Abstract
Chlamydia pecorum causes subclinical infections in cattle, but sporadic, bovine encephalomyelitis cases have been reported in calves and documented in two instances in European buffalo. An outbreak of Chlamydia pecorum-induced encephalomyelitis and serositis occurred in 3-month-old buffalo calves from Brazil. Initially presenting with pelvic limb incoordination, the calves progressed to lateral recumbency, depression, and death. Necropsies of two calves revealed encephalomyelomalacia, fibrin deposition on the external surface of the pericardium (case 1) and pleural and pericardial fibrosis (case 2). Microscopically, a multifocal to coalescing, necrotizing, neutrophilic and lymphocytic meningoencephalomyelitis with fibrinoid vasculitis and thrombosis was present. Anti-Chlamydia antibody labeling was demonstrated by immunohistochemistry. Bacteriological examination yielded no pathogenic bacteria in the brain or lungs. Chlamydia pecorum was confirmed by PCR. This work describes the gross, histopathological, microbiological, and molecular findings in two cases from an outbreak of Chlamydia pecorum-induced disease in buffalo calves.
The genus Chlamydia comprises a group of gram-negative, obligate intracellular bacteria that can infect a wide range of hosts, including domestic and wild animals.2,11 Chlamydia species exhibit varying degrees of virulence, leading to a spectrum of outcomes from asymptomatic infections to severe systemic diseases. 2
In cattle, C. abortus and C. pecorum are the most common chlamydial species. 17 Infections caused by C. pecorum are typically subclinical, but occasionally manifest as arthritis, 19 abortion,6,9 enteritis, 14 pneumonia, 20 encephalomyelitis, or serositis.7,8,18 Bovine sporadic encephalomyelitis, associated with C. pecorum and documented since 1940, presents with encephalomyelitis, vasculitis, and thrombosis.7,8,12,18 Buffalo cases are rare, with only reports in Bulgaria 13 and Italy. 5 This present report aims to describe the gross, histopathological, microbiological, and molecular findings in two cases from an outbreak of encephalomyelitis and serositis caused by Chlamydia pecorum in Brazilian buffalo calves.
The outbreak occurred between March and April 2022 when four, 3-month-old Murrah water buffalo calves (Bubalus bubalis) belonging to a dairy farm in the municipality of Bandeirantes (19°55'04"S 54°21'50"W), Mato Grosso do Sul state, displayed clinical signs of incoordination in their pelvic limbs, which progressed to lateral recumbency, depression, and death. These animals were part of a group of 40 calves housed in a 100 m2 paddock. They were weaned at 3 months of age and received milk during the day, along with concentrate feed at night. All the affected animals received treatment, including antibiotics from the cephalosporin group, corticosteroids, vitamin B1, and Mercepton (Laboratório Bravet LTDA); however, only one of the animals showed clinical improvement. Two of these buffalo calves (cases 1 and 2) were referred to the Pathological Anatomy Laboratory at the Federal University of Mato Grosso do Sul (LAP/UFMS). Case 1 had been in recumbency for 3 days, with reduced pelvic limb reflexes and 8% dehydration. One day after the initial clinical intervention the animal died and was subsequently subjected to necropsy. Ten days after the necropsy of case 1, case 2 was admitted to the veterinary hospital with a history of pelvic limb incoordination, progressing to lateral recumbency and depression within 4 days. The clinical examination revealed the absence of a threat response and swallowing reflex, right eyelid ptosis, and decreased tone in the tongue and tail. The blood count showed a mild thrombocytosis (683,000/mm3; reference interval [RI], 253,790–516,830/mm3). Biochemical tests revealed elevated serum levels of aspartate aminotransferase (720.6 UI/L; RI, 104–191.6 UI/L), creatine kinase (8353 UI/L; RI, 88–292 UI/L), creatinine (3.2 mg/dL; RI, 0.4–0.9 mg/dL), and urea (141 mg/dL; RI, 7–19 mg/dL). 4 Cerebrospinal fluid analysis revealed marked neutrophilic pleocytosis and elevated globulins. Because of the poor prognosis, the animal was euthanized and necropsied.
