Abstract
This study examined immunohistochemical staining patterns for several meningioma variants involving either the brain or spinal cord of dogs. Formalin-fixed, paraffin-embedded tissue from 15 tumors was obtained. The selected tumor group included seven meningothelial, three transitional, two malignant (anaplastic), one myxoid, one papillary, and one osteomatous meningiomas. Tumors were evaluated for reactivity to the following six immunohistochemical markers: vimentin, pancytokeratin, glial fibrillary acidic protein (GFAP), S100, neuron-specific enolase (NSE), and synaptophysin. Vimentin expression was detected in all meningiomas, and 14 of 15 tumors demonstrated intense vimentin staining in more than 50% of the neoplastic cells. Pancytokeratin expression was present in 11 of 15 neoplasms; however, positive staining frequently was focal and often involved a small percentage of the neoplastic cells. GFAP expression was detected in a single, anaplastic meningioma. Although expression of NSE and S100 was detected in 12 of 25 meningiomas, the intensity of the staining and the percentage of positive neoplastic cells was highly variable. Synaptophysin was uniformly negative. These results will help to establish immunohistochemical profiles for meningiomas that will improve our ability to correctly differentiate these neoplasms of meningeal origin from central nervous system tumors originating from other sites.
Meningiomas are a common neoplasm of the central nervous system (CNS) in both human and veterinary medicine. They occur most frequently external to the dorsal cerebrum near the falx, and other common locations include the tentorium cerebelli, other external surfaces of the brain, and the interventricular region near the choroid plexus of the lateral and third ventricles.19,45 Meningiomas arise from the arachnoid cells (also termed meningothelial cells, meningocytes, or meningoblasts) located within arachnoid villi.4,54 These tumors are histologically diverse and can undergo both mesenchymal and epithelial differentiation, likely as a consequence of their development from the multipotential arachnoid cells.4,19,20
Normal arachnoid cells have the ability to perform many diverse and important functions within the leptomeninges. In some instances, they form intertwining cytoplasmic membranes that have a protective role and function similarly to epithelial and mesothelial cells.4 Arachnoid cells may also be involved in the fibrous response of leptomeninges to injury, and they have the ability to form several structurally important glycoproteins (fibronectin, laminin, collagen type IV, and procollagen type III). In addition, arachnoid cells may have a secretory role related to circulation of cerebrospinal fluid and are considered to be part of the mononuclear phagocyte system.4 Light and electron microscopy coupled with immunohistochemical staining have been utilized to determine the immunophenotype of arachnoid cells. Several human studies have described positive staining for vimentin, desmoplakin, epithelial membrane antigen (EMA), and some cytokeratins.2,21,29
Ultrastructural analyses have revealed that nonneoplastic arachnoid cells are joined through desmosomal attachments and hemidesmosome-like junctions, as well as gap, intermediate, and tight junctions.19,28,54 All of these structures also have been identified in meningiomas, although the number of cell junctions typically is decreased.54 An ultrastructural study by Mennel et al.30 concluded that interdigitating membranes, desmosomes, and intermediate filaments are found in all meningiomas, irrespective of the predominant morphological pattern.
