produce a sarcoma with the phenotype of alveolar RMS. 58,165 Further study is needed to better clarify the origin of RMS in human and veterinary medicine.
Currently, there are so few cases of canine RMS that there is no information available regarding molecular pathogenesis of this neoplasm. Although genetic mutations may prove useful in future diagnosis of canine RMS, the relative rarity of this neoplasm and the difficulty in making a diagnosis make genomic analysis less useful as a diagnostic tool. At present, immunohistochemistry is the most feasible adjunct to standard histopathology for diagnostic purposes. The diagnostic utility of myogenin and MyoD1 immunohistochemistry can be investigated from archived cases and be readily applied to diagnostic laboratories throughout the country.
Pathogenesis of Metastatic Disease in Human RMS
Mutations in the oncogene FGFR4 have been found in 7.5% of human alveolar and embryonal RMS. 80 This mutation is associated with advanced stage of disease in alveolar RMS in particular, whose uncontrolled expression leads to cell cycle progression and has a role in metastasis that is not well defined. 143 Interleukin-4 (IL-4) helps recruit myoblasts to form myotubes in normal mammalian myogenesis and has been associated with childhood and mouse RMS differentiation, mitogenesis, and metastasis. 59
Immunohistochemical identification of receptors downstream of Hedgehog signaling such as Smoothened (SMO) and glioblastoma transcription factor 2 (GLI2) are common in human RMS and are the result of increased Hedgehog signaling in these tumors. In fact, inhibition of SMO by cyclopamine slows the growth of RMS cell lines in vitro. Inhibition of GLI1 by GANT61 (inhibitor of Hedgehog signaling by interfering in DNA binding with GLI1) also slowed growth of in vitro RMS cell lines. 63,148 Inhibitors of Hedgehog signaling have been suggested as a therapeutic method for human gliomas and may be useful in the treatment of RMS as well. 29
