Abstract
The roster of amdoparvoviruses (APVs) in small carnivores is growing rapidly, but in most cases, the consequences of infection are poorly understood. Red panda amdoparvovirus (RPAV) is highly prevalent in zoo-housed red pandas and has been detected in both healthy and sick animals. Clarifying the clinical impact of RPAV in this endangered species is critical, and zoological collections offer a unique opportunity to examine viral disease association in carefully managed populations. We evaluated the potential impact of RPAV in captive red pandas with a combination of prospective and retrospective analyses. First, we collected feces from 2 healthy animals from one collection over a 6-year period and detected virus in 72/75 total samples, suggesting that RPAV can be a long-term subclinical infection. We next investigated the infections using a retrospective study of infection status and tissue distribution in a cohort of necropsied animals. We performed polymerase chain reaction and in situ hybridization on 43 necropsy cases from 4 zoo collections (3 from the United States, 1 from Europe, 1997–2022). RPAV was present in these populations for at least 2 decades before its discovery and is detectable in common and significant lesions of zoo-housed red pandas, including myocarditis (3/3 cases), nephritis (9/10), and interstitial pneumonia (2/4). RPAV is also detectable in sporadic lesions, including multisystemic pyogranulomatous inflammation, oral/pharyngeal mucosal inflammation, and dermatitis. The colocalization of virus with lesions supports a role in causation, suggesting that despite the apparently persistent and subclinical carriage of most infections, RPAV may have a significant impact in zoo collections.
Parvoviruses are small (approximately 26 nm diameter), with an approximately 5 kb ssDNA genome encoding 2 open reading frames devoted to proteins for replication (2.2 kb) and capsid construction (2.3 kb).9,10,26 Their small size and simple genomic organization belies a diverse, successful, and ancient family of viruses with highly variable biological behavior and host impact.11,17 The severe and sometimes fatal disease caused by the protoparvovirus carnivore parvovirus-2 (CPV-2) is notorious and well-studied in several carnivores, but this type of acute, fulminant infection characterizes only a subset of the Parvoviridae. 17 Other, more insidious parvoviruses, such as the recently recognized mouse kidney parvovirus, can cause persistent or lifelong infections with consequences that do not coincide with initial infection, but can result in the gradual accumulation of damage over time. 29 A growing body of epidemiologic and pathologic evidence demonstrates that persistent parvovirus infections can have dire consequences for the health of the host.8,16,29
The prototypical and archetypical amdoparvovirus (APV), Aleutian mink disease virus (AMDV), is well known to cause persistent infections and a wide range of clinical outcomes that are modulated by host factors, such as immune status, genetics, stress, coinfections—and viral factors, such as genotype. 8 AMDV was discovered in the 1960s in mink housed for the fur industry and has been reviled for decades for its impact on commercial mink farming. Infections target a variety of tissues and cell types, resulting in a spectrum of acute and chronic clinical sequelae, including renal failure, vasculitis, neurologic disease, and/or systemic disease termed “fatal wasting syndrome.”4,13,28 In mink kits, permissive infection of pneumocytes results in abundant virus production, cytolysis, and fulminant, fatal, interstitial pneumonia; however, adult mink pneumocytes restrict viral replication, and adults do not typically develop pneumonia.5,22 Despite sensitive diagnostic tests and extensive research on AMDV, no effective prophylaxis is available, and mink farms continue to struggle with controlling infections. 23 AMDV was the only known APV for decades, but the genus has expanded rapidly in recent years. Novel APVs have been detected in foxes, skunks, raccoon dogs, and red pandas, but studies to date examine individual or small numbers of animals and the consequences of these infections are not well understood.1,3,6,8,21,30
There is urgency in identifying disease threats to zoo-housed red panda populations. Estimates suggest that fewer than 10,000 red pandas (Ailurus spp.) remain in the wild, and ex situ conservation efforts, which include nearly 1000 red pandas in zoos across the world, represent critical safeguards for the survival of the species.
15
The welfare and sustainability of these managed populations is paramount and relies on understanding and mitigating disease threats. RPAV is a novel APV that has only been reported in red pandas. First described in 2018, RPAV shares up to 85% nucleotide identity with AMDV and has similarities in biological behavior, including high prevalence (
In this study, we sought to clarify the potential impact of RPAV infections in zoo-housed red panda populations using (1) a prospective evaluation of viral persistence (shedding in feces, collected over 6 years), (2) a retrospective assessment of infection status in necropsied animals (43 animals, 4 cohorts, spanning 25 years), and (3) analysis of viral distribution in RPAV-positive animals (29 animals). Drawing upon a recent comprehensive mortality review and observations of APV-associated disease in mink and skunk APV cases, we establish that RPAV codistributes with lesions such as myocarditis, nephritis, and pneumonia, which are known to be impactful in this population.3,12 RPAV is a persistent virus with prevalence in healthy red pandas so the visualization of virus in regions of inflammation/tissue damage is fundamental evidence for establishing a causal relationship of virus with disease. Viral association alone is not conclusive for causality, but given that experimental infection of this endangered species is indefensible; in concert—the establishment of viral distribution in areas of cell and tissue damage and the similarity to other APV infections (mink, skunk) are evidence that RPAV can cause impactful, even fatal, disease in zoo-housed red pandas.
Materials and Methods
Longitudinal Study Quantitative Polymerase Chain Reaction
Repeated fecal samples from 2 infected, un-related, cohoused red pandas 31 were collected over a 6-year timespan. Throughout the sampling period, the size of this cohort fluctuated between 2 and 6 animals due to mortalities and interinstitutional transfers. Samples that could be confidently attributed to individuals were collected opportunistically from enclosures by animal care staff and stored at –20°C until use. Timepoints for collection each year are presented in Supplemental Table S1. DNA extractions were performed on 1 g of feces using a commercial kit (DNeasy Blood and Tissue Kit, Qiagen), with a final elution volume of 200 µL. The quantitative polymerase chain reaction (qPCR) assay was adapted from studies of related viruses targeting the capsid-coding (VP) gene, using forward primer 5’-CTGTAACAGAAACCAACCAAGGTA-3’ and reverse primer 5’ -GGTTGGTTTGGTTGCTCTCCA-3’.’ 32 Samples were assayed on an ABI 7500-fast cycler (Applied Biosystems), and amplifications were performed using Maxima SYBR green with ROX as the reference dye (ThermoFisher Scientific). Each sample of extracted DNA was tested in triplicate, and the mean Ct value for each sample was determined. A standard curve was generated from a segment of RPAV nucleic acid cloned into a pUC53 plasmid vector, with a lower limit for detection of 3 genome copies per 5 µL sample. Samples with mean copy numbers above this threshold were considered positive, and samples below this threshold were considered negative. At least, 6 negative (no-template) control wells were placed randomly on each 96-well plate.
