Abstract
Choroid plexus tumors (CPT) are intraventricular neoplasms accounting for 10% of all primary central nervous system tumors in dogs. They are frequently classified according to the human WHO classification into choroid plexus papilloma (CPP, grade I), atypical CPP (aCPP, grade II), and choroid plexus carcinoma (CPC, grade III). Histological features observed in canine CPT such as increased vascular density (IVD) and glomeruloid microvascular proliferation (GMVP) are not part of the WHO classification. This multi-centric study aimed to investigate tumor-associated vascular hyperplasia in dogs by determining the prevalence of GMVP and IVD in 52 canine CPT and their association with tumor grade. In addition, the expression of angiogenic factors was assessed by immunohistochemistry in 25 tumors to investigate the pathogenesis of tumor-associated vascular hyperplasia. Based on the classical histological hallmarks, this study of 52 CPT identified 22 (42%) CPP (grade I) and 30 of (58%) CPC (grade III). GMVP was more prevalent in CPC (13/30; 43%) than CPP (1/22; 4%), whereas IVD occurred to a similar extent in CPP and CPC. Desmoplasia was more common in CPC (19/30; 63%) than CPP (2/22; 9%), and similarly, the proliferative index (PI) of neoplastic epithelium was significantly higher in CPC (5.14%) than CPP (0.94%). The majority of CPT expressed platelet-derived growth factor (PDGF), PDGFRα, PDGFRβ, and vascular endothelial growth factor (VEGF) irrespective of tumor grade or tumor-associated vascular hyperplasia. These results suggest that tumor-associated GMVP, desmoplasia, and PI may serve as histological indicators of malignancy in CPT.
Keywords
Choroid plexus tumors (CPT) are intraventricular neoplasms arising from the choroid plexus epithelium. In animals, these tumors occur most commonly in dogs and account for approximately 10% of all canine primary brain tumors. 10 Due to their histological similarity to the human counterpart, in the current literature, canine CPT 7,10,17,20,24,31 are frequently graded in analogy to the human WHO current classification of central nervous system (CNS) tumors 14 into choroid plexus papilloma (CPP, grade I), atypical choroid plexus papilloma (aCPP, grade II), and choroid plexus carcinoma (CPC, grade III). 31 In humans, CPC is a highly aggressive malignant tumor compared to CPP and aCPP, and tumor grade currently dictates the use of appropriate treatment modalities. 13 Little, though, is known about the predictive value of the human grading system regarding the biological behavior and prognosis of canine choroid plexus tumors due to lack of prospective studies in this species.
Angiogenic supply within a neoplasm is fundamental for tumor growth and metastatic potential. 1,34 Tumor neovascularization occurs through several mechanisms, including vessel cooption, classical angiogenesis, and vasculogenesis, 14 and it is well established that tumor progression from a premalignant to a malignant phenotype is dependent on angiogenesis. 5 Accordingly, tumor-related vascular patterns such as increased vascular density (IVD) and glomeruloid microvascular proliferation (GMVP) have been described for human CNS tumors, primarily occurring in gliomas. 26 While IVD is described in low-grade infiltrating astrocytomas (grade II) and characterized by increased density of normal-appearing vessels compared to the normal brain, 5 GMVP occurs in high-grade gliomas like oligodendroglioma grade III and glioblastoma both in humans and dogs. 10 Hence, it is used as a histopathological hallmark of malignancy. 14 Similarly, GMVP in human cancer patients is associated with aggressive tumor behavior, shorter survival, and increased clinical recurrence rates in non-CNS tumors like melanoma, breast, endometrial, and prostate cancers 27 and chemoresistance in breast cancer. 1
Morphologically, GMVP consists of tortuous vascular channels lined by multilayered and mitotically active endothelial cells, pericytes, and smooth muscle cells and is also known by the term glomeruloid bodies due to its resemblance to renal glomeruli. 14 Ultrastructural analysis of GMVP demonstrated that vascular channels are lined with hyperplastic endothelial cells, a basal lamina, and an incomplete layer of pericytes. 5 Pathogenetically speaking, GMVP ontogenesis is likely the result of an upregulation of vascular endothelial growth factor (VEGF) 5,9 and platelet-derived growth factor (PDGF). 18,33 We observed vascular hyperplasia in a subset of canine CPT. 17 Therefore, the aims of the current study are to describe the prevalence and characteristics of GMVP and IVD in canine CPT, investigate their association with malignant forms of CPT, and yield insights into tumor-associated vascular ontogenesis by investigating the expression of angiogenesis-stimulating growth factors (VEGF, PDGF) and tyrosine kinase receptors.
Material and Methods
Cases and Tissues
Fifty-two cases of canine CPT collected at postmortem were included in this retrospective study (1960–2015). The cases were collected from the diagnostic neuropathology archives of the Division of Neurological Sciences, Department of Clinical Research and Veterinary Public Health, University of Bern; Department of Pathology, Microbiology and Immunology, School of Veterinary Medicine, University of California–Davis; and Department of Veterinary Medicine, University of Perugia. Inclusion criteria were a histological diagnosis of choroid plexus tumor and brain or spinal tissue adjacent to the tumor to assess tumor-related vascular patterns. Six histologically normal choroid plexuses (NCP) from dogs without neurological disorders were included as control material.
Histology-Based Tumor Grading
Tumors were available as formalin-fixed and paraffin-embedded tissue. Three μm-thick sections were cut from paraffin blocks and routinely stained with HE. All available slides containing brain tissue were included in the evaluation. Tumors were graded according to the human WHO 2016 classification 14 (Table 1).
System for Grading Choroid Plexus Tumors in Dogs (according to Westworth et al. 31 ).