In patients with distant metastasis, there is high expression of FOXF1 (Forkhead family of transcription factors) and LMO4 (LIM domain transcription factor), which are thought to aid in tumor cell migration. 4 Overexpression of MET by some tumors may provide RMS cells with the same property as embryonal myoblasts to migrate into surrounding connective tissues. 36
Snail1 is highly expressed in alveolar RMS, E-cadherin is downregulated, and expression of MMP2 and MMP9 is upregulated in alveolar RMS. 117 Because of Snail1’s known roles in the metastasis of other tumors, it is presumed, yet unproven, to aid in metastasis of RMS. The role of E-cadherin and MMP2 in the pathogenesis of RMS and metastasis is less well known. Loss of E-cadherin is thought to reduce sensitivity of cell growth to external factors such as contact with other cells or the extracellular matrix. Increased amounts of matrix metalloproteases presumably aid in cell migration, local invasion, and metastasis. 59,117
Case Reports in Veterinary Literature
Frequency of Published Cases
The frequency of (nonlaryngeal/noncardiac) canine rhabdomyosarcoma in veterinary medicine is low, with 65 total case reports published (Table 4) and available by a PubMed search and in the textbook Tumors of Domestic Animals. 31 Pleomorphic RMS is the least common (n = 2; 3%), 8,31 and botryoid RMS is the most common (n = 28; 43%),3,11,31,47,54,64,65,68,73,83,108,115,131,138,140,141,144,155 followed by embryonal RMS (n = 15; 23%).7,31,35,61,62,65,153,166,167 Unclassified RMS (RMS NOS) is prevalent in the literature (n = 13; 20%),14,16,21,49,74,75,99,102,103,113,162 and probably reflects the lack of knowledge of RMS classification in veterinary medicine. Alveolar RMS (n = 7; 11%) is less frequently diagnosed in canines second only to pleomorphic RMS.11,31,65,100,125,128,134 In human medicine, embryonal RMS and alveolar RMS are by far the most common tumors in children. 78
Veterinary Case Reports and Literature Review of Canine Rhabdomyosarcomas (Noncardiac)
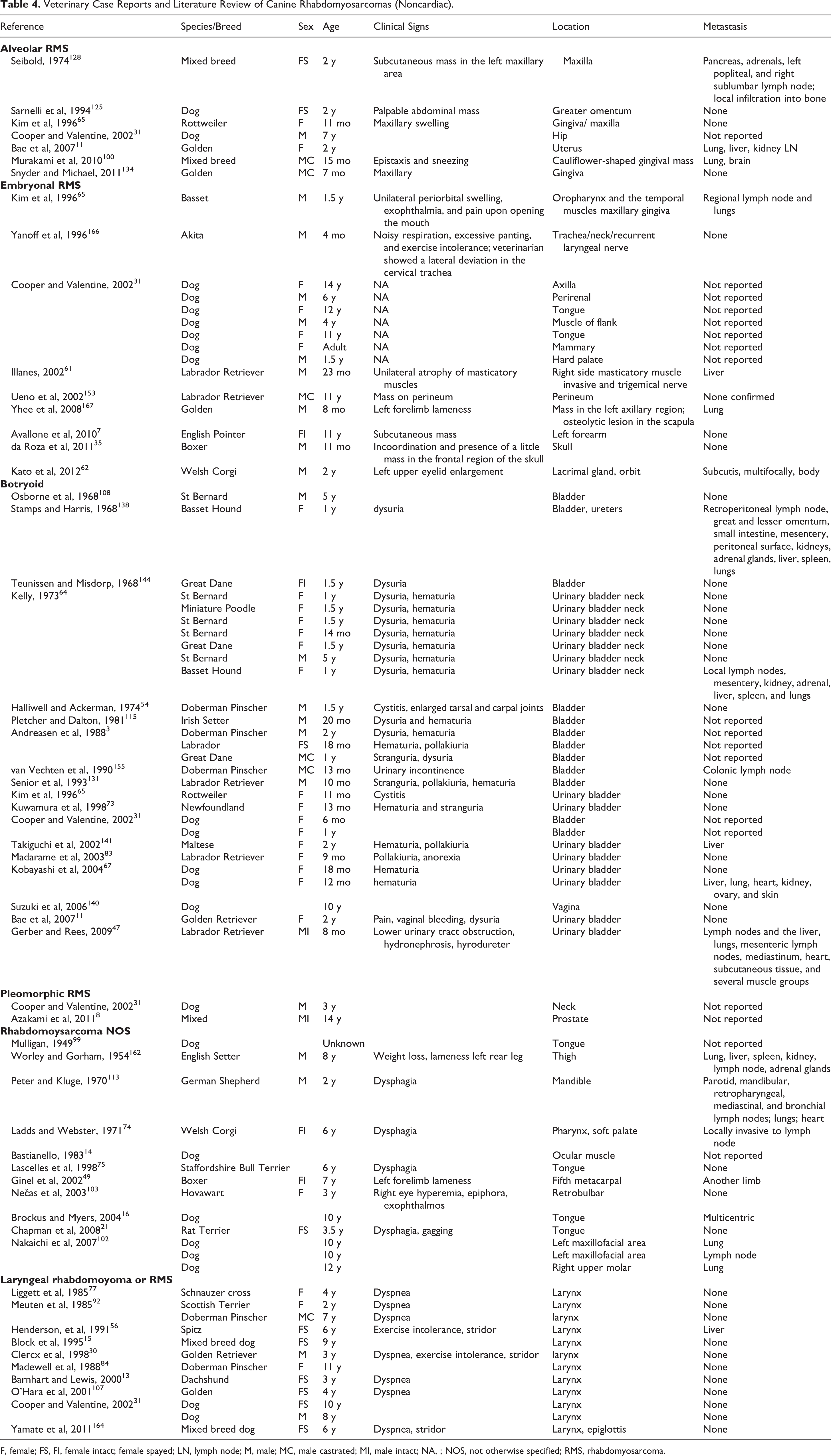
F, female; FS, FI, female intact; female spayed; LN, lymph node; M, male; MC, male castrated; MI, male intact; NA, ; NOS, not otherwise specified; RMS, rhabdomyosarcoma.
