Abstract
14-3-3σ is a protein expressed in many epithelial tissues associated with essential cell functions, including cell-cycle control, apoptosis, and cytoskeletal integrity. There is a paucity of knowledge of the tumorigenesis of canine renal cell carcinomas (RCCs), and the histological origin of this tumor has not been established. This study analyzed the expression of 14-3-3σ, Ki-67, cytokeratins, and vimentin in 40 canine RCCs. Aberrant expression of 14-3-3σ was demonstrated in 15 (38%) cases and was associated with a significantly shorter survival time (P < .002). In contrast to canine RCC, normal kidney did not express 14-3-3σ. The Ki-67 proliferation index did not show utility as a prognostic factor. The distal convoluted tubular epithelium in normal kidneys coexpressed cytokeratins and vimentin, and thus maintenance of this coexpression pattern in canine RCC suggests that most tumors arise from the distal segment of the nephron. These results suggest that 14-3-3σ is a potential negative prognostic factor and a possible therapeutic target.
Keywords
The 14-3-3 family is a group of small, highly conserved, and ubiquitously expressed acidic proteins with molecular weights between 28 and 33 kDa. They are found in all eukaryotic species and include 7 different 14-3-3 protein isoforms in mammalian cells, each designated by a Greek letter (beta, β; epsilon, ∊; gamma, γ; eta, η; theta, θ; sigma, σ; and zeta, ζ). Multiple 14-3-3 ligands are involved in various cellular processes including DNA damage signaling, cell proliferation and survival, cytoskeleton rearrangement, transcriptional control, and cell cycle regulation. Specifically, the ability of 14-3-3 proteins to bind and regulate several oncogenic as well as various tumor suppressor gene products suggests they have a potential role in tumorigenesis. 13,17
Unlike other family members, which are broadly expressed in the brain and play a pivotal role in neurodegenerative diseases, 14-3-3σ (also known as stratifin or human mammary epithelial marker 1) is expressed only in epithelial cells and is closely associated with tumor development. The expression of this isoform was originally identified as a response to DNA damage and TP53 activation. TP53 encodes the protein p53, which subsequently induces the production of 14-3-3σ in the cytoplasm. 14-3-3σ is able to bind and sequester specific cyclin-dependent kinases, CDK4, CDK2, and CDK1, that regulate the G1/S and G2/M cell cycle checkpoints, preventing initiation of mitosis and allowing DNA repair. 14 Furthermore, 14-3-3σ has been shown to play an antiapoptotic role and promote cell survival 29 and recently has been identified as a promising therapeutic target. 2,40 In veterinary medicine, 14-3-3σ has been reported as an oncoprotein in canine mammary and urinary bladder carcinomas. 33 –35
Carcinomas are the most common canine primary renal tumors, comprising 4 different histological patterns—solid (clear cell or chromophobe type), tubular, papillary, and multilocular cystic 9,19,20 —that display heterogeneous biological behaviors and present an overall median survival time (MST) ranging from 359 to 743 days. 3,5,9 Clinical outcome is associated with histological subtype, mitotic index, and Cox-2 expression. 5 In contrast to humans, there is a dearth of knowledge with regard to the mechanisms and molecules that contribute to the development and progression of canine renal cell carcinomas (RCCs). Additionally, the histological origin of these tumors in the dog has not been clarified, and there is evidence that their immunophenotype differs from the human counterpart. 8
To elucidate whether 14-3-3σ plays a role in canine RCC oncogenesis, as has been demonstrated for other canine tumors, this study aimed to investigate the expression of the protein and its association with outcome and with various clinicopathological variables, including Ki-67 labeling index. Immunohistochemical labeling was used to compare the distribution of 2 major cell markers (cytokeratins and vimentin) in the normal kidney to their expression pattern in tumors to determine the cell of origin of canine RCC.
Materials and Methods
Case Selection
Cases included in this retrospective study were selected from a previously published series of dogs with canine RCC that underwent nephrectomy (n = 39). 5 One dog presented with metastatic disease and was immediately euthanized, and diagnosis was made at necropsy; this dog was not included in the survival analysis. A complete medical history from dogs containing signalment, clinical presentation, laboratory findings, clinical stage, histological diagnosis, use and type of adjuvant therapy, and overall MST was available. Samples from 5 normal canine kidneys were used as controls.