Case Criteria for Retrospective Studies
Retrospective cases were identified from San Diego Zoo Wildlife Alliance (San Diego, CA, USA), the Sacramento Zoo (William R. Pritchard Veterinary Medical Teaching Hospital, University of California Davis, Davis, CA, USA), Aalborg Zoo (Aalborg, Denmark), and Bronx Zoo (Wildlife Conservation Society, New York, NY, USA). All 4 collections had previous RPAV-positive cases detected in previously published and unpublished studies. 2 One case of significant myocarditis was present in the collections evaluated. To evaluate a potential association of RPAV with myocarditis, we added 2 additional individual cases of myocarditis from other zoo collections.
Because splenic tissue has been determined to be a sensitive sample for systemic APV infection, spleen was chosen to determine individual infection status. 33 Cases were excluded if fresh, frozen or formalin-fixed, paraffin-embedded (FFPE) splenic tissue was not available.
Animal histories, including birth date, death date, sex, and (sub)species were obtained from the Zoological Information Management System (ZIMS). 3 The manner of death (euthanasia or natural death) and cause of death were obtained from necropsy reports or histopathology reviews.
PCR detection in retrospective cases
RPAV PCR on FFPE (n = 42) or frozen (n = 1) splenic tissue was performed as previously described, targeting a 187 nucleotide segment of the viral nonstructural (NS) gene.2,3 Cases were considered positive if a band of the correct size was obtained. PCR products were further confirmed to be RPAV by sequencing of 10 cases. Amplicons were purified with ExoSAP-IT (ThermoFisher) according to manufacturer instructions, then submitted for bidirectional Sanger sequencing at the UCDNA Sequencing Facility (UC Davis sequencing core facility). Resulting sequences were aligned using the MUSCLE 14 algorithm in Geneious, along with reference sequences obtained from GenBank. 18 The optimal substitution model was determined using a model test in MEGA-X 20 prior to generating a phylogenetic tree. The tree was generated using a Kimura 2-parameter model with discrete gamma distribution.19,20,32
For PCR, FFPE samples (25-µm-thick scrolls) were deparaffinized with a series of xylene and graded ethanol washes. DNA was extracted using a commercial kit (DNeasy Blood and Tissue Kit, Qiagen). PCR targeting a housekeeping gene (GAPDH) was performed on every sample to confirm DNA quality.
In Situ Hybridization
Colorimetric in situ hybridization (ISH) was performed in 29/34 PCR-positive cases. Five cases were excluded either due to autolysis, limited tissue for evaluation, or prolonged formalin fixation (>1 week). Of the 29 cases tested, select tissues were examined by ISH based on (1) lesions identified as important causes of death in zoo-housed red pandas and/or (2) lesions recognized as APV-associated in other carnivores.3,1,8
To localize RPAV in tissues, we performed ISH using the RNAscope® 2.5 HD Reagent Kit—RED (Advanced Cell Diagnostics, Newark, CA, USA) and followed the manufacturer’s instructions to visualize the signal in FFPE tissues. A custom anti-sense probe was designed targeting the VP gene of RPAV obtained from RpAPV-CA1 (GenBank accession NC_031751). 3 To confirm adequate DNA/RNA integrity of the FFPE samples, a housekeeping gene (peptidylprolyl isomerase B) probe was applied to a subset of samples as a positive control. At least 1 slide with previously ISH-detected and PCR-confirmed viral nucleic acid was included in every run. Healthy red panda tissues and a probe targeting the dapB gene (Advanced Cell Diagnostics, Newark, CA, USA) were used as negative controls.
Results
Individual Red Pandas Can Shed RPAV for >6 Years
To understand patterns of RPAV shedding, we amplified RPAV DNA from feces of 2 live animals from one zoo collection over a 6-year timespan. Viral DNA was present in 35/38 samples from case A and 37/37 samples from case B over the 6-year period (Table 1). Dates of collection and mean target copy numbers detected per sample are given in Supplemental Table S1.
Frequent detection of RPAV in fecal samples from two apparently persistently infected red pandas.
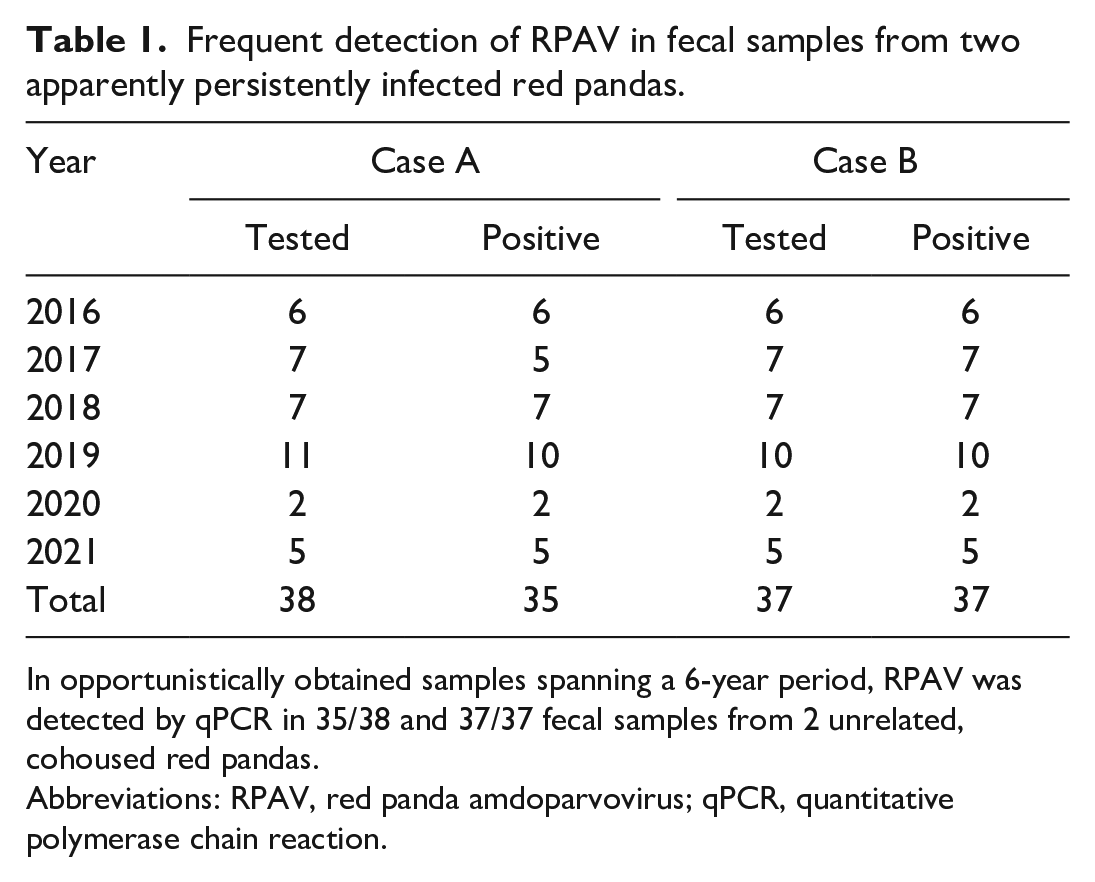
In opportunistically obtained samples spanning a 6-year period, RPAV was detected by qPCR in 35/38 and 37/37 fecal samples from 2 unrelated, cohoused red pandas.