Twelve laryngeal rhabdomyomas/rhabdomyosarcomas have been reported in dogs, all but one in dogs older than 2 years. It is generally believed that laryngeal rhabdomyomas/rhabdomyosarcomas are a distinct clinical entity of dogs, being locally invasive yet rarely metastatic.13,15,30,31,56,77,84,92,107,164,166 Complete excision is often difficult due to local invasion, and recurrence of these tumors often leads to euthanasia. Histologically, these neoplasms contain large cells with granular eosinophilic cytoplasm containing large amounts of periodic acid-Schiff (PAS)–positive glycogen and mitochondria. Although some have been diagnosed as oncocytomas based on ultrastructural findings of large numbers of mitochondria, laryngeal rhabdomyomas can be distinguished from oncocytomas by positive staining for myoglobin and desmin or by the ultrastructural presence of myofibers in addition to glycogen and mitochondria. 76,92,161
Cutaneous rhabdomyomas are rarely reported. Two cases of cutaneous rhabdomyoma were reported by Copper and Valentine, 31 one occurring in the skin and subcutis of a leg and the other in the skin of a foot in 11-year-old and 14-year-old dogs, respectively. Cutaneous RMS can occur in the dermis or subcutis of humans. 86 These occur mostly in adults and are diagnosed as pleomorphic RMS. Those that occur in children are usually embryonal and alveolar RMS. In 1 report, 6 of 12 cases metastasized. 86 Primary cutaneous RMS can occur in dogs, and some cutaneous RMS that occurs in the dermis is likely misdiagnosed as anaplastic or poorly differentiated soft tissue sarcoma if immunohistochemical studies are not pursued. Despite being excluded from the category of soft tissue sarcomas by some investigators, 37,72,130 others have included them in their soft tissue sarcoma category on the basis of similar behavior. 156 Some subcutaneous masses undoubtedly arise from muscles of the appendicular skeleton but invade superficially, making determination of origin difficult from a biopsy.
Cardiac muscle tumors are rare in dogs, but the majority of those reported and available through PubMed are RMS (6/12), 1,6,52,69,114 3 of which had metastasized. Four (4/12) cardiac rhabdomyomas are reported, 50,66,85,119 as well as 1 myocardial hamartoma of the right atrium. 81 One malignant mixed mesenchymal tumor (mesenchymoma) with rhabdomyosarcomatous differentiation was reported in the heart of an 8-year-old female Pug. 82
One rhabdomyoma of the tongue was reported in a 9-year-old, castrated male, mixed breed dog. 122 One mesenchymoma with rhabdomyosarcomatous differentiation was reported in the spleen of a dog. 159 There are 15 reports of cats with RMS,20,31,57,88,96,98,110,132,137 8 in sheep, 41,106,142 and 15 in the horse.18,28,44,45,55,147,151 Feldman recounts 1 rhabdomyoma in the shoulder of a 13-year-old horse. 44
In addition to the skeletal muscle RMS mentioned in pigs, cardiac rhabdomyomas are found in pigs more often than in other species. A peculiar tumor of cattle appears histologically as cuboidal epithelioid cells arranged in glandular acini in a collagenous stroma. These have been termed myocardial epithelial inclusions or myocardial adenomatoid tumors. 12,135,152 They are hamartomas composed of pericardial mesothelial inclusions.
Poorly differentiated sarcomas are occasionally reported in the literature. These microscopically resemble embryonal tumors, with small cells and scanty cytoplasm, and have hyperchromatic nuclei. Immunohistochemical studies often do not include myogenin or MyoD1 but are negative for desmin. 33 Young age, or genitourinary location, should indicate a possible RMS despite negative desmin staining. Increased availability of myogenin, MyoD1, or other primitive myogenic markers may help diagnose alveolar and embryonal RMS in these circumstances.
