Abstract
Feline infectious peritonitis (FIP) is a serious, widely distributed systemic disease caused by feline coronavirus (FCoV), in which ocular disease is common. However, questions remain about the patterns of ocular inflammation and the distribution of viral antigen in the eyes of cats with FIP. This study characterized the ocular lesions of FIP including the expression of glial fibrillary acidic protein and proliferating cell nuclear antigen by Müller cells in the retina in cases of FIP and to what extent macrophages are involved in ocular inflammation in FIP. Immunohistochemistry for FCoV, CD3, CD79a, glial fibrillary acidic protein, calprotectin, and proliferating cell nuclear antigen was performed on paraffin sections from 15 naturally occurring cases of FIP and from controls. Glial fibrillary acidic protein expression was increased in the retina in cases of FIP. Müller cell proliferation was present within lesions of retinal detachment. Macrophages were present in FIP-associated ocular lesions, but they were the most numerous inflammatory cells only within granulomas (2/15 cats, 13%). In cases of severe inflammation of the ciliary body with damage to blood vessel walls and ciliary epithelium (3/15, 20%), some macrophages expressed FCoV antigens, and immunolabeling for calprotectin on consecutive sections suggested that these FCoV-positive macrophages were likely to be recently derived from blood. In cases of severe and massive inflammation of most ocular structures (4/15, 26%), B cells and plasma cells predominated over T cells and macrophages. These results indicate that gliosis can be present in FIP-affected retinas and suggest that breakdown of the blood–ocular barrier can allow FCoV-bearing macrophages to access the eye.
Keywords
Feline infectious peritonitis (FIP) is one of the most serious viral diseases of domestic and wild Felidae and is a leading infectious cause of mortality in young cats. 32,35,39 Feline coronavirus (FCoV), the causative agent of this disease, occurs in 2 pathotypes: avirulent, feline enteric coronavirus, which can persist for a long time in the colon of carrier animals, and virulent, feline infectious peritonitis virus. 35 In cats infected with feline enteric coronavirus, the virus persists not only in the colon but also in macrophages in the lymphatic organs. 29 There is strong evidence that FCoV mutates from feline enteric coronavirus to feline infectious peritonitis virus after infecting cats and that the avirulent type is found in macrophages in the blood of healthy animals. 35,43
Morphologically, the disease is characterized by fibrinous, pyogranulomatous, or granulomatous inflammation of different organs with protein-rich effusions in body cavities. 28 The most common lesions are granulomas and pyogranulomas accompanied by granulomatous to necrotizing phlebitis and periphlebitis, and these are considered a morphologic hallmark of the disease. 4,25,28,34,45 For diagnosis, it can be valuable to classify FIP into 3 different forms: the effusive (wet) form, the noneffusive (dry) form, and a mixture of both. 7,21,24 However, it is recognized that effusion and granulomatous changes are nearly always present to some extent, and individuals transition from one form to the other. 7,24
Ocular lesions are frequent in cats with FIP. 24,28 In natural cases of FIP, those with ocular lesions are more likely to have the dry form than the wet form of the disease and more likely to have concurrent brain lesions. 3,9,28,35
Various forms of ocular inflammation have been described in cats with FIP. Pyogranulomatous or granulomatous inflammation has been found throughout the uvea and in the sclera, conjunctiva, retina, and optic nerve. Macrophages, lymphocytes, plasma cells, and neutrophils were focally or diffusely distributed in these affected tissues. 3,9,15,27,36,45,46 Granulomatous phlebitis and periphlebitis occur in the iris, choroid, retina, and sclera and around the optic nerve. 27 In naturally infected cats, macrophages containing viral antigens are present within inflammatory infiltrates around the choroidal veins, in the connective tissue of the third eyelid, and in the conjunctiva. 25,27,41
This study investigated the patterns of ocular inflammation and the distribution of FCoV-bearing macrophages in the eyes of cats with FIP. The first objective was to determine whether expression of glial fibrillary acidic protein (GFAP) by retinal Müller cells is increased in cases of FIP with retinitis as it is in response to retinal infections in other species and during some ocular disorders in cats such as retinal detachment, progressive retinal atrophy in Abyssinian cats, and autosomal dominant feline rod/cone dysplasia. 10,11,16,17,19,33 In all of these conditions, Müller cells react by increasing GFAP expression; this upregulation is a sensitive, nonspecific marker of Müller cell gliosis. The second objective was to characterize Müller cell proliferation in the retina in FIP, as also occurs in retinal detachment in cats and retinal injuries in other species. 2,10,18 The third objective was to characterize the involvement of macrophages in ocular inflammation in FIP. In general, macrophages are thought to play an important role in the pathogenesis of FIP due to massive replication of FCoV inside these cells, and macrophages seem to be the predominant inflammatory cells in FIP lesions in other organs. 7,25,27,28,34 However, the blood–ocular barrier normally makes the eye an immune-privileged organ, and it has not been determined whether macrophages predominate within ocular lesions in cats with FIP and how many of these macrophages bear the virus.
Materials and Methods
Animals and Tissues
The study was performed on 15 cats of different breeds (Canadian Sphinx, n = 4; Persian, n = 1; European shorthair, n = 10) between 1 and 3 years of age, which were spontaneously infected with FCoV. The cats were presented with clinical manifestations of FIP at different private veterinary practices and at the Veterinary Polyclinic of the University of Warmia and Mazury in Olsztyn (Poland) between 2008 and 2015. The cats were selected for this study irrespective of their ocular signs. However, in 6 animals, obvious ocular signs such as hyphema, iris rubeosis, keratic precipitates on the corneal endothelium, and fibrin accumulation in the anterior chamber were observed (Supplemental Figs. S1–S4). Serum samples from all cats were tested for the presence of feline leukemia virus (FeLV) antigen and feline immunodeficiency virus (FIV) antibody with commercially available tests (VetExpert FIV Ab/FeLV Ag; VetPlanet, Warsaw, Poland) and all were negative. Additionally, antibody titers against FCoV were measured in serum of 7 cats, and the serum albumin to globulin ratio was measured in 6 cats. The decision to euthanize the cats with FIP was based on their clinical condition including most or all of the following signs: anorexia, depression, neurological signs, dyspnea, and general cachexia. The cats were euthanized by intravenous pentobarbital overdose and prepared for the histologic and immunohistochemical investigations. For negative controls, eyes were obtained from age-matched cats that were euthanized for reasons unrelated to ophthalmologic diseases. All procedures were performed in strict accordance with Polish regulations for animal welfare and approved by the Local Ethical Commission for Animal Experimentation.
Histology and Immunohistochemistry
After euthanasia, the eyes and samples of other organs (liver, spleen, kidneys) were immediately fixed in 4% paraformaldehyde. The eyes were sectioned on the sagittal plane (these sections included the optic nerve of all but 3 animals), dehydrated, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin or with methyl green-pyronin for plasma cell identification.
Immunohistochemical examination of the globe sections was performed using a panel of primary antibodies against FCoV, CD3, CD79a, calprotectin, GFAP, and proliferating cell nuclear antigen (PCNA), and a visualization system based on the immunoperoxidase method, with 3,3-diaminobenzidine or 3-amino-9-ethylcarbazole as the substrate (Supplemental Table S1). The specimens were counterstained with Mayer’s hematoxylin and coverslipped using Mounting Glycergel Medium (Dako, Glostrup, Denmark) or Canadian balsam. For negative controls, the primary antibody was replaced with mouse IgG1 or IgG2a (Dako) at the appropriate dilution (FCoV, CD79a, calprotectin, GFAP, PCNA) or omitted (CD3). Feline lymphatic tissues served as positive controls for CD3 and CD79a. Sections of internal organs (liver, spleen, and kidneys) were stained with hematoxylin and eosin and immunohistochemically to confirm the presence of FCoV antigens within the FIP lesions.
