Abstract
Wild banded mongooses (Mungos mungo) in northeastern Botswana and northwest Zimbabwe are infected with a novel Mycobacterium tuberculosis complex (MTC) pathogen, Mycobacterium mungi. We evaluated gross and histologic lesions in 62 infected mongooses (1999–2017). Many tissues contained multifocal irregular, lymphohistiocytic to granulomatous infiltrates and/or multifocal or coalescing noncaseating to caseating granulomas with variable numbers of intralesional acid-fast bacilli. Over one-third of nasal turbinates examined had submucosal lymphohistiocytic to granulomatous infiltrates, erosion and ulceration of the nasal mucosa, bony remodeling, and nasal distortion. Similar inflammatory cell infiltrates expanded the dermis of the nasal planum with frequent ulceration. However, even in cases with intact epidermis, acid-fast bacilli were present in variable numbers among dermal infiltrates and on the epidermal surface among desquamated cells and debris, most commonly in small crevices or folds. In general, tissue involvement varied among cases but was highest in lymph nodes (50/54, 93%), liver (39/53, 74%), spleen (37/51, 73%), and anal glands/sacs (6/8, 75%). Pulmonary lesions were present in 67% of sampled mongooses (35/52) but only in advanced disseminated disease. The pathological presentation of M. mungi in the banded mongoose is consistent with pathogen shedding occurring through scent-marking behaviors (urine and anal gland secretions) with new infections arising from contact with these contaminated olfactory secretions and percutaneous movement of the pathogen through breaks in the skin, nasal planum, and/or skin of the snout. Given the character and distribution of lesions and the presence of intracellular acid-fast bacilli, we hypothesize that pathogen spread occurs within the body through a hematogenous and/or lymphatic route. Features of prototypical granulomas such as multinucleated giant cells and peripheral fibrosis were rarely present in affected mongooses. Acid-fast bacilli were consistently found intracellularly, even in regions of necrosis. The mongoose genome has a unique deletion (RD1mon) that includes part of the encoding region for PPE68 (Rv3873), a gene co-operonic with PE35. These proteins can influence the host’s cellular immune response to mycobacterial infections, and it remains uncertain how this deletion might contribute to observed patterns of pathology. M. mungi infection in banded mongooses is characterized by both a unique transmission and exposure route, as well as accompanying pathological features, providing an opportunity to increase our understanding of MTC pathogenesis across host-pathogen systems.
Keywords
Mycobacterium mungi is a newly discovered emerging tuberculosis (TB) pathogen within lineage 6 of the Mycobacterium tuberculosis complex (MTC). 2 M. mungi is closely related to Mycobacterium africanum West African 2 (Maf 2), a significant cause of TB among humans in West Africa. 3,20 M. mungi infects banded mongoose (Mungos mungo) populations in northeastern Botswana and northwest Zimbabwe. 4 Banded mongooses are small, cooperative breeding carnivores with a wide distribution in Africa. 4
M. mungi was confirmed as a member of the MTC through full genome sequencing, placing the organism in the lineage 6 wildlife-associated group, 2,3 together with Mycobacterium suricattae (meerkats, Suricata suricatta 22 ), Dassie bacillus (rock hyrax, Procavia capensis 24 ), and chimpanzee bacillus (chimpanzee, Pan troglodytes spp 8 ). Previously, MIRU-VNTR analyses of M. mungi isolates identified substrains within and between mongoose troops and sample years, indicating possible evolution of the organism over time and transmission complexity among mongoose social groups. 2 Evidence presently suggests that the banded mongoose is the definitive host; the full host spectrum, however, remains unknown.
M. mungi has many unique ecological characteristics that set it apart from other members of the MTC complex. First, M. mungi causes significant mortality among banded mongooses, with disease-mediated losses threatening the persistence of smaller social groups experiencing other sources of mortality and group size depression. Second, rather than relying on primary aerosol or oral transmission routes, as is characteristic of most other MTC species, M. mungi is transmitted via an environmental pathway through infected urine and anal gland secretions. 4 These secretions are used in olfactory communication behavior, with pathogen invasion occurring through breaks in the skin, nasal planum, and/or snout of the mongoose host. Such a mode of transmission would allow the pathogen to spread between social groups where territorial behavior would normally limit contact and would potentially allow transmission among troops across a landscape. Third, the disease outbreaks have a seasonal pattern, with onset in the dry season (April to October) and, generally, few cases in the wet season (November to March), 5 although an increasing number of cases are now occurring in the wet season. Last, the period between clinical presentation and death in affected mongooses is generally short (<3 months) compared with other MTC pathogens where infection can last years. 21
Since the first outbreak in 1999, 93% (14/15) of radio-tracked and intensively studied troops are now infected. The prevalence of M. mungi disease within mongoose troops currently ranges from 3% to 17% (7.3% ± 4.4%, mean ± SD) of the total troop, including pups and juveniles, although, historically, disease incidence has been as high as 37.5%. The 15th intensively studied group has not shown any evidence of infection over the past 18 years of observations and is not obviously isolated by any geographical barriers from surrounding troops.