Histological Classification
Hematoxylin and eosin–stained tissue sections were histologically classified according to the WHO Histological Classification of Tumors of the Urinary System of Domestic Animals by 3 veterinary pathologists (A.S.B., A.L.S., and S.L.P.). 19 In cases of disagreement between 2 or more pathologists, a consensus was reached. To further classify the solid tumors into clear cell and chromophobe-type carcinomas, additional tissue sections were stained with Hale’s colloidal iron stain. In addition to subtype, other histopathological features including mitotic activity index (MAI) (mitoses per 2.37 mm 2 ), 21 presence of necrosis, inflammation, and vascular invasion were assessed as previously described. 5
Immunohistochemistry and Evaluation
Immunohistochemical analysis was performed in 40 cases. Four-micrometer-thick fresh-cut tissue sections from formalin-fixed, paraffin-embedded tissue blocks were labeled with mouse-monoclonal antibodies against 14-3-3σ (clone 5D7, 1:40; Santa Cruz Biotechnology, Heidelberg, Germany), Ki-67 (MIB-1, 1:150; Dako, Ely, UK), cytokeratins (clone AE1/AE3, 1:100; Dako), and vimentin (clone V9, 1:500; Dako) on a BondMax Autostainer (Leica; Milton Keynes, UK). The 14-3-3σ antibody has been previously shown to identify the appropriate canine molecular weight antigens by Western blot and to cross-react with canine tissues. 27,34,36 Heat-induced antigen retrieval was performed using a pH 6 buffer (Bond ER1; Leica) for 10 minutes (vimentin), 20 minutes (14-3-3σ), and 30 minutes (AE1/AE3) at 90°C and a pH 9 buffer (Bond ER2; Leica) for 10 minutes at 90°C for Ki-67. The Bond Polymer Refine Detection kit (Leica) was used for visualization with hematoxylin counterstain. As positive controls, normal canine urinary bladder (for 14-3-3σ), normal canine skin (for cytokeratins and vimentin), and a hyperplastic lymph node (for Ki-67) were used in every assay. All positive controls showed cytoplasmic immunolabeling of the urinary bladder urothelium for 14-3-3σ, nuclear immunolabeling for Ki-67, and cytoplasmic immunolabeling for cytokeratins and vimentin. When present, surrounding histologically normal renal tissue was used as an internal positive control. Negative controls were prepared by replacing the primary antibody with Leica Antibody Diluent (Leica) only. A positive reaction was indicated by the presence of distinct brown cytoplasmic or nuclear labeling in neoplastic cells.
Immunoreactivity was independently evaluated by 2 of the authors (A.S.B. and S.L.P.), and discrepancies were discussed and assessed jointly by use of a double-headed microscope. To evaluate correlations with clinical features, pathological variables, and outcome, positive labeling for 14-3-3σ was assessed using a previously published semiquantitative scoring system. 35 The scores for percentage (1, ≤10%; 2, 11%–50%; 3, 51%–80%; and 4, >80%) and staining intensity (0, negative; 1, weak; 2, moderate; and 3, intense) of positive cells were recorded, and a total score (TS) ranging from 0 to 12 was calculated by multiplying these 2 parameters for each of the studied cases. The count of Ki-67-positive cells was performed in 10 high-power fields (40×) counting at least 1000 cells for each case, using the TMARKER software. 28 Ki-67 expression was evaluated as the labeling index and was defined as the percentage of Ki-67-positive cells. For cytokeratins and vimentin, positivity was recorded when there was distinct cytoplasmic labeling of the neoplastic epithelial cells and was scored semiquantitatively (0, negative; 1, weakly positive; 2, moderately positive and 3, strongly positive) as previously described. 12
Statistical Analysis
Expressions of 14-3-3σ, Ki-67, cytokeratins, and vimentin were compared with histological subtype, MAI, metastasis at diagnosis, necrosis, and inflammation. A chi-square test and a Fisher’s exact test were used for studying categorical variables. Nonparametric analysis was performed with the Kruskal-Wallis 1-way analysis of variance. Overall median survival time was defined as time from nephrectomy for canine RCC treatment until death. Dogs that were lost to follow-up, were still alive at the end of the study period, or died were censored.
Survival analysis using the Kaplan-Meier product limit method was conducted to estimate disease-related MST for the whole population of canine RCC dogs. To compare estimated disease-related MST between categories, the log rank test (univariate analysis) was used. Multivariate analysis using Cox regression analysis was performed to demonstrate combined predictor variables found significant in univariate analysis. All statistical analyses were carried out using SPSS software (version for Windows 22.0; IBM Corp, Armonk, NY). In all comparisons, P < .05 was accepted as defining statistical significance.