On gross examination, both buffaloes had distended urinary bladders filled with urine. Case 1 had a moderate amount of fibrin deposited on the external surface of the pericardium, while case 2 had thickening of the pericardium with whitish material and white strands attached to the pleura (chronic pleuritis). In some areas, the interstitial septae of the lungs were expanded by fibrosis, and the pulmonary parenchyma had multifocal dark red areas of atelectasis (Fig. 1a). In both cases, the fresh brain had slight softening of the telencephalons and diffuse congestion of leptomeningeal blood vessels. Following fixation in formalin, sections of the spinal cord, telencephalon, and brainstem revealed multifocal dark red areas associated with loss of the parenchyma and the formation of variably sized cavities (Fig. 1b).
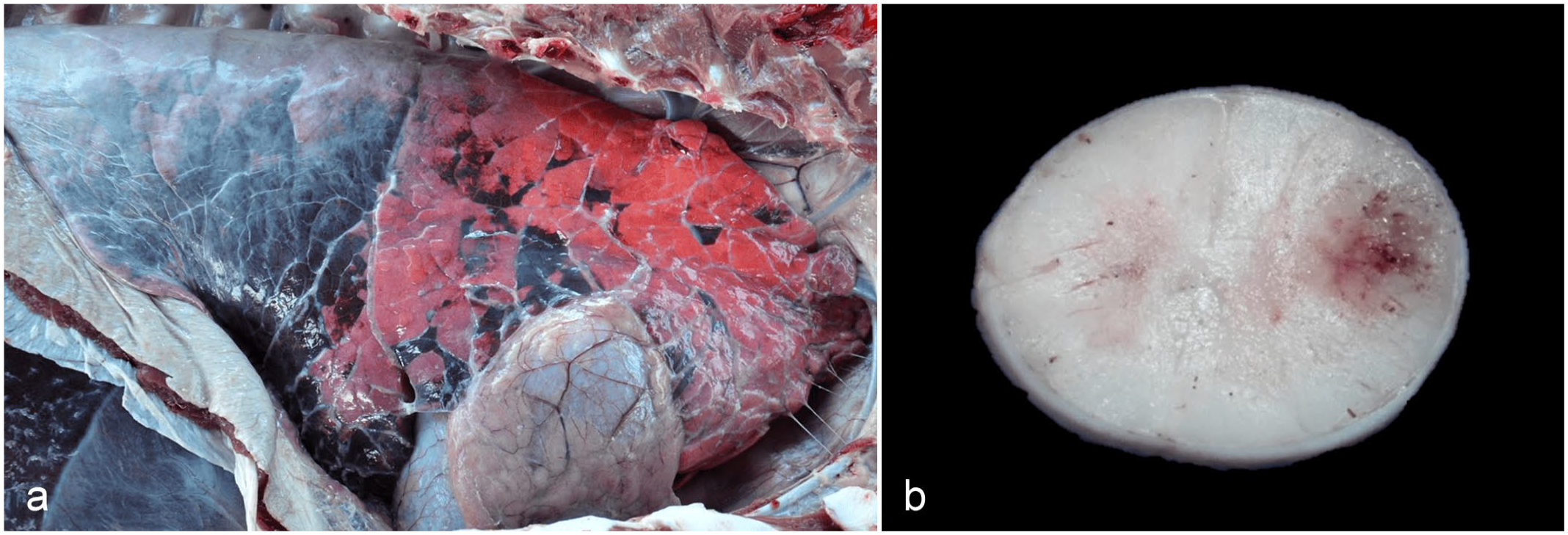
Gross lesions in buffalo calves associated with Chlamydia pecorum infection. (a) Right lateral view of the thoracic cavity. There is multifocal white material (fibrosis/fibrous granulation tissue) on the pleural and pericardial surfaces; a few fibrous/fibrinous strands adhere the visceral and parietal pleura (bottom right corner). Irregular, multifocal dark red areas are observed in the cranial lobes that are diffuse in the caudal lobes (atelectasis). Case 2. (b) Cross-section of the cervical spinal cord after 24 hours of fixation in formalin. There is a focally extensive red area with small cavitations (malacia) that mostly affects the white matter. Case 1.
During necropsy, samples of various organs were collected in 10% formalin and processed according to standard procedures for histopathological evaluation. Formalin-fixed, paraffin-embedded sections of the spinal cord, brain, lungs, and heart of the two buffaloes were subjected to immunohistochemistry (IHC) for Chlamydia spp. (Supplemental Method S1).