A scoring system was used to evaluate the intensity of ocular inflammation based on the number of inflammatory cells in ocular anatomic structures with lesions. A score ranging from 0 to 4 was assigned to each ocular structure based on the number of inflammatory cells that were present within all of the structure that was visible in a single section. A score of 0 indicates absence of inflammation (0-3 inflammatory cells); 1 indicates slight inflammation (4-50 inflammatory cells); 2, moderate inflammation (51-100 inflammatory cells); 3, severe inflammation (101-200 inflammatory cells); and 4, massive inflammation (over 200 inflammatory cells) (see Supplemental Table S2 for more information). The data analyzed in this study are not available as supplemental materials.
Results
In all cases of FIP, the necropsy revealed fibrinous peritonitis and/or pleuritis with fibrin deposition on the serosal surfaces. In 13 of 15 cases, examination revealed an accumulation of fibrinous, clear to yellowish fluid in the body cavities. In 8 of 15 cases, multifocal granulomatous lesions of variable size (3-10 mm) were found in the liver, spleen, and kidneys and on the serosal surfaces.
Histologically, in the 7 cats with fluid accumulation in the body cavities (effusive FIP) (Table 1), multifocal pyogranulomas consisting of central macrophages, peripheral neutrophils, and scattered plasma cells were detected in all examined internal organs. Two cats did not have fluid accumulation in the body cavities (noneffusive FIP), and these had multifocal granulomas in all examined organs composed of central macrophages and peripheral plasma cells. In 6 cats with the mixed form of the disease, both multifocal pyogranulomas and granulomas were revealed in the examined organs. Immunohistochemistry showed the presence of FCoV antigens in all examined organs in each case.
Signalment and Clinical Features of 15 Cats With Feline Infectious Peritonitis.
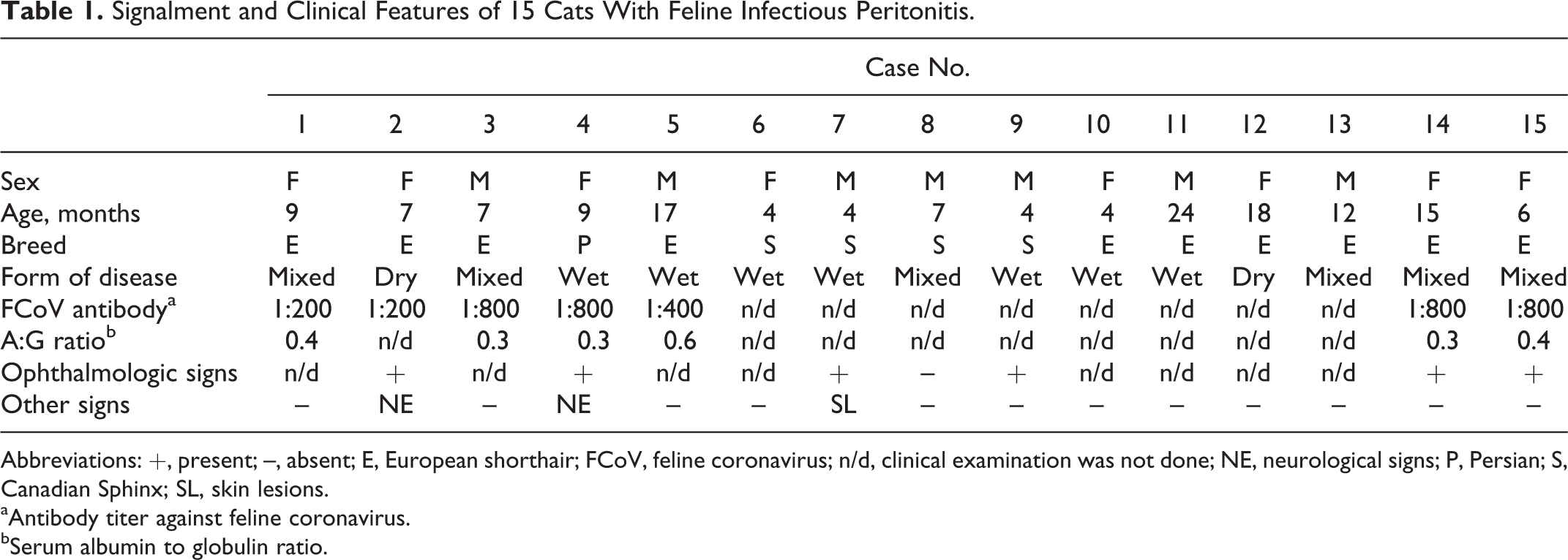
Abbreviations: +, present; –, absent; E, European shorthair; FCoV, feline coronavirus; n/d, clinical examination was not done; NE, neurological signs; P, Persian; S, Canadian Sphinx; SL, skin lesions.
aAntibody titer against feline coronavirus.
bSerum albumin to globulin ratio.
Histologic examination of the eyes from cats with FIP revealed that, in most of the examined eyes, there were both diffuse and multifocal inflammatory infiltrates. The intensity of inflammation differed between cats and between individual ocular structures in the same animal. The inflammation was most intense in the ciliary body (mean score = 2.4), the retina (mean score = 2.3), and the choroid (mean score = 2.2).
Uvea
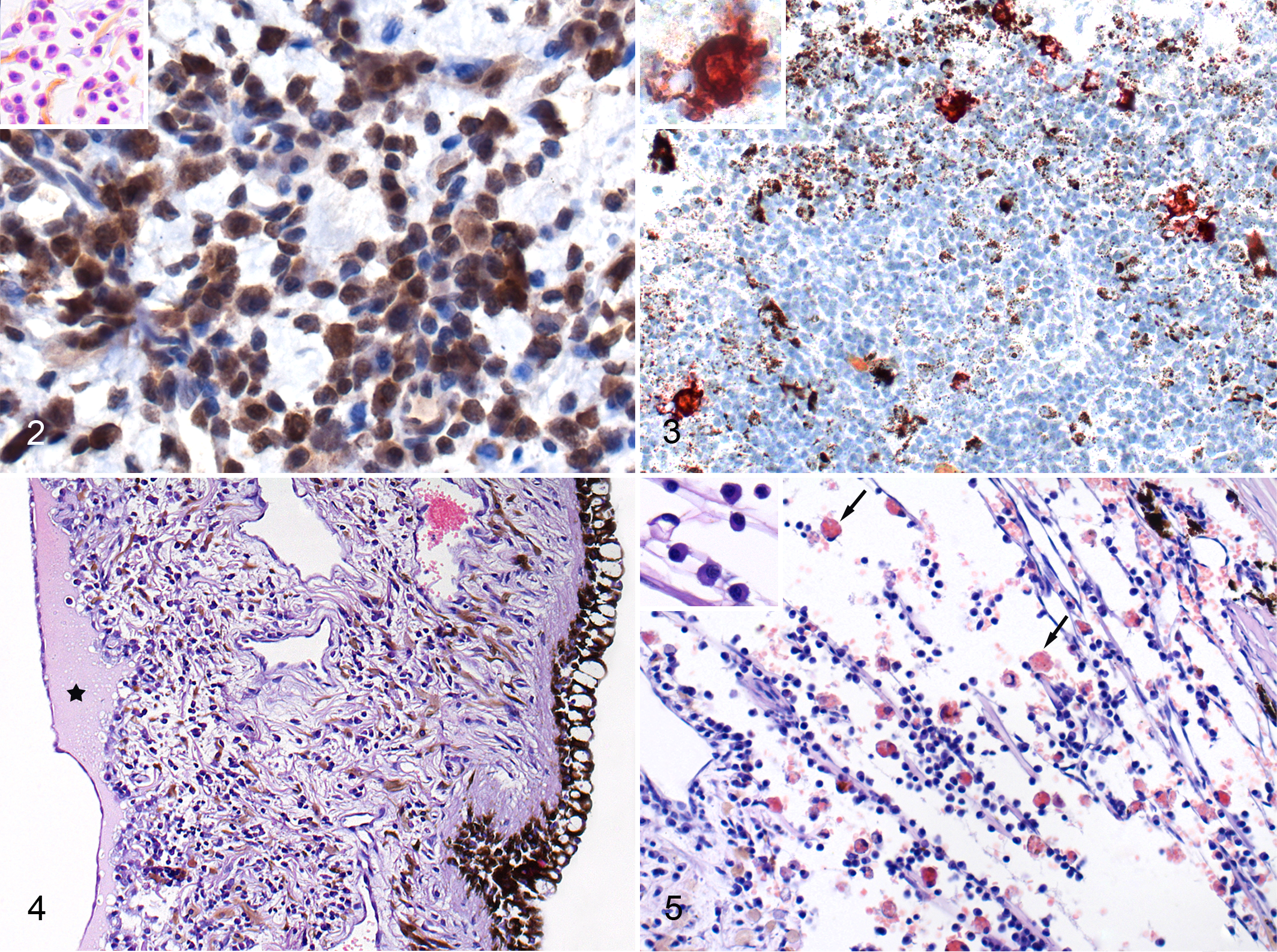
In the iris, the average intensity of inflammation was slight to moderate (mean score = 1.8), but 4 cats did have severe or massive inflammation, and these 4 cats had lesions of damage to blood vessels in the ciliary body and conjunctiva manifested as endothelial necrosis and loss of vessel wall continuity (Fig. 1). In these 4 cats, the most numerous inflammatory cells were B cells (CD79a+) and plasma cells (Fig. 2), and macrophages and T cells (CD3+) were also present. In some of these cases, the inflammatory cells had densely infiltrated the iris stroma and formed nodules composed mostly of B cells. Most of the macrophages present on the sections expressed calprotectin (indicating that they were recently blood-derived 9 ), and a lesser number of macrophages also expressed FCoV antigen (Fig. 3). In cats with moderate to massive inflammation of the iris, lesions were often accompanied by an accumulation of protein-rich fluid in the iris stroma, with vacuolization and damage to the iris epithelium (Fig. 4). In nearly all cats, the inflammatory cells extended from the base of the iris into the trabecular meshwork, and in cases of massive inflammation, this area contained numerous erythrophagocytic macrophages (Fig. 5).