Abbreviations: RPAV, red panda amdoparvovirus; qPCR, quantitative polymerase chain reaction.
Retrospective Detection of RPAV in Four Zoo Cohorts
All necropsied red pandas from cohorts 1 and 2 were evaluated (n = 27). A subset of necropsied red pandas (based on available tissues) from cohorts 3 and 4 were evaluated (n = 16). The entire tested population from these cohorts comprised 19 males and 24 females. Ages at death ranged from < 1 day to 19 years (mean = 6.5 years), and necropsies were performed between 1997 and 2022. A summary of cases is provided in Supplemental Table S2.
In total, PCR testing of splenic tissue demonstrated RPAV in 32/43 (74%) animals. This included 13/16 necropsied animals from San Diego Zoo Wildlife Alliance, 8/11 cases from Sacramento Zoo, 6/6 cases from Aalborg Zoo, and 5/10 cases from the Wildlife Conservation Society. The positive cases included a wide age range from neonates to geriatric animals (mean = 7.7 years) and included detection in some of the oldest necropsy samples available from these collections (1997). All sequenced amplicons (n = 10) were phylogenetically closely related to previously identified RPAV sequences (85.8%–100% identity, Fig. 1). The negative cases ranged from neonates to geriatric animals (mean = 2.75 years), with the oldest negative case dating to 2001.
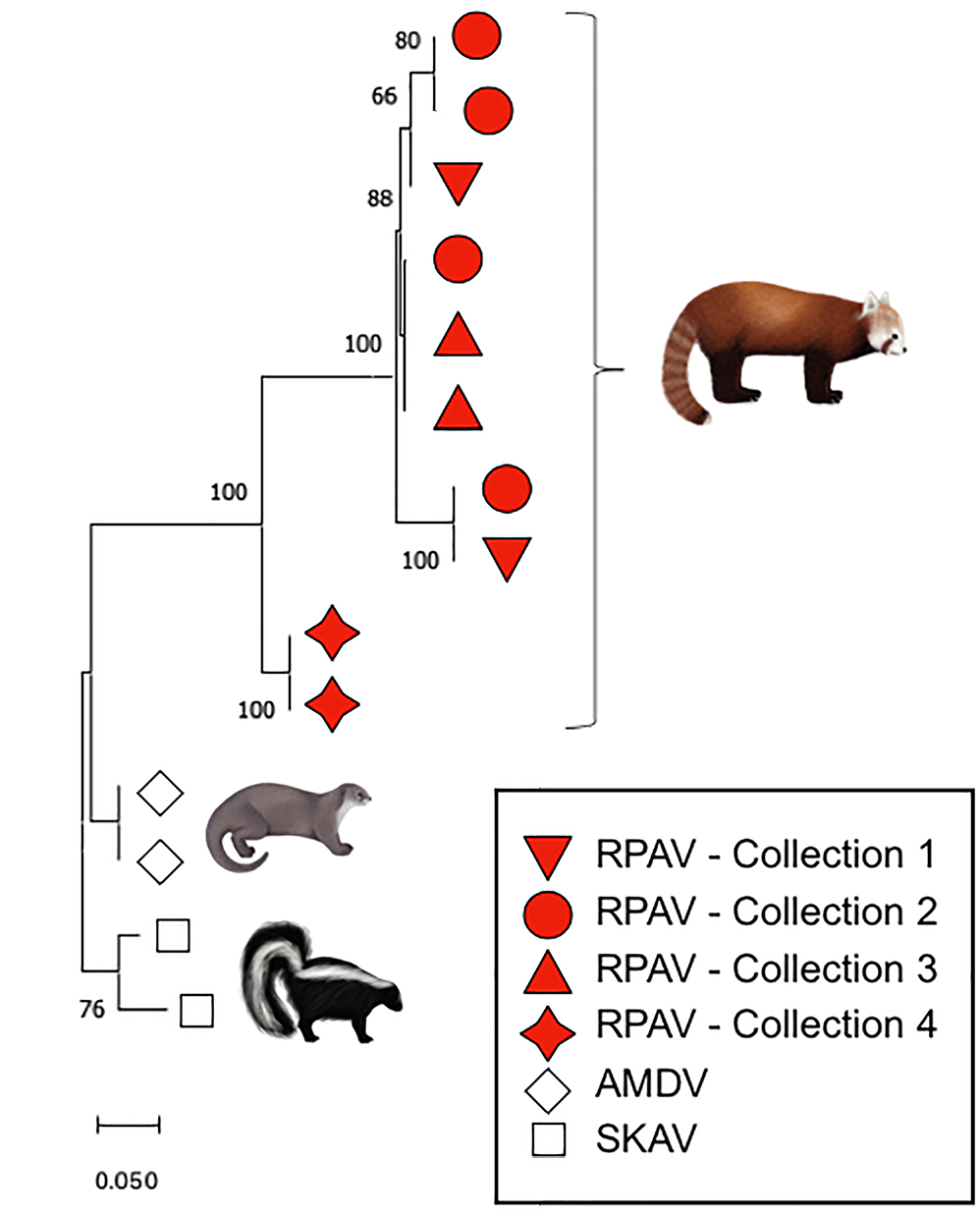
Specific detection of red panda amdoparvovirus (RPAV) in cases from multiple zoo collections. Maximum likelihood phylogeny of 113nt amplicons from each of 4 included cohorts belong to a single viral species within genus Amdoparvovirus. In this short (113 nucleotide [nt]) segment of the nonstructural gene, sequences from disparate cohorts are sometimes more closely related than sequences within cohorts. Reference sequences obtained from GenBank for this analysis are, for Aleutian mink disease virus (AMDV), JN040434 and KU856562, and for SKAV, KX981924 and OL889869.1. SKAV, skunk amdoparvovirus.
RPAV Association With Diseases of Red Pandas
From PCR-positive cases, we evaluated viral distribution and the potential contribution of RPAV to morbidity/mortality by histopathology and ISH.
Myocarditis: Sections of myocardium from 10 cases were evaluated by ISH (Table 2). Five cases (cases 16, 19, 21, 26, and 35) were histologically normal. Two cases (cases 34 and 43) had endocardial and/or myocardial fibrosis with minimal inflammation. Three cases (cases 22, 44, and 45) had severe myocarditis.
Detection of RPAV by ISH in cases of myocarditis.