The objective of the present study was to evaluate the pathological presentation of M. mungi infection in the banded mongoose host in relation to the unique mechanisms of pathogen shedding and transmission observed in this emerging MTC species. We compared pathological presentation to other lineage 6, wildlife-associated organisms, Mycobacterium bovis and Mycobacterium tuberculosis, and their hosts and discuss implications to our understanding of the pathogenesis of M. mungi infection.
Materials and Methods
In Chobe District, Botswana (approximately 25.163°E and 17.828°S), banded mongoose troops have been the focus of a long-term study since 2000 after the discovery of M. mungi in 1999. 2 In each troop, one to two animals were radio-collared and troops were monitored weekly for infection and other variables as previously described. 2 Necropsies were performed on opportunistically obtained banded mongoose carcasses found in the study area during this same period (hit by car, attacked by dogs, killed by wildlife management officers, etc). Samples of all major organs and any macroscopically visible lesions were collected and fixed in 10% buffered formalin for histological examination. Tissue availability varied among necropsies according to the condition of the carcass being examined. Formalin-fixed tissue sections were prepared using routine techniques and stained with hematoxylin and eosin (HE). A Ziehl-Neelsen (ZN) staining method was used to visualize acid-fast bacilli (AFB). Infection status was also confirmed by polymerase chain reaction (PCR) as previously described. 4
Ethical Treatment of Animals
All animal handling and sampling activities in this study were conducted under approval from the Virginia Tech Institutional Animal Care and Use committee (IACUC 13-164-FIW), as well as the Botswana Ministry of Environment, Wildlife, and Tourism (EWT 8/36/4 XXVI (24)).
Results
We evaluated gross pathologic and histopathologic findings associated with M. mungi infection in 62 banded mongooses (1999–2017). Positive infection status was determined through gross examination (lesions consistent with mycobacteriosis), histology (presence of AFB), and PCR (detection of M. mungi–specific DNA). At present, there are no antemortem diagnostics available for diagnosis of M. mungi infection. There was no statistical difference in infection levels between males and females (χ2(1, N = 184); P = .32). It was not possible to estimate the percentage of affected mongooses sampled in this study as the population size and total number affected fluctuated over time and could not be accurately estimated. Clinical signs in M. mungi–infected mongooses included fearlessness, lethargy, lagging behind the group, severe weight loss, hunched body posture, matted fur, drooping and/or enlarged testicles, epiphora, nasal distortion, rhinorrhea, and sneezing.