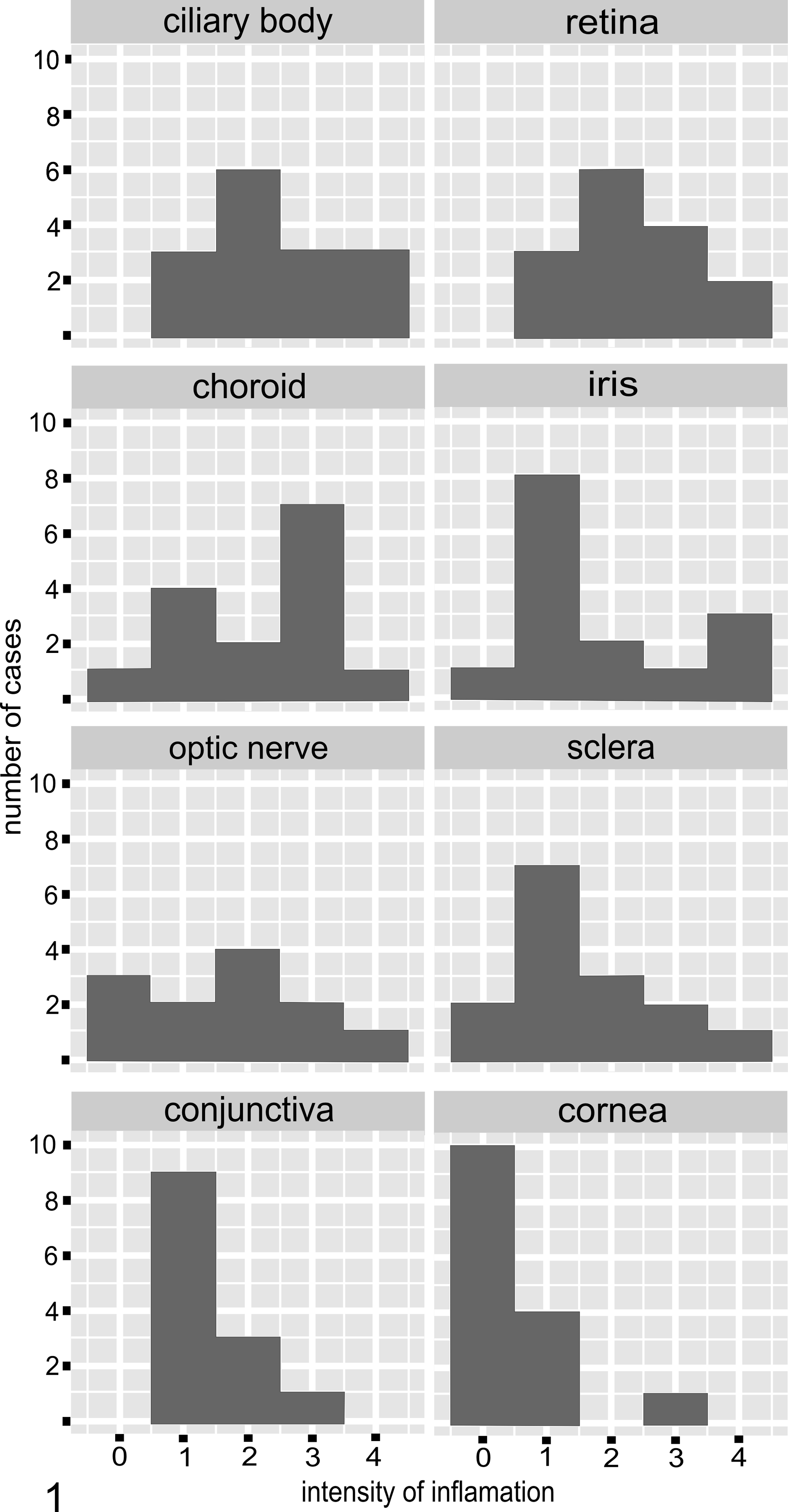
Variation in intensity of ocular inflammation in cats with feline infectious peritonitis. The vertical axis shows the number of cases and the horizontal axis shows the score for intensity of inflammation (as defined in the Materials and Methods section).
All 15 cases had inflammation of the ciliary body that involved both the ciliary stroma and the ciliary veins. The pars plicata of the ciliary body (ciliary processes) in all cats with slight and moderate inflammation contained B cells (CD79+) intermingled with T cells (CD3+) in roughly equal proportions, with fewer macrophages, some of which were calprotectin positive (recently blood-derived), present on the epithelial surface. In severe and massive inflammation, there were more B cells than T cells (Fig. 6a, b). Calprotectin-positive macrophages were more common in cellular infiltrates in cases where there was necrosis to both layers of the epithelium of the ciliary processes, and even more common when necrosis was also present in their stroma (Fig. 7a, b). In the damaged areas, some of the macrophages expressed FCoV (Fig. 7b); this was most common in the samples where both the stroma and the epithelium were damaged. Immunohistochemistry on consecutive sections showed that some FCoV-positive macrophages also expressed PCNA (suggesting that they were proliferating). Some FCoV-negative macrophages were also PCNA positive. All cases with massive inflammation of the ciliary body also had marked edema of the ciliary body with accumulation of exudate and rupture of the ciliary processes.
