Abstract
Multisystem inflammatory syndrome in children is an emerging pediatric illness associated with severe acute respiratory syndrome coronavirus 2 infection. The syndrome is rare, and evidence-based guidelines are lacking. This report reviews a patient who presented for medical care multiple times early in the course of his illness, thus offering near-daily documentation of symptoms and laboratory abnormalities. The patient did not have thrombocytopenia, anemia, or myocardial inflammation until the fifth day of fever. These laboratory abnormalities coincided with the onset of rash, conjunctival injection, vomiting, and diarrhea: clinical signs that could serve as indicators for when to obtain blood tests. The timing of this patient’s onset of multisystem involvement suggests that testing for multisystem inflammatory syndrome in children after only 24 h of fever, as the Centers for Disease Control and Prevention recommends, may yield false-negative results. Testing for multisystem inflammatory syndrome in children after 4 days of fever may be more reliable.
Introduction
Most children experience mild or asymptomatic severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) disease, with rare reports of multisystem inflammatory syndrome in children (MIS-C), a life-threatening illness with diverse clinical phenotypes in patients, 1 developing several days to weeks after an initial SARS-CoV-2 infection or exposure. The pathogenic mechanism causing MIS-C is unknown. MIS-C may involve immune activation, 2 and inflammatory monocyte-activating SARS-CoV-2 IgG antibodies may play a role in the disease severity of MIS-C patients. 1 The unpredictable clinical presentation of the MIS-C can include fever, gastrointestinal symptoms, myocardial dysfunction, as well as Kawasaki Disease (KD) and toxic shock–like features; however, it is distinct from these inflammatory and infectious conditions3,4 and challenges the clinician to make a timely diagnosis.
As a novel condition, specific diagnostic criteria and management recommendations for MIS-C are evolving. In this study, we provide evidence that understanding the timeline of clinical, laboratory, and radiological abnormalities in MIS-C is essential to decipher the pathological mechanisms of this novel illness, establish guidelines for its diagnosis and management, and predict its long-term health implications. This case report discusses a patient who did not have thrombocytopenia, anemia, or evidence of hepatic and myocardial inflammation until the fifth day of fever, despite checking for these earlier in his illness. These laboratory abnormalities coincided with the onset of rash, conjunctival injection, vomiting, and diarrhea: clinical signs that could serve as indicators for when to obtain blood tests. The timing of this patient’s onset of multisystem involvement suggests that testing for MIS-C after only 24 h of fever, as the Centers for Disease Control and Prevention (CDC) recommends, may yield false-negative results and may falsely reassure medical providers. Testing for MIS-C after 4 days of fever may be more reliable in some patients.
Case
A 4-year-old male with past medical history of platelet function defect, sleep apnea with prior tonsillectomy/adenoidectomy, prematurity, and developmental delays was admitted from the emergency department (ED) after 5 days of fevers and abdominal pain with progressive development of sore throat, decreased appetite, vomiting, diarrhea, conjunctivitis, and rash.
He initially presented on day of illness (DOI) 3 with fever and abdominal pain in the context of a positive SARS-CoV-2 exposure 2.5 weeks prior. Apart from fever, vital signs were within normal range, and his physical exam was unremarkable. Complete blood count (CBC), erythrocyte sedimentation rate (ESR), c-reactive protein (CRP), ferritin, complete metabolic panel (CMP), disseminated intravascular coagulation (DIC) screen, troponin I, n-terminal prohormone beta-type natriuretic peptide (NT-pro-BNP), urinalysis, chest x-ray, and SARS-CoV-2 polymerase chain reaction (PCR) were ordered. Workup was significant for modest elevations in ESR, CRP, ferritin, and prothrombin time (PT) (Table 1). The patient was well-appearing and without evidence of focal bacterial infection. He was discharged home with a documented diagnosis of “fever in pediatric patient,” and parents were advised to continue supportive care.
Patients’ blood test results from day of illness 3, 5, 6, 7, 8, 9, and 10. Numbers highlighted in red are outside the normal range. Normal range for each result is listed in italics below each blood test in the first column.

DOI: day of illness; AST: aspartate transaminase; ALT: alanine transaminase; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; PT: prothrombin time; PCR: polymerase chain reaction; NAAT: nucleic acid amplification test; NT-pro-BNP: N-terminal prohormone b-type natriuretic peptide; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2.