Results
Clinical Findings
For the 39 dogs with clinical follow-up, the median age at diagnosis was 8 years (range, 2–12 years). There was a male to female ratio of 1.05:1, including 20 males (9 intact) and 19 females (5 intact). Breed distribution among dogs was as follows: 12 cross breeds, 7 Labrador Retrievers, 5 Boxers, 3 Golden Retrievers, 2 German Shepherd Dogs, 2 Dalmatians, and 1 each of Cocker Spaniel, Rottweiler, Weimaraner, Japanese Akita, Galgo Español, Bullmastiff, English Bulldog, and Collie (Suppl. Table S1).
The overall MST for dogs diagnosed with RCC was 540 days (95% confidence interval [CI], 407–673). Of the 39 dogs, 29 were dead (74%) at the end of the data collection period, 8 (21%) were alive, and 2 (5%) were lost to follow-up. Death due to neoplastic disease occurred in 18 of 39 dogs (46%). Death was documented as unrelated to RCC in 5 (13%) and of unknown cause in 6 dogs (15%).
The most commonly reported presenting signs were weight loss in 15 dogs (38%), hematuria in 12 dogs (31%), inappetence or anorexia in 11 dogs (28%), and lethargy in 8 dogs (20%). A palpable abdominal mass was evident on physical examination in 17 dogs (43%). In 3 dogs (8%), the renal tumor was an incidental finding during a health screening or staging prior to surgery. Other less common presenting signs included, vomiting, abdominal pain, and diarrhea. For the 18 cases in which tumor size was provided, the average size was 10.5 cm in largest diameter with a range of 5 to 16 cm.
Histological Findings
Forty RCC cases were available for assessment. Following histological examination these were categorized as tubular (n = 21, 53%), papillary (n = 9, 22%) or solid (n = 10, 25%). Within the group of tumors exhibiting a tubular pattern, 12 of 21 (57%) cases were well-differentiated (composed of easily recognizable tubules formed by cells with mild to moderate atypia) and 9 of 21 (43%) were poorly differentiated (composed of closely packed sheets of cells with rare tubules formed by epithelial cells with marked atypia). Using colloidal iron stain, the solid group was subclassified into chromophobe RCCs (n = 9, 90%) that were positive with colloidal iron stain, and clear cell RCCs (n = 1, 10%) that exhibited large cytoplasmic vacuoles and eccentric nuclei and were negative with colloidal iron stain.
14-3-3σ Expression
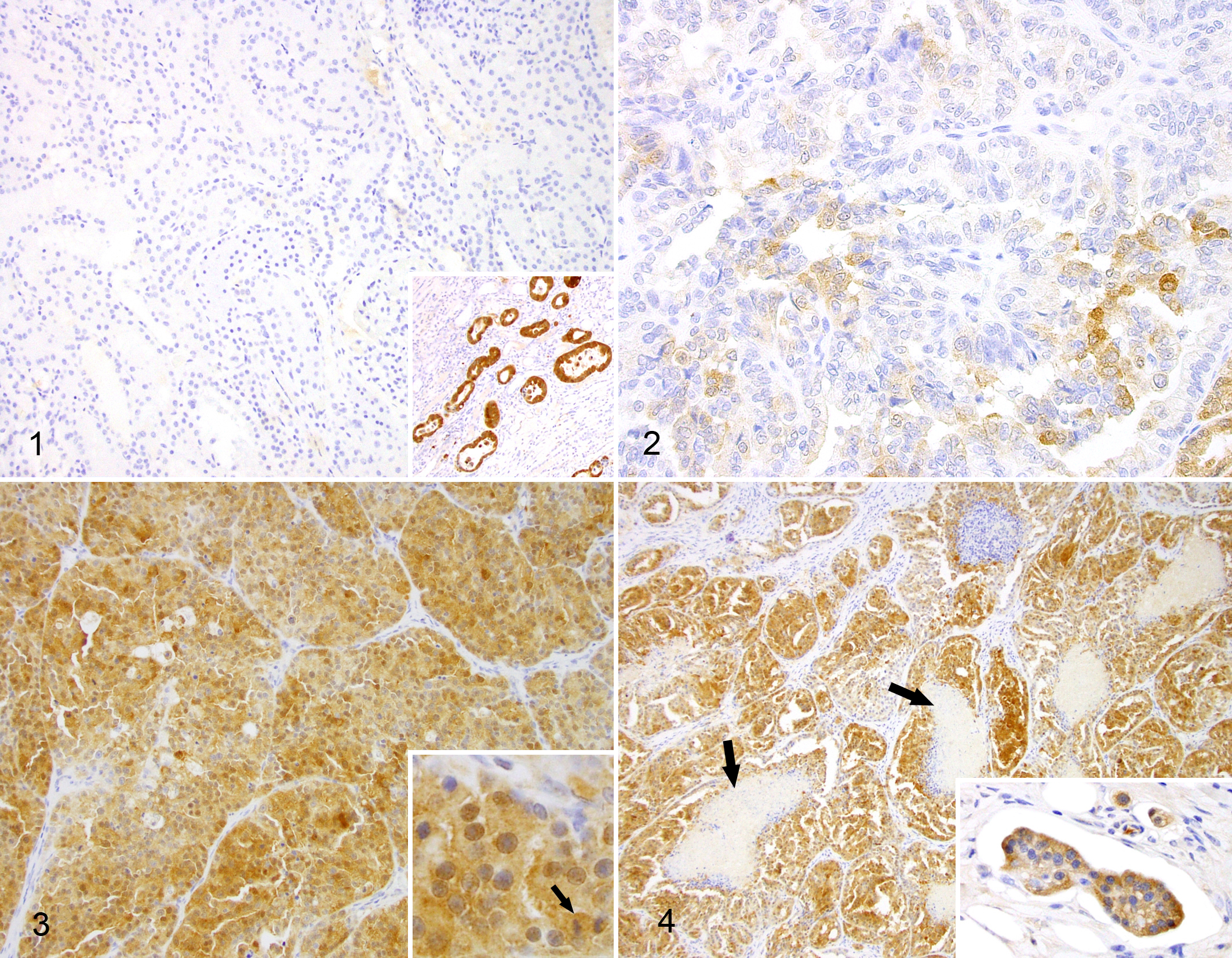