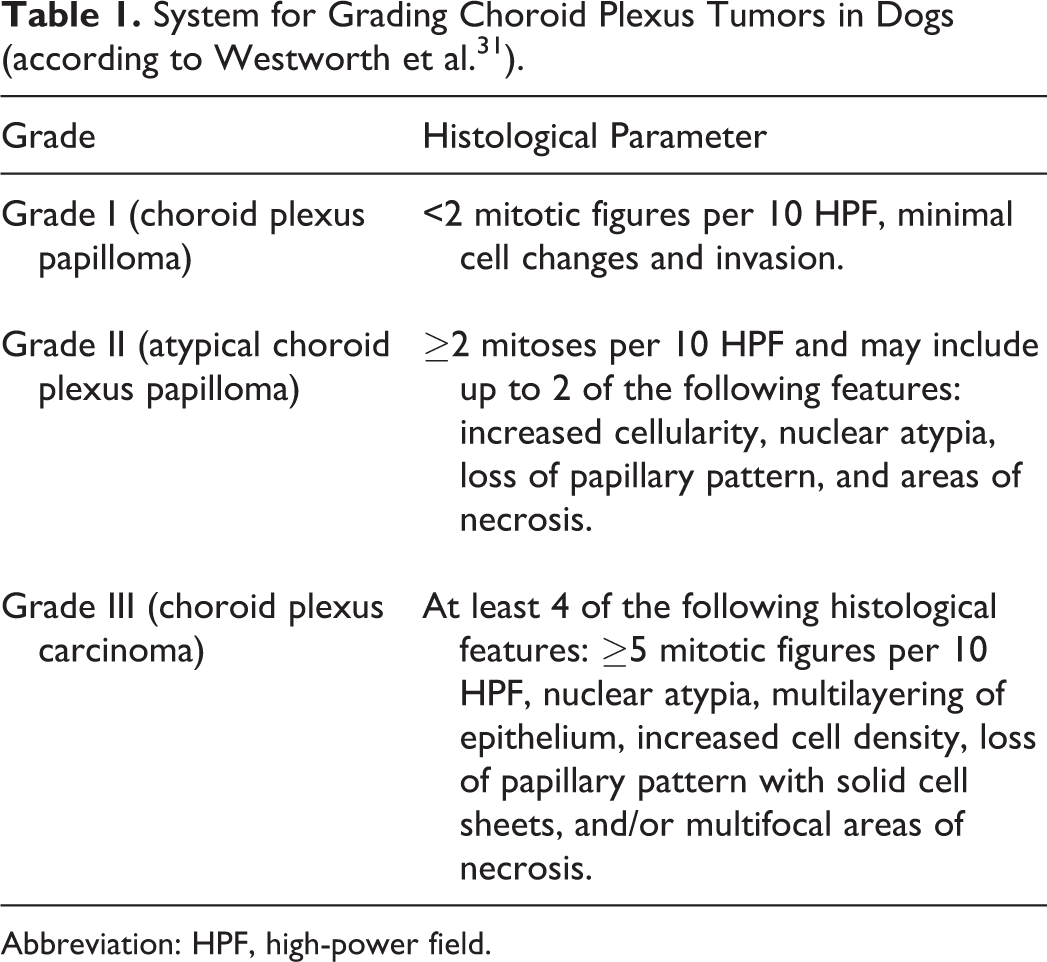
Abbreviation: HPF, high-power field.
Microscopically, CPP closely resembles the healthy non-neoplastic choroid plexus, but cells tend to be more crowded or stratified. The neoplastic epithelium loses the normal cobblestone appearance of choroid plexus epithelium, being more columnar and organized in papilliform projections supported by delicate fibrovascular connective tissue fronds. 14 Atypical CPP is defined by an increased mitotic count and may exhibit up to 2 additional features of malignancy. 14 CPC shows frank features of malignancy as listed in Table 1. 14 Additional features recorded included brain invasion, intraventricular and subarachnoid spread, ependymal changes, hydrocephalus, and abundant tumor-associated collagenous stroma (desmoplasia). Since benign CPP may seed neoplastic cells into the cerebrospinal fluid (CSF), distant tumor spread (drop metastasis) and minimal brain invasion are not canonically used to differentiate CPP from CPC. 14 The mitotic count was performed with a light microscope using a 40× objective and lenses with a field number of 20 (area per field of 0.196 mm2). Tumor size was measured in millimeters along the major axis.
This study classified tumor-associated angiogenesis into either IVD, characterized by an increased number of vessels with a single lumen and normal vessel wall anatomy, or GMVP, characterized by a glomerulus-like tortuous aggregation of blood vessels. 5 The presence/absence of IVD and GMVP in the tumor stroma and within the parenchyma adjacent to the tumor were evaluated.
Western Blot
Normal choroid plexus, lung, and brain (VEGF, PDGF, PDGFRα) were used to test the cross-reaction of antibodies with canine tissues. Each sample consisted of 100 mg of tissue mechanically homogenized in 900 ul of RIPA buffer (sodium deoxycholate 0.1%, 5 mM Tris-HCl pH 7.2, 15 mM NaCl, 1 mM EDTA pH 8, 0.005% SDS, 0.1% NP-40) to obtain 10% (w/v) tissue homogenates. Homogenates were centrifuged at 1000 g for 5 minutes to pellet cell debris. Supernatants were resuspended in Laemmli buffer and heated at 99°C for 5 minutes. Protein electrophoresis was done at 150 volts for 85 minutes using 15% sodium dodecylsulphate–polyacrylamide (SDS-PAGE) gels for VEGF/PDGF and 8% SDS-PAGE gels for platelet-derived growth factor receptor alpha (PDGFRα) and platelet-derived growth factor receptor beta (PDGFRβ). Proteins were transferred to nitrocellulose membranes by applying 85 volts for 1 hour. Nonspecific binding sites were blocked with 5% milk solution for 1 hour, and proteins were probed overnight at 4°C with antibodies against PDGF, PDGFRα, PDGFRβ, and VEGF (Suppl. Table S1). Proteins of interest were detected by incubation with a secondary HRP-labeled anti-rabbit antibody diluted at 1:3000 (DAKO P0448) for 30 minutes. Binding sites were revealed with a commercial chemiluminescent substrate (ECL prime, GE Healthcare Life Sciences). A membrane without the primary antibody was used as negative control. Western blot membranes were imaged with the LAS3000 luminescent image analyzer (Fuji) and the AIDA software package (Raytest, Version 5.1).
Immunohistochemistry
Twenty-five canine CPTs and normal choroid plexuses from 6 non-neurologic dogs were immunohistochemically tested for alpha smooth muscle actin (αSMA), Von Willebrand factor (vWF), Ki67, VEGF, PDGF, PDGFRα, and PDGFRβ (see Suppl. Table S1 for antibody details) to label smooth muscle and endothelial cells and proliferating cells and evaluate the expression of growth factors, respectively. Slides contained tumor tissue or NCP plus surrounding brain parenchyma. Slides were deparaffinized in xylol and rehydrated after immersion in different gradients of ethanol. The endogenous peroxidase was blocked by immersion in 3% H2O2 in methanol for 30 minutes at room temperature. For the detection of PDGF, PDGFRα, PDGFRβ, and vWF, enzymatic antigen retrieval consisted of digestion with 50 μl Proteinase K (19.2 mg/ml, Roche) in 50 ml TBS for 15 minutes in a water bath at 37°C. For the detection of Ki67, αSMA, and VEGF, heat antigen retrieval was performed by boiling tissue slides in pH 6.0 citrate buffer at 95°C (Ki67, αSMA: 20 minutes, VEGF: 45 minutes) using a laboratory microwave, followed by cooling at room temperature for 20 minutes. All tissue sections were incubated with the primary antibodies overnight at 4°C. Binding sites were revealed using the streptavidin-biotin-peroxidase reaction (LSAB DAKO Kit K500) and 3-amino-9-ethylcarbazole (AEC) as the chromogen (DAKO Kit K500). Slides were counterstained with Ehrlich’s haematoxylin. As negative control, the primary antibody was replaced with an irrelevant, isotype-matched antibody to control for nonspecific binding of the secondary antibody. Positive control canine tissue to test the specificity and sensitivity of antibodies included a lymph node (anti-Ki67), brain vessels (anti-PDGFRβ), and lung (for anti-PDGF, anti-PDGFRα, anti-VEGF, anti-αSMA, and anti-vWF).