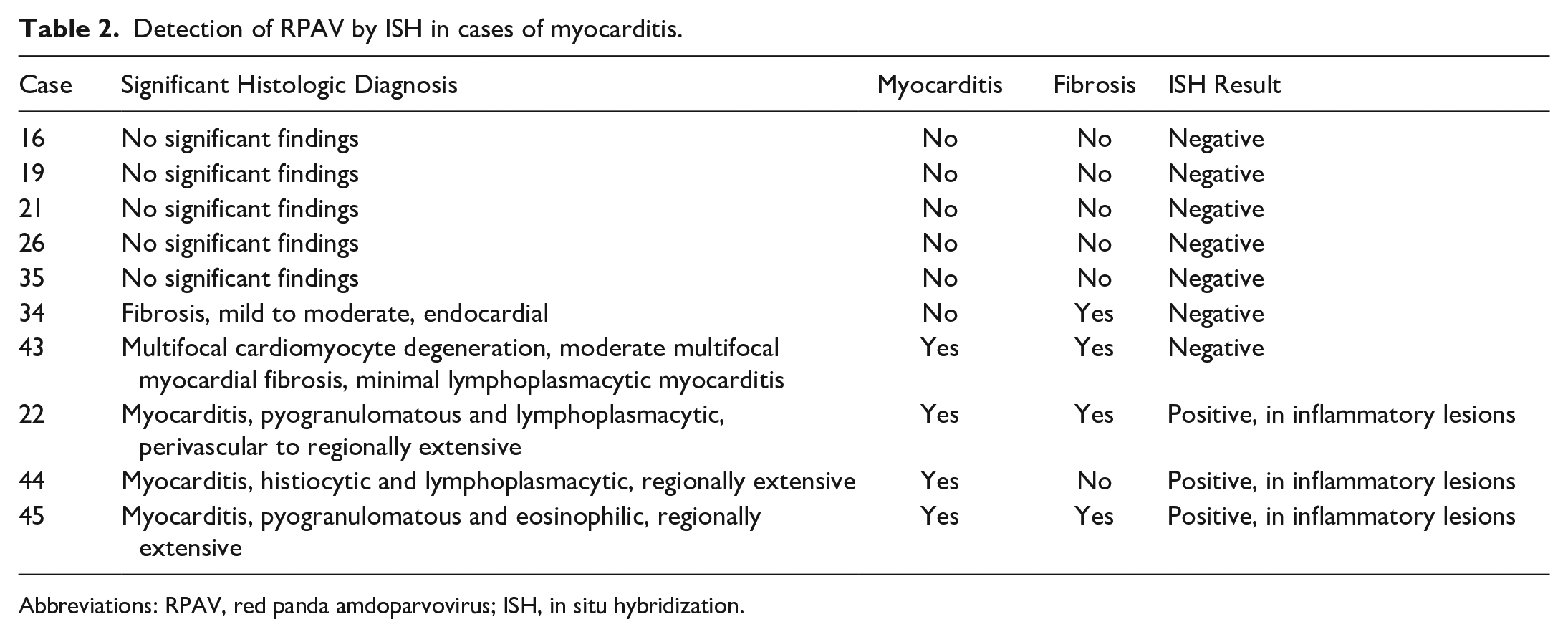
Abbreviations: RPAV, red panda amdoparvovirus; ISH, in situ hybridization.
All 3 myocarditis cases had patchy areas of inflammation with separation of cardiomyocytes by edema, plump reactive pericytes and infiltrating leukocytes. In affected areas, rare, scattered cardiomyocytes were hypereosinophilic, fragmented, or replaced by eosinophilic acellular material and karyorrhectic debris. In case 22, which was previously reported, these foci were scattered and primarily centered around small-caliber blood vessels. 3 In the other 2 cases (cases 44 and 45), inflammation was regionally extensive and coalescing, involving large zones of the myocardium and up to ~75% of the thickness of the affected wall (Fig. 2a, d). There were minor differences in the composition of the inflammatory infiltrate. In case 22, inflammation was primarily histiocytic and neutrophilic, while cases 44 and 45 were predominantly histiocytic, lymphocytic, and plasmacytic, with fewer neutrophils and eosinophils admixed.
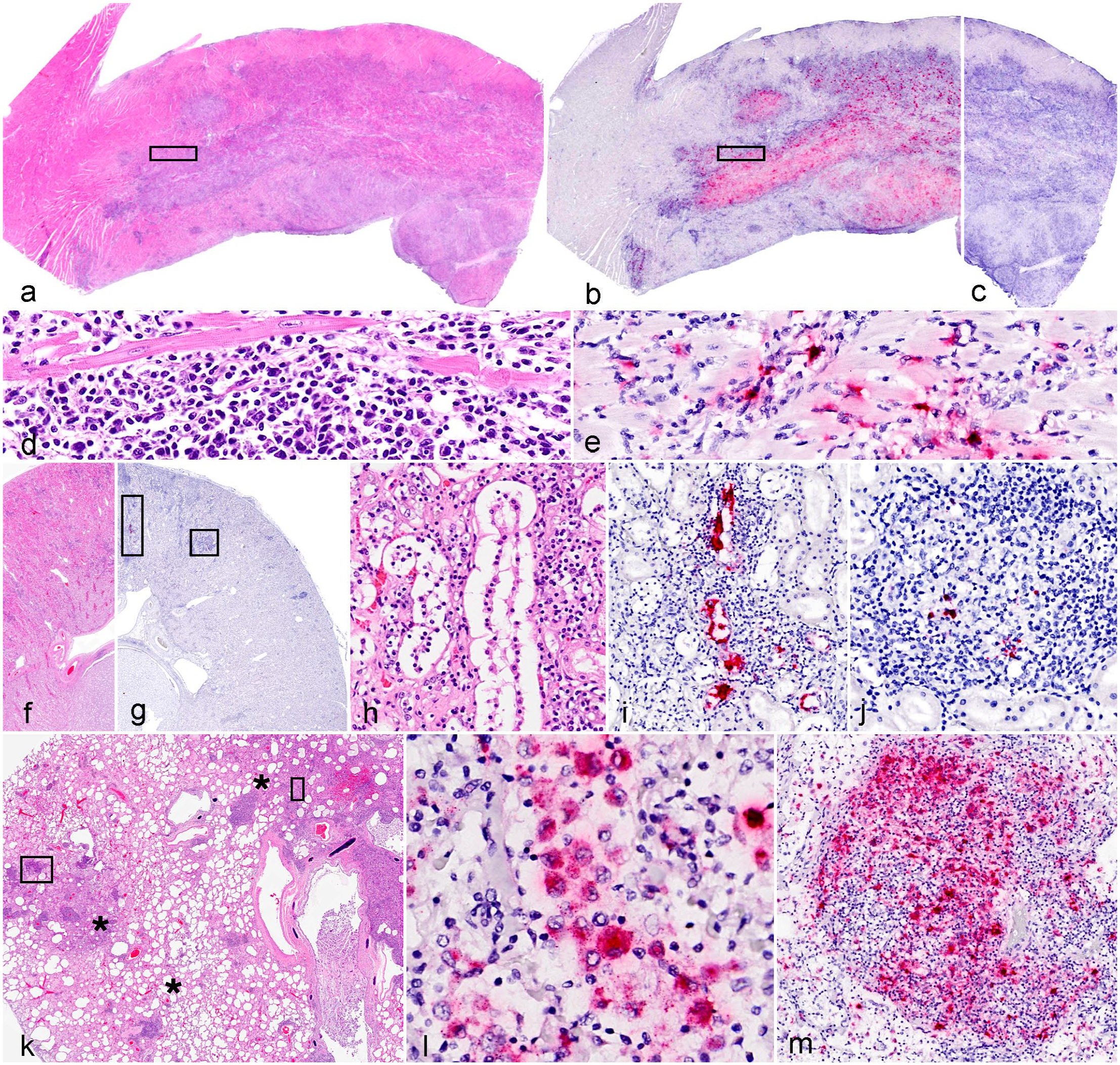
Red panda amdoparvovirus colocalization with lesions in red pandas. In situ hybridization (ISH) demonstrates viral colocalization with inflammation in cases of myocarditis, chronic interstitial nephritis, and pneumonia. (a–e) Heart (interventricular septum), myocarditis, 6-year-old red panda. (a) Dense infiltrates of inflammatory cells multifocally disrupt or replace regions of the myocardium. Hematoxylin and eosin (HE). (b) Red panda amdoparvovirus (RPAV) ISH demonstrates probe hybridization (red) colocalizing to areas of inflammation and necrosis. ISH, RPAV probe. (c) No probe hybridization is detected with an unrelated (dapB, negative control) probe. (d) Higher-magnification image of the boxed area in (a). The inflammation separates cardiac myofibers and consists of histiocytes, plasma cells, and lymphocytes. HE. (e) Higher magnification of the boxed area in (b), with signal limited to infiltrating cells. ISH, RPAV probe. (f–j) Kidney, mild, chronic tubulointerstitial nephritis, 6-year-old red panda. (f) Multifocal infiltrates disrupt the renal cortex. HE. (g) Two boxed regions on a replicate ISH slide using the RPAV probe. (h) Rectangular boxed area from (g). There is interstitial inflammation and entrapped tubules. HE. (i) RPAV is detected in epithelium of an entrapped tubule. ISH, RPAV probe. (j) Square boxed area from (g). Chronic interstitial inflammation with follicular formation and scattered cells containing RPAV nucleic acid. ISH, RPAV probe. (k–n) Lung, pneumonia, 7-year-old red panda. (k) Regionally, alveolar septa are thickened, and dense inflammatory foci are scattered haphazardly within the pulmonary interstitium (asterisks). HE. (l) Rectangular box in (k). Stippled to punctate viral probe hybridization is present in the cytoplasm of intraalveolar cells (presumed macrophages). ISH, RPAV probe. (m) Square boxed region of (k), densely cellular focus of inflammation. By ISH, RPAV signal is present in many inflammatory cells.
Among the 10 hearts analyzed, RPAV ISH probe hybridization was only detected in the 3 cases with active myocarditis. RPAV probe hybridized only to regions of inflammation and was not detected in the adjacent histologically normal myocardium (Fig. 2a–e). ISH signal was present within cells morphologically consistent with endothelial cells and macrophages (Figs. 2d, e).
Chronic kidney disease: Although renal disease was not considered the primary cause of death in any case in this cohort, chronic interstitial nephritis was a common comorbidity, and the kidney is a known and significant target for APV infections in mink and skunk. 3 We tested sections of kidney from 20 cases representing a spectrum of disease. Findings grouped by histologic diagnoses are summarized in Table 3, and results for individual cases are presented in Supplemental Table S3.