Expression of 14-3-3σ was not detected in histologically normal canine kidney (glomeruli, proximal convoluted tubules, distal convoluted tubules, loop of Henle, and collecting ducts) surrounding carcinomatous areas (n = 12) or in 2 normal canine kidney samples used as controls (Fig. 1). In contrast, compressed and reactive convoluted tubules, located at the periphery of renal carcinomas, occasionally displayed moderate to intense cytoplasmic immunoreaction (Fig. 1 inset), but these positive areas were not considered in the generation of a TS. As there was no protein labeling in normal kidney, cases with a TS ≥ 1 were considered positive and consequently to neoexpress 14-3-3σ. Within the tumors, positive protein expression occurred in 15 of 40 RCCs (38%) (12 tubular, 2 papillary, and 1 solid), with TS ranging from 1 to 12 (median, 5.2), and 26 of 40 cases were negative (62%). The positive to negative ratios between the 3 histological tumor subtypes (tubular, papillary, and solid) were 12:9, 2:7, and 1:9, respectively. Seven cases had a TS < 3, considered as low 14-3-3σ expression (Fig. 2), and 8 cases had a TS ≥ 4, considered as high 14-3-3σ expression (Figs. 3 and 4). Of this latter group, 6 cases were tubular, 2 cases were papillary, and 1 case was solid (clear cell type). Two tubular carcinomas and 1 papillary carcinoma showed the highest TS (12). The predominant staining pattern was cytoplasmic, but in 13 of 15 positive cases (86%) the nucleus also exhibited positive labeling. There were no cases with an exclusive nuclear staining pattern. In all positive cases, the immunoreaction varied from those with heterogeneous staining, characterized by groups of cells without expression or mildly positive, or cases with multifocal strongly positive cells, to cases with diffuse and intense staining. Within the positive cases, mitotic figures including those with bizarre morphology exhibited moderate to strong immunopositivity to 14-3-3σ (Fig. 3 inset). Positive cases that contained areas with clusters and solitary bizarre neoplastic cells at the front of invasion into the adjacent renal parenchyma and vascular invasion within lymphatics showed intense expression of the protein (Fig. 4 inset). Furthermore, 1 of the positive cases had tissue from a lung metastasis available that was positively labeled. Stromal tissue, fibroblasts, and areas of necrosis were not immunolabeled for 14-3-3σ.