Due to persistent fever, he returned to the ED later that same day. A second evaluation on DOI 3 was notable for pharyngeal erythema; a rapid strep was obtained and negative. Acetaminophen was given for fever with improvement noted in mild tachycardia, and the patient tolerated oral fluids without difficulty. He was diagnosed with “fever in pediatric patient,” likely attributable to viral illness, and discharged home with a plan for supportive care to include oral hydration, acetaminophen as needed for fever, and close outpatient follow-up.
On DOI 5, the patient presented to the ED with persistent fever and new symptoms including four episodes of non-bloody, non-bilious emesis, five episodes of non-bloody diarrhea, poor appetite, conjunctivitis, and a rash that developed in the 24 h preceding his third ED visit. Vital signs were significant for tachycardia (150 beats/minute) and tachypnea (56 breaths/minute). He was afebrile with age-appropriate blood pressure and normal oxygen saturation. On exam, he appeared ill and uncomfortable. He had bilateral conjunctival injection (Figure 1), erythematous lips, bilateral anterior cervical lymphadenopathy, tachycardia without murmur, rub or gallop, diffuse abdominal tenderness, and a generalized erythematous macular rash that was most pronounced on bilateral palms and soles, posterior thighs, and back. Laboratories returned grossly abnormal, notable for anemia, hyponatremia, transaminitis, and elevated inflammatory markers, ferritin, D-dimer, and fibrinogen (Table 1). Potassium, chloride, total bilirubin, blood urea nitrogen, and creatinine remained normal throughout the child’s illness. Blood cultures were sent.

Conjunctival injection.
Cardiac testing revealed evidence of myocardial injury with elevated troponin and NT-pro-BNP. Electrocardiogram (EKG) showed sinus tachycardia and normal intervals including a QTc of 425. ST elevations were noted diffusely. T-wave inversion seen in V1 and V2 is normal in this age range (Figure 2). An echocardiogram was obtained and demonstrated diminished left ventricular function with ejection fraction of 48%. There was structurally normal cardiac anatomy and normal coronary anatomy without evidence of coronary artery dilation or ectasia. The left main coronary artery measured 2.5 mm (Z-score of −0.39) and the proximal right coronary artery measured 2.1 mm (Z-score of −0.05). 5 There was no pericardial effusion. SARS-CoV-2 PCR returned positive. He was given a 10 mL/kg normal saline bolus, IV morphine for pain, and empiric IV ceftriaxone. He was admitted to the Pediatric Intensive Care Unit with a diagnosis of MIS-C.