Histologically, the pericardium and pleura of case 1 was thickened due to the presence of fibrin and a marked inflammatory infiltrate composed of macrophages, lymphocytes, plasma cells, and cellular debris (Fig. 2a). In case 2, there was thickening of the pleura due to fibrosis and activated mesothelial cells, as well as endothelial thickening in proliferated blood vessels. In the central nervous system, lesions were observed in all parts of the brain and spinal cord, with more pronounced lesions in the cerebral cortex and cervical spinal cord.
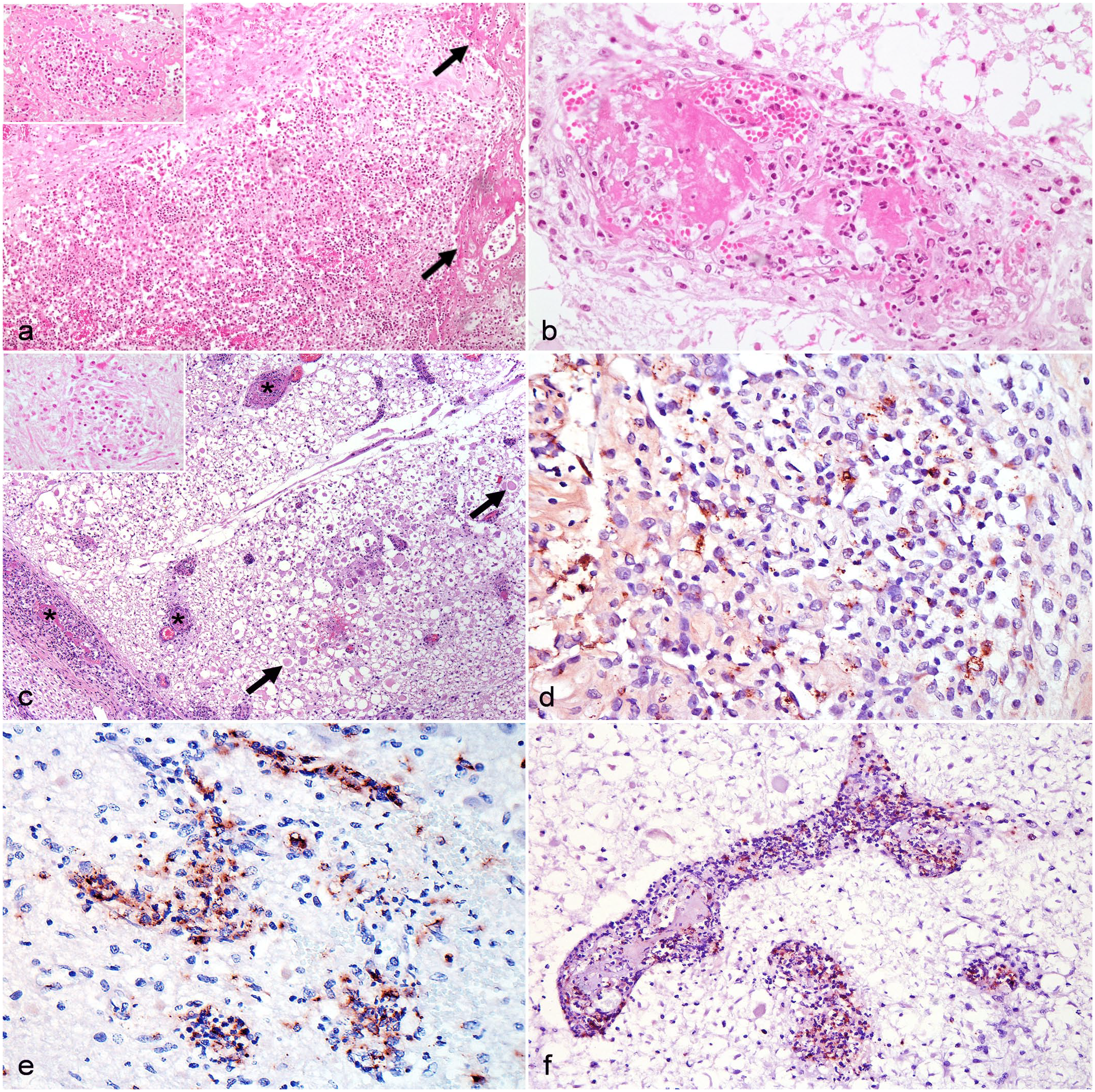
Histological and immunohistochemical aspects of encephalomyelitis and fibrinous pericarditis caused by Chlamydia pecorum in buffalo calves. (a) Pericardium. There is thickening by fibrin (arrows) and a marked mononuclear inflammatory infiltrate (inset). Case 1. Hematoxylin and eosin (HE). (b) Spinal cord. The wall of the blood vessel is infiltrated by degenerate neutrophils and discrete mononuclear cells and is associated with a central fibrin thrombus obliterating the lumen and vessel wall. Case 2. HE. (c) Cervical spinal cord. Multiple blood vessels are occluded by fibrin thrombi (*). There is extensive necrosis of the adjacent neuroparenchyma with vacuolization of the white matter and axonal swellings (spheroids, arrows). Case 2. HE. Inset: glial and inflammatory cell infiltration in the neuroparenchyma. Case 2. HE. (D) Pericardium, immunolabeling for Chlamydia spp, in the cytoplasm of macrophages. Case 1. Immunohistochemistry (IHC). (E) Brain, immunolabeling for Chlamydia spp. in the inflamed neuroparenchyma. Case 1. IHC. (F) Spinal cord, immunolabeling for Chlamydia spp. in endothelial cells and macrophages within the lumen of the blood vessels. Case 2. IHC.