In a single case report, 16 a dog developed multiple rhabdomyosarcomas in multiple locations that were categorized as embryonal RMS at one location and alveolar RMS at another. It is unclear whether these neoplasms arose separately or are the product of a metastatic process and subsequent divergence in phenotype. Kim et al 65 diagnosed an alveolar RMS in the gingiva of an 11-month-old Rottweiler, and upon necropsy, although no metastasis was reported, a botryoid RMS was diagnosed in the urinary bladder. These tumors were considered to arise independent of each other. A report of orbital embryonal RMS in a dog described metastasis to the cervical region with a histologic pattern of alveolar RMS. 62 Clearly, these incidences warrant further investigation into the diagnosis, pathogenesis, and the identification of prognostic markers of RMS in dogs. In the meantime, comparison to human RMS pathogenesis and prognosis should be made with caution.
Clinical Features of Rhabdomyosarcomas
Human Rhabdomyosarcoma
Childhood rhabdomyosarcoma primarily affects children younger than 15 years19,36,47,58,60,101,109,116 and comprises about two-thirds of soft tissue sarcomas in children. 146 Because of the significant difference in survival times in humans between alveolar and embryonal rhabdomyosarcomas, it is necessary for human pathologists to reliably differentiate the two, even when both appear as small round cell neoplasms. With current chemotherapy treatments, the 5-year survival of embryonal RMS in children is about 67% to 70%, whereas for alveolar RMS, it is about 40% to 49%. 116,158 Age and location also have prognostic implications. Children 1 to 4 years old have a 5-year survival of 77%, and for those with localized disease, it is even higher at 83%. 116 Orbital and genitourinary locations have a 5-year survival of 86% and 80%, respectively. 116 Poor prognosis is associated with RMS diagnosed at infancy or adolescence (47%-48%, respectively) and locations including the limbs (50%), trunk (52%) and retroperitoneum (52%). 116
In humans, there is slightly better survival with embryonal histology (67%) compared with alveolar histology (49%). Lowest 5-year survival is correlated to metastasis at the time of presentation (31%) and alveolar histology (49%). 116 Prognosis of patients with regional lymph node metastasis is similar to distant metastasis in alveolar RMS but not embryonal RMS. 123 Botryoid rhabdomyosarcomas in humans are locally infiltrative and are second only to the less common spindyloid variant embryonal RMS in favorable prognosis. 94 Most human embryonal rhabdomyosarcomas fall into the intermediate risk category (55%), with a 3-year survival of 59% to 85% 94 and a 5-year survival of 67%. 116 In contrast, even though spindyloid RMS comprises 3% of all RMS, it has a 95% survival at 5 years. Survival of people with alveolar RMS ranges from 49% to 66% at 5 years. 94,116 Embryonal and botryoid RMS are frequently reported to have lower metastatic rates than alveolar RMS, but precise figures are not often reported. 118 Only about 15% to 20% of patients have clinically detectable distant metastasis at the time of diagnosis, but all are considered to have micrometastatic disease. 94
Canine Rhabdomyosarcoma and Soft Tissue Sarcomas
Rhabdomyosarcomas are occasionally grouped together within the broad category of canine soft tissue sarcomas, 89 yet they often have a different clinical signalment and location. Soft tissue sarcomas graded by the Kuntz et al 72 system are restricted to subcutaneous neoplasms and exclude hemangiosarcomas and rhabdomyosarcomas. Table 5 compares the biological behavior of canine RMS and soft tissue sarcomas. The most significant difference between the two is the age of onset, location, and survival.