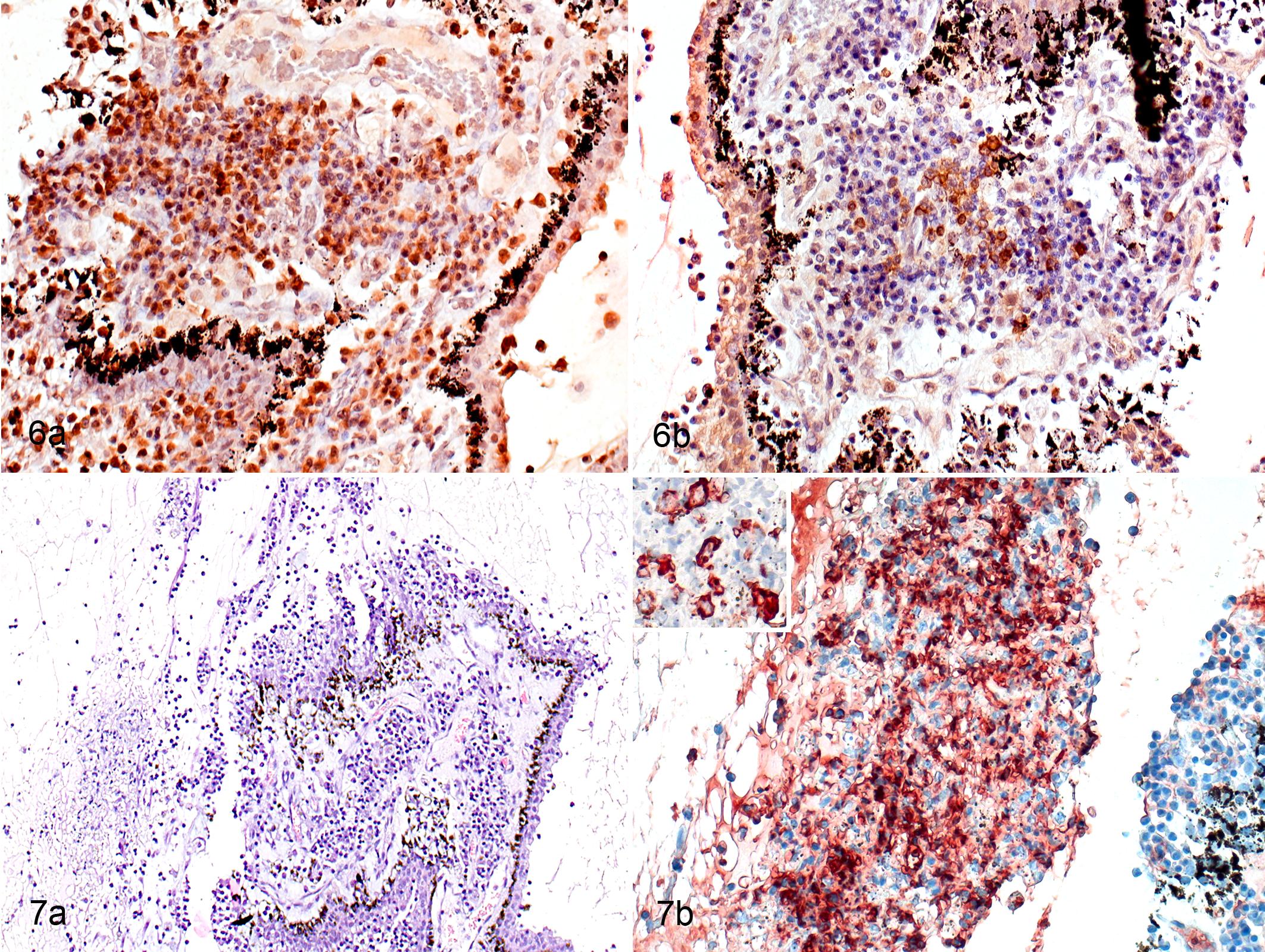
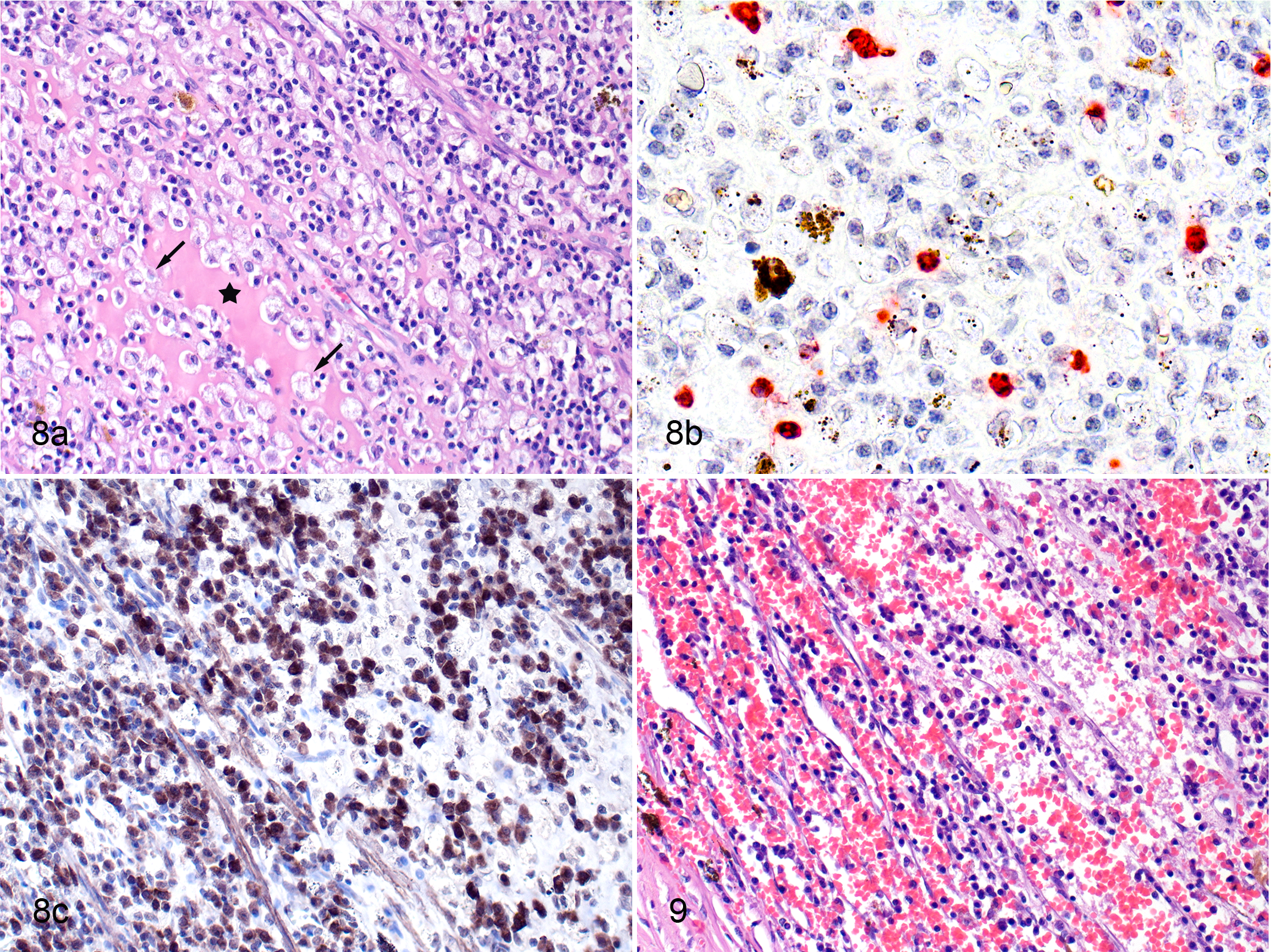
In the pars plana of the ciliary body, the 2 cats (cat Nos. 7 and 14) with granulomatous inflammation differed from those with nongranulomatous inflammation. The cats with granulomas did not have necrosis in the areas of inflammation, and the granulomas consisted of numerous B cells intermingled with foamy macrophages, some of which were calprotectin positive (Fig. 8a–c). Immunohistochemistry with consecutive sections showed that it was likely that a few calprotectin-positive (recently blood-derived) macrophages were PCNA positive (ie, proliferating). In 2 cats with massive inflammation, there was extensive extravasation into the surrounding tissue (Fig. 9). In the 13 cats with nongranulomatous inflammation, numerous B cells with fewer T cells and macrophages were present, and some of the macrophages were calprotectin positive.
The choroid contained some degree of inflammation in 14 of the 15 cats with FIP (Fig. 1). In the 8 cats with severe or massive choroiditis, the choroid had degenerative changes with vacuoles in the cellular tapetum lucidum (Fig. 10). In 5 of these 8 cases, exudate had accumulated between the retinal pigment epithelium and the sensory retina (Fig. 10) or between the choroid and the retina. In some of these cases, where the retina was detached and exudate was present, there were other abnormalities of the retinal pigment epithelium such as hypertrophy.