Expression of PDGF, PDGFRα, PDGFRβ, and VEGF was evaluated in neoplastic and healthy choroid plexus epithelium if available, GMVP, IVD, and normal vessels of the brain following a subjective scoring system established by Nupponen et al. 18 Accordingly, the percentage of immunostained cells was semiquantitatively determined as follows: 0 (absent), 1 (<10%), 2 (10%–50%), 3 (51%–80%), and 4 (81%–100%), and the intensity of the immunostaining as 0 (absent), 1 (weak), 2 (distinct), and 3 (strong).
The immunolabeling of αSMA and vWF in GMVP and IVD was qualitatively recorded as either present or absent. The Ki67 proliferative index (PI) (ratio of Ki67-positive nuclei from the total number of nuclei present) was determined by examining 10 high power fields (0.237 mm2) in CPT and normal choroid plexus and from the evaluation of at least 100 cells in GMVP, using the cell counter plugin in the ImageJ software (http://imagej.nih.gov/ij/). Unfortunately, in IVD or normal blood vessels, a comparable number of cells could not be evaluated due to the small number of cellular nuclei (less than 100), and therefore, Ki67 positivity was assessed by counting individual cells. The immunohistochemical slides were evaluated by a blinded single examiner (L.V.M.) and cross-checked by a second examiner (A.O).
Statistical Analysis
The statistical analysis was performed with the GraphPad Prism 5 statistical software (GraphPad Software, Inc.). Because data were not normally distributed, continuous data were expressed as median value and analyzed with the nonparametric Mann-Whitney test to compare 2 groups and with the nonparametric Kruskal-Wallis test followed by Dunn’s multiple comparisons test to compare more than 2 groups. Categorical data were examined either with the chi-square or Fisher’s exact test depending on the number of groups and the sample size. A P value ≤.05 was considered as significant.
Results
The Proliferative Index Correlates With Histological Grade
This study identified 22 of 52 (42%) CPP and 30 of 52 CPC (58%) in dogs by following the human WHO histological classification of CPT. 14 No canine aCPP was diagnosed. Histological features of NCP, CPP, and CPC are shown in Suppl. Figs. S1, S2, and S3. Anatomic localization and histological features are given in Table 2. No primary site was detected in the brain in 5 cases with multiple spinal subarachnoid masses.
Size, Location, and Histological Features of Canine Choroid Plexus Tumors.
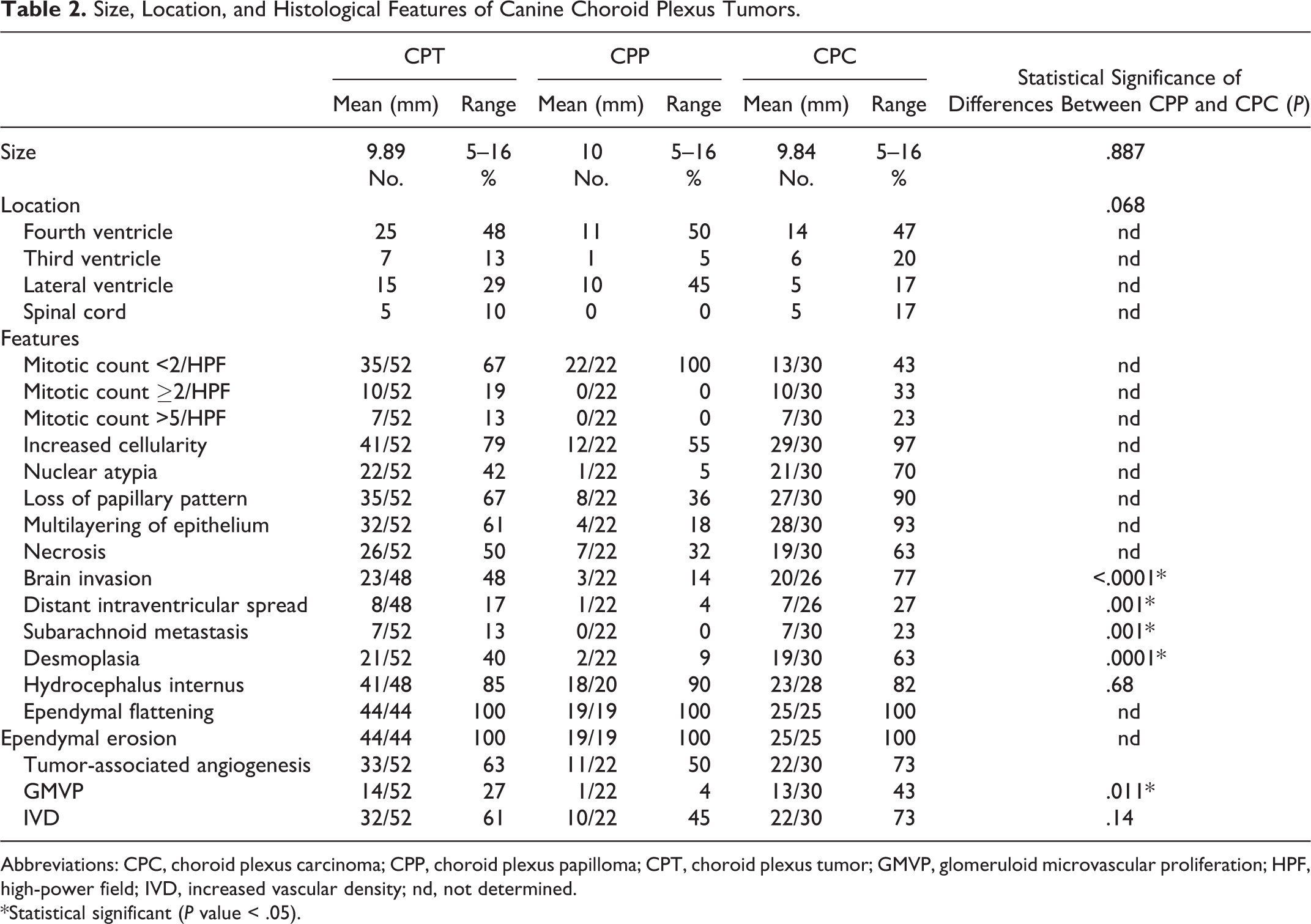
Abbreviations: CPC, choroid plexus carcinoma; CPP, choroid plexus papilloma; CPT, choroid plexus tumor; GMVP, glomeruloid microvascular proliferation; HPF, high-power field; IVD, increased vascular density; nd, not determined.
*Statistical significant (P value < .05).
Desmoplasia was more frequent in CPC (65%) (Suppl. Fig. S3) than CPP (9%; P = .0001 Fisher’s exact test). Similarly, brain invasion (CPC = 77%; CPP = 22%; P < .0001 Fisher’s exact test) and intraventricular (CPC = 27%; CPP = 4%) and subarachnoid (CPC = 23%; CPP = 0%) spread (P = .001 Fisher’s exact test) were more prevalent in CPC. In contrast, the histological grade was not associated with tumor size or location. Additionally, the prevalence of hydrocephalus internus was similar in CPP (90%) and CPC (82%; P = .68 Fisher’s exact test), and ependymal changes (flattening and erosion) were present in all CPTs.