Ki-67 Expression
Ki-67 was detected only in cell nuclei without cytoplasmic immunolabeling in any of the samples. Normal canine kidney did not express Ki-67, and histologically normal kidney adjacent to carcinomas sporadically showed positive cells in the tubular lining epithelium. Ki-67 proliferative index (PI) ranged from 0.0% to 56.0% (mean, 15.7%) of cells. The mean Ki-67 values for each tumor type (tubular, papillary, and solid) were 20.2%, 14.5%, and 3.0%, respectively. In 16 of 40 (40%) cases there was no immunolabeling for Ki-67. There was no correlation between Ki-67 expression and MAI (P > .05).
Cytokeratin and Vimentin Expression
In normal canine kidney and in the nonneoplastic renal tissue of the dogs with carcinoma, the cytokeratin cocktail (which immunoreacts with keratins 1, 2, 3, 4, 5, 6, 7, 8, 10, 13, 14, 15, 16, and 19) labeled the epithelial cells forming the distal convoluted tubule and the collecting tubule and duct, while the proximal convoluted tubules and the glomeruli were negative (Fig. 5). Variable proportions of neoplastic cells were immunolabeled in 33 of 40 (82.5%) cases, whereas 7 (17.5%) cases were negative. Within the positive cases, 15 (45.5%) were moderately to strongly positive and 18 (54.5%) were weakly positive in randomly distributed cell clusters. The staining pattern was mainly cytoplasmic and diffuse to finely granular, with frequent membranous highlighting (Fig. 6).
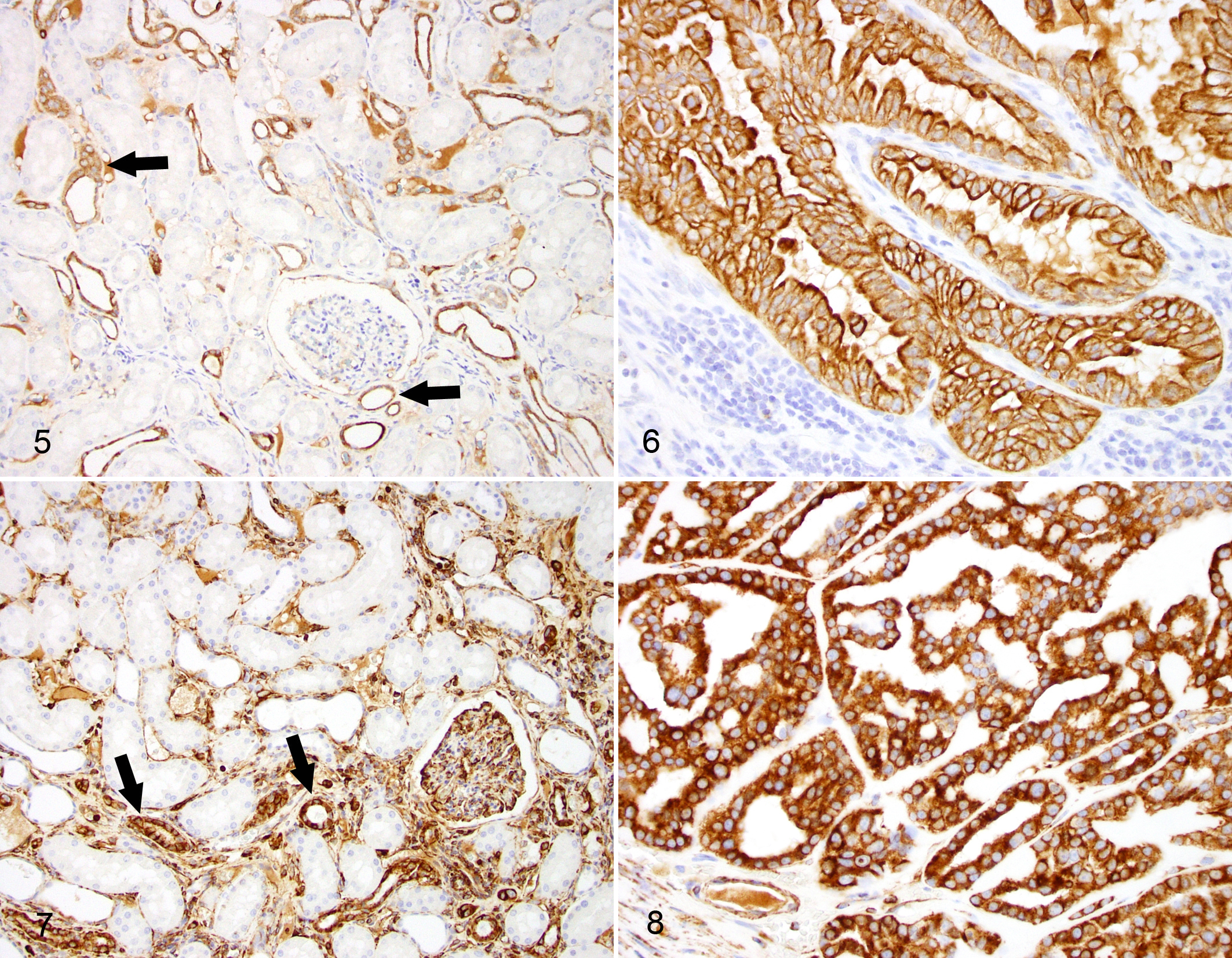
In normal renal parenchyma, vimentin was expressed in glomerular endothelial cells, interlobular arteries and veins, peritubular capillaries, vasa recta, and the interstitium as well as in epithelial cells lining distal convoluted tubules (Fig. 7). In canine RCC, vimentin was expressed in neoplastic epithelial cells in 34 cases (85%) while 6 cases (15%) did not express this marker. Of the positive cases, 26 (76.5%) were moderately to strongly positive (Fig. 8) and 8 (23.5%) were weakly positive.