Comparison of Signalment and Biologic Behavior of Canine Rhabdomyosarcoma (RMS) and Canine Soft Tissue Sarcomas
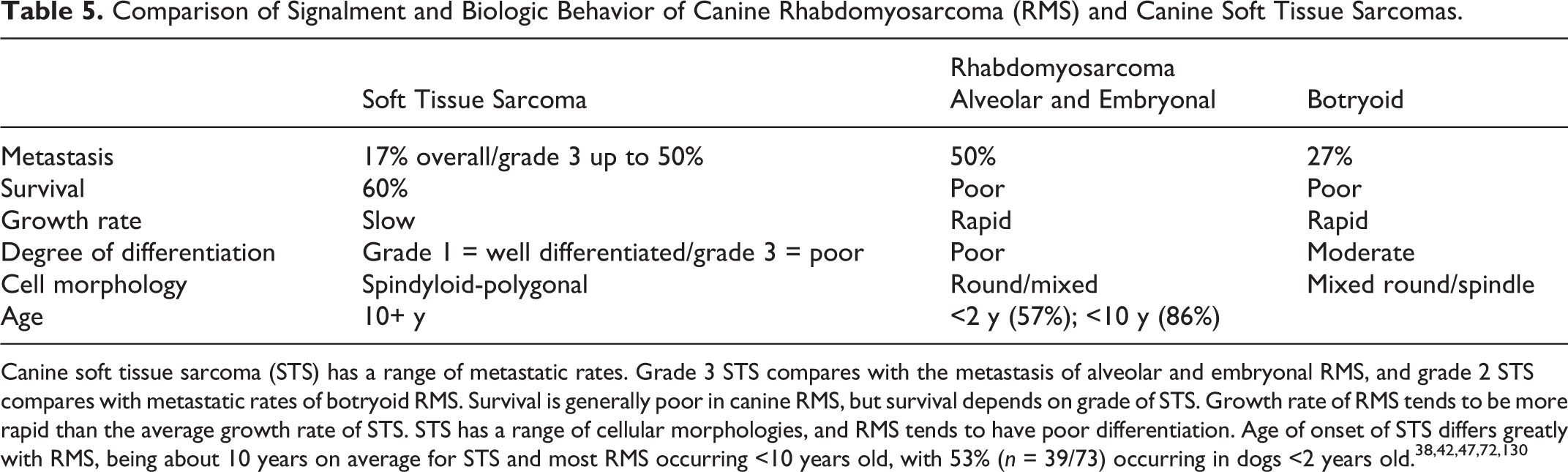
Canine soft tissue sarcoma (STS) has a range of metastatic rates. Grade 3 STS compares with the metastasis of alveolar and embryonal RMS, and grade 2 STS compares with metastatic rates of botryoid RMS. Survival is generally poor in canine RMS, but survival depends on grade of STS. Growth rate of RMS tends to be more rapid than the average growth rate of STS. STS has a range of cellular morphologies, and RMS tends to have poor differentiation. Age of onset of STS differs greatly with RMS, being about 10 years on average for STS and most RMS occurring <10 years old, with 53% (n = 39/73) occurring in dogs <2 years old. 38,42,47,72,130
RMS is often reported as appearing most often in younger dogs, 2 years old or younger.§§ A survey of the reported cases of nonlaryngeal RMS indicates that 63% (n = 39/62) occurs in young dogs (≤2 years old) (Fig. 14). In dogs, 89% (n = 25/28) of botryoid RMS occurs in dogs 2 years old or younger. Similarly, in alveolar RMS, 86% (n = 6/7) occurs in young dogs. By contrast, only 47% of embryonal RMS occurs in dogs 2 years old or younger. Only 8% (n = 1/13) of RMS NOS occurs in young dogs, and no reported cases of pleomorphic RMS have occurred in young dogs. Although this seems to correspond to the high prevalence of this subclass in human adults, the frequency of this diagnosis is very low (n = 2) (Fig. 15).