In view of the wide array of functions performed by arachnoid cells and their ability to develop structural features of both mesenchyme and epithelia, it is not surprising that meningiomas exhibit highly variable morphologic patterns. The World Health Organization (WHO) classification for human tumors currently contains 15 distinct meningeal variants, which are separated into three grades (Table 1).27 The classification of meningiomas in veterinary medicine is not as elaborate and well defined. Many of the human categories and terms are utilized, but no universal classification system has been established. The most recent classification by Koestner et al.25 describes nine histologic patterns in domestic animals that parallel some of the variants included in the human classification system (Table 2). A list of patterns common to both human and veterinary medicine includes meningothelial (meningotheliomatous), fibrous, transitional, psammomatous, angiomatous, papillary, and anaplastic tumors.45
World Health Organization (WHO) classification of human meningiomas. 27
World Health Organization (WHO) classification of meningiomas in domestic animals. 25

In addition to these general categories, other variants have been described in the veterinary literature and mixtures of the patterns are common.44 Additional meningeal variants that have been documented include microcystic, atypical, lipomatous, myxoid, granular cell, angioblastic, endotheliomatous, and osteomatous tumors.11,25,40,45,48 According to WHO classification for human tumors, the lipomatous and osteomatous variants would be included in the metaplastic meningioma category.9,27 Although the terms angioblastic and angiomatous often are used synonymously in veterinary medicine, human researchers have concluded that neoplasms previously described as angioblastic meningiomas are actually hemangiopericytomas.17 Angiomatous refers to an actual meningeal variant, whereas the term angioblastic is now considered obsolete. The same trend for angioblastic meningiomas possibly exists in veterinary medicine, but this concept has yet to be investigated. Myxoid meningioma is a rare variant that has been described in both the human and veterinary literature. Although it has been included in the recent classification of meningiomas in domestic animals, myxoid meningioma is not currently included in human classification schemes.3,13,22,45,48 An evaluation of canine intracranial tumors by Patnaik et al.36 revealed that the meningotheliomatous (meningothelial) and transitional types are the most commonly diagnosed.
Vimentin is the predominant intermediate filament of human meningiomas, and continued investigation into the immunophenotype of these neoplasms also has revealed significant epithelial membrane antigen expression as well as desmoplakin and cytokeratin expression in many of the histologic subclassifications.5,12,26,31,43
Although meningiomas are a common neoplasm of dogs, particularly older dogs, few immunohistochemical studies on canine meningiomas have been reported in the literature.41 This study describes the staining patterns of canine meningiomas for six immunohistochemical markers that are commonly utilized to diagnose neoplasms of the CNS.
Materials and Methods
Twenty-nine canine meningiomas diagnosed at the Texas A&M University Veterinary Medical Teaching Hospital between 1993 and 2000 were evaluated, and 15 tumors were chosen for analysis. Only tumors that were unequivocally diagnosed as a meningioma were selected. In addition, several tumor blocks contained bone and had been previously decalcified. Because this process is known to interfere with antigen detection, these tumors were excluded from the study. The neoplasms were then screened with vimentin to determine if the preservation of the archived specimens was sufficient for detection and analysis of antigen expression. Tumors with poor vimentin labeling were excluded. The age of the tissue blocks selected for the study ranged from several months to 7 years; however, the majority had been stored at room temperature for less than 3 years. Tumors were classified morphologically with hematoxylin and eosin (HE)–stained sections and the WHO classification system for human meningiomas.27
A panel of six immunohistochemical markers that included vimentin, pancytokeratin, S100, neuron-specific enolase (NSE), glial fibrillary acidic protein (GFAP), and synaptophysin was selected (Table 3). All tissues were routinely processed with 10% neutral buffered formalin and embedded in paraffin. Five-micrometer–thick sections from the tissue blocks were placed onto poly
Antibodies used for immunohistochemistry.

All antibodies were obtained from Dako.
Immunohistochemical results were categorized based on the number of positively stained tumor cells as follows: − = no tumor cells stained; (+) = less than 10% or isolated positive tumor cells; + = 10–50% of the tumor cells stained positive; ++ = 50–90% of the tumor cells stained positive; and +++ = greater than 90% of the tumor cells stained positive. Staining intensity was reported as either light, moderate, or high. Positive results were further evaluated for the presence of focal or diffuse staining patterns, and the location of the stain either in the cytoplasm, nucleus, or on the cell surface.
Results
Several staining trends for meningiomas were observed. Table 4 provides a detailed list of the staining characteristics determined for each meningioma.
Reactivity against antibodies.∗

GFAP = glial fibrillary acidic protein; NSE = neuron-specific enolase; F = female; S = spayed; M = male; N = neutered. +++ = more than 90% of tumor cells positive; ++ = 50–90% of tumor cells positive; + = 10–50% of tumor cells positive; (+) = <10% or isolated positive tumor cells; − = no tumor cell reactivity.