Of the 40 cases, 27 (67.5%) coexpressed cytokeratins and vimentin to some extent, 7 cases (17.5%) were cytokeratin negative and vimentin positive, and 6 cases (15%) were cytokeratin positive and vimentin negative.
Statistical Analysis
Univariate analysis showed a statistically significant association between high 14-3-3σ expression (TS > 4) and a shorter survival time (P = .002) (Fig. 9). Furthermore, shorter survival time was significantly associated with a high percentage (percentage scores 3 and 4) of positive cells (P = .001) and high stain intensity (stain intensity scores 2 and 3; P = .000). A higher 14-3-3σ TS was positively correlated with the poorly differentiated tubular subtype (P = .03) and with a higher (>30) MAI (P = .003). Furthermore, in multivariate analysis, 14-3-3σ expression remained an independent predictor of prognosis (P = .013). The expression of 14-3-3σ was not correlated with metastasis at the time of diagnosis, necrosis, or type of inflammation nor with expression of Ki-67, vimentin, or cytokeratins (P > .05). No association was observed between Ki-67 index and MST.
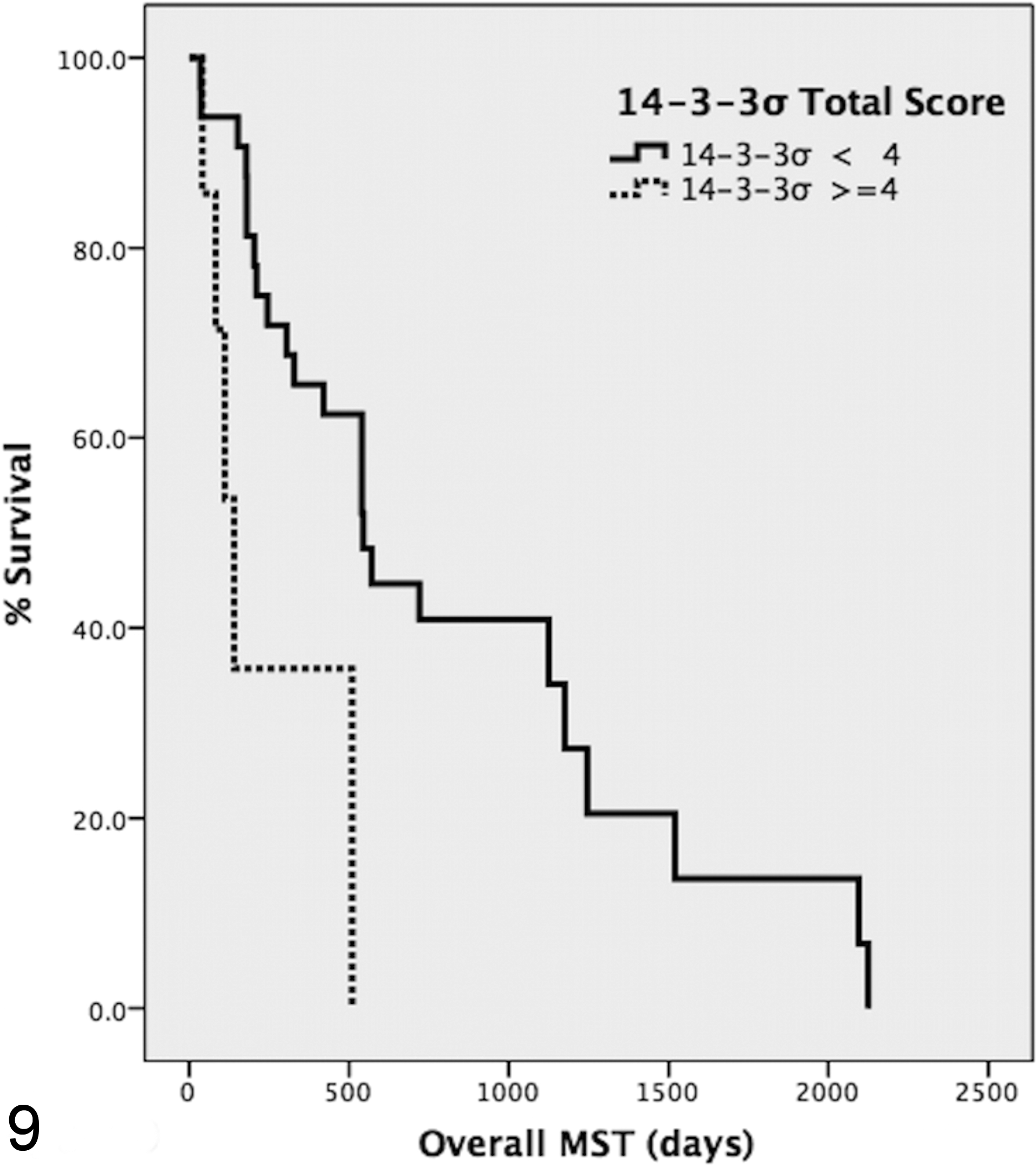
Kaplan-Meier survival curve for overall median survival time (MST) according to 14-3-3σ total score (TS). A TS greater than 4 (solid line) is significantly associated with a shorter survival time (P = .002).
Discussion
In the present study, the expression and prognostic significance of various markers of cell proliferation, tumor progression, and cell characterization were investigated in a large case series of canine RCC. To the authors’ knowledge this is the first analysis of 14-3-3σ in canine RCC and the first evidence that this protein may be a significant prognostic factor in canine neoplasms.
Expression of 14-3-3σ has become the focus of much research in human medicine. The levels of mRNA, protein expression, and methylation status have been studied in a variety of human cancers. 