While Ki67 was only expressed in 0.15% (range, 0%–0.37%) of normal choroid plexus epithelial cells, the PI was higher in neoplastic choroid plexus epithelium (Suppl. Figs. S4–S6). The Ki67 PI was higher in CPC than CPP (P = .0033, Mann-Whitney t test), with 5.14% positive cells (range, 1.55%–24.75%) in CPC compared to 0.94% cells (range, 0.59%–1.64%) in CPP (Fig. 1).
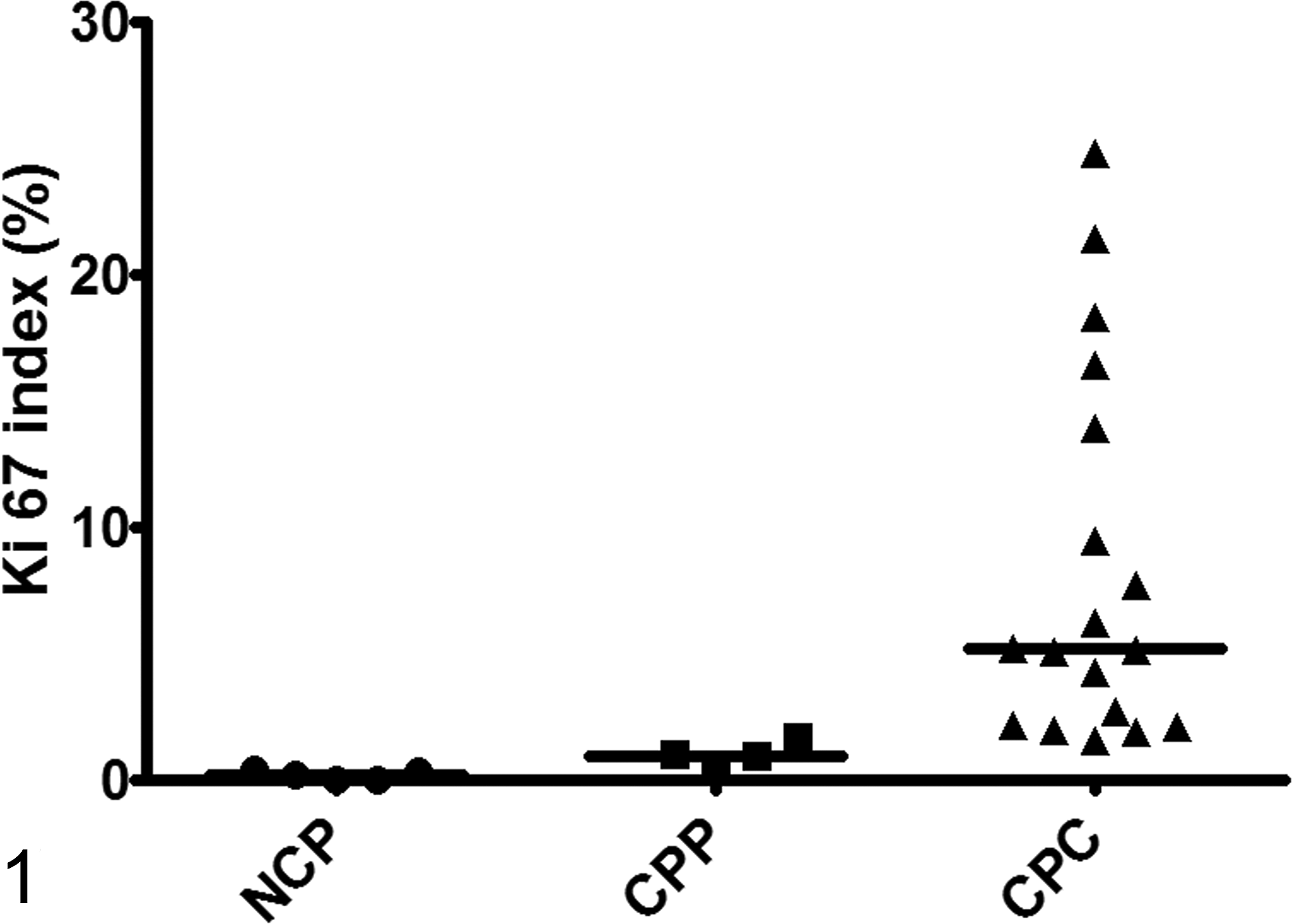
Proliferation indices in normal choroid plexus (NCP), choroid plexus papilloma (CPP), and choroid plexus carcinoma (CPC). Ki67 index is markedly increased in CPC compared to CPP and NCP. The scatterplot shows the individual data points as dots (NCP), squares (CPP), and triangles (CPC). The horizontal line bar indicates the median value.