Positive staining for vimentin was identified in all tumors, regardless of subclassification, and 11 (73%) of 15 tumors displayed positivity in greater than 90% of the neoplastic cells. Only one tumor contained less than 50% positive cells. In all tumors, cells positive for vimentin displayed diffuse cytoplasmic staining, and the intensity was generally very high. Figures 1–3 illustrate vimentin staining from a meningothelial, anaplastic, and transitional meningioma, respectively.
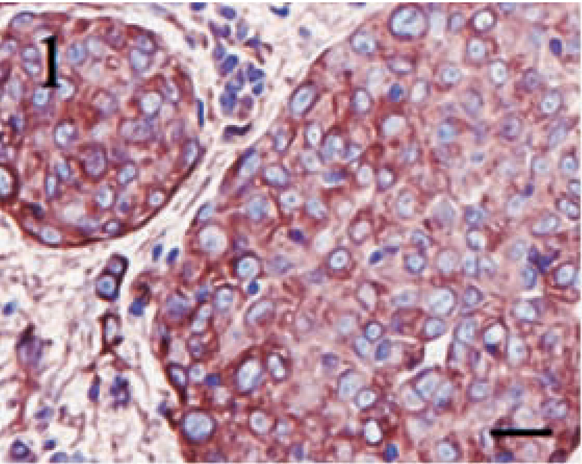
Meningothelial meningioma. High-intensity, diffuse cytoplasmic staining for vimentin in nearly 100% of the neoplastic cells. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
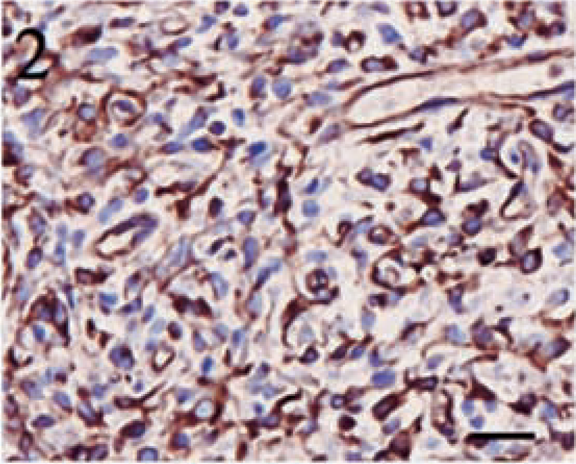
Anaplastic meningioma. High-intensity, diffuse cytoplasmic staining for vimentin in a majority of the neoplastic cells. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
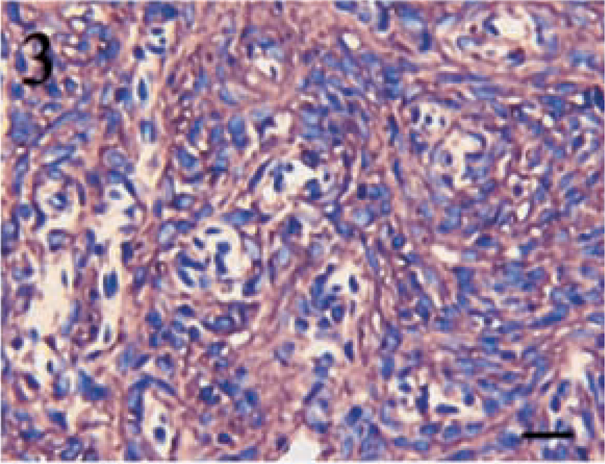
Transitional meningioma. High-intensity, diffuse cytoplasmic staining for vimentin throughout the cellular whorls typical of a transitional meningioma. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
Pancytokeratin staining showed a much higher degree of variability than did vimentin. Four tumors failed to display a positive reaction for cytokeratin. No tumor contained greater than 50% positive cells, and the majority of the tumors had only 5–15% positive cells. Despite the lower percentages, the intensity of the cytokeratin stain was typically high. Positive cells were often randomly scattered throughout the sections, and they frequently formed small, multifocal aggregates (Figs. 4–6).
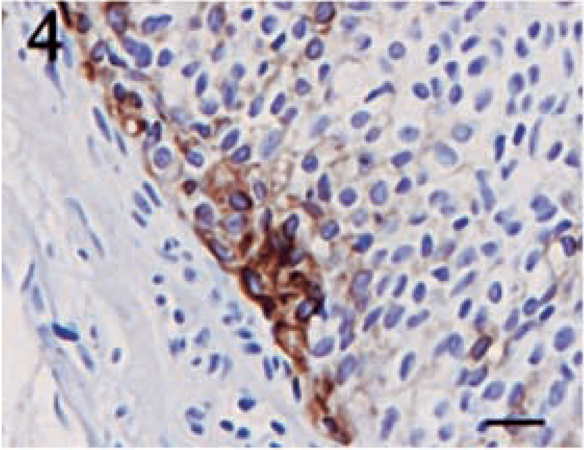
Meningothelial meningioma. Focal area of pancytokeratin positivity displaying a membranous staining pattern. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
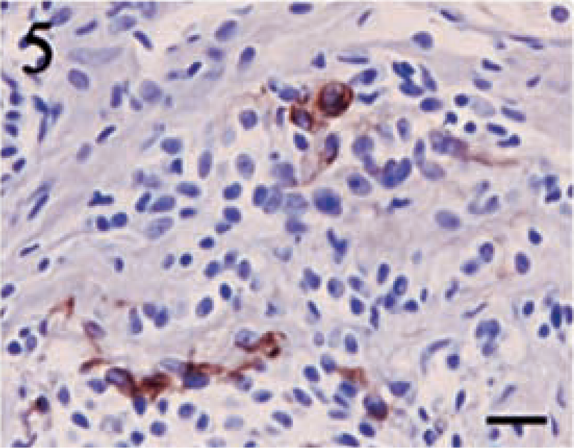
Meningothelial meningioma. Scattered cells demonstrating intense pancytokeratin reactivity are irregularly identified throughout the neoplasm. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
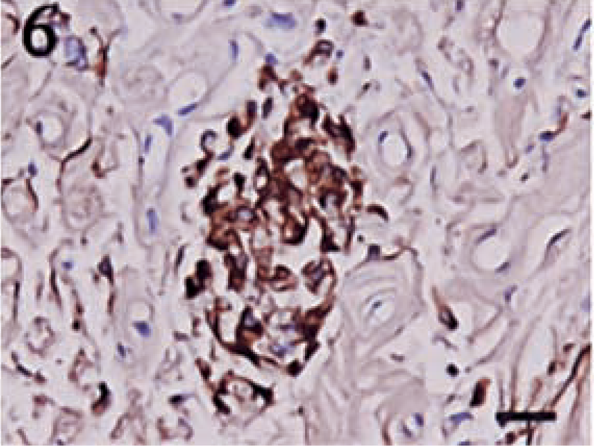
Anaplastic meningioma. A focal area of cells staining intensely for pancytokeratin. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
The two most common meningeal variants in this study, meningothelial and transitional, appeared to have differences in cytokeratin expression. Cytokeratin staining was detected in six (86%) of seven of the meningothelial meningiomas, whereas one of three meningiomas contained positive cells. In addition, the number of positive cells in this transitional tumor was very low (<5%). The only other variant that failed to display cytokeratin positivity was the papillary meningioma (n = 1). The staining pattern for cytokeratin of all tumors was membranous with the exception of one meningothelial meningioma that exhibited a diffuse, punctate, cytoplasmic pattern.
GFAP expression was noted in only one anaplastic meningioma. The staining pattern was diffusely cytoplasmic, and the intensity varied from moderate to high. Positive cells were present in focal to locally extensive aggregates, in which approximately 30–40% of the neoplastic cells displayed GFAP expression (Fig. 7).