Detection of RPAV by ISH in chronic interstitial nephritis.

Abbreviations: RPAV, red panda amdoparvovirus; ISH, in situ hybridization.
Seven cases were histologically normal. Nine cases were mild chronic interstitial nephritis, with interstitial lymphocytes, plasma cells, and macrophages, and minimal fibrosis affecting < 5% of the renal parenchyma. Two cases were diagnosed with moderate chronic interstitial nephritis, characterized by similar inflammation but affecting 5% to 30% of the renal parenchyma. Glomeruli were normal in all but 2 cases; 1 had mild membranoproliferative glomerulonephritis in addition to moderate chronic interstitial nephritis and 1 had mild membranoproliferative glomerulonephritis with histologically normal interstitium. Arteritis was present in 2 cases; 1 with mild nephritis and 1 who was otherwise histologically normal. Affected renal, interlobar, and arcuate arteries were segmentally, transmurally disrupted by lymphocytes, accompanied by regional vacuolation and separation of smooth muscle cells and accumulation of brightly eosinophilic, acellular, fibrillar material (fibrinoid necrosis). Most arteries had dense, asymmetric or circumferential lymphocytic cuffs within their periphery (adventitia/vasa vasorum).
In total, RPAV was detected by ISH in renal tissue in 14/20 cases tested. Among histologically normal kidneys, RPAV was detected in 2/7 cases. ISH signal in these cases was localized to the interstitium in rare, scattered individual cells that, based on morphology and location, were presumed to be endothelial cells and/or circulating leukocytes. RPAV was detected by ISH in 8/9 cases with mild chronic interstitial nephritis. In these cases, in addition to scattered individual cells throughout the interstitium, signal was present in cells within inflammatory cell aggregates (Fig. 2f). In 2 cases, signal was also detectable in tubular epithelial cells, typically adjacent to foci of interstitial inflammation (Fig. 2g–i). Where probe hybridization was detectable in tubular epithelium, cells exhibited features of tubular injury (karyorrhexis, necrosis, intraluminal cellular debris/casts) or epithelial attenuation. In many of these tubules, ISH signal was also observed in intraluminal cellular debris or casts (Fig. 2i). In both cases with moderate chronic interstitial nephritis, RPAV was detected in organized, chronic inflammatory cell aggregates (Fig. 2j) where there was nephron loss. Both cases diagnosed with membranoproliferative glomerulonephritis exhibited ISH signal in scattered cells in the interstitium, and 1 case additionally had ISH signal in the pelvic urothelium. In these 2 cases, probe hybridization was not detected in affected glomeruli.
Pneumonia
We evaluated lung tissue from 13 red pandas by ISH: 4 neonates (less than 30 days), 2 juveniles (30 days–1 year), 3 adults (1–10 years), and 4 geriatric (greater than 10 years) animals. No mortalities in this set were attributed to pneumonia, but pulmonary lesions were comorbidities in several cases. RPAV was detected by ISH in the lungs of 7/13 animals tested. Histologic findings and ISH results are summarized in Table 4. Of the 4 neonates tested, 1 had histologically normal lungs, 1 had intraalveolar hemorrhage and edema attributed to trauma, and 2 had regionally extensive to diffuse interstitial pneumonia. The latter was characterized by expansion of alveolar walls with edema and fibrin, intraalveolar fibrin exudation, accumulations of intraalveolar macrophages and cellular debris, and type 2 pneumocyte hyperplasia. RPAV was not detected in the lung of any neonate by ISH.
Detection of RPAV by ISH in lungs.