The walls of multiple blood vessels, particularly in the white matter and leptomeninges, were completely or partially replaced by eosinophilic fibrillar material (fibrinoid necrosis) along with an inflammatory infiltrate comprising degenerate neutrophils and, to a lesser extent, macrophages, lymphocytes, and occasional plasma cells (Fig. 2b). Adjacent to these vessels, areas of necrosis were identified, characterized by intense white matter vacuolation, axonal spheroids, hemorrhage, cellular debris, and frequent obliteration of the vascular lumen due to fibrin thrombi. These areas also contained an infiltrate of neutrophils and lymphocytes (Fig. 2c). Macroscopically and histologically, no lesions were observed in other organs, including the joints.
In the immunohistochemical assessment of both buffaloes, sections of the pericardium and visceral pleura showed multifocal positive chlamydial immunolabeling within the cytoplasm of macrophages (Fig. 2d). Moreover, in sections of the brain and spinal cord, there was pronounced multifocal immunolabeling in the necrotic and inflamed neuroparenchyma (Fig. 2e), particularly within the affected vessels, present both within macrophages and endothelial cells or freely within the vessels (Fig. 2f).
The brain, spinal cord (cervical, thoracic, lumbar, and sacral portions), cerebrospinal fluid, joint capsule, liver, and lung samples of case 2 were processed using standard culture media, including brain heart infusion, MacConkey, and mannitol, with the spinal cord cultured in enriched media (chocolate agar) under reduced O2 atmosphere, 5% CO2, and pH of 7.6. These samples were then incubated at 37°C for a maximum of 72 h. No bacterial growth was observed. Fresh samples from the central nervous system were collected for direct immunofluorescence and biological testing through intracerebral inoculation in mice to test for rabies. These samples were subsequently sent to the Official Diagnostic Laboratory (IAGRO, Mato Grosso do Sul, Brazil). Virological tests for rabies yielded negative results.
Fresh tissues (brain, spinal cord, pericardium, pleura, lung and joint capsule) from case 2 underwent testing at the Molecular Biology Laboratory at UFMS using PCR for the Chlamydiaceae family, after DNA extraction using the DNeasy blood & tissue kit (Qiagen). The primers used were CHYF (5′-GCCTACCGGCTTACCAAC-3′), and CHYR (5′-GGCGC AATGATTCTCGAT-3′), which amplify a 238 bp fragment of the 23S rRNA gene. Amplification was observed for all tissues analyzed, except for the joint capsule (Supplemental Figure S1). The PCR product underwent DNA sequencing (Supplemental Method S2). The DNA sequence obtained in this study has been deposited in GenBank under the accession number OQ734980.