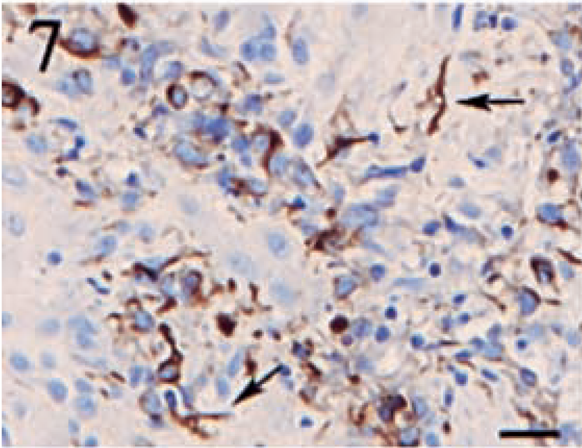
Anaplastic meningioma. A focal area of oval, neoplastic cells displaying intense, cytoplasmic staining for GFAP. Arrows denote several GFAP-positive stellate cells indicative of reactive astrocytes. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
Positive staining for S100 was detected in 12 (80%) of 15 tumors. The intensity of the staining and the distribution of positive cells was highly variable. Although the number of positive cells was 70% in one tumor, most of the tumors contained 5–25% positive cells. The staining intensity was typically light to moderate. The pattern of S100 expression was variable. In some tumors, individual positive cells were scattered throughout the sections, whereas other tumors contained focal to locally extensive aggregates of positive cells. Granular staining in both the nucleus and cytoplasm was commonly noted for this marker (Figs. 8 9).
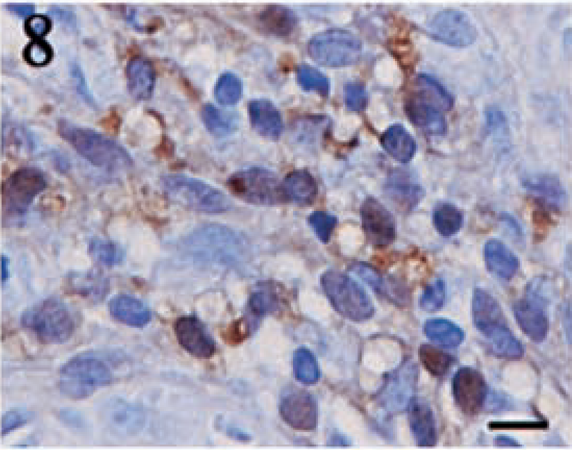
Anaplastic meningioma. Moderate to highly intense, granular staining for S100 is detected in both the cytoplasm and the nucleus. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 10 μm.
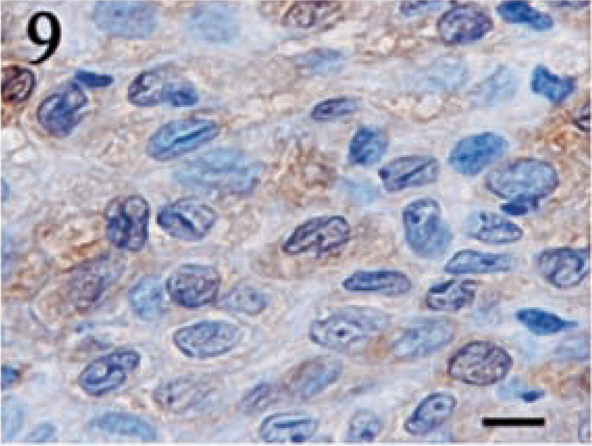
Meningothelial meningioma. Weak to moderately intense, granular staining for S100 is detected in both the cytoplasm and the nucleus. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 5 μm.
Immunostaining for NSE was highly variable in both distribution and intensity. Twelve (80%) of fifteen tumors demonstrated NSE expression. Similar to S100, the intensity of the staining was inconsistent, with the majority of the neoplasms displaying only a light to moderate level of positivity. Tumor sections contained a range of 5–60% positive cells; however, in most cases, the number of positive cells varied from 5 to 30%. Staining was typically cytoplasmic and granular (Figs. 10, 11). An interesting feature of NSE expression was the occurrence of positive cells in a bandlike fashion that occasionally centered around vessels or connective tissue cells (Fig. 12). These areas were located within the body of the tumor and not attributed to edge artifact.