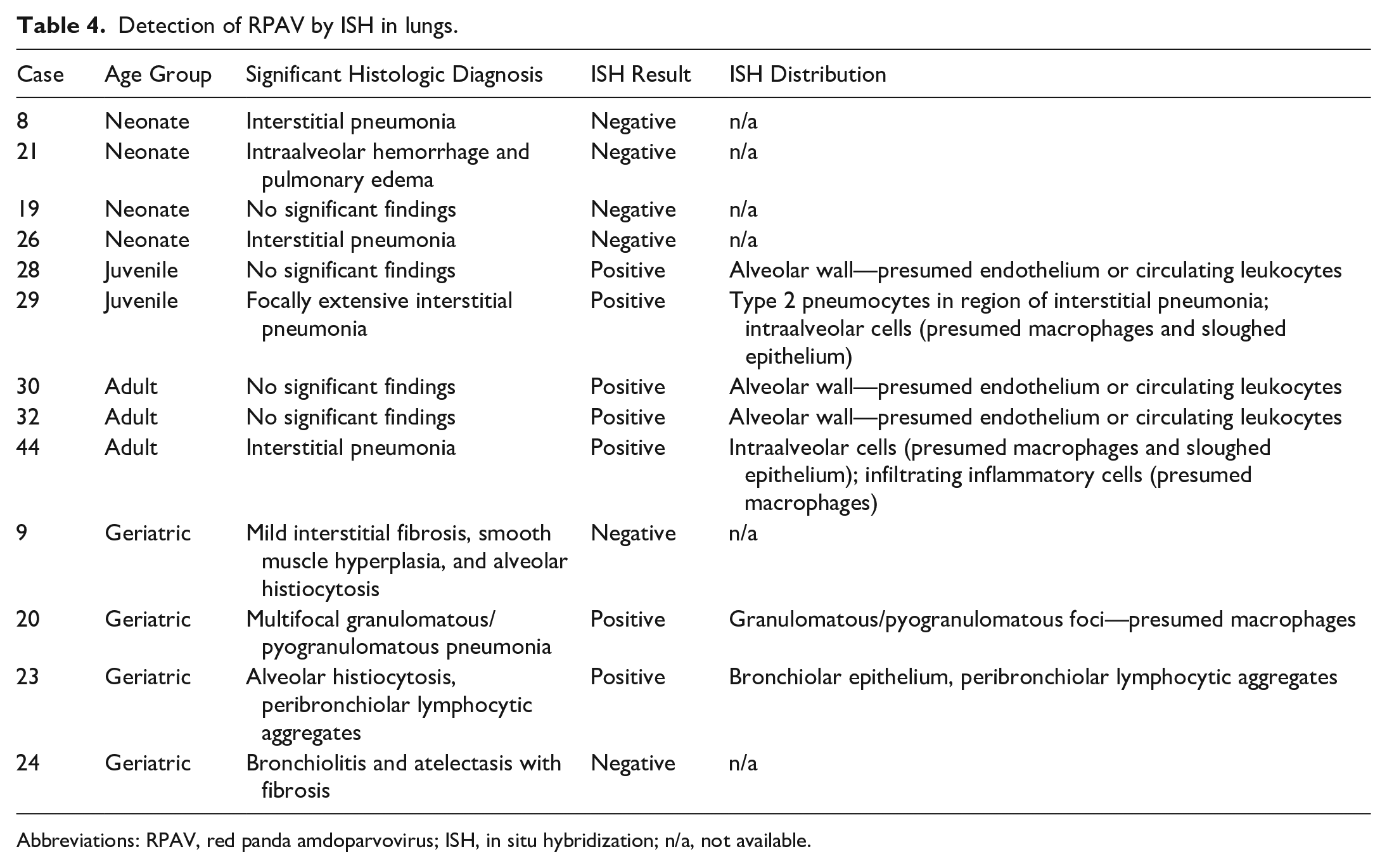
Abbreviations: RPAV, red panda amdoparvovirus; ISH, in situ hybridization; n/a, not available.
Among lungs of juveniles and adults (n = 5 total), 3 (1 juvenile and 2 adults) were histologically normal, and 2 (1 juvenile and 1 adult) had interstitial pneumonia. RPAV was detected in the lungs by ISH in all 5 cases. In these normal lungs, signal was detected in rare, scattered individual cells in the alveolar wall. In both cases with interstitial pneumonia, signal was colocalized to the sites of inflammation where it was present in lining pneumocytes, and in free cells within alveolar lumen.
Lungs from 4 geriatric animals were tested. All 4 had pulmonary lesions, but the lesions were distinct in each case. The first case (case 9) was essentially normal except for mild, multifocal interstitial fibrosis with smooth muscle hyperplasia and mild alveolar histiocytosis. The second case (case 20) had a mild suppurative bronchiolitis and mild interstitial fibrosis. The third case (case 23) had multifocal granulomatous to pyogranulomatous foci randomly distributed throughout the lungs, with no other pathogens detected in a battery of special stains (Gram, Giemsa, Gomori methenamine silver, Ziehl-Neelsen acid-fast). The fourth case (case 24) had moderate alveolar histiocytosis and prominent (subjectively increased or hyperplastic) bronchus-associated lymphoid tissue (BALT). ISH was negative in the first 2 geriatric cases. In the case with granulomatous/pyogranulomatous pneumonia (Fig. 3k), ISH signal was consistently detected within macrophages in the lumen of alveoli (Fig. 3l) and within cell dense granulomas (Fig. 2m). In the case with BALT hyperplasia, signal was detected in most BALT aggregates as well as segmentally in histologically normal bronchiolar epithelial cells.