7,22,32,39 As 14-3-3σ can be induced in a p53-dependent manner, preventing mitotic catastrophe, and because its expression is silenced in various cancers, such as breast and head and neck carcinoma, it has been regarded as a tumor suppressor. 6,18 Subsequent studies have suggested that its role in tumorigenesis is tumor-dependent, as an increasing number of investigations have found overexpression and neoexpression of the protein being associated with malignancy. 16,23,32,39 Similarly, the relevance of this protein as a tissue differentiation marker and as an oncoprotein has been reported in canine mammary and urinary bladder carcinomas. 33 –35 In the current study, immunohistochemical analysis revealed the absence of 14-3-3σ immunolabeling in normal kidney and is in accordance with previous studies using Western blot and immunohistochemistry in both humans and dogs. 22,24,34,36 Interestingly, 14-3-3σ was neoexpressed in 37.5% of canine RCCs, which is more than double the 16.4% rate described in human RCCs. 22 Additionally, positive cases that had vascular invasion showed positive labeling in embolic neoplastic cells. While in humans the role of 14-3-3σ remains to be confirmed, we have demonstrated that its neoexpression in a subset of canine RCC is significantly associated with a shorter survival time and is an independent prognostic factor. These results are compatible with previous studies in human and canine mammary basal carcinomas, characterized by a poor prognosis, that also overexpress 14-3-3σ. 16,36 It has now been established that 14-3-3σ can promote lung, liver, and pancreatic carcinomas. 31,32,38,39 Similar to our findings, these tumors arise from tissues that do not constitutively express the protein. These findings could support the hypothesis that a mutant form of 14-3-3σ, which retains antiapoptotic and proliferative abilities and favors invasion and metastasis but lacks tumor-suppressive activity of the wild-type protein, may be produced during tumorigenesis. Further studies analyzing ligands of 14-3-3σ and possible mutations are required to clarify its precise role in canine RCC.
The histological origin of canine RCC has not been established. In a previous study 12 including tubulopapillary, papillary, papillary-cystic, sarcomatoid, and solid variants, all cases were positive for uromodulin, also known as Tamm-Horsfall glycoprotein, which is exclusively synthesized in the kidney. 10 It was suggested that because bovine renal cell tumors were also positive for uromodulin, 15 the origin of canine RCC was most likely the distal convoluted tubule. However, the bovine study also reported that in the normal bovine kidney, uromodulin was present in epithelial cells of both the proximal and the distal convoluted tubule and, consequently, it was not possible to establish the histogenesis of RCC with this antibody. In the current study we attempted to provide insights into the origin of canine RCC by immunohistochemistry for broad-spectrum keratin (AE1/AE3) and vimentin in both normal and canine RCC. To the best of our knowledge, the expression pattern of vimentin and cytokeratins in normal kidney in comparison with renal carcinomas has not been studied to date. This comparison is essential in order to validate the use of a given antibody as a differentiation marker as this allows pathologists to identify whether its expression is modified (the antigen may be lost, under- or overexpressed, or even neoexpressed) in a neoplasm. 27 Interestingly, here we show that cytokeratins and vimentin are coexpressed in the distal convoluted tubule and, for cytokeratins, this expression extends to the collecting tubule and duct. The coexpression was preserved in 67.5% of the tumors. Vimentin, a marker of mesenchymal cells, is expressed in a large proportion of canine RCC and it has been proposed that this could represent the acquisition of an epithelial to mesenchymal phenotype. 12 However, given that we demonstrated the coexpression of cytokeratins and vimentin in the normal distal convoluted tubule epithelium, this supports the hypothesis of this cell type as the possible origin of canine RCC. This would be in agreement with the above study that used uromodulin expression to support the same hypothesis. 12 In humans, clear cell RCCs account for 70% to 80% of renal carcinomas, and papillary carcinoma is second in frequency accounting for 10% to 15% of renal carcinomas. These tumors share a similar immunoprofile, and most of them express cytokeratins, vimentin, and CD10 (a marker of proximal convoluted tubules) and are negative for CD117 (positive in chromophobe carcinomas). Based on this combination of markers, it is known that clear cell RCCs arise from proximal convoluted tubules. 30,37 In addition, papillary carcinomas are thought to arise from distal convoluted tubules. 1 Taken together, these data suggest that many canine RCCs arise from a different segment of the nephron than that of human RCCs.
Ki-67 protein is expressed in all active phases of the cell cycle (G1, S, G2, and M phases) and is a well-documented marker of cell proliferation in different malignancies. Although there was no significant correlation between Ki-67 expression and clinicopathological features, the Ki-67 index tended to be higher in 14-3-3σ-positive cases. In 40% of cases there was no immunolabeling for Ki-67; nonetheless, these tissues were reactive for cytokeratins and vimentin. One explanation for this lack of labeling may reside in different sensitivity to fixation, and it is known that fixation times can affect some antigens more than others. There is a single case report of a metastatic canine RCC that labeled with Ki-67, but these authors did not specify the source of the antibody or provide a labeling index. 4 In human RCC there are conflicting results regarding Ki-67. In one study Ki-67 served as an independent predictor of oncological outcomes in patients with localized clear-cell renal cell carcinoma, 11 while another study found no correlation between grade, stage, and outcome. 26 Overall it seems that Ki-67 is not yet recognized as a reliable predictor of prognosis for RCC. 25
In summary, this study shows for the first time that 14-3-3σ protein is neoexpressed in a significant number of canine RCCs and its presence is positively associated with a shorter survival time. Furthermore, it seems that in contrast to humans, most canine RCCs may arise from distal segments of the nephron.
Footnotes
Acknowledgements
Dr Alejandro Suárez-Bonnet received a travel grant from the Journal of Comparative Pathology Educational Trust to visit and develop this research in the Department of Pathobiology and Population Sciences at the Royal Veterinary College, University of London, UK.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