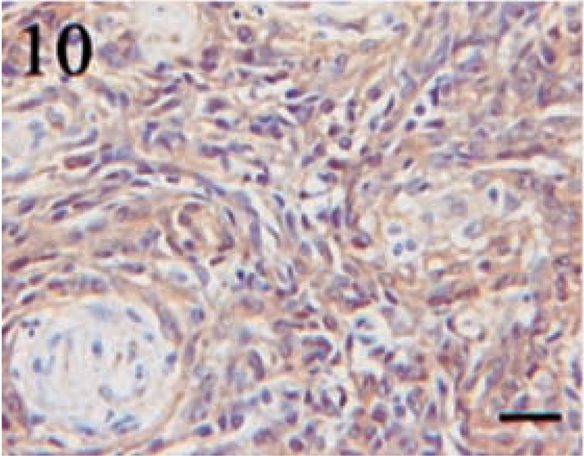
Transitional meningioma. Diffuse, moderately intense, granular, cytoplasmic staining for NSE. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
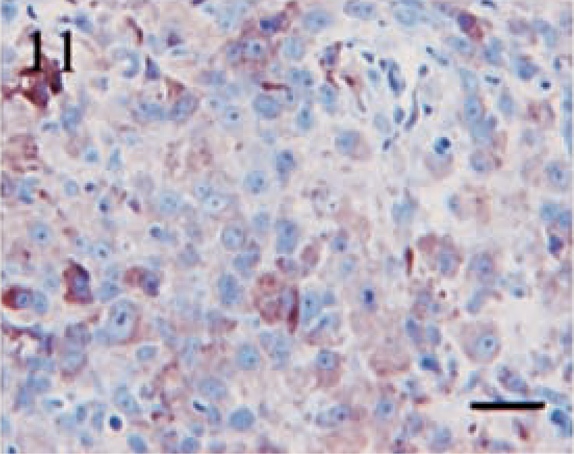
Anaplastic meningioma. Diffuse, granular cytoplasmic staining of variable intensity for NSE. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 20 μm.
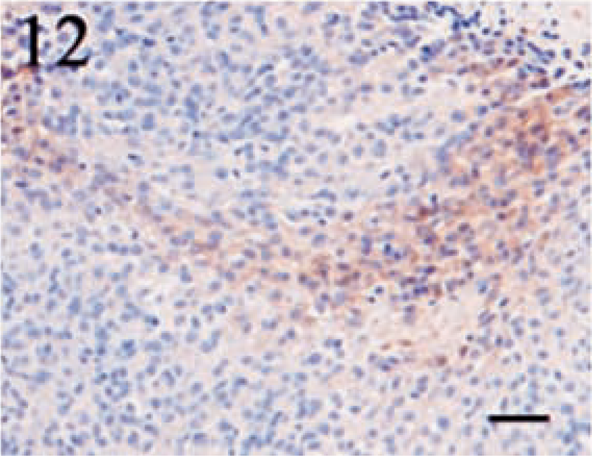
Meningothelial meningioma. This section illustrates the bandlike pattern of NSE-positive cells that was characteristic of several tumors. Streptavidin–biotin complex, DAB–horseradish peroxidase, Mayer's hematoxylin counterstain. Bar = 40 μm.
Synaptophysin stained normal canine neural tissue, but it was uniformly negative in all tumors.
Discussion
Information on the immunophenotype of CNS neoplasms, including meningiomas, in veterinary medicine is extremely limited. Currently, the majority of the information regarding immunohistochemical phenotyping of CNS tumors has been derived from case reports involving only one or two animals. Table 5 summarizes previous studies and case reports that describe the immunophenotype of canine meningiomas. The present study was designed to elicit a more complete understanding of the immunohistochemical staining characteristics for six different morphologic patterns found in canine meningiomas and to determine similarities between immunohistochemical staining patterns in human meningiomas.
Previous reports describing the immunophenotype of canine meningiomas.∗

NSE = neuron-specific enolase; GFAP = glial fibrillary acidic protein; FF/PE = formalin-fixed/paraffin-embedded; NA = not available.
Multiple tumor variants were included in the study.