Tumor-Associated Angiogenesis
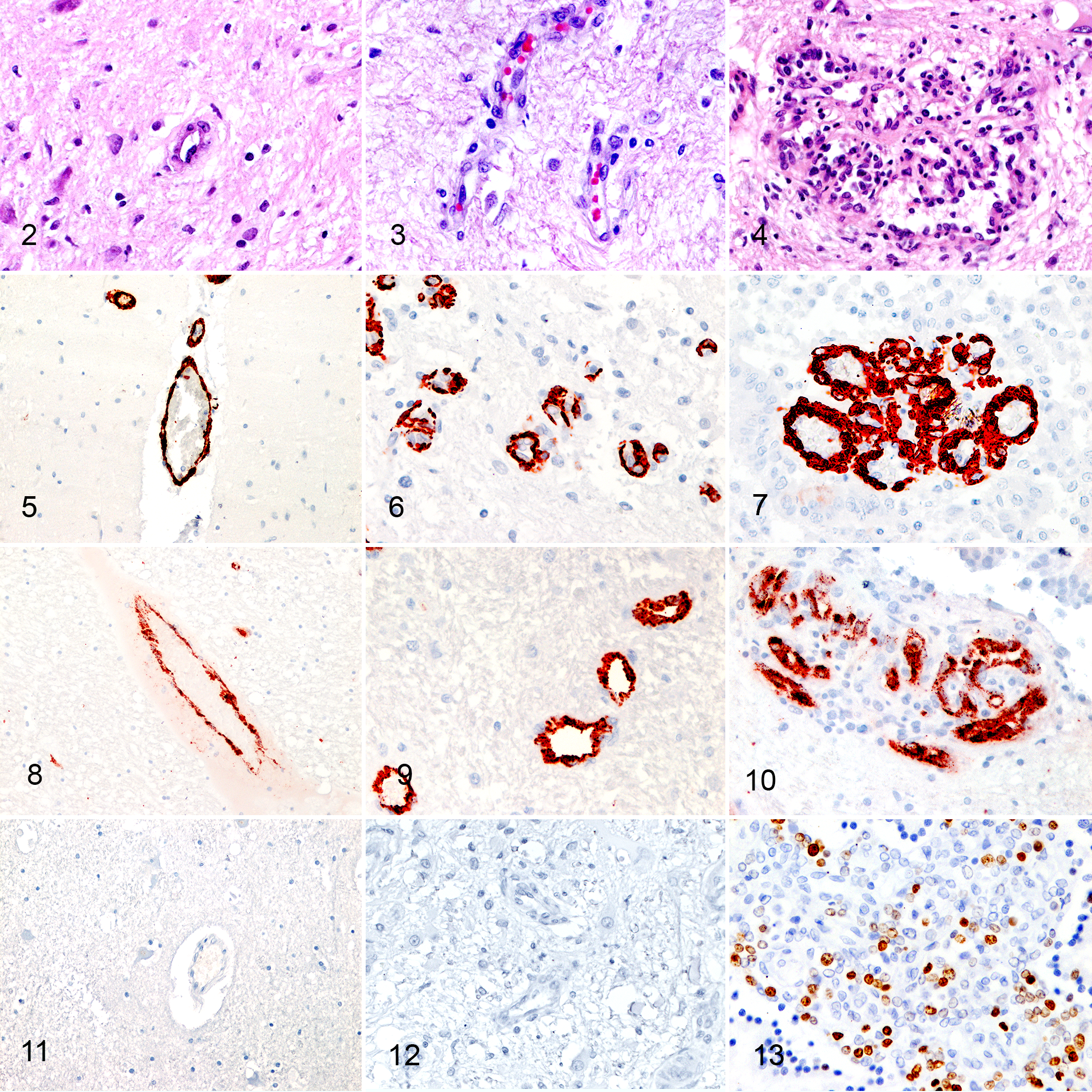
Tumor-associated angiogenesis was observed in 63% of CPT and occurred in 2 forms: IVD (61%) and GMVP (27%) (Figs. 2–4) (Table 2). While IVD was not associated with tumor grade (P = .14, Fisher’s exact test), GMVP occurred more frequently in CPC (43%) compared to CPP (4%) (P = .011, Fisher’s exact test). All tumors with GMVP exhibited concurrent IVD. Both IVD and GMVP were consistently observed in the subependymal brain parenchyma adjacent to the tumor and occasionally within the tumor stroma. In 5 cases, extensive GMVP was observed in the subependymal parenchyma along the ventricular system at sites distant from the primary tumor and occasionally lining the entire ventricular system of the brain.
GMVP occurred more frequently in CPT with necrosis (46%) than tumors without necrosis (12%) (P = .012, Fisher’s exact test). However, necrotic areas had no apparent spatial relationship (co-localization) with GMVP. No association was found between increased vascular density and necrosis (P = .17, Fisher’s exact test). In addition, tumor-associated angiogenesis was not associated with tumor size (P = .06 Mann-Whitney test) or hydrocephalus (P = .39 Fisher’s exact test). Of the histological features occurring most frequently in CPCs, only GMVP was associated with tumoral brain invasion (P = .024, chi-square) and PI (P = .006, Mann-Whitney test) but not with spread along the CSF spaces. Desmoplasia was not associated with any of these features.
All endothelial cells regardless of the types of vasculature expressed αSMA in mural cells (Figs. 5–7). Similarly, all types of vasculature were positive for vWF (Figs. 8–10). However, compared to normal vasculature and IVD, GMVP vessels were lined by multiple layers of αSMA positive mural cells. Additionally, while normal vessels and IVD expressed Ki67 in single cells only (Fig. 11, Fig. 12), GMVP showed a PI of 5.75% in mural and endothelial cells (range, 0%–8.5%) (Fig. 13).
Intratumoral and Vascular Expression of Growth Factors and PDGF Tyrosine Kinase Receptors
Antibodies against VEGF, PDGF, PDGFRα, and PDGFRβ produced bands at the expected molecular weights in Western blot analysis and a specific signal in paraffin-embedded control tissues indicating their cross-reaction with the canine antigens. Anti-PDGFRα did not produce any specific WB band in the brain, while with anti-PDGF a specific band was lacking in NCP (Suppl. Figs. S7–S10).
Expression of growth factors and receptors were immunohistochemically assessed in 6 NCP and 25 CPT (6 CPP, 19 CPC). Generally, growth factors and their receptors were expressed in choroid plexus tumors independently of the grade. No association was found between the expression of angiogenesis factors and the Ki67 labeling index, the size of the neoplasm, or the presence of IVD and GMVP.
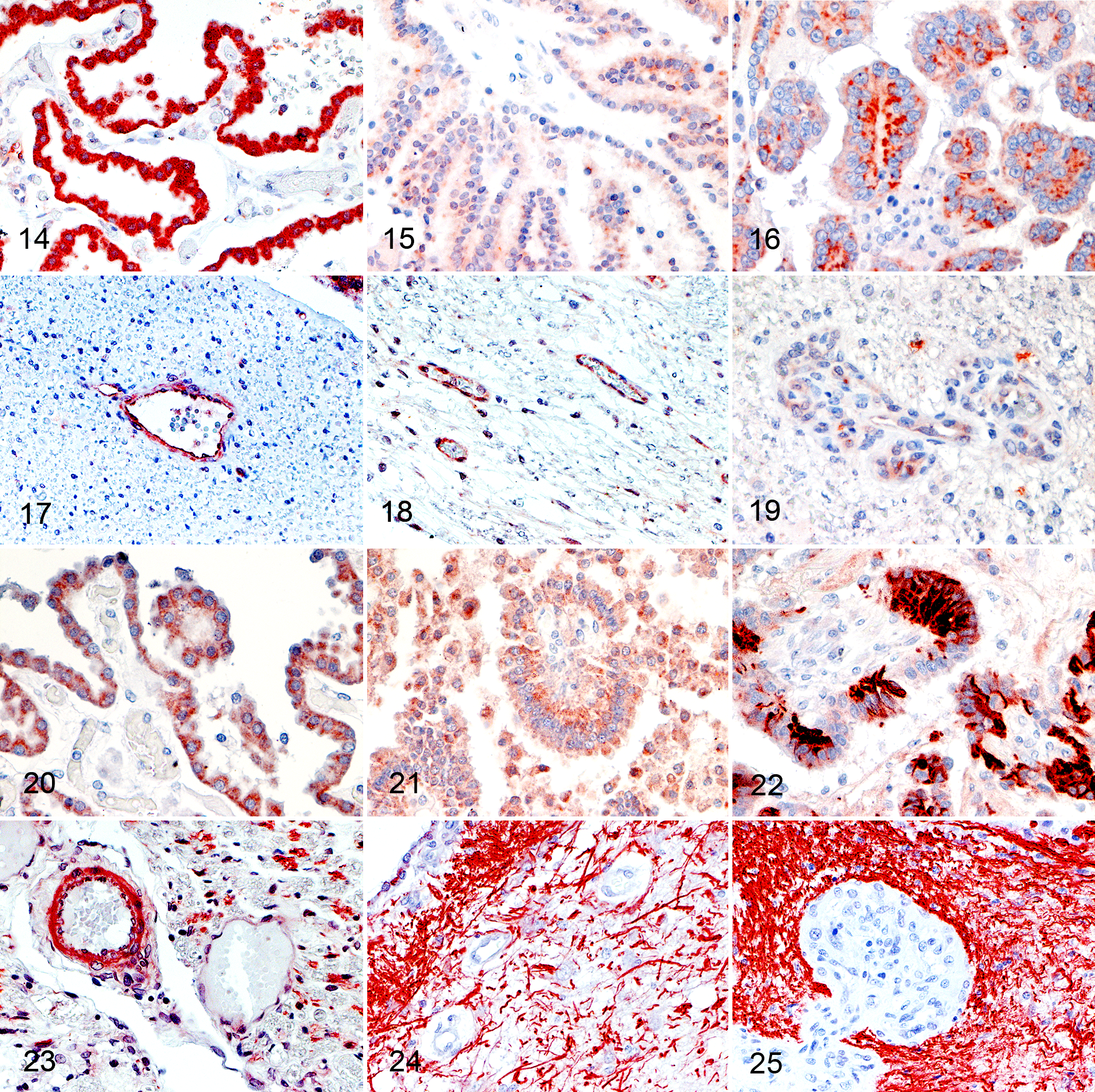
VEGF was strongly expressed in the cytoplasm of normal choroid plexuses (Fig. 14). Similarly, VEGF was intracytoplasmically expressed in most CPT (Figs. 15, 16). However, 12% of tumors were negative for VEGF, and immunohistochemically, the intensity of VEGF expression was lower in CPP (P = .018, Kruskal-Wallis test) and CPC (P = .004, Kruskal-Wallis test) than NCP. No difference in expression intensity was observed between CPP and CPC (Table 3).