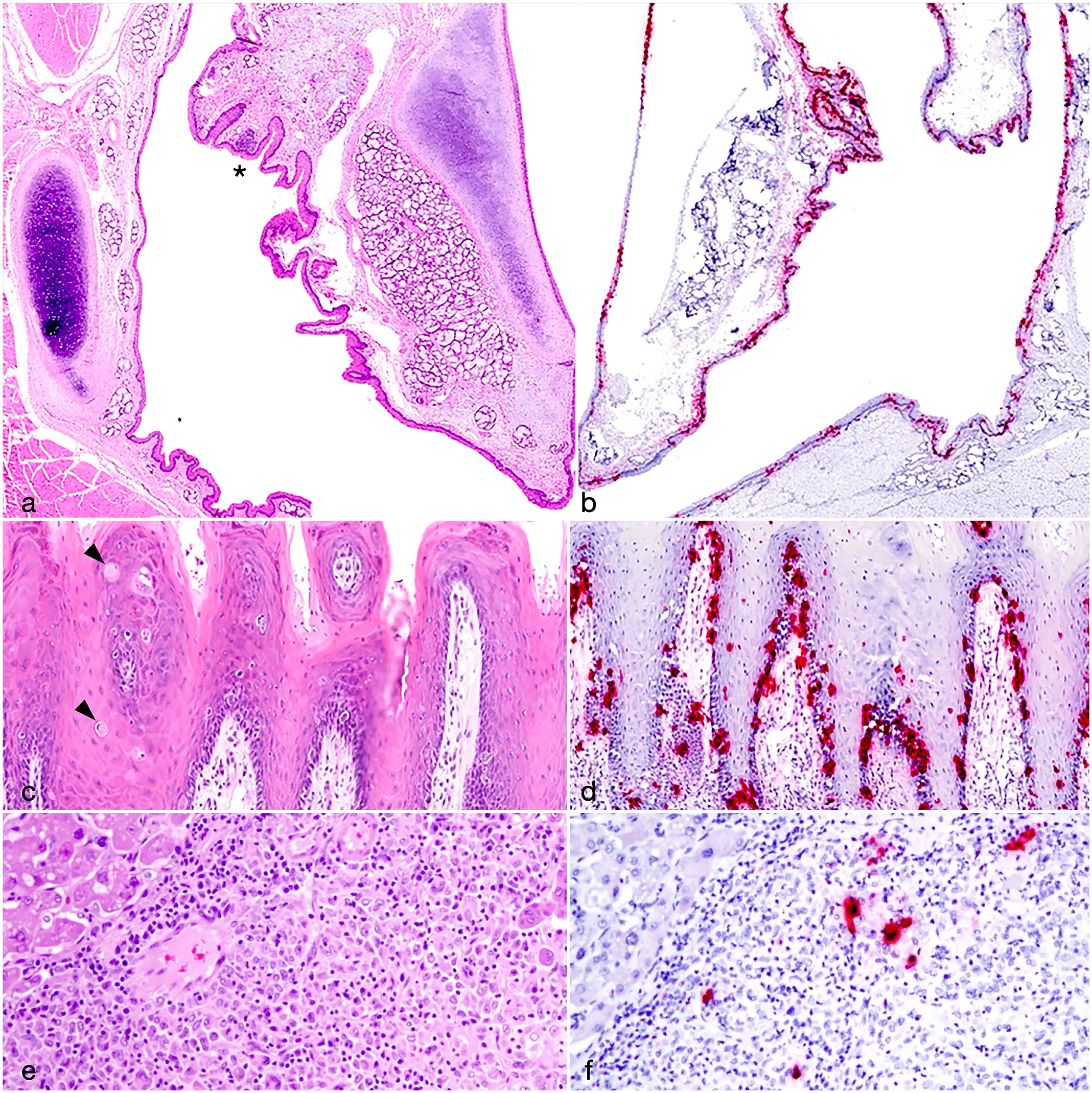
Novel manifestations of amdoparvovirus disease. Red panda amdoparvovirus (RPAV) is detected in multisystemic pyogranulomatous lesions and oropharyngeal mucosal inflammation. (a–b) Larynx, level of arytenoids, 6-month-old red panda (a) Minimal laryngitis (asterisk). Hematoxylin and eosin (HE). (b) Intense, segmental to vast probe hybridization within laryngeal epithelium. In situ hybridization (ISH), RPAV probe. (c–d) Tongue, 6-month-old red panda (c) Inflammation is present in the superficial subepithelium, with scattered transmigration of lymphocytes disrupting the basilar layer. Few individual epithelial cells are swollen and pale (arrowheads). HE. (d) RPAV is primarily in the basilar epithelium with scattered signal present in individual cells of upper stratum. ISH. (e–f) Liver, multifocal, histiocytic, lymphocytic hepatitis, 17-year-old male red panda. (e) In this portion of a loosely organized granuloma (HE), (f) RPAV probe hybridization demonstrates viral nucleic acid within centralized inflammatory cells (presumed macrophages).
Other diseases
We previously reported a possible role of RPAV in inducing several additional lesions, including fulminant multisystemic pyogranulomatous inflammation and oral mucosal inflammation. 14 In addition, the epidermis has been demonstrated as a site of APV tropism, 15 and pilot studies on red panda tissue identified dermatitis as being potentially RPAV-associated. Therefore, although these are not recognized as common causes of mortality in captive red pandas, these lesions were evaluated by ISH.
We tested oral/pharyngeal and/or esophageal mucosal tissue from 11 cases. Five cases had evidence of RPAV in the mucosal epithelium, in a distribution similar to that which was previously reported.7,24,25 Virus was often detectable in the basal layers of the lingual and/or oropharyngeal epithelium, with individual cells in the stratum basale and stratum spinosum exhibiting hypereosinophilia and nuclear pyknosis (presumed apoptosis), and a minimal subepithelial inflammatory infiltrate. Histologically, these sites of infection varied from essentially normal (most cases) to eroded (rare). In the most severe cases, virus was abundant throughout the oral and pharyngeal mucosa, which was subtended by multifocal clusters of mononuclear cells (larynx, Fig. 3 a–b), or more broadly subtended by inflammatory cells in the superficial epithelium and basilar epithelial layer (tongue, Fig. c–d).
We tested skin tissue from 8 red pandas for evidence of RPAV tropism to this tissue. These ranged from histologically unremarkable to moderately inflamed. Inflammation, where present, was characterized by scant to moderate subepithelial infiltrates of lymphocytes and plasma cells. In more heavily infiltrated sections, inflammatory cells occasionally extended into the overlying epidermis and scattered individual epithelial cells were swollen and hypereosinophilic with pyknotic nuclear material (presumed apoptosis). A subset of cases (4/8) had detectable virus in the skin, ranging from scattered, individual epidermal cells to widespread dispersion throughout the basal epidermis, follicular epithelium, subepithelial inflammatory cells, and basal cells of sebaceous glands.
Three red pandas in this study died with severe, pyogranulomatous inflammation affecting multiple organ systems, with no other pathogens detected using a variety of special stains (Gram, Giemsa, Gomori methenamine silver, Ziehl-Neelsen acid-fast). In 1 case (case 16), these lesions were limited to liver and spleen. In the second case (case 20), pyogranulomatous lesions were present in liver, spleen, lymph nodes, lung, and bone marrow. In the third case (case 22), pyogranulomatous lesions were present in liver, pancreas, intestine, urinary bladder, and epicardium/myocardium. RPAV was detected by PCR and ISH in all 3 cases. At least, 2 tissues with representative lesions from each case were tested by ISH. In all cases, RPAV was detectable by ISH within macrophages in area of inflammation (liver, Fig. 3 e–f).
Discussion
Healthy, sustainable zoo populations represent a critical safeguard for the survival of endangered species. Threatened by habitat loss, increasing domestic animal populations (and their communicable pathogens), and illegal trade, red pandas in the wild now number less than 10,000.8,28,27 The roughly 1000 red pandas in zoos worldwide represent approximately 10% of the overall population. Characterizing disease threats to zoo populations is imperative. Drawing on our understanding of AMDV and the results of a recent, comprehensive red panda mortality study, this study provides information about the potential impact of RPAV infection, and characterizes widespread viral distribution, demonstrating an apparent association with common or fatal diseases of zoo-housed red pandas. 12
A high prevalence of infection (approximately 50%) based on conventional PCR and a one-time fecal sampling of US zoo populations suggests that the pathogenesis of RPAV, like AMDV, includes persistent infection. In this study, long-term viral persistence was confirmed in 2 animals with a longitudinal qPCR spanning over 6 years. Despite irregular (opportunistic) timespans between fecal collections, nearly all samples from these animals were positive, and the study covered 1/3 to 1/2 the lifespan of a zoo panda. We propose that, like AMDV, RPAV infection can be lifelong. We cannot exclude the possibility that the high frequency of detection in longitudinal sampling could be sequelae to repetitive re-infection or due to environmental contamination of fecal samples, given the remarkable environmental stability of parvoviruses. Red pandas are frequently transferred between zoos for breeding or management purposes, essentially comprising a single, widely dispersed population. Interinstitutional transfers are an essential component of a carefully managed Species Survival Plan, but are also potential opportunities for viral transmission between institutions. RPAV has been circulating among zoo-housed red pandas for decades, but we do not know whether infection exists in the wild population.