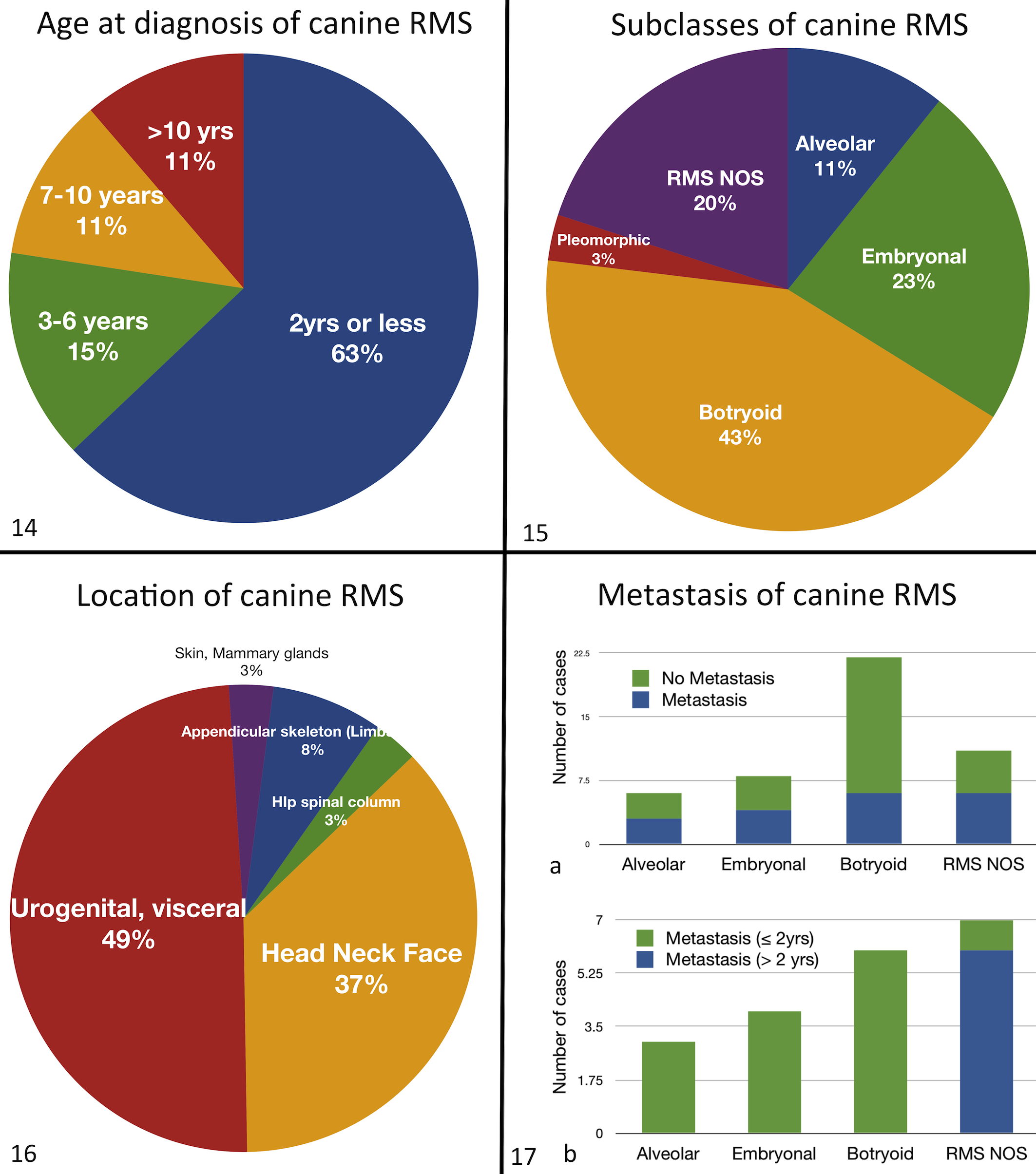
Age of diagnosis of canine rhabdomyosarcoma (RMS). A literature search shows 63% (n = 39/62; in 2 cases, the age was not reported) of canine RMS (excluding laryngeal rhabdomyomas/rhabdomyosarcoma) occurs in dogs younger than 2 years. The remaining 37% is divided between animals 3 to 6 years old (n = 9/62; 15%), 7 to 10 years old (n = 7/62; 11%), and >10 years old (n = 7/62; 11%). Eighty-nine percent (n = 55/62) of cases occur in dogs younger than 10 years.