Immunohistochemical Expression of Growth Factors and Tyrosine Kinase Receptors in Normal Choroid Plexuses, Choroid Plexus Papilloma, and Choroid Plexus Carcinoma.a
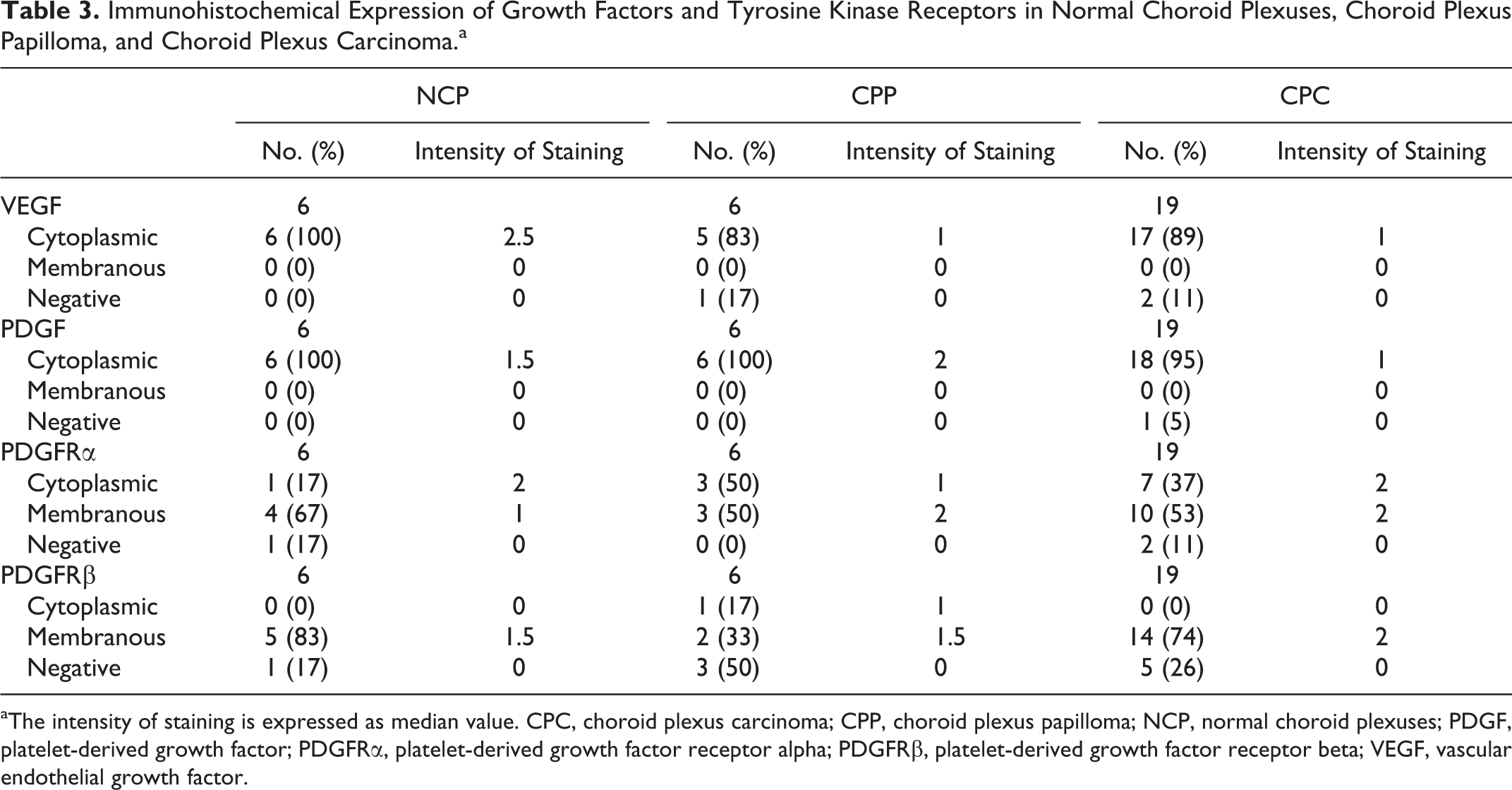
aThe intensity of staining is expressed as median value. CPC, choroid plexus carcinoma; CPP, choroid plexus papilloma; NCP, normal choroid plexuses; PDGF, platelet-derived growth factor; PDGFRα, platelet-derived growth factor receptor alpha; PDGFRβ, platelet-derived growth factor receptor beta; VEGF, vascular endothelial growth factor.
In normal brain capillaries, pericytes and endothelial cells expressed VEGF (Fig. 17). Expression intensity was apparently lower in pericytes and endothelial cells of IVD (Fig. 18) and GMVP (Fig. 19) than normal capillaries but did not reach statistical significance (P = .095 and P = .095, Kruskal-Wallis test).
