Abstract
Congenital ocular abnormalities in cervids have been previously reported as individual cases from various regions of the United States and include microphthalmia, anophthalmia, congenital cataracts, dermoids, and colobomata. A common underlying cause for these abnormalities, such as nutritional deficiencies, environmental toxin exposures, or genetic mutations, has not been established. This retrospective study summarized and compared cases of suspected congenital ocular abnormalities in free-ranging white-tailed deer (Odocoileus virginianus) submitted to the Southeastern Cooperative Wildlife Disease Study (SCWDS) in Athens, Georgia, to the preexisting literature. Of 3645 accessions of white-tailed deer submitted to SCWDS, 15 qualifying case records were found. An additional 15 cases were reported previously in the literature. Conditions described in SCWDS cases included microphthalmia (8/15), congenital cataracts (3/15), anophthalmia (2/15), colobomata (1/15), anterior segment dysgenesis (1/15), ectopic lacrimal gland tissue (1/15), and congenital blindness with corneal opacity (1/15). Most (11/15; 73%) of the SCWDS cases were male fawns with an average age of 4 months at presentation, consistent with previously described cases. Most animals had bilateral abnormalities with few extraocular congenital abnormalities, also consistent with existing reports. Cases were variably tested for various infectious agents at the time of submission; 2 cases were seropositive for bluetongue virus. Spatiotemporal clustering of cases was not evident. This study provided a concise and systematic summary of known existing cases of congenital ocular defects in fawns but did not identify a cause.
Congenital ocular defects, including microphthalmia, anophthalmia, congenital cataracts, and colobomata (Table 1), have been well documented in domestic ruminants, particularly sheep and cattle. 5,13,16,26,29,32 There are several suspected viral 2 and nutritional 27 causes for these defects, and in some cases, specific genetic mutations or breed predispositions have been identified. 1,5,8,26,29 These defects can be the result of local disruption of eye development in utero or part of a more generalized developmental anomaly. 29 Microphthalmia and anophthalmia result from the partial to complete failure of formation of the primary optic vesicle or its early regression. Microphthalmia is often accompanied by various other intraocular anomalies, including congenital cysts, aphakia, congenital cataracts, and intraocular cartilage. 7,29 In cattle, the primary infectious agent associated with these conditions is bovine viral diarrhea virus. 2,29 Colobomata can form in many parts of the eye but typically result from a failure of optic fissure closure, most commonly seen at the caudal pole of the optic disc. 7,29 This defect results in an outpouching of dysplastic neuroectoderm that creates a bulge in the sclera. Colobomata are often associated with choroidal hypoplasia (resulting from induction failure by a defective retinal pigmented epithelium), segmental or diffuse iris hypoplasia, or ciliary hypoplasia. 7,29
Glossary of Terms for Congenital Ocular Conditions.
In humans, the birth prevalence of anophthalmia is reported from 0.6 to 4.2 per 100,000 births, 2 to 17 per 100,000 births for microphthalmia, and 2 to 14 per 100 000 births for coloboma. 24 A wide array of monogenic changes and chromosomal abnormalities have been identified in humans, with SOX2 and OTX2 being the most frequently identified mutated genes in cases with monogenic causes. 20,24 Despite significantly more intensive studies in humans than in veterinary species, an underlying genetic cause for these abnormalities remains poorly understood, but the genetic component is thought to be heterogenous, leading to a spectrum of variable presentations of these congenital defects. These conditions may also be associated with a syndromic set of developmental defects or may be restricted to the ocular tissues. Overall, there is a broad spectrum of genetic conditions that can produce an appropriately broad spectrum of pathologic conditions, and the underlying genetic associations are incompletely characterized. Current opinions in human medicine propose that the detailed phenotypic characterization and classification of affected cases may better direct genetic etiology research. 24 Outside of genetic causes, environmental influences have frequently been suggested as a cause for sporadic cases of colobomata in humans, but very few specific embryopathic agents have been identified. Vitamin A deficiency is suspected of being a factor in the development of these anomalies, as its involvement in optic fissure closure and embryonic retinal development is well established. 23,27 In animal models, colobomatous and noncolobomatous microphthalmia have been induced in rats using vitamin A deprivation during pregnancy, folic acid deficiencies, irradiation, and exposure to nickel carbonyl. 20 Congenital cataracts, which are distinct from cataracts secondary to ocular injury postpartum, are also thought to be influenced by environmental factors that cause oxidative stress within the globe, potentially implicating vitamin E and A deficiencies, heavy metal toxicities, and irradiation. 13,27
Similar congenital ocular defects to those described in humans and domestic animals have been sporadically reported in wild cervids, but underlying causes remain unknown. Cases in white-tailed deer (WTD) have been most frequently reported in the literature, 7,9,12,15,25,31 but reports of affected red deer (Cervus elaphus) 10,19 and moose (Alces alces) 14 are available. Specific findings in WTD have included unilateral and bilateral anophthalmia and microphthalmia, 7,9,12,25,31 cataracts, 12 retinal dysplasia, 9,12,31 colobomata, 12 and corneoscleral choristomas. 15 Results of the literature search are summarized in Supplemental Table S1, including 15 animals described in 5 publications. 9,12,15,25,31 These sporadic reports are all either individual case reports or small case series, leading to an obfuscated understanding of the condition in this species.
The recognition and reporting of these congenital ocular defects in wild mammals, such as deer, will likely become more relevant and identified more frequently as human contact with wildlife increases with urban expansion. Examining existing cases may help to identify predisposing factors or potential contributing causes. In addition, most of the previous reports of congenital ocular defects in WTD have been from the northeastern (New York) 9,25,31 and midwestern (Wisconsin, Michigan) 7,12 United States. In the current study, our objective was to conduct a retrospective search of WTD necropsies with reported congenital ocular anomalies from the southeastern United States to systematically summarize and compare our findings to the preexisting literature.
Materials and Methods
Literature Search
The National Center for Biotechnology Information’s PubMed search engine was used to search for the terms congenital, blindness, microphthalmia, anophthalmia, coloboma, and dermoid in connection with white-tailed deer to identify previously published reports of similar cases. The year of publication, state of origin, approximate age, sex, and gross and microscopic findings were summarized when available.
Retrospective Case Search
The diagnostic database of the Southeastern Cooperative Wildlife Study (SCWDS) was searched for cases of white-tailed deer necropsies using the terms congenital, blindness, microphthalmia, anophthalmia, coloboma, and dermoid. Case reports were reviewed, and cases were identified that fit the search criteria. The state of origin, approximate age, sex, gross and microscopic findings, and ancillary testing for each case were compiled. Gross images and histologic slides of the eyes were reviewed by 2 board-certified anatomic pathologists (L.L.C. and H.F.), when available, to confirm report findings (Table 1). Eyes were not collected routinely for histopathology, and histology was not performed in all cases. Cases with histologic sections of eyes available also had gross ocular lesions or at least a history of the animal being blind and/or having neurologic signs.
Results
Sixteen cases of WTD with suspected congenital ocular abnormalities were diagnosed between 1976 and 2016 from a total of 3645 WTD accessions in the SCWDS database. Demographic data for these cases and diagnoses are summarized in Supplemental Table S2. One of these cases was previously published as a case report and will be discussed separately. 15
Of the remaining SCWDS cases identified, most (11/15; 73%) were male fawns from 1 day to 1 year of age at the time of presentation. The average age was 4 months. The lone corneoscleral choriostoma case that was previously reported from SCWDS was in a 1.5-year-old doe. 15 The location of origin and date of submission for the SCWDS cases revealed no obvious spatiotemporal trends, implying that all of these cases were separate, sporadic occurrences. Three of the SCWDS cases had concurrent extraocular congenital abnormalities (scoliosis, meningoencephalocele, or metatarsal deformities with piebaldism). In 2 SCWDS cases, only the head was submitted.
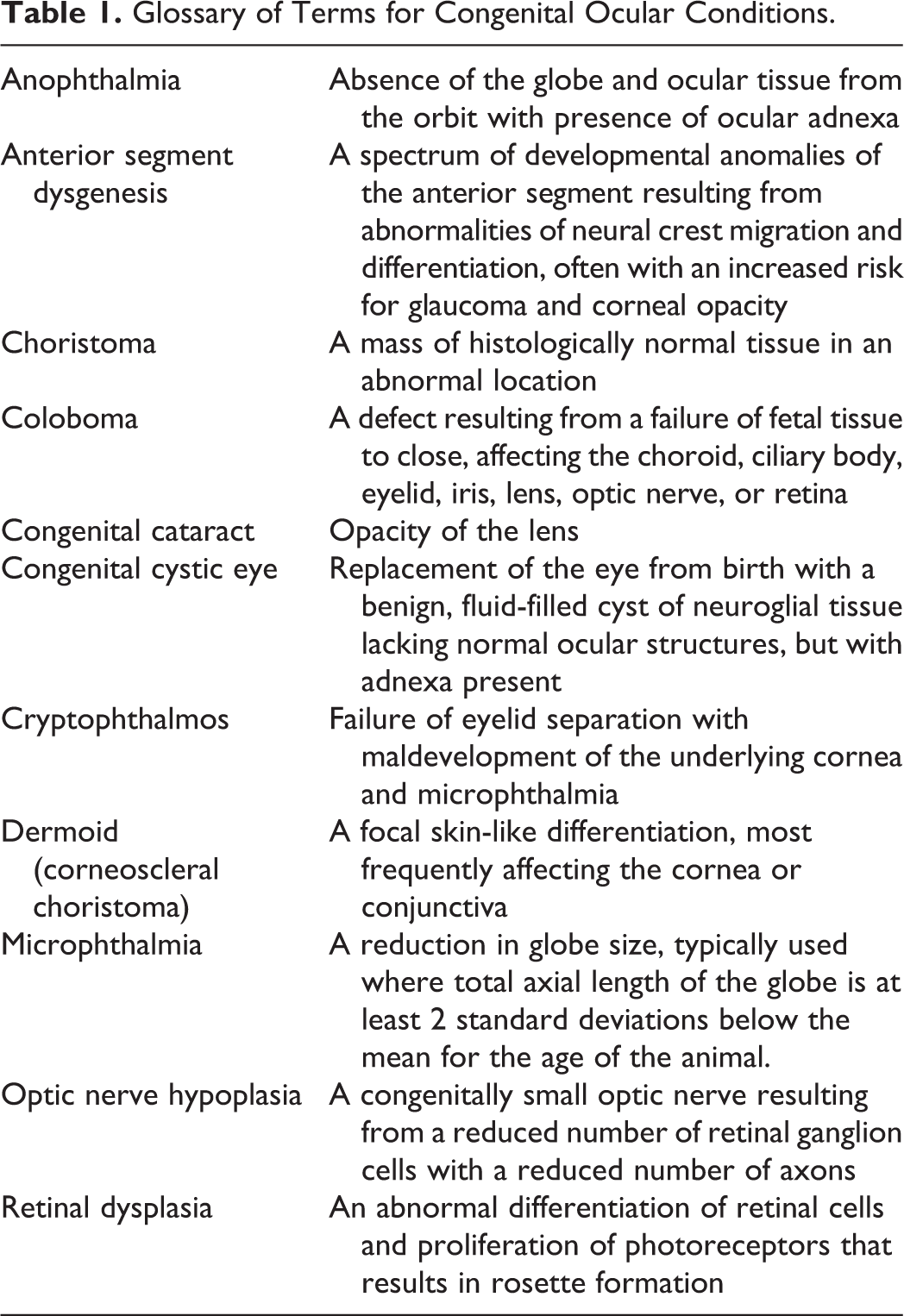
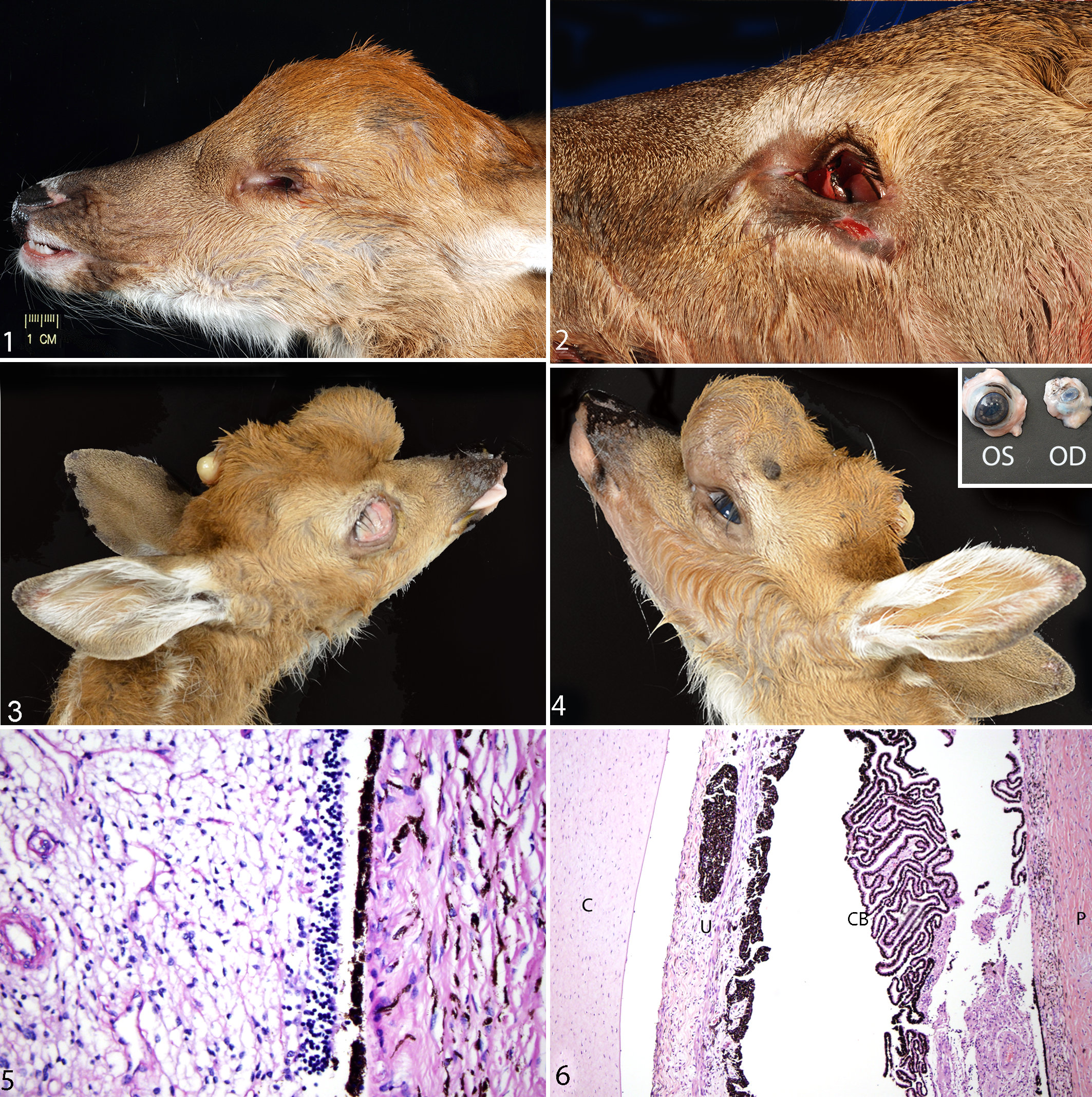
The most commonly reported condition was microphthalmia, which was described in 8 SCWDS cases. Gross descriptions of this condition include diffuse shrinkage of the globe (approximately 10%–50% smaller than normal), corneal opacities or dark brown pigmentation, a reduced optic nerve, and occasional defects of the bony rim of the orbit (Figs. 1, 2). Microphthalmia was bilateral (n = 6), unilateral with contralateral anophthalmia (n = 1), or unilateral with a normal-sized contralateral eye (n = 1). One of the unilateral cases had a concurrent meningoencephalocele and subcutaneous epithelial cyst (Figs. 3, 4). Microscopic lesions included retinal dysplasia, aphakia, intraocular lacrimal gland and cartilage tissue, generalized anterior segment dysgenesis, and optic nerve hypoplasia (Figs. 5, 6). One case of bilateral microphthalmia had mild lymphocytic ophthalmitis and meningitis.
Testing for infectious diseases was done on several SCWDS cases, including immunohistochemistry for bovine viral diarrhea virus (BVDV), serology for epizootic hemorrhagic disease virus (EHDV), bluetongue virus (BTV), bovine herpesvirus-1 (BHV-1), parainfluenza virus-3 (PI-3), Brucella spp, and Leptospira spp, and virus isolation for EHDV and BTV. One fawn that had bilateral microphthalmia was seropositive for BVDV, PI-3, and BTV, and an orbivirus consistent with either BTV or EHDV was isolated from the spleen. Dam serum and tissues were unavailable for testing for all SCWDS cases.
Anophthalmia was recognized in 2 SCWDS cases, one bilateral and the other with contralateral microphthalmia. The fawn with bilateral anophthalmia was also diagnosed with midthoracic scoliosis. In both cases, the associated skeletal muscle, lacrimal glands, and eyelids were reported as normal.