AMDV-associated mortality often results from plasmacytic nephritis and membranous glomerulopathy. Kidney was similarly a common target of RPAV, but renal disease in infected red pandas was seldom a primary cause of death, although it remains the single most common comorbidity in necropsied red pandas. 12 RPAV was detectable both in histologically normal kidneys, in rare, scattered tubular epithelial cells or individual cells in the interstitium, and in diseased kidneys, where it frequently colocalized with interstitial inflammation and degenerate or necrotic tubular epithelium. Given the polyphasic nature of the renal damage in this cohort, we propose that in some cases, RPAV contributes to chronic kidney disease in red pandas through piecemeal accumulation of tubular injury and parenchymal loss over the lifetime of the animal. Glomerulopathy, which is a characteristic feature of AMDV infections in mink, was rare in this study. 28 For the purpose of diagnosis, this is an important difference in pathogenesis among carnivores: membranoproliferative glomerulonephritis in AMDV is attributed to an exuberant, nonneutralizing humoral immune response with antigen-antibody complex deposition, which does not appear to play a significant role in RPAV-associated renal disease. 13
The spectrum of lesions analyzed in this study, in addition to considering known APV-related disease, relied on known causes of morbidity and mortality among zoo-housed red pandas. While active myocarditis is an uncommon diagnosis in necropsied red pandas, cardiomyopathies, especially myocardial fibrosis, are frequent contributors to mortality in adult and geriatric animals. 12 We found 3 cases with severe myocarditis, and in all 3, we demonstrated an abundance of RPAV nucleic acid in close association with inflammatory lesions. As an end-stage lesion, the cause or causes of myocardial fibrosis are typically unknown. Given that RPAV is associated with myocardial inflammation, it is possible that some cases of myocardial fibrosis could represent chronic sequelae of nonfatal episodes of RPAV-associated damage. 12
We were especially interested in the high rate of neonatal/juvenile mortalities in zoo-housed red pandas, with only ~45% of zoo-housed red pandas living beyond their first year. 12 Pneumonias is the most common cause of red panda infant mortality, and these cases were additionally of interest given that AMDV is a well-recognized cause of interstitial pneumonia in mink kits.5,12 We found no strong evidence to suggest that RPAV-associated pneumonia was important in young red pandas. RPAV was present in 1 case of interstitial pneumonia in a juvenile. In other young pandas, RPAV was detected by ISH in scattered individual cells in the interstitium or within alveoli, in regions with no apparent damage, and although the cell target was not definitively determined, the morphology and distribution was consistent with alveolar or circulating macrophages.
We also recognized convincing viral colocalization in sporadic lesions in other tissues. Three animals died with severe, multisystemic pyogranulomatous disease affecting the liver, spleen, pancreas, intestinal tract, heart, and urinary bladder.19,20,31 Virus distribution was central to inflammatory lesions by ISH, suggesting that this is a distinct presentation of RPAV-associated disease. Other tissue targets included oral mucosal epithelium, which was originally reported in the index case and was detected in numerous additional cases in this study. Viral infection of epithelium ranged from segmental, histologically subtle, to vast tracts of infection with cell degeneration/apoptosis and attendant subepithelial inflammation. In concert with evidence of intestinal infection and demonstration of virions in lingual epithelium and feces by electron microscopy, we suggest that the gastrointestinal system is a site of viral persistence and shedding. 3 Intestinal disease was not often reported in this cohort, but oral ulceration was present in 2 cases. Finally, RPAV was present within the arterial wall and surrounding affected vessels in cases of arteritis. This lesion has been reported for AMDV-infected mink where, like the kidney lesions, it is considered to be from immune complex deposition. 28 It is unclear whether a similar pathogenesis underlies the lesions in affected red pandas, where virus is detectable in cells within the inflamed arterial wall or vasa vasorum.
Establishing a causal relationship of RPAV infection with disease is a significant challenge due to the high prevalence of infection, clinically silent infections, and low overall case numbers. We recognize limitations of this study, including a small sample size and the largely descriptive nature of lesion association by ISH. The confounding nature of a retrospective study on natural disease in an endangered population further complicates interpretation, especially given that factors, such as host immunity, viral genetics, or coinfections can influence distribution and effects of any pathogen. We also consider that detection could be underrepresented due to viral sequence variation, since RPAV sequences can be highly variable in infected red pandas—up to ~14% nucleotide sequence divergence—and we rely on techniques (PCR, ISH) that depend on sequence stability.2,3
The convincing colocalization of virus with lesions suggests that at least in some cases, significant diseases in red pandas may be attributed to RPAV-associated damage. The significance of viral colocalization with inflammation could be over-interpreted if RPAV were, for example, simply targeting inflammatory cells or any mitotically active population of cells. This is unlikely given that, among PCR-positive animals, mitotically active cell populations, nor inflammation predicted viral detection, with only subsets of dermatitis, stomatitis, glossitis, enteritis, and pneumonia cases RPAV-positive by ISH analysis. Furthermore, in other mitotically active cell populations, as examples bone marrow and a single infected animal with lymphoma, no virus was present by ISH analysis. We do not know whether there are risk factors or triggers for development of disease. Epidemiologic studies of zoo animals and/or further diagnostic evaluation could more definitively assert a causal relationship, but recognizing this wide tissue distribution and a spectrum of potential lesions is a critical first step for understanding RPAV-associated disease.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231196860 – Supplemental material for Amdoparvovirus-associated disease in red pandas (Ailurus fulgens)
Supplemental material, sj-pdf-1-vet-10.1177_03009858231196860 for Amdoparvovirus-associated disease in red pandas (Ailurus fulgens) by Charles E. Alex, Pavel Kvapil, Martin D. M. Busch, Trine Jensen, Kenneth Conley, Kenneth Jackson, Eric L. Stubbs, Jenessa Gjeltema, Michael M. Garner, Steven V. Kubiski and Patricia A. Pesavento in Veterinary Pathology
Footnotes
Acknowledgements
We thank the keepers and veterinary staff from Sacramento Zoo for assistance with fecal sampling. We acknowledge the pathology and histopathology technical staff from University of California Davis Veterinary Medical Teaching Hospital, San Diego Zoo Wildlife Alliance, Bronx Zoo/Wildlife Conservation Society, and Aalborg Zoo, and Vet Med Labor GmbH, Division of IDEXX Laboratories for assistance with tissue samples and sectioning.
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a philanthropic grant from the McBeth Foundation via San Diego Zoo Wildlife Alliance. C.E.A. is supported in part by NIH T32 training grant 1T32CA251007.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