Soft tissue sarcomas arise mostly in middle-aged to older dogs and occur in the skin and subcutis of the trunk and extremities and oral cavity. 38,42 RMS occurs often in the urogenital tract (n = 32/65; 49%), and these are most frequently botryoid RMS of the urinary bladder. The second most common location is the head, neck, and face (n = 24/65; 37%), and a small percentage occurs in the hip/pelvis/spinal column (n = 2/65; 3%), appendicular skeleton (n = 5/65; 8%), and skin (including mammary gland) (n = 2/65; 3%) (Fig. 16).
It is difficult to compare metastatic rates between canine RMS subgroups because in some cases, these data were not reported or euthanasia was elected at the time of diagnosis. In veterinary medicine, the prognosis depends on the severity and extent of invasiveness, as well as the presence of metastatic disease at the time of diagnosis. In cases of extensive local infiltration or radiographic evidence of metastatic disease, euthanasia may be sought instead of radical surgery or adjunct therapy. 31,65 Metastasis reported at the time of RMS diagnosis or at necropsy shows that metastatic rates of RMS in dogs is highest in RMS NOS (n = 6/11; 55%), followed closely by embryonal RMS (n = 4/8; 50%) and alveolar RMS (n = 3/6; 50%). Botryoid RMS has the lowest metastatic rate (n = 6/22; 27%) (Fig. 17). In reported literature, botryoid RMS is reported to rarely metastasize. 65,149 There are no data on metastasis of pleomorphic RMS (n = 2). It is interesting that of the metastatic cases of canine botryoid, embryonal, and alveolar RMS, all are ≤2 years old, whereas the metastatic cases of RMS NOS are mostly older than 2 years (Fig. 17).
High-grade soft tissue sarcomas have similar rates of metastasis and recurrence to reported incidence of canine RMS and therefore may carry the same prognosis. Kuntz et al 72 reported the metastatic rate of grade 3 soft tissue sarcomas without adjuvant therapy as 46% (ranging from 41%–50%), which is roughly equivalent to embryonal RMS, alveolar RMS, and RMS NOS but is greater than botryoid RMS. 130 Grade 1 and 2 soft tissue sarcomas fare better at 11% and 19%, respectively. In addition, high-grade soft tissue sarcomas may be poorly differentiated, resembling alveolar and embryonal RMS, and yet may also resemble pleomorphic RMS with numerous cells having bizarre mitotic figures and having cells with a high degree of anisocytosis and anisokaryosis. 42,72 Drawing conclusions from these data is difficult because of the paucity of clinical cases, lack of follow-up and survival data, and in many cases unknown frequency of metastasis. Improved diagnostic techniques for RMS may lead to more diagnosed cases and better data on survival and prognosis.
Summary
Rhabdomyosarcomas are a diverse and histologically variable set of highly malignant neoplasms occurring predominately in younger individuals. Subclassification has been attempted based on histologic morphology, but these classification schemes in human and veterinary medicine fall short when dealing with very homogeneous cell populations with no particularly identifying characteristics. Identification of particular gene mutations is useful in human medicine and may become the standard for distinguishing between alveolar and embryonal tumors. The use of IHC to identify desmin, α-actins, myogenin, and MyoD1 and electron microscopic identification of sarcomeric structures can be used to identify RMS in relatively undifferentiated neoplasms. In veterinary medicine, much work needs to be done to evaluate the prognostic significance of subclassifications and to identify new methods to characterize these neoplasms to make classification effective and useful for veterinary oncologists and pathologists. Further investigation is needed to better understand the biology of these neoplasms that may aid in diagnosis and prognosis. Unless veterinary medicine can identify prognostically significant markers or mutations in dogs, the classification system currently used may continue to yield poorly prognostic diagnoses.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