Congenital cataracts were described in 3 SCWDS cases. One had a unilateral cataract with concurrent bilateral retinal dysplasia. This animal was serologically positive for both BTV and Leptospira interrogans. This was the only animal to have any notable changes to the retina or other ocular structures in addition to cataracts. One case was a piebald fawn with concurrent bilateral metatarsal deformities. Another animal with bilateral cataracts was tested for eastern equine encephalitis virus, West Nile virus, BTV, and EHDV by virus isolation; rabies virus by fluorescent antibody testing; adenovirus by polymerase chain reaction (PCR); and salmonellosis or bacterial sepsis by PCR and culture. All test results were negative in this case.
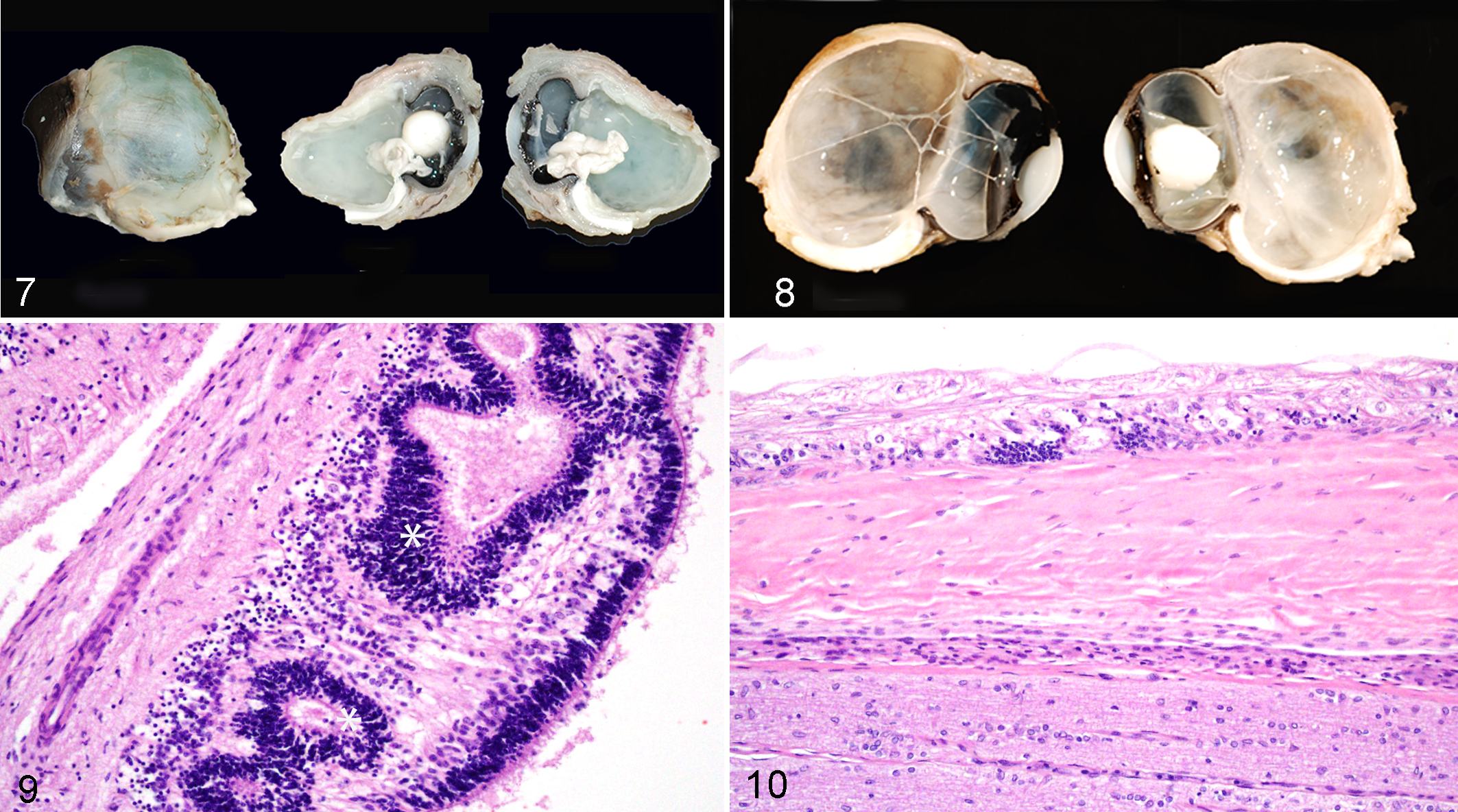
Single SCWDS cases were found for colobomata, anterior segment dysgenesis, congenital blindness with corneal opacity and a corneal defect in normal sized globes (no histologic examination was done), and the previously described corneoscleral choristomas. 15 The animal with colobomata also had scleral ectasia, retinal dysplasia, and choroidal hypoplasia (Figs. 7–10). There was a single case of normal-sized eyes that had partial to complete bilateral anterior segment dysgenesis with ectopic lacrimal gland tissue present in the ciliary body and in the root of the iris. Perivascular mononuclear cuffing and gliosis were identified in the brain of this animal. It was subsequently serologically tested for BTV, EHDV, and BVDV but was negative for all 3 viruses.
Discussion
The objective of this study was to systematically summarize the occurrence and description of assumed congenital ocular defects in WTD fawns submitted to SCWDS in comparison to preexisting reports of similar conditions. Over a 40-year period, a similar number of cases (16) of presumed congenital ocular defects in WTD were found at SCWDS compared with the number reported in the literature over the same time period (15). Demographic data for the cases examined were similar in several aspects, namely, that these conditions were largely recognized in very young animals and were described with relatively consistent macroscopic and microscopic pathologic findings. The estimated average age in previous reports varied more widely, from 2 weeks to 7 months (see supplemental tables). The exception to this was the 1.5-year-old doe previously reported with corneoscleral choristomas. 15 This doe was significantly older than the other fawns identified in this study, which may be attributed to the retention of some degree of vision, unlike many of the other defects described. The incidence of various conditions between the 2 sample sets was roughly comparable; microphthalmia was the most commonly identified defect, followed by congenital cataracts and anophthalmia. Colobomata and corneoscleral choriostomas were reported only as single cases. Most animals affected had bilateral involvement of ocular structures to a variable degree. Concurrent extraocular congenital anomalies were rare. Some cases, however, were poorly characterized because of the retrospective nature of the study. It should be noted that many of the changes described, particularly the retinal changes described in cases with dysplasia, could also be indicative of postinflammatory scarring and could occur concurrently with congenital anomalies. Given the young age of the animals affected, congenital ocular anomaly was the consensus diagnosis.
Of the cases identified at SCWDS, male fawns were more commonly affected (73%). This predilection was not evident in previously reported cases, where sex was identified in only 10 of 14 cases in young fawns and only 5 of those were reported as male (see supplemental tables). If the predilection of male WTD to develop ocular anomalies is verified, this may strengthen the evidence for a potential X-linked hereditary etiology for some anomalies, as has been described in humans. 20 A single herd case report of bilateral congenital cataracts in Ayrshire cattle in Northern Ireland also noted male predominance; however, a pattern of inheritance was not established. 13 A similar trend was seen in Swiss veal calves potentially exposed to radiation from mobile phone base stations, where a higher percentage of male calves developed cataracts. 11
Reports of congenital defects in deer are rare and are primarily restricted to skeletal abnormalities. 4,30 These anomalies are often associated with piebaldism, which is characterized by a white to patchy brown coat color and variable bowing of the nose (Roman nose), metacarpal and metatarsal bowing, scoliosis, and prognathia. 22 Ocular defects have not previously been reported with piebaldism to the authors’ knowledge. Only 1 of the SCWDS fawns in this study had piebald coloration. Piebaldism reportedly affects less than 1% of the WTD population, but this may vary regionally based on hunting restrictions on white or partially white deer.
Reports of congenital anomalies in other mammalian wildlife species are also uncommon, likely because of a combination of a low survival rate in affected animals and a lack of regular surveillance. A case report described 2 raccoons with microphthalmia. One of the raccoons had a concurrent meningoencephalocele and the other had a chorioretinal coloboma with retinal dysplasia and folding. 21 Congenital cataracts and corneal dermoids were reported in a 5-week-old camel. 17 Horses, particularly Quarter horses, have occasionally been diagnosed with colobomata, microphthalmia, and corneal dermoids. 6,18,28 In captive wildlife, multiple ocular colobomata have been documented in captive-bred snow leopards, many of which were surgically corrected. 3
Discussion of nonheritable causes for the congenital ocular defects described is largely limited by the retrospective nature of this study and sporadic spatiotemporal incidence of cases. SCWDS cases were tested for a variety of infectious diseases on a case-by-case basis. Two fawns were found to be serologically positive for exposure to BTV, and a hemorrhagic virus (either BTV or EHDV) was isolated from the splenic tissue of 1 of these fawns. BTV is an orbivirus known to cause profound corneal edema, blindness, and hydrocephalus in calves or hydranencephaly and retinal dysplasia in lambs infected in utero. 2,29 None of the previous reports of microphthalmia in WTD include any mention of infectious disease testing. More systematic testing of affected fawns and dams may be useful to identify further associations with infectious agents.
Noninfectious causes, specifically intoxication from agricultural chemicals, were a proposed cause for the development of cases of microphthalmia in WTD from central Wisconsin. 7 These cases were not included in our sample set as details of individual cases were not available. None of the SCWDS cases were tested for potential teratogenic chemicals, nor were vitamin A or E tissue levels obtained. These congenitally apparent anomalies could also be the result of a maternal insult affecting the fetus in utero; thus, future studies attempting to identify exposure to a potential causative agent would necessitate monitoring of does.
The findings of this report may be strongly influenced by its retrospective nature. Because of the need for specific fixatives and additional costs, eyes are not routinely collected for histopathology at SCWDS and were collected in the cases presented because of the presence of gross lesions. This implies that some number of cases with microscopic ocular lesions were likely missed by our case selection criteria. In addition, eyes may not have been initially examined upon submission because of poor preservation resulting from postmortem autolysis or freeze-thaw artifact. The frequent lack of descriptive histories in wild animals can lead to assumptions regarding causation. Many of the gross and histologic lesions described here could also result from traumatic or inflammatory ocular injury, evidence of which is not always readily recognized. Given the age of the animals described, a congenital defect was often assumed as the most likely cause. In addition, it is difficult to assess if the ocular defects described here are part of syndromic developmental abnormalities, as necropsy reports may be incomplete or concurrent conditions may not have been recognized at the time of necropsy. The combination of these factors emphasizes the necessity of a thorough postmortem examination and an awareness of these congenital ocular conditions in young fawns, potentially assisting in the recognition of additional cases.
In the lack of a clearly definable trend in the development of the congenital anomalies discussed, the potential for a multifactorial cause at the population level, combining both genetic and heritable traits with influence from environmental conditions, must be strongly considered. This hypothesis has been proposed in conjunction with congenital ocular anomalies in other species as well 11,24 and may explain the sporadic occurrence and rarity of these cases. As with many conditions in veterinary medicine, and wildlife population-level medicine in particular, increased surveillance and reporting will help to counteract the current dearth of information regarding congenital ocular defects in this species and potentially allow for better analysis of existing cases.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818759771 - Congenital Ocular Abnormalities in Free-Ranging White-Tailed Deer
Supplemental Material, DS1_VET_10.1177_0300985818759771 for Congenital Ocular Abnormalities in Free-Ranging White-Tailed Deer by Lorelei L. Clarke, Kevin D. Niedringhaus, K. Paige Carmichael, M. Kevin Keel, and Heather Fenton in Veterinary Pathology
Supplemental Material
Supplemental Material, DS2_VET_10.1177_0300985818759771 - Congenital Ocular Abnormalities in Free-Ranging White-Tailed Deer
Supplemental Material, DS2_VET_10.1177_0300985818759771 for Congenital Ocular Abnormalities in Free-Ranging White-Tailed Deer by Lorelei L. Clarke, Kevin D. Niedringhaus, K. Paige Carmichael, M. Kevin Keel, and Heather Fenton in Veterinary Pathology
Footnotes
Acknowledgements
Thank you to the University of Georgia histology laboratory for processing the slides evaluated in this study and to the Athens Veterinary Diagnostic Laboratory for performing the reported virus isolation, molecular, and serology assays. Thanks to Megan Kirchgessner, James Crum, and other SCWDS submitters for providing case materials. Thanks to Charlotte Quist, Sonia Hernandez, Jennifer Ballard, Janet Steiner, Ed Couvillon, Cynthia Tate, Elizabeth Howerth, Rick Gerhold, Victor Nettles, Kaitlin Fiske, and other SCWDS students and diagnosticians for providing necropsy reports.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the sponsorship of the Southeastern Cooperative Wildlife Disease Study by the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, Nebraska, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, United States. Support from the states to SCWDS was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