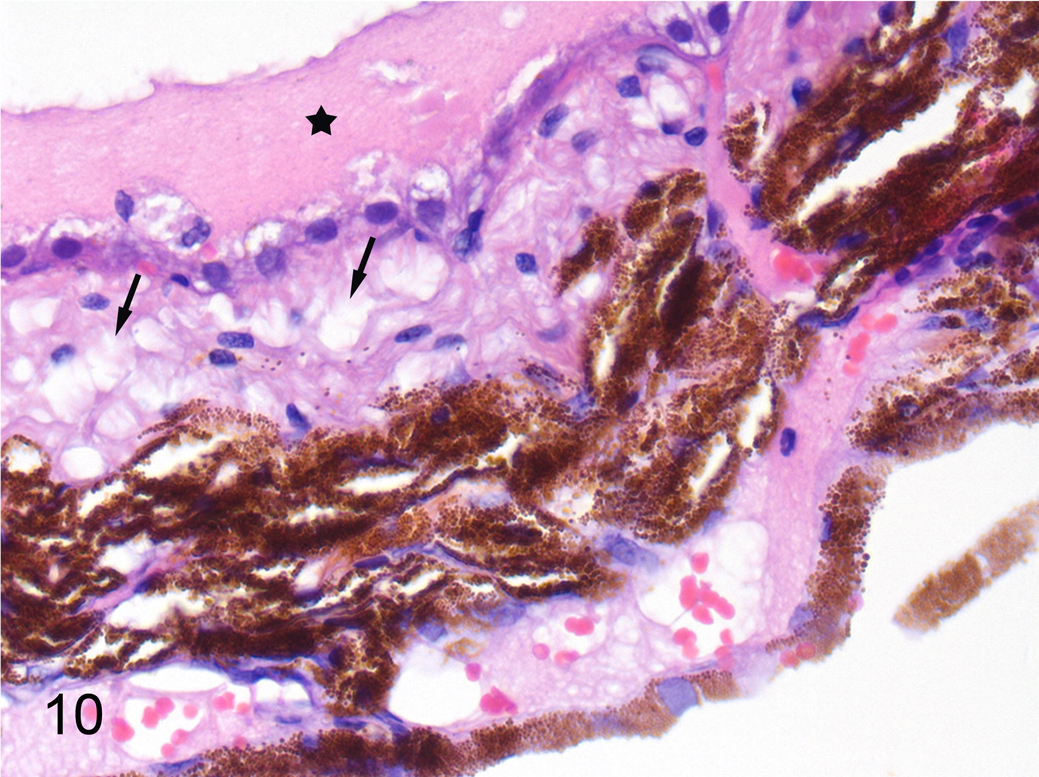
Feline infectious peritonitis, choroid, cat. Degeneration of the tapetum lucidum (arrows) and accumulation of proteinaceous fluid on the surface of the retinal pigment epithelium (star). Hematoxylin and eosin.
Retina and Optic Nerve
In most cases, inflammation of the retina in cats with FIP was scored as moderate (Fig. 1). Inflammatory cells were located in all layers of the retina including the perivascular areas of the innermost regions (Fig. 11a). The infiltrates consisted of B cells (Fig. 11b) with only occasional T cells and macrophages (mostly calprotectin positive). In 3 cats with massive inflammation of most ocular structures, the retina had degenerative changes with loss of the photoreceptor layer and destruction of the retinal architecture (Fig. 11a).
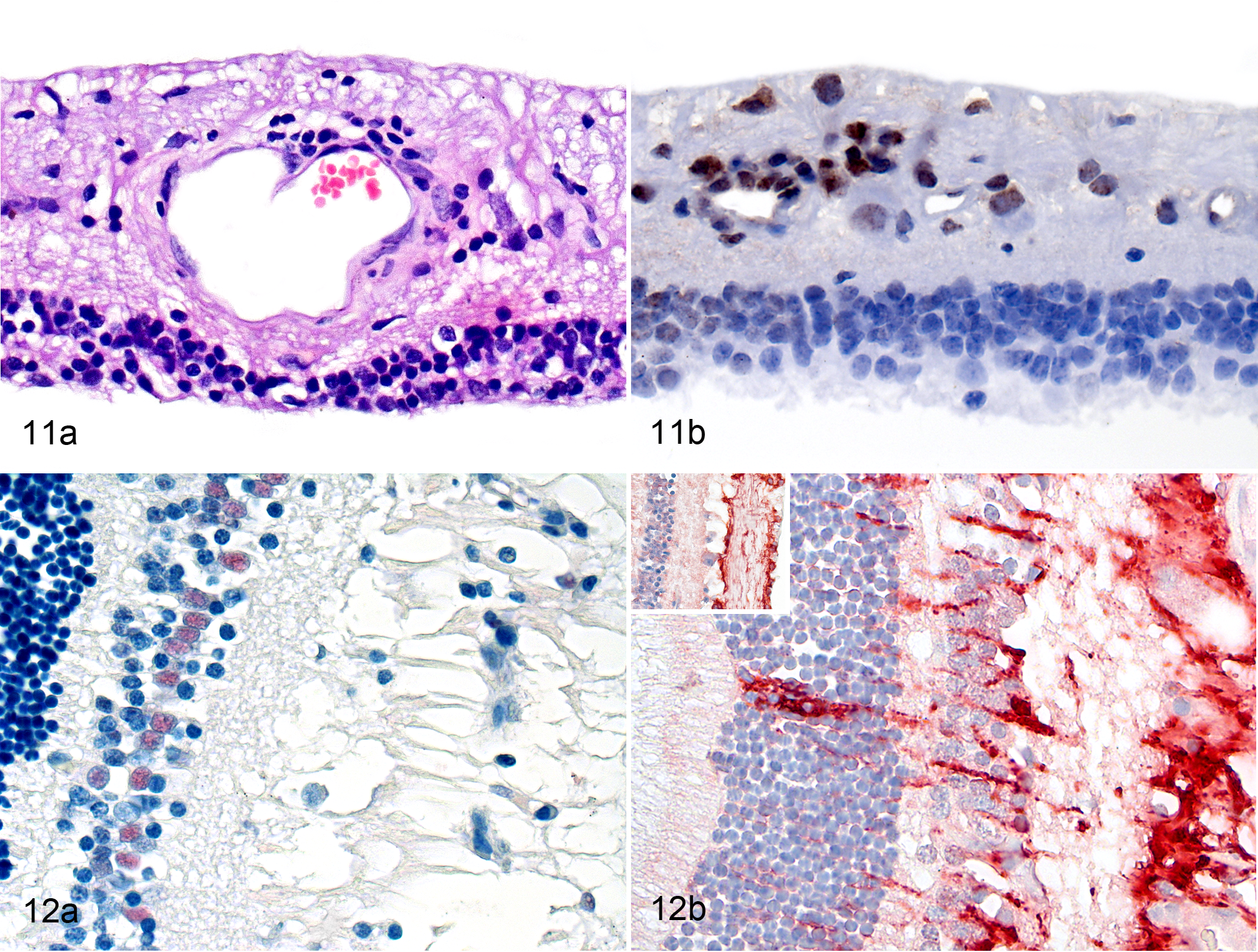
In the 4 cases where the retina was focally detached, nuclear immunolabeling for PCNA was detected in cells with oval nuclei located in the inner nuclear layer that were identified as Müller cells based on their location and shape (Fig. 12a). In all other cases of FIP and in the retinas of control cats, there were no indications of Müller cell proliferation (data not shown).
Glial fibrillary acidic protein immunoreactivity was stronger and distributed more widely in the retina of all cats with FIP compared with that in control cats (Figure 12b). Glial fibrillary acidic protein immunoreactivity was identified in the outer nuclear layer, ganglion cell layer, inner nuclear layer, outer plexiform layer, and inner plexiform layer (Fig. 12b). In 2 cases of FIP where the photoreceptor layer had been lost, GFAP immunoreactivity was also found in the side branches of Müller cells and on the outer surface of the retina.