Gross Lesions
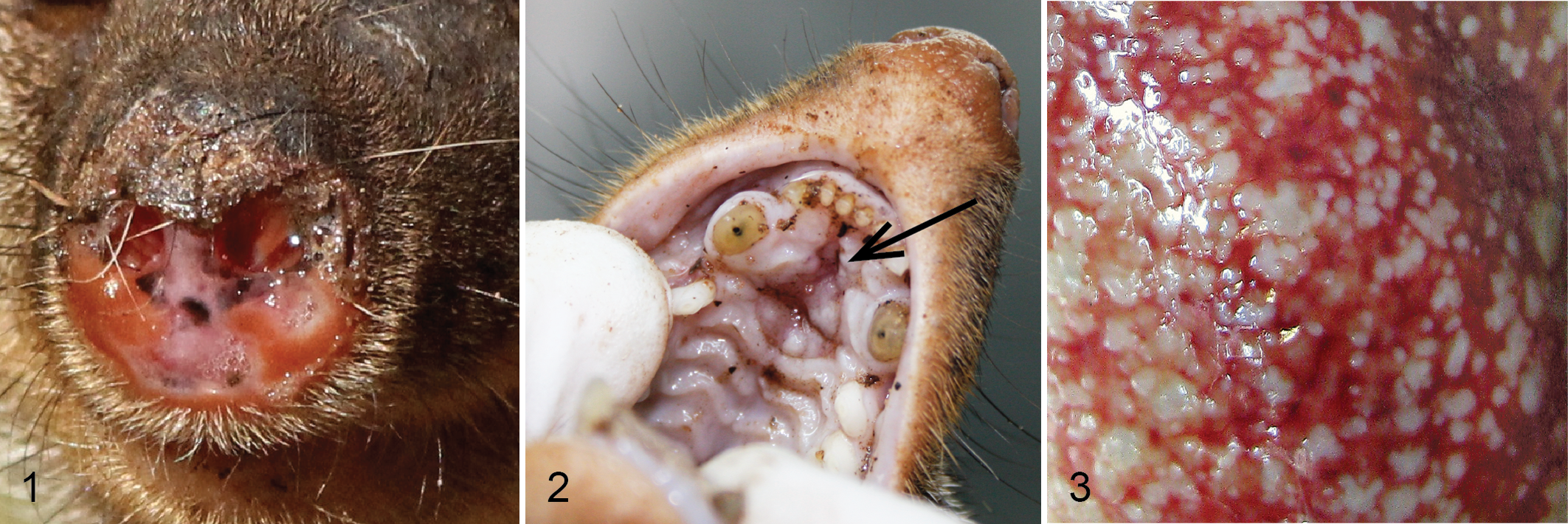
Nasal distortion was commonly identified with occasional erosion of the nasal planum (Fig. 1). Erosion of the hard palate was noted in two cases (Fig. 2). In most animals, lymph nodes were enlarged and pale, with multifocal necrosis and indistinct separation of cortex and medulla. Lungs, liver, spleen, and kidney had focal to multifocal pale nodular to irregular masses (0.1–2 cm in diameter) on the capsular or pleural surface as well as on the cut surface (Fig. 3).
Microscopic Lesions
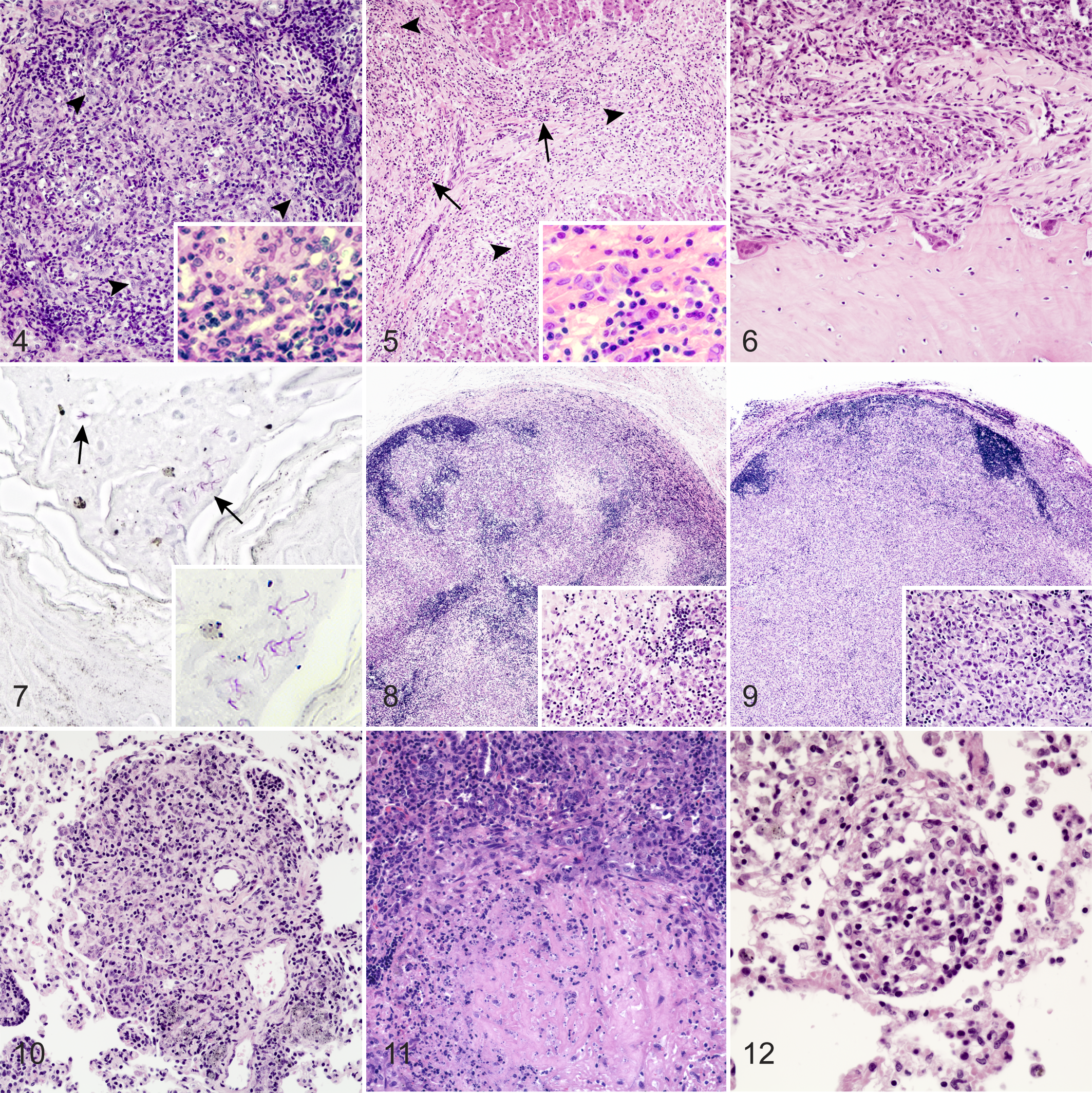
Among the 62 infected mongooses, 806 separate tissues were examined microscopically. Tissue involvement varied among affected mongooses but was highest in lymph nodes (50/54 mongooses, 93%), liver (39/53, 74%), spleen (37/51, 73%), and anal glands/sacs (6/8, 75%; Table 1). Tissue changes were characterized by the presence of irregular lymphohistiocytic (unorganized infiltrates of roughly equal numbers of lymphocytes and macrophages) to granulomatous infiltrates (macrophages and activated epithelioid macrophages with fewer lymphocytes, arranged in variably sized aggregates or sheets) or multifocal to coalescent noncaseating to caseating granulomas. In general, organs (liver, spleen, kidney, adrenal gland, salivary glands, pancreas, stomach, intestines, and bladder) had multifocal, nodular to irregular noncaseating granulomas with infiltrates of macrophages, epithelioid macrophages, lesser numbers of lymphocytes, and rare multinucleated giant cells arranged in round to oval fashion (Fig. 4). Outermost regions of the nodular arranged infiltrates were more lymphocyte rich than central regions. In other cases, mild to moderate central necrosis was identified (caseating granuloma). Mineralization and peripheral fibrosis were absent. Three affected livers had multifocal, bridging-portal, lymphohistiocytic inflammation with mild to moderate fibrosis (Fig. 5). In all tissues, AFB were variable in number.
Lesion Distribution in 62 Mycobacterium mungi–Infected Banded Mongoose (Mungos mungo).a
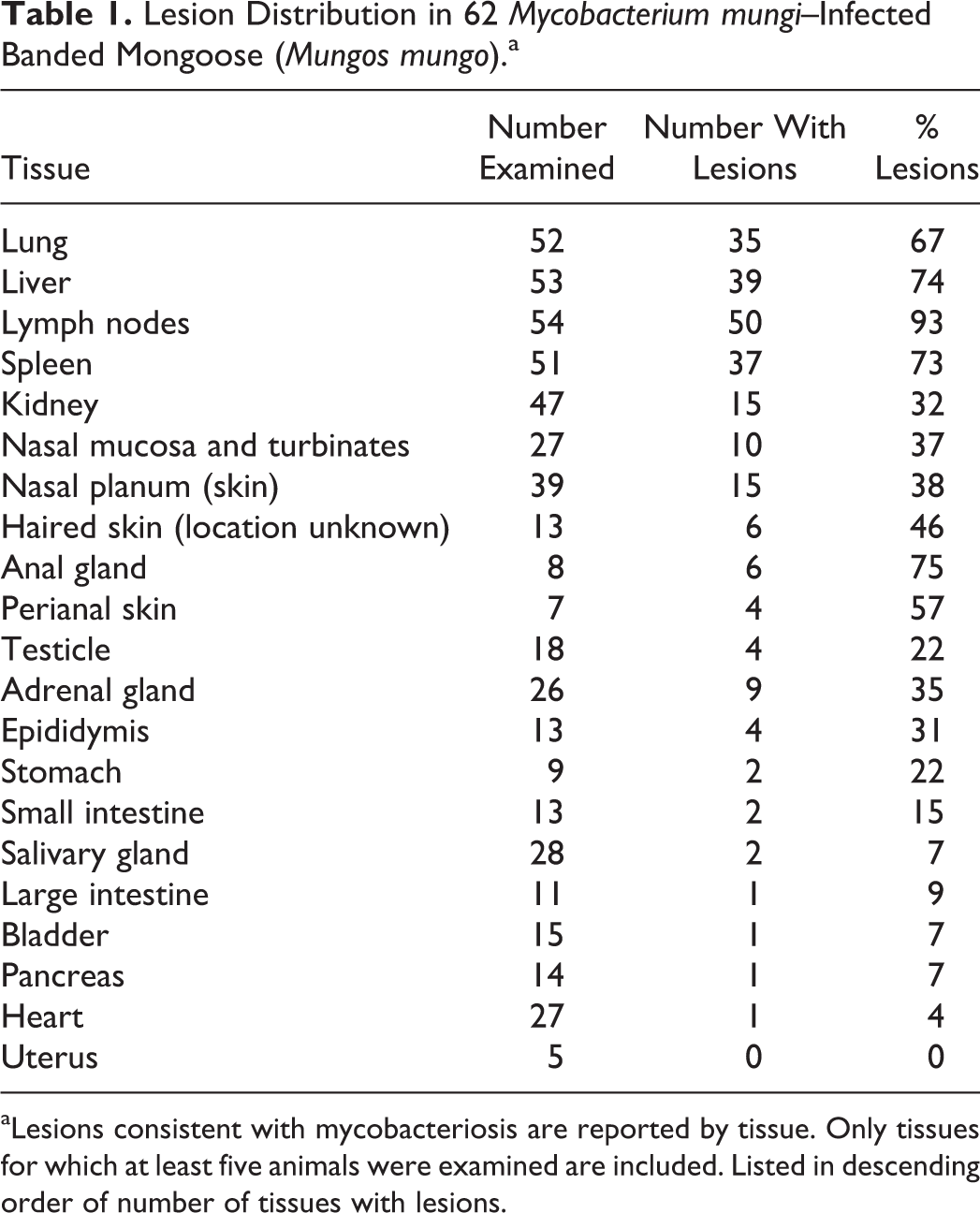
aLesions consistent with mycobacteriosis are reported by tissue. Only tissues for which at least five animals were examined are included. Listed in descending order of number of tissues with lesions.
Of 27 nasal turbinates examined, 10 (37%) had submucosal lymphohistiocytic to granulomatous infiltrates. Erosion and ulceration of the nasal mucosa was commonly noted. Multifocally, nasal turbinate bone had scalloped edges and periosteal fibrosis characteristic of bony remodeling (Fig. 6). Intracellular AFB were frequently located in macrophages in submucosal inflammatory lesions. Similar infiltrates expanded the dermis of the nasal planum. The epidermis of the nasal planum was ulcerated in 38% (15/39) of the cases in which it was examined, with moderate numbers of neutrophils and fibrin associated with the ulcerated areas. Even in cases with intact epidermis, AFB were present in variable numbers among dermal infiltrates and in small numbers among desquamated cells and debris on the epidermal surface, most commonly in small crevices or folds (Fig. 7). Similar dermal and epidermal lesions with surface AFB were detected in sampled skin lesions (6/13, 46%) and scrotum (2/4, 50%). In some cases, testicular and epididymal involvement included multifocal to diffuse granulomatous infiltrates that effaced normal tissue architecture in more advanced lesions. The number of AFB in testicular and epididymal lesions ranged from few to myriad.
A total of 605 lymph nodes from 63 infected mongooses were examined, including the mandibular, mesenteric, superficial cervical, inguinal, popliteal, anal, medial retropharyngeal, lumbar, mediastinal, gastric, hepatic, and parotid lymph nodes. Lymph node lesions were characterized by multifocal to coalescent noncaseating to caseating granulomas (Fig. 8) or focally extensive granulomatous infiltrates arranged in sheets of cells (mostly macrophages) that effaced normal lymph node architecture (Fig. 9). While necrosis was observed in approximately one-third of granulomas, peripheral fibrosis was not a feature of lymph node lesions, and dystrophic mineralization of necrotic caseum was only rarely seen (two cases). Irrespective of the anatomic location of the lymph nodes examined or the stage of disease, the lesion size and severity were generally consistent among all lymph nodes in an infected mongoose.
Pulmonary lesions were found in 67% (35/52) of lungs examined and were characterized as disseminated, nodular (30–500 μm) lymphohistiocytic to granulomatous infiltrates, or noncaseating to caseating granulomas. Pulmonary lesions were mainly present in animals with disseminated disease. In 94% (33/35) of the cases in which pulmonary lesions were identified, at least 1 other primary organ (ie, liver, spleen, or kidney) contained lesions suggestive of hematogenous or lymphatic dissemination of the pathogen. Of the two cases without involvement of liver, spleen, or kidney, one had similar lesions in multiple lymph nodes and the anal glands/sacs consistent with hematogenous or lymphatic dissemination.
Microscopic lesions within the affected lung were of three general types. The first was characterized by perivascular and subpleural distribution of lymphohistiocytic to granulomatous infiltrates that varied from individually separated nodules to expansile coalescing lesions (Fig. 10). Perivascular and peribronchiolar macrophages often contained darkly colored, granular material suggestive of silicosis. The second lesion type presented as multifocal, noncaseating to caseating granulomas with no apparent vasocentric or bronchocentric distribution (Fig. 11). The third lesion type was characterized by small nodular lymphohistiocytic infiltrates within the alveolar septa, which filled variable amounts of the alveolar space (Fig. 12). Intracellular AFB were noted in all lesion types. It was noteworthy that even in necrotic foci, AFB were invariably intracellular and not free within the necrotic caseum. Other infrequent findings in the lungs were focal to multifocal hemorrhages, pulmonary edema, and nematode parasites within bronchiolar lumens.
Anal gland and sac tissue often contained lymphohistiocytic to granulomatous infiltrates with large numbers of AFB (6/8, 75%). The keratinized surface of the duct of the anal sac contained small numbers of AFB in cellular debris found in small superficial epidermal crevices, as well as within the epidermis itself.
Discussion
M. mungi–infected banded mongoose had the highest number of lesions in the lymph nodes, liver, spleen, and anal glands/sacs, with the lung only involved in cases of advanced disease. In other MTC hosts, including humans and animals, aerosol transmission is believed to be the primary route of pathogen transmission. In these species, lesions centered on airways (ie, bronchi and bronchioles) predominate, and lesion expansion, airway invasion, and expulsion of bacilli through breathing or coughing are critical to disease transmission. In mongooses infected with M. mungi, a different picture emerges. While tuberculous lesions were common in the lungs of severely affected mongooses, these were centered on pulmonary vasculature rather than associated with the bronchi or bronchioles, suggesting hematogenous or lymphatic dissemination to and within the lung. Common involvement of the lymph nodes, liver, and spleen is also consistent with hematogenous or lymphatic spread acting as the primary mechanism of pathogen movement within the mongoose host. Consistent with pathogen shedding occurring through infected anal gland and urine secretions, 4 granulomatous lesions with AFB were seen in 75% of the anal gland/sac tissues and 32% of the kidneys of infected animals.
Lymphohistiocytic to granulomatous lesions were seen in 37% of the nasal cavities examined in M. mungi–infected banded mongoose. Upper respiratory lesions are uncommon in tuberculosis caused by other MTC pathogens, such as M. tuberculosis and M. bovis. 14 In humans, primary nasal tuberculosis is exceedingly rare, with only 40 cases reported worldwide. 14 Granulomatous lesions were also found in the skin of the nasal planum. Lesions in the nasal cavity and on the nasal planum could result from mongooses sniffing scent marks containing M. mungi organisms (urine and/or anal gland secretions) or foraging in the soil in locations where M. mungi–containing secretions have been deposited. The presence of AFB in the crevices and hair follicles of intact skin, nose, scrotum, anal sac, duct of the anal sac, and perineum is consistent with anal marking behavior of mongooses where anal gland secretions are placed directly on conspecifics or used in overmarking behaviors, where mongooses will deposit anal gland secretions on other anal marks of potentially infected mongooses.