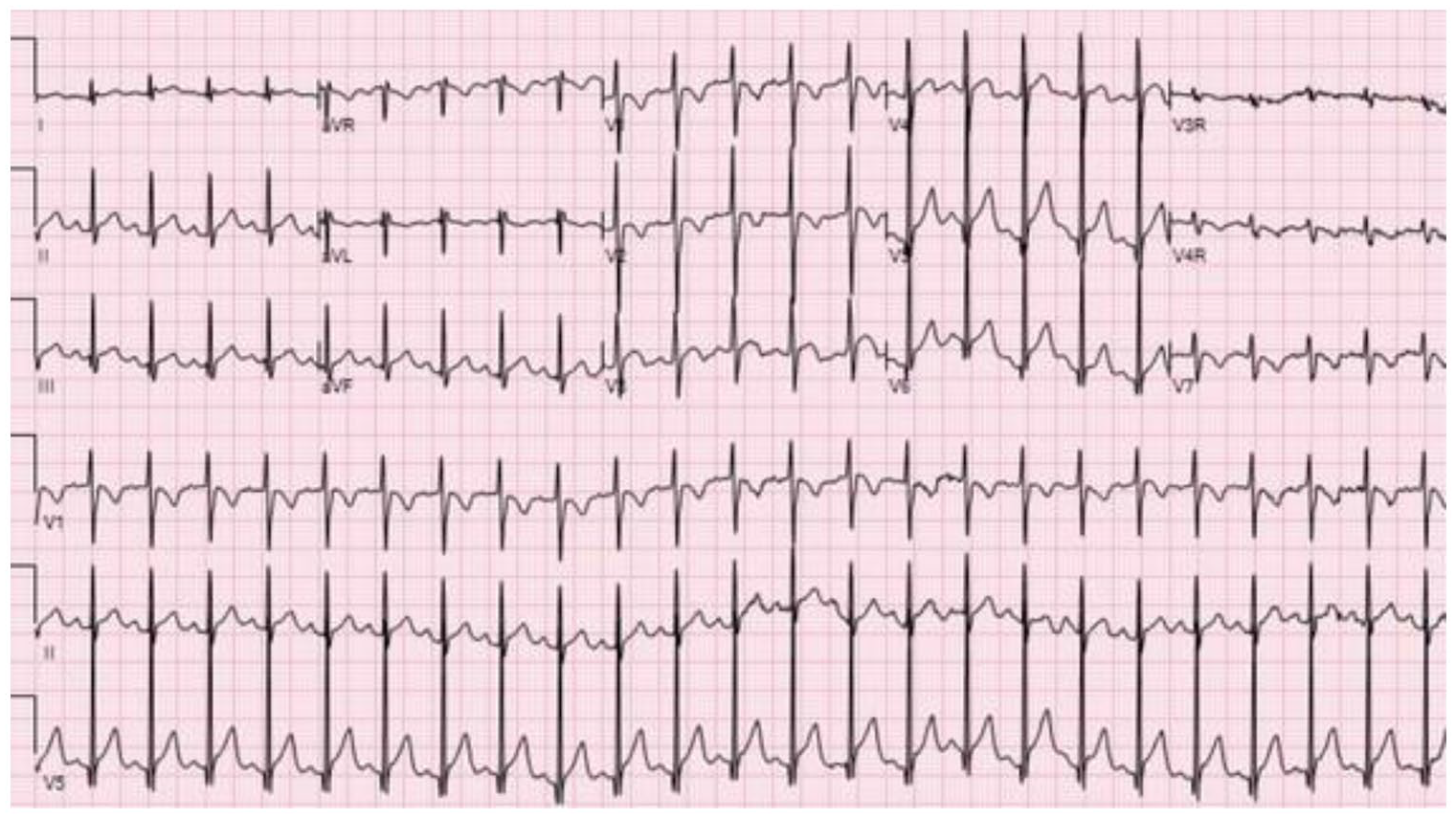
The patient’s electrocardiogram showing sinus tachycardia and diffuse ST elevation.
Per pediatric rheumatology recommendations, the patient was started on intravenous immunoglobulin (IVIG) 2 g/kg and IV methylprednisolone 2 mg/kg/day. He was started on famotidine to prevent steroid-related ulcers. IV ceftriaxone was continued until blood cultures were negative at 48 h. The pediatric hematology service was consulted regarding aspirin dosing, given his thrombocytopenia and history of platelet function defect. Consistent with the American College of Rheumatology’s guidance for the management of MIS-C in pediatric COVID-19 and KD, aspirin was recommended for its anti-platelet 6 and anti-inflammatory effects. He was prescribed 50 mg/kg/day of aspirin until afebrile for 2 days and transitioned to 3–5 mg/kg/day. Patients with MIS-C have typically been kept on low-dose aspirin for 6–8 weeks due to the increased risk of thrombotic complications but the patient was kept on aspirin for 4 months due continued clinical concerns. The patient never required respiratory, vasoactive, or inotropic support. Cardiac enzymes were down-trending at discharge. Cardiac magnetic resonance imaging (MRI), a component of our cardiac evaluation in MIS-C, 7 was deferred due the need for anesthesia in the setting of positive SARS-CoV-2 PCR and clinical diagnosis of myocarditis made based on serum and echocardiogram findings. Ejection fraction normalized to 65% on repeat echocardiogram on DOI 8. Repeat EKG demonstrated resolution of ST changes.
Daily labs were trended and continued to show recovery (Table 1). Interestingly, serial rapid NAAT SARS-CoV-2 testing remained negative and “confirmatory” PCR was only intermittently positive (Table 1). The patient was clinically stable and discharged on the sixth day of hospitalization with activity restriction, outpatient follow-up, and cardiac MRI scheduled with pediatric cardiology.
Discussion
When the SARS-CoV-2 pandemic began in December 2019, the incidence of pediatric cases was relatively low and most infected children exhibited mild clinical courses, such as a minor upper respiratory infection. In April 2020, the United Kingdom reported a case series of children with incomplete KD or toxic shock syndrome4,8 that seemed to be temporally related to SARS-CoV-2. Since then, this syndrome has been named MIS-C and is increasingly being reported in Europe and North America.9,10 As of August 27, 2021, 4661 cases of MIS-C and 41 deaths have been reported in children in the United States. 11
MIS-C is defined as a person <21 years old presenting with fever ⩾24 h, laboratory evidence of inflammation and clinically severe illness involving >2 organ systems with no alternative plausible diagnoses, and a recent SARS-CoV-2 infection/exposure. 12 The presentation of MIS-C is very similar to KD. One case series reported that 40–50% of children with MIS-C also met criteria for complete or incomplete KD. 13 However, there are key differences between MIS-C and KD that may help clinicians with diagnosis. MIS-C tends to affect older children, while KD tends to affect infants and younger children.14,15 MIS-C tends to disproportionally affect African American and Hispanic children, while KD tends to affect children of Asian descent.14,16 Gastrointestinal symptoms including abdominal pain, nausea, vomiting, and diarrhea are more common in MIS-C than in KD, with MIS-C case series reporting anywhere from a 60–100% prevalence of abdominal symptoms. 17 Myocardial dysfunction and cardiogenic shock are more common in MIS-C than in KD. Inflammatory markers including CRP, ferritin, and d-dimer tend to be higher in MIS-C than in KD, and absolute lymphocyte and platelet counts tend to be lower. 14 Ultimately, however, the key distinction between MIS-C and KD centers on SARS-CoV-2 exposure history and testing.
While there are clear risk factors for MIS-C including gender, race, and age, the exact pathophysiology of MIS-C is still unknown. Due to the clinical similarities between MIS-C and KD, it has been proposed that MIS-C results from an abnormal immunologic response to the novel 2019 coronavirus, like KD. 18 Children with MIS-C maintain highly inflammatory monocyte-activating IgG antibodies 1 that may contribute to monocyte activation in the kidney and heart weeks after the initial SARS-CoV-2 infection. It has also been postulated that MIS-C is a post-infectious process, as some children test negative for SARS-CoV-2 via PCR but have positive serology.13,17 Our patient’s only known exposure to the virus was via household contact more than 2 weeks prior to symptom onset, suggesting that his illness was a post-infectious process. However, he had a negative SARS-CoV-2 PCR on DOI 3 and 8 but positive testing on DOI 5 and 9. This may reflect issues with diagnostic test sensitivity rather than active ongoing infection; three different PCR tests were used over his hospital course, with varied results.
A survey of MIS-C surveillance and treatment guidelines at 40 US institutions conducted by Dove et al. in June of 2020 found that most institutions (~53%) initiate MIS-C screening with minimum 1 day of fever, consistent with current CDC recommendations. Of the remaining institutions, 38% required at least 3 days of fever before considering MIS-C.19,20 While our patient was examined and assessed multiple times early in his illness, the majority of lab abnormalities hit their peak/nadir at DOI 4-5, for example, NT-pro-BNP, liver enzymes, thrombocytopenia, and anemia. This coincides with onset of clinical signs including rash, conjunctival injection, vomiting, and diarrhea as well. This may suggest that testing for MIS-C may yield false-negative results if patients have had fever for less than 4 days.
As a new clinical entity with non-specific symptoms and laboratory results, diagnosis of MIS-C is difficult to make until other diseases have been ruled out or the illness evolves. Since this patient’s troponin, NT-pro-BNP, and CBC were all normal at his first ED visit, the diagnosis of MIS-C was excluded on initial screening. As the illness progressed, he presented with conjunctivitis and mucositis that was suggestive of MIS-C or KD. Ultimately, subsequent evaluation demonstrated prolonged fever, rash, elevated inflammatory markers, and cardiac involvement consistent with the diagnosis of MIS-C. It is important to be able to distinguish MIS-C from other infectious or inflammatory conditions, like KD or severe acute SARS-CoV-2 infection, for early diagnosis and management.
Conclusion
This case demonstrates that early screening for MIS-C may contribute to premature diagnostic closure and delayed diagnosis. As MIS-C remains a clinical diagnosis, clinicians should be mindful of when to initiate screening and how symptoms and laboratory findings may evolve over time. MIS-C should not be eliminated early from the clinician’s differential diagnosis and the disease should be reconsidered if the patient’s condition fails to improve in order to facilitate timely treatment and prevent negative outcomes.
Footnotes
Acknowledgements
The authors would like to thank the patient and his family for their willing participation in this report. They would like to acknowledge the cardiology, rheumatology, critical care, and hospitalist teams at the Medical College of Wisconsin for their collaboration in this patient’s care.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