Diffuse optic neuritis of differing intensity was observed in 9 of 15 cats with FIP (Fig. 1). Inflammatory cells had extensively infiltrated the optic nerve sheath and, to a lesser extent, the optic nerve itself (Fig. 13). Macrophages were most abundant (and many were calprotectin positive; Fig. 13), followed by B cells and single T cells. In some cases, the inflammation was accompanied by swelling of the optic nerve and thickening of the endoneurium (data not shown).
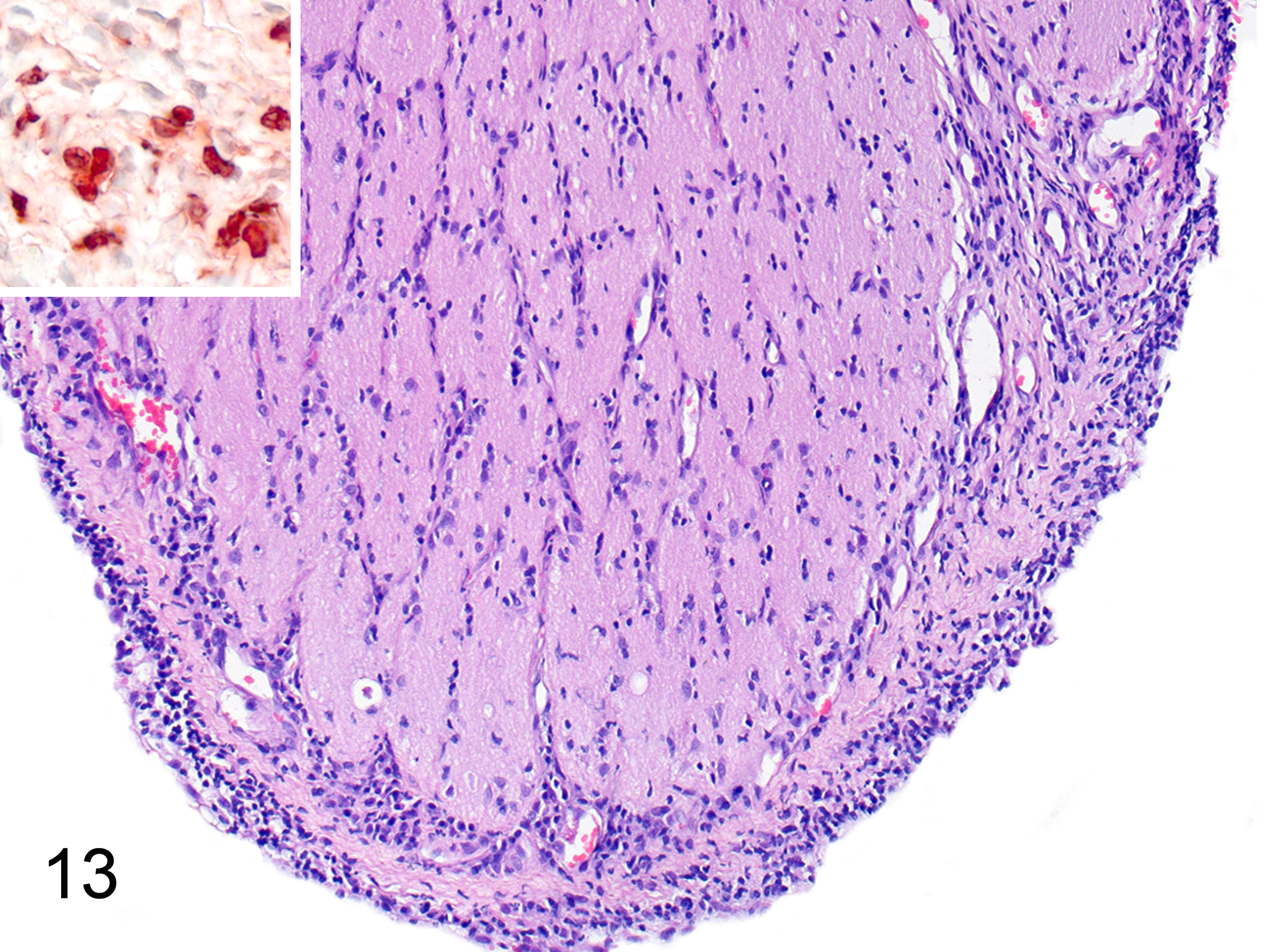
Feline infectious peritonitis, optic nerve, cat. Inflammatory cells infiltrate the optic nerve sheath and, to a lesser extent, the optic nerve itself. Hematoxylin and eosin. Inset: In the optic nerve sheath, numerous macrophages are calprotectin positive (suggesting that they were recently blood-derived). Immunohistochemistry for calprotectin.
Cornea and Sclera
In the cornea, the inflammatory cells consisted of lymphocytes, plasma cells, and macrophages. The inflammatory infiltration was found predominantly in the middle and deep layers of the corneal stroma (Fig. 14). Some of the macrophages were calprotectin positive (Fig. 14). Mild vascularization of the stroma was present in 3 cases. In 2 animals with massive inflammation of the ciliary body, extravasation with clot formation in the anterior chamber was accompanied by the presence of erythrophagocytic macrophages (Fig. 15).
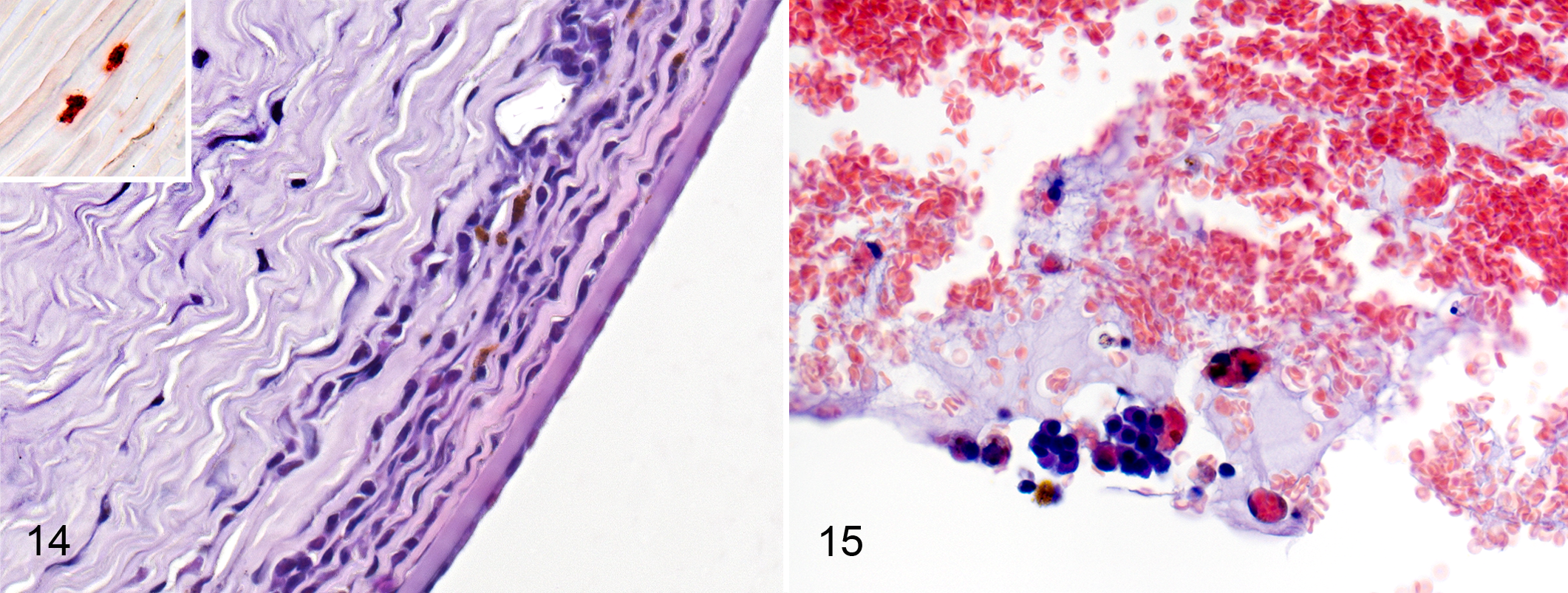
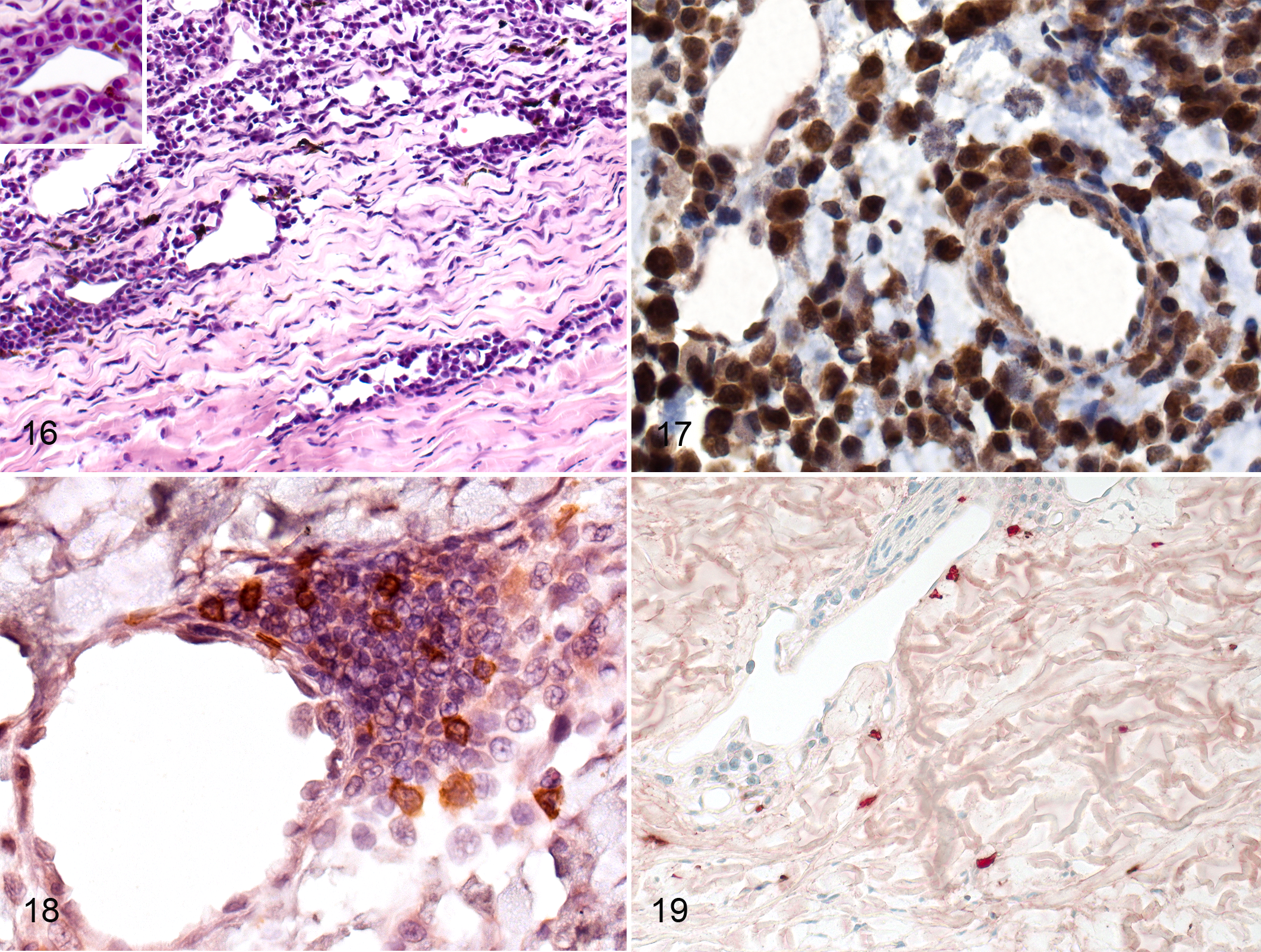
Thirteen of 15 cats with FIP had scleritis that was slight or mild in 10 cases (Fig. 1). Scleritis was mostly present adjacent to the ciliary body and was mostly localized around the scleral veins (Fig. 16).
Vascular Lesions in the Eye
Vasculitis (phlebitis) and periphlebitis were identified histologically in 15 of 15 cases and were evidenced by peri- and intravascular infiltrates accompanied by necrosis of the endothelial cells and loss of continuity of walls of the veins. Phlebitis and periphlebitis most noticeably affected scleral veins (Fig. 16) and were identified in all ocular structures (Supplemental Table S2). The composition of the cellular infiltrates in and around the vessel walls depended on the intensity of the inflammation. In mild and moderate inflammation, B cells and T cells were present in roughly equal proportions along with a few macrophages. In severe and massive inflammation, many more B cells and plasma cells were present than T cells and macrophages (Figs. 16–19), and in these cases, the infiltrates formed thick perivascular cuffs and extended into the surrounding tissue (Fig. 16). Occasionally, plasma cells with acidophilic cytoplasm were observed within the infiltrates. In all grades of inflammation, most of the macrophages were calprotectin positive (recently derived from blood monocytes; Fig. 19). Neutrophils were rare in vascular lesions, and when present, they were found mostly in lesions of the sclera and choroid. In 2 cases of massive inflammation, the ciliary body and conjunctiva had damage to the walls of veins evidenced by loss of continuity, and erythrocytes had extravasated into the surrounding tissue. Although veins were mostly affected, cellular infiltrates comprising mostly B cells were also present around small iridial arterioles in cases of severe iris inflammation (Fig. 17).
Discussion