Formalin and embedded in paraffin blocks tissues (brain and spinal cord) from both cases were positive using a species-specific C. pecorum qPCR targeting a 76 bp sequence within the ompA gene (Supplemental Method S3). 16
Encephalomyelitis and serositis in these buffaloes were diagnosed as C. pecorum-induced based on epidemiological data, clinical signs, gross and histologic findings, immunohistochemistry, molecular identification, and exclusion of other potential causes. Histological and immunohistochemical observations in affected buffaloes mirrored those seen in C. pecorum-infected bovine calves7,8 and aborted fetuses, 18 including mononuclear vasculitis, neutrophil aggregates, thrombus formation, malacia, and endothelial and cytoplasmic immunolabeling in inflammatory cells of the brain, spinal cord, pleura, and pericardium. Similar lesions in the serosa of the abdominal organs are also described.7,8
The buffaloes’ ages, clinical presentation, and morbidity rates mirrored those reported in prior cattle3,7 and buffaloes’ cases.5,13 Calves were primarily affected, with few instances of disease in adults. Clinical signs included limb paralysis, prolonged recumbency, and depression, with morbidity rates around 10%. Hematological and serum biochemical findings were not detailed in previous reports, and were absent in case 1 due to the rapid disease progression. However, minor changes were observed in case 2, such as elevated creatine kinase and aspartate aminotransferase levels, likely due to muscle injuries from prolonged recumbency, along with other parameters indicating severe dehydration of this animal. All brain and spinal cord samples from the two buffaloes were positive in the species-specific C. pecorum qPCR.
Only two prior instances of Chlamydia encephalomyelitis in buffaloes have been documented, occurring in Bulgaria in 197413 and Italy in 1998. 5 The first diagnosis relied solely on serological testing and antibody detection, while the second involved the isolation of C. pecorum from inoculating brain samples from affected calves into cell culture. Hence, this current report stands as the first to diagnose the disease via PCR directly from affected buffalo tissues and immunohistochemistry.
Possible viral causes of neurological lesions in calves include rabies virus, ovine herpesvirus-2 (linked to malignant catarrhal fever), and bovine viral diarrhea virus. Rabies was ruled out based on negative virological tests. Molecular tests for other viruses were not performed, but distinct lesions associated with these diseases were absent. Histological lesions of malignant catarrhal fever in buffaloes resemble those in cattle, including disseminated lymphocytic arteritis-phlebitis and ulcerative lesions in mucosal surfaces from various tissues, with fibrinoid arteritis being uncommon in buffaloes. 15 Concerning bovine viral diarrhea virus, the most commonly reported brain injury in calves with transplacental infection is cerebellar hypoplasia, while meningoencephalitis cases are rare. 1 Although bovine viral diarrhea virus can cause vasculitis/fibrinoid arteriolitis in some tissues, we have not found reports of vasculitis caused by this virus in the central nervous system. Conversely, the bacterium Histophilus somni induces lesions similar to Chlamydia, including vasculitis, thrombosis, suppurative inflammation, and central nervous system malacia, often causing confusion among pathologists. 2 However, it was ruled out through microbiological examination in this case.
Most C. pecorum infections in ruminants are asymptomatic, with clinical diseases influenced by factors like host stress, 2 co-infections, and genotypic variability. 8 Dairy farms, like the outbreak site, share similarities with locations reporting C. pecorum encephalomyelitis in cattle.7,8,18 Dairy production systems pose risks for Chlamydia infections due to factors such as introducing animals from external sources, using breeding bulls, insufficient pen segregation, and poor environmental hygiene. 10 Overcrowding in the affected herd and the lack of separate calving pens may have contributed to the disease outbreak.
This study emphasizes C. pecorum-induced lesions as a differential diagnosis for multifocal necrosuppurative encephalitis with vasculitis, thrombosis, pleuritis, and pericarditis in buffaloes. Pathological, molecular, and immunohistochemical analyses confirm the C. pecorum DNA in affected tissues, providing insights into this pathogen in buffalo livestock.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858241288116 – Supplemental material for Encephalomyelitis and serositis caused by Chlamydia pecorum in buffalo calves from Brazil
Supplemental material, sj-pdf-1-vet-10.1177_03009858241288116 for Encephalomyelitis and serositis caused by Chlamydia pecorum in buffalo calves from Brazil by Larissa L. de Souza, Saulo P. Pavarini, Marcele B. Bandinelli, Nicole Borel, Rayane C. Pupin, Carlos A.N. Ramos, Ricardo A.A. Lemos and Danilo C. Gomes in Veterinary Pathology
Footnotes
Acknowledgements
The authors are grateful to Sandra Schneider, Institute of Veterinary Pathology, Vetsuisse Faculty, University of Zurich, for her support with the laboratory work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support was supplied by Universidade Federal de Mato Grosso do Sul (UFMS/MEC) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Brazil)—Finance Code 001. Two of the authors (Ricardo A.A. Lemos and Saulo P. Pavarini) had a fellowship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