Two types of immunohistochemical staining patterns were observed with PDGF. Both normal and neoplastic choroid plexus epithelium exhibited similar diffuse cytoplasmic PDGF expression, which ranged between weak and moderate (Figs. 20, 21) (Table 3, P > .999, Kruskal-Wallis test for both CPP and CPC). The second staining pattern was characterized by multifocal neoplastic cell clusters with strong cytoplasmic PDGF immunostaining (Fig. 22) in 5 out of 19 CPCs. This staining pattern was not observed in either NCP or CPP. PDGF was expressed by astrocytes in the surrounding parenchyma and mural cells in the majority of normal brain vessels (Fig. 23). In contrast, expression was only rarely observed in tumor-associated vessels (in 2/25 CPTs) (Figs. 24, 25). Also, the intensity of PDGF was higher in normal vessels than GMVP (P = .001, Kruskal-Wallis test), but no differences between normal vessels and IVD were detected (P = .163, Kruskal-Wallis test).
PDGFRα was intracytoplasmically expressed by most NCP (Fig. 26). Additionally, a weak multifocal membranous expression was observed in 50% (Table 3) (Fig. 26). Similarly, cytoplasmic expression was observed in CPTs, and approximately half of these tumors expressed the receptor on the epithelial apical surface (Figs. 27, 28). Although the intensity of expression appeared to be increased in CPPs and CPCs (Figs. 27, 28) (Table 3), the difference was not statistically significant (P = .687 and P = .111, Kruskal-Wallis test). Pericytes of normal capillaries weakly and inconsistently expressed PDGFRα in 24 of 25 cases (Fig. 29), whereas the receptor was not expressed in pericytes of tumor-associated vessels (Figs. 30, 31). Its expression was not picked up by WB of the brain possibly due to the multifocal and weak expression of PDGFRα in vessels (Suppl. Fig. S9).
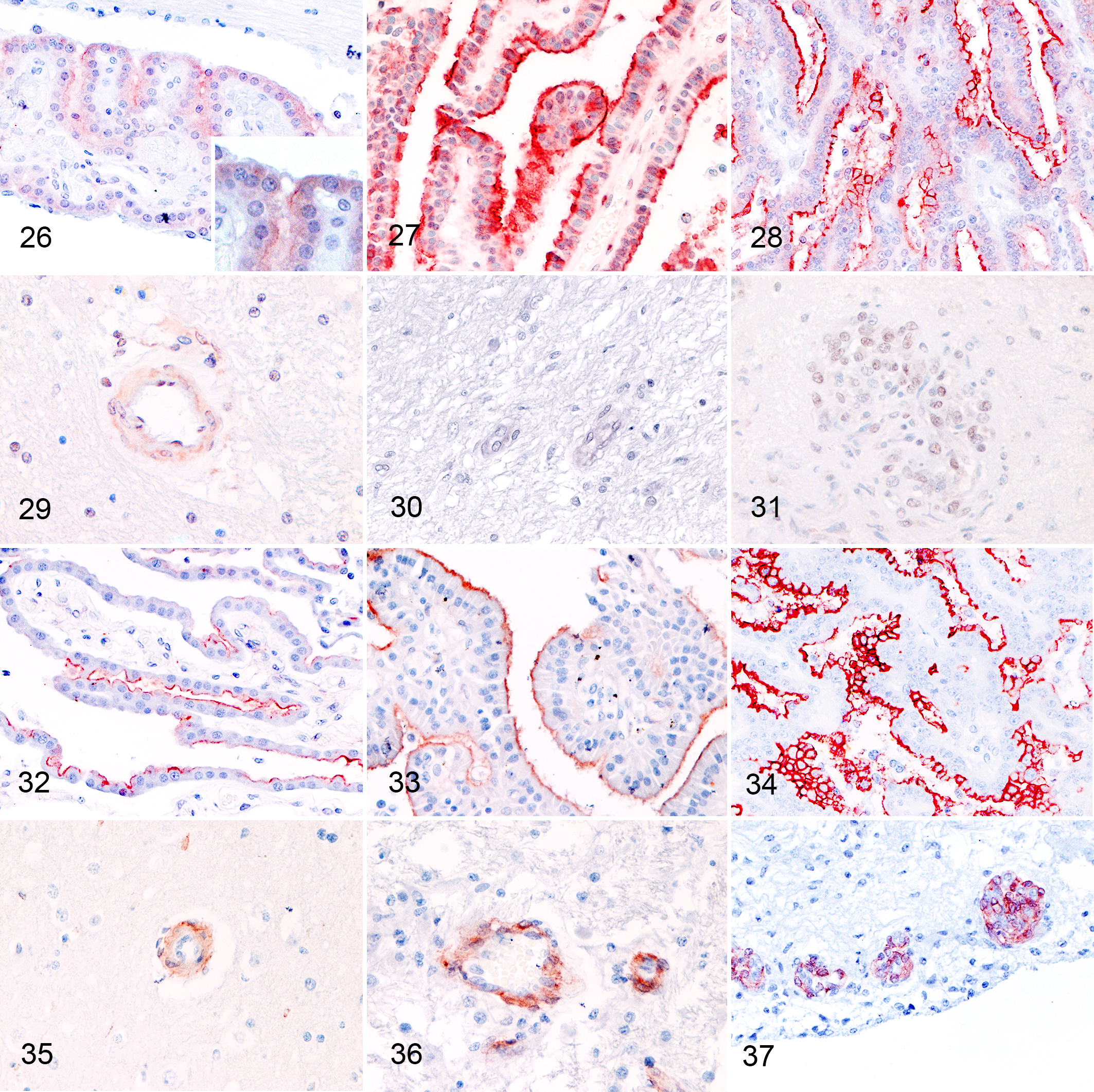
PDGFRβ was expressed on the apical membrane of normal choroid plexuses and CPT. Expression intensity of PDGFRβ was subjectively higher in CPP and CPC than NCP (Figs. 32–34), but the differences were not statistically significant (P = .789 and P > .999, Kruskal-Wallis test). PDGFRβ was expressed in pericytes of normal brain vessels and tumor-associated vasculature (Figs. 35–37). The expression was equally intense between normal vessels, IVD, and GMVP (P = .243 and P = .393, Kruskal-Wallis test).
Discussion
Observations of tumor-associated angiogenesis have been previously reported in canine CPT 17,21 and rarely in human CPT 4 with no associated significance. This study shows that GMVP is a prevalent feature found in canine CPCs. Morphologically, GMVP was characterized by tortuous vascular channels encased by thick layers of cells with an increased PI. Mural cells expressing markers of reactive pericytes αSMA and PDGFRβ accounted for the majority, while vWF-expressing endothelial cells provided the minority of cells present in these proliferations. Such findings suggest pericyte abundance in GMVP as reported for human gliomas. 3,28
Based on the human WHO classification of brain tumors, 14,31 which is frequently applied to canine CPT, our study included slightly more CPC than CPP. These results are concordant with previous studies on canine CPT. 10,31 CPC exhibited significantly higher Ki67 indices than CPP indicating that the increased proliferative activity of canine CPC is a potential indicator of malignancy. These data are in line with a recent report on canine CPT 20 and parallel human CPT data showing that the Ki-67 PI is an important prognostic indicator associated with the aggressiveness of human CPT, response to treatment, and clinical outcome. 6,7,23,28,32 Additionally, desmoplasia and GMVP were observed clearly more frequent in CPC than CPP, suggesting that these 3 features could be included in the histological grading system as criteria for malignancy in CPT. Indeed, we found an association between GMVP and brain invasion suggesting that GMVP occurs in tumors with more aggressive behavior but not between GMVP and spread within the CSF spaces. Our observations regarding the occurrence of GMVP are in line with a study of human CPT that reported microvascular proliferation in carcinomas only 4 and previous reports on canine CPTs, which found an increased frequency of vascular proliferation in high grade tumors. 17,21 However, the canine studies also regularly observe vascular proliferation in CPP, which might be due to the fact that these studies did not distinguish between IVD and GMVP. Clearly, all canine CPT studies, including ours, suffer from the low number of tumor cases, and the partially different grading systems applied preclude the comparison of studies. Additionally, it is currently not clear whether the human WHO classification of human brain tumors reflects the biological behavior of canine CPT. Therefore, large-scale prospective studies are needed to ultimately define morphological features that correlate with the biological behavior of canine CPTs to confirm PI, desmoplasia, and GMVP as factors of malignancy.
The pathogenetic mechanism of GMVP formation is a matter of debate. In experimentally induced brain metastases, GMVP originate from preexisting capillaries that are induced to bend and loop their walls following adhesion of tumor cells, and there is no evidence of sprouting. 8 Strikingly, in our case population, tumor-associated angiogenesis was frequently located in the subependymal brain parenchyma, also at distant sites, which does not support induction of GMVP by tumor cell adhesion. There was no association of subependymal GMVP with tumor size, hydrocephalus, or ependymal disruption, which argues against GMVP induction by mechanical stimulation. The subependymal localization of distant GMVP in absence of ependymal disruption at these sites argues for production and secretion of a soluble (growth) factor by CPT into the CSF causing GMVP following permeation through the ependymal lining. Indeed, intradermal GMVP has been experimentally induced by VEGF injections through the mechanism of sprouting angiogenesis. 29 Additionally, the induction of GMVP by VEGF is known in human astrocytomas. 5 In these tumors, hypoxia and necrosis cause VEGF upregulation resulting in vascular hyperplasia including GMVP, 5 and hence, GMVP is frequently observed in the neighborhood of necrosis. In canine CPT, GMVP was more frequent in tumors with necrosis than tumors without, possibly suggesting that either GMVP is induced by hypoxia-induced tissue necrosis or GMVP are leaky, nonfunctional vessels causing hypoperfusion of neoplastic cells. However, unlike GMVP in gliomas, we did not observe any clear topographic association between GMVP and necrosis, making a causal link between these 2 events in CPT unlikely. In our study, VEGF was expressed in both normal canine choroid plexus epithelium and CPT cells, yet CPT exhibited only low VEGF expression compared to NCP. Additionally, VEGF expression was not associated with intratumoral necrosis or GMVP, and only few endothelial cells were present in GMVP. Altogether, these results indicate that unlike gliomas, GMVP is independent of hypoxia-induced necrosis and VEGF expression in canine CPTs and its pathogenesis remains to be clarified. Similarly, GMVP formation is independent of VEGF expression in human melanoma, endometrial carcinoma, 27 and non–small cell lung cancer. 30 VEGF expression in the normal choroid plexus may be explained by its angiogenic phenotype that ensures the maintenance of endothelial fenestrations by the expression of pro-angiogenic factors. 15,16 The decrease of VEGF expression intensity observed in CPT compared to NCP may therefore suggest tumor dedifferentiation.
Two staining patterns were observed with anti-PDGF. The first pattern was observed in canine NCP and CPT and was characterized by rather weak cytoplasmic granular expression of PDGF. As the antibody did not produce a specific band in the WB analysis of NCP, the signal might be considered a nonspecific reactivity of the antibody. Alternatively, the signal might be too weak to be detected by WB analysis. The second expression pattern was restricted to CPCs (5/19) and consisted of a strong intracytoplasmic signal in multifocal clusters of neoplastic cells, suggesting a possible (over)expression in a subset of malignant tumors, which warrants further investigation. Prospective studies using WB analysis of fresh CPT tissue, not available in our study, are required to correctly interpret the PDGF staining patterns.
The expression of PDGFRα and –Rβ expression in CPT was similar to that in normal plexuses, which is in contrast to human CPT, in which increasing overexpression of the receptors has been found between grades. 11,18,25 PDGFRβ protein overexpression in human CPT is related to PDGFRB gene amplification 18 and associated with a shorter overall survival. 19 We did not observe any association between expression of VEGF, PDGFB, PDGFRα, and PDGFRβ and tumor grade or size, suggesting that these pathways are not exclusive growth factor signals for the proliferation of cancer cells and tumor progression in canine CPTs. However, this has to be confirmed by further, more extensive studies.
Physiologically, angiogenesis is promoted by various growth factors. 12 A well-known signaling pathway operating the endothelial-pericyte cross-talk is the PDGF/PDGFR–dependent pathway during wound healing and in tumors. 22 In the course of angiogenesis, PDGF is produced by sprouting endothelial cells and activates the receptor β on pericytes, suggestive of a paracrine mode of interaction between these 2 cell types. 2,3,22 PDGF promotes the recruitment and proliferation of pericytes precursor cells. 22 In our tumor population, PDGF was basically not expressed in tumor vessels but in neoplastic epithelium of a few CPC, representing a potential source of PDGF that may act on PDGFRβ expressing pericytes of intratumoral and intracerebral vessels and stimulating their proliferation. However, no association was observed between strong tumoral PDGF expression and GMVP.
In conclusion, the histological grade of canine CPTs correlates with higher Ki67 proliferative indices, and the prevalence of GMVP and desmoplasia is strikingly higher in CPC. In contrast, no association was found between IVD and tumor grade. These results suggest that GMVP, desmoplasia, and Ki67 PI might be promising prognostic markers for canine CPT. Large-scale prospective studies based on clinico-pathological correlations and survival rates of canine patients with CPT should confirm our findings. The signaling pathways involved in GMVP of CPT await identification.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985817754124 - Glomeruloid Microvascular Proliferation, Desmoplasia, and High Proliferative Index as Potential Indicators of High Grade Canine Choroid Plexus Tumors
Supplemental Material, DS1_VET_10.1177_0300985817754124 for Glomeruloid Microvascular Proliferation, Desmoplasia, and High Proliferative Index as Potential Indicators of High Grade Canine Choroid Plexus Tumors by Luisa Vera Muscatello, Giancarlo Avallone, Fabienne Serra, Torsten Seuberlich, Maria Teresa Mandara, Silvia Sisó, Barbara Brunetti, and Anna Oevermann in Veterinary Pathology
Footnotes
Acknowledgements
We thank Camille Monney, Manuela Bozzo, Erika Bürgi, and Evelyne Rohrer for excellent technical support. Furthermore, we are grateful to Istvan Vajtai and Daniela Gorgas for constructive discussions.
Author Contribution
Brunetti B, Oevermann A, equal contribution.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