The results of the present study indicate that GFAP expression is increased in the retina in cases of FIP. In cases with retinal detachment, Müller cells proliferate in the retina, as demonstrated by immunolabeling for PCNA. We found that macrophages were present in FIP-associated ocular lesions, but in most cases, they were not the most numerous inflammatory cells. Some of the macrophages expressed FCoV antigen as shown by immunohistochemistry, but this was found only in cases of severe or massive inflammation. The macrophages that expressed FCoV antigen appeared to be recently blood-derived. In most ocular structures, B cells and plasma cells predominated over T cells and macrophages when there was severe and massive inflammation.
The increase in GFAP expression and the proliferation of Müller cells are both likely to result from retinal injury caused by FCoV-induced ocular inflammation. In the healthy feline retina, GFAP expression is almost exclusively limited to the end foot of Müller cells and to astrocytes located in the innermost layer of the retina. 17 In contrast, in the cats with FIP, we observed GFAP expression in almost all retinal layers. This finding is consistent with the increase in GFAP expression in the feline retina in various other retinal disorders. 11,16,17,19,33 In all of these disorders, Müller cells react by increasing GFAP expression consistent with hypertrophy of these cells, and Müller cells also proliferate in some of these disorders. 16,17,19 Glial fibrillary acidic protein upregulation is a nonspecific marker of Müller cell gliosis, which is one of the retina’s responses to pathogenic (infectious) factors. 10 Other components of this response include microglial activation and, as observed in our study, the breakdown of the blood–retinal barrier and the migration of macrophages and lymphocytes into the retinal tissue. 10
The proliferation of Müller cells in the retina of the cats with FIP was probably induced by the detachment of this structure, which was likely a result of exudate accumulating in the subretinal space. Retinal detachment results in a decreased oxygen and nutrient supply of the photoreceptor cells and activated Müller cells, which begin to proliferate within 1 day of detachment. 10 In many species, detachment of the neural retina is associated with hypertrophy of the retinal pigment epithelium, 10 which was found in some of the cats with FIP in this and other studies. 5
Our results suggest that macrophages are usually not the predominant inflammatory cell in FIP-associated ocular lesions, unlike what is thought about other organs with FIP lesions. 7 The lower number of macrophages in the eye may be due, at least in part, to the fact that this organ is immune privileged. 40
In the 2 cats where macrophages predominated in ocular lesions, the walls of the blood vessels in the eye had been damaged based on necrosis of the endothelium and loss of continuity. This vascular damage could have disrupted the blood–ocular barrier and made it easier for macrophages to enter the structures of the eye. This supposition is supported by the fact that, in these cats, we found damage to both layers of the ciliary epithelium and to the blood vessel walls in the ciliary processes, as well as lesions in the retinal pigment epithelium and the choroid. In these 2 cats, more of the macrophages appeared to be recently blood-derived (based on expression of calprotectin) than in the other cats.