The hallmark lesion of tuberculosis in natural hosts is the granuloma. The prototypical tuberculoid granuloma has a central region of caseous necrosis, which may contain foci of mineralization, surrounded by a zone of epithelioid macrophages and multinucleated giant cells, with the outermost zone containing increasing numbers of lymphocytes. The entire mass of necrotic caseum and inflammatory cells is often encased in a fibrous capsule of variable thickness. 1 M. mungi–induced lesions differed slightly in that multinucleated giant cells were rarely present and peripheral fibrosis was lacking. Such findings have also been features of M. bovis–induced lesions in Eurasian badgers (Meles meles), brushtail possums (Trichosurus vulpecula), and meerkats. 11,13,19
In MTC-susceptible species where aerosol is the primary route of infection, secondary routes can include oral and percutaneous transmission (e.g., bite wounds). Cutaneous and subcutaneous granulomas are commonly observed in the Eurasian badger and are thought to be the result of fighting and biting behaviors. 7 Cutaneous lesions may also be associated with draining sinuses from affected superficial lymph nodes. Such lesions have been reported in M. suricattae–infected meerkats, as well as M. bovis–infected brushtail possums, fallow deer (Dama dama), and red deer (Cervus elaphus). 6,15 Although cutaneous and subcutaneous granulomatous inflammation was seen in mongooses examined in this study, draining sinuses from affected superficial lymph nodes were not observed.
M. mungi infection in the mongoose host was characterized by lymphohistiocytic to granulomatous inflammation, as well as the presence of caseating and noncaseating granulomas. Features common in prototypical granulomas, such as multinucleated giant cells and peripheral fibrosis, were rarely present. Moreover, AFB were consistently found intracellularly, even in regions of necrosis. This contrasts with many other species where necrotic caseum often contains large numbers of both free and intracellular bacilli. Fibrous encapsulation of granulomas is generally considered to be a sign of disease control or healing, while expansion of necrosis with little or no encapsulation is considered a sign of disease progression. 12,17 In the zebrafish (Danio rerio) model of human tuberculosis, M. tuberculosis–infected macrophages migrate within a granuloma, as well as exit the granuloma to produce secondary granulomas and disseminated disease. 9 In similar fashion, macrophages with intracellular bacilli may escape poorly encapsulated granulomas in M. mungi–infected mongoose, resulting in disease dissemination and progression.
Differences in lesion morphology and disease progression may be related to genetic variations, such as the unique deletion in the M. mungi genome, RD1mon. 5 The RD1mon deletion is comparatively small (1610 bp) compared to other RD1 deletions in MTC organisms (RD1das/RD1sur, 4132 bp; RD1BCG, 9456 bp; RD1mic, 14 120 bp). 4,10,16,18,23 The RD1mon deletion encompasses part of the encoding region for PPE68 (Rv3873), a gene present in both lineage 6 dassie bacillus and M. suricattae. PPE68 is co-operonic with gene PE35, and together these gene products have an important immunomodulatory role on host-pathogen interactions, influencing cellular immune responses to mycobacterial infections. 25 Experimentally, PPE68/PE35 is seen to increase levels of anti-inflammatory cytokine interleukin (IL)–10 and chemokine monocyte chemoattractant protein 1 (MCP-1). 4,25 Evidence suggests that this interaction may control granuloma formation regulating the pathophysiology in M. tuberculosis infections, 25 possibly contributing to the more unique pathological features of M. mungi infection in the banded mongoose.
M. mungi infection in banded mongoose is characterized by both a unique transmission and exposure route, as well as accompanying pathological features, providing an opportunity to increase our understanding of MTC pathogenesis across host-pathogen systems. An important area of future research is the examination of the potential role of the unique RD1mon deletion in the pathophysiology of M. mungi infection in the banded mongoose host.
Footnotes
Acknowledgement
We thank the Botswana Department of Wildlife and National Parks for facilitating this work. We also thank CARACAL staff (M. E. Vandewalle, R. Sutcliffe, C. A. Nichols, S. Hill, and N. LaHue) for their assistance with field and lab investigations, as well as A. Lasley, J. Wiarda, J. Stasko, and V. Montgomery from the Bacterial Diseases of Livestock Research Unit, National Animal Disease Center, for their technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the Morris Animal Foundation (grant D14ZO-083) and the National Science Foundation (grant 1518663) as part of the joint NSF-NIH-USDA Ecology and Evolution of Infectious Diseases program. K. A. Alexander was also supported in part through the National Institutes of Health and National Institute of General Medical Sciences—Models of Infectious Disease Agent Study (grant 5U01GM070694-13). The funders had no role in study design, data collection and analysis, or the decision to submit the work for publication.