Numerous reports describe the immunophenotype of human meningiomas. The majority of these studies were performed retrospectively on formalin-fixed, paraffin-embedded tissues, and the most commonly evaluated immunohistochemical markers include vimentin, cytokeratin, GFAP, S100, NSE, and EMA. Several additional studies also have utilized frozen tissues, cytological specimens, and cultured cells for immunophenotyping. The results of these investigations are summarized in Table 6.
Previous reports describing the immunophenotype of human meningiomas.∗

NSE = neuron-specific enolase; GFAP = glial fibrillary acidic protein; AF/PE = alcohol-fixed/paraffin-embedded; FF/PE = formalin-fixed/paraffin-embedded; cyto = impression smears or cerebrospinal fluid; NA = not available.
Study conducted on a specific meningeal variant.
Vimentin is the most widely distributed intermediate filament found in mesenchymal tissues, and vimentin immunostaining is commonly used to delineate tissues of mesenchymal origin.35 The high level of vimentin expression detected in 100% of the meningiomas in the present study closely paralleled the results obtained from human studies. With the exception of a single report that revealed vimentin staining in only 9 (18%) of 50 tumors, all investigators detected strong vimentin staining in nearly 100% of the tumors examined.28 The conflicting report was obtained from one of the oldest studies, and less than optimal tissue fixation for immunohistochemistry may explain the unusually low level of vimentin staining.
A single study in the veterinary literature reported vimentin staining in 26 (79%) of 33 canine meningiomas.41 Two case reports involving papillary meningiomas had conflicting results. One tumor was vimentin positive, whereas the other did not show vimentin expression.10,42 Vimentin staining also was reported previously in microscopic sections from four myxoid meningiomas and cytologic impression smears from meningiomas in which the variant was not described.48,49
In the human literature, numerous studies have characterized the degree of epithelial differentiation in meningiomas by use of several markers, primarily cytokeratins and EMA. Human studies have consistently reported a high level of EMA expression.1,32,34,39 Unfortunately, effective antibodies for EMA currently have not been established in veterinary medicine. Cytokeratin expression displays a higher degree of variability than EMA expression in human meningiomas. Studies have reported a range of 0–100% positivity, but some of this variation may reflect the type of meningiomas that predominated. For example, a study performed entirely on secretory meningiomas concluded that all 31 tumors expressed cytokeratin.38 Another factor that researchers believe has contributed to the conflicting reports of cytokeratin staining in human meningiomas is the inconsistent use of different individual cytokeratin markers. Overall, 25% of human meningiomas have been estimated to have focal cytokeratin expression.9
In the present study, the staining pattern noted with the pancytokeratin antibody varied between different types of canine meningiomas. Although transitional meningiomas apparently are less likely to express cytokeratin, it is difficult to make a strong conclusion because of the low number of tumors examined (n = 3). A multifocal staining pattern involving less than 50% of the tumor cells was a common feature of cytokeratin expression. The only other comprehensive study evaluating the immunohistochemistry of canine meningiomas demonstrated that 42% (14/33) of the tumors had cytokeratin staining that was frequently focal in distribution.41 In a report detailing four myxoid meningiomas, two tumors displayed focal cytokeratin positivity.48 Immunocytochemical analysis performed on impression smears of meningiomas, in which the variant was not reported, also demonstrated cytokeratin staining in focal cell clusters.49 Thus, our data indicate that many cells in canine meningiomas will express cytokeratins in addition to vimentin.
GFAP is the characteristic intermediate filament of both normal and reactive glial cells, and GFAP production frequently is maintained in glial neoplasms.35 Although GFAP intermediate filaments are not considered a normal structural component of meningeal cells, GFAP expression has been rarely reported in human meningiomas44,52 and other nonglial tissues. Because reactive astrocytes are often associated with neoplasms, it may be difficult to determine whether GFAP-positive cells are infiltrating astrocytes or a true component of the tumor. Several convincing reports have described GFAP positivity in chorvp.39-3-311d and papillary meningiomas in the human literature.5,44,52
The present study also revealed strong evidence of GFAP expression in a malignant meningioma. Although reactive astrocytes were present, the majority of the GFAP-positive cells resembled neoplastic cells and not reactive astrocytes. Based on morphologic features of the neoplasm that were strongly indicative of a meningioma and concurrent cytokeratin expression, it was considered unlikely that this tumor represented an anaplastic glioma. Currently, the basis for GFAP expression in human meningiomas has not been established. This highly anaplastic tumor likely represents an aberration, and we postulate that GFAP expression will be a rare occurrence in meningiomas.
Originally, the S100 protein was found exclusively in neural cells, but subsequent investigations have demonstrated that many nonneural tissues, both neoplastic and nonneoplastic, react with antibodies to the S100 protein.35 Immunohistochemical evaluation of S100 protein in human meningiomas has demonstrated variable positivity that is rarely a prominent feature of the tumor.4,9,27 The staining pattern is inconsistent, and scattered foci of positive cells are often identified. One study described the intensity of S100 staining in several meningeal variants as weak.14 Staining for S100 may occur diffusely in both the cytoplasm and the nucleus.29,38,39 In the present study, very similar results for S100 were obtained. Although S100 often was detected, the intensity was rarely marked, and the positive cells often were scattered in a multifocal and irregular pattern throughout the tumor sections. Both nuclear and cytoplasmic staining were observed. Consequently, this marker may be of ancillary assistance in ruling out certain neoplasms, but will not help to establish a firm diagnosis of meningioma by itself.
Few investigators have attempted to evaluate the presence of NSE in human meningiomas, and significant discrepancy exists between the studies that have examined NSE expression. NSE preferentially localizes to neurons and endocrine cells of the amine precursor uptake and decarboxylation system, and this is frequently used to identify neural and neuroendocrine tumors. Despite the known specificities, NSE also has been identified in many other normal, reactive, and neoplastic tissues.51 Two studies of human meningiomas found no NSE expression, whereas three others found NSE staining in 67–91% of the tumors evaluated. The two studies that reported no staining were performed on either cultured cells or cytologic samples from a total of 14 meningiomas, whereas the other three studies included formalin-fixed, paraffin-embedded samples from 139 tumors.1,15,18,24,29 The differences in NSE expression may be attributable to the type of sample used for analysis; however, given the large percentage of the 139 tumors that demonstrated NSE staining, it is likely that NSE positivity is a real feature of human meningiomas.
In the canine meningiomas, NSE-positive cells seemed to be a consistent feature; however, the number of positive cells in each tumor was typically low (approximately 5–30%) and staining was often light to moderate and not a prominent feature. The significance of the bandlike pattern of positivity identified in some tumors could not be determined.
Synaptophysin, a highly specific marker for presynaptic vesicles found in neural and neuroendocrine tissues, is utilized to diagnose primary neural tumors (e.g., medulloblastoma and ganglioneuroma) in the brain and spinal cord.35 In addition, synaptophysin occasionally is detected in oligodendrogliomas.8,53 All of the canine meningiomas in this study were negative, a feature consistent with the absence of synaptophysin staining in reported cases of human meningiomas.
In summary, the immunohistochemical phenotypes identified for major morphologic variants of canine meningiomas are very similar to those described for human meningiomas. Vimentin is the predominant intermediate filament, and strong vimentin expression in a majority of neoplastic cells is expected. Cytokeratin staining is variable but common, and frequently focal. Although some meningiomas will fail to express cytokeratin, the presence of dual positivity for vimentin and cytokeratin is a useful diagnostic feature that helps distinguish meningiomas from other tumors of the CNS. In general, GFAP staining is indicative of a glial tumor, but one meningeal neoplasm expressed GFAP. Consequently, GFAP expression does not unequivocally exclude the possibility of a meningioma. S100 and NSE staining often are reported for meningiomas; however, they are rarely the predominant feature, and the presence or absence of these markers cannot be utilized to confirm the diagnosis of meningioma. A lack of synaptophysin expression is expected for all meningeal variants. The results of this study suggest that canine meningiomas have antigenic expression that is comparable to their human counterparts, and the immunoreactivity profiles provide a basis for further study of this disease.