This damage to the walls of the blood vessels was likely a consequence of vasculitis. A number of our observations regarding FIP-associated vasculitis confirm observations made in previous studies, whereas other observations are reported here for the first time. First, we observed that vascular inflammation mainly caused periphlebitis and that most cats had lymphoplasmacytic periphlebitis. 3,8,27,34,36 A novel finding was that periphlebitis was most common in the sclera and most intense above the ciliary body. Although we observed granulomas in the ciliary body, we did not find granulomatous and pyogranulomatous vasculitis in the eyes of cats with FIP, unlike some previous reports. 5,15 In other organs in cats with FIP, granulomatous to necrotizing vasculitis and lymphoplasmacytic vasculitis (phlebitis and periphlebitis) have been reported. 7,22,25,27,34,44
Second, we found that in cases of massive ocular inflammation, perivascular infiltrates were composed mostly of B cells and plasma cells (as shown by positive staining for CD79 antigen), which had not previously been observed in the ocular tissues of cats with FIP. This finding demonstrates that, in cats with this disease, perivasculitis in the eyes is similar to perivasculitis in the leptomeninges and mesentery. 25,27
Third, in some cases of massive ocular inflammation, we found periarteritis that was confined to the iridal arterioles. This finding contrasts with that of a previous study, 27 in which the arteries in various organs were not affected in FIP-associated vasculitis. The infiltrates observed around the iridal arteries in the present study may represent a spillover from periphlebitis and from focal infiltrates present in the iris tissue.
Finally, we did not find a positive reaction to FCoV antigens in the macrophages that were sparsely present in FIP-associated vasculitis. This is in contrast to previous findings, 25,27 where FCoV antigens were detected in macrophages located around choroidal blood vessels and in FIP-associated granulomatous vasculitis in other organs. The absence of FCoV-positive macrophages in the present study may simply mean that the number of macrophages in the lesions was low. The internalization, disassembly, and replication of the virus is limited to a very small proportion of monocytes/macrophages. 13,30 Furthermore, the point in the time course of the inflammatory process at which the eyes were examined may also influence the number of FCoV-positive macrophages that were found.
In our study, 10 cats with FIP had additional signs of increased permeability of the blood–ocular barrier. Six of them had 1 or more of the following clinical signs before euthanasia: hyphema in the anterior chamber, fibrin exudate in the anterior chamber, keratic precipitates on corneal endothelium, or iris rubeosis. When examined histologically, all of these cats and 4 others had accumulation of proteinaceous exudate in the ciliary processes, iris stroma, and subretinal space. Damage to the ciliary processes—the structures responsible for aqueous humor production—may explain the low intraocular pressure that is commonly observed in uveitis including FIP. 3,4,12 The notable accumulation of exudate even in cases of slight or moderate inflammation could be partially explained by the fact that, even in healthy cats and especially in kittens, the blood vessels of the iris and choroid are more permeable to circulating proteins than they are in other species, to the extent that molecules even larger than serum proteins can pass through the vessel wall. 6 However, the extent to which exudate had accumulated makes it seem likely that the permeability of blood vessels in the eyes, which is already higher in cats than in other species, may be further increased in cases of FIP-associated vasculitis.
With regard to inflammation of other ocular structures such as the iris and the ciliary body, we found FCoV antigen in these structures in only 3 of the 15 cats with FIP; each of these 3 cats had severe or massive ocular inflammation. We detected a strong positive reaction to FCoV antigen in macrophages within granulomas of the ciliary body and in a few macrophages present in cellular infiltrates in the iris. Staining on the consecutive sections of the same eye revealed that these macrophages were recently blood-derived based on calprotectin immunolabeling. In previous reports, FCoV antigen was detected in the conjunctiva 23,25 and in macrophages around choroidal blood vessels. 25 Our findings in the eyes of cats with FIP are similar to those in other tissues in this disease. In the connective tissue of the third eyelid, macrophages bearing FCoV antigen were detected in 9 of 108 cats. 41 In other organs, FCoV antigen was detected in variable amounts and mostly in macrophages associated with granulomas and pyogranulomas. 4,7,25 There are 2 likely explanations for the low numbers of FCoV-positive macrophages in our study. First, as mentioned above, the virus is limited to a very small proportion of monocytes/macrophages, and additionally, these cells can have difficulty accessing the eye due to the presence of immunosuppressive biochemical mechanisms. 13,28,42 Second, there is evidence for antigenic heterogeneity of the coronavirus nucleocapsid protein in FIP lesions. 37 Thus, some FCoV may have been present that our immunostaining could not detect. 37
Our observation that there was occasionally positive staining for PCNA in the nuclei of macrophages present in the aqueous humor of cats with massive ocular inflammation suggests that macrophages may proliferate in the eyes of cats with FIP. Some of these potentially proliferating macrophages may also be infected with FCoV because we found that, in consecutive sections from the same eyes, some of the PCNA-positive macrophages were also positive for FCoV antigens. Previous studies have reported that FCoV-infected, activated monocytes/macrophages might induce proliferation of myelomonocytic cells and macrophages by releasing cytokines such as interleukin 1β and tumor necrosis factor. 1,26
In the 2 cats with the granulomas in the ciliary body, the morphology of these lesions was similar to that of FIP-associated lesions in other organs, in that they contained numerous macrophages intermingled with B cells. 7,25,34 The granulomas in our study may be the result of a chronic inflammatory process, because only some of the macrophages were calprotectin positive and therefore recently blood-derived. 38 Other studies have shown that the B cells surrounding macrophages in FIP-associated granulomas express coronavirus-specific immunoglobulin on their cell surface, which indicates that they constitute a feline infectious peritonitis virus–specific immune response. 25,28 It is likely that B cells gradually replace the macrophages in the granulomas, limiting disease progression to some extent. 28 Similar granulomas have been reported not only in the eyes of cats with FIP but also in the eyes of cats with other ocular diseases, such as fungal, bacterial, and parasitic infections. 8,9,14,15,25,27,31 ,36
There are several possible explanations for our findings that both the intensity of inflammation and the types of lesions (granulomatous, lymphoplasmacytic) in the eyes varied from cat to cat and also from structure to structure within the same eye. First, the various types of lesions within a single eye could reflect different stages in the process of inflammation. It is worth noting that although many cats had slight inflammation in the conjunctiva (mean score = 1.38), they were not tested for herpes virus, so we cannot exclude this pathogen as the cause of the conjunctivitis. Second, the differences between cats may be due to the heterogeneity of cellular immune responses in different cats. 25,28 Our finding that neutrophils were rarely present in eye lesions is in agreement with reports of other organs. 5,7,25,41,45 Although some previous reports on the eyes of cats with FIP did find a greater abundance of neutrophils, the low number of neutrophils in our study can be explained simply by rarely observed tissue necrosis in our samples or by the fact that tissues were sampled after the abundance of these cells had peaked.
The predominance of B cells and plasma cells in the infiltrates in cases of severe and massive inflammation may indicate that the humoral immune response plays an important role in ocular inflammation in cats with FIP. For many years, there was a hypothesis that cats with strong cell-mediated immune response would clear the virus and prevent FIP, whereas those with a predominantly humoral response would develop wet FIP. This seems consistent with our findings of many B cells and plasma cells in severe forms of FIP, if the antibody response is nonprotective or exacerbated the disease. 28,35 On the other hand, a few reports have indicated that the humoral immune response can sometimes limit the progression of the disease to some extent and for a limited time. 28 This could be the reason that ocular signs manifest earlier in some cases of FIP than in others. 3
In the present study, T cells constituted a minority of the inflammatory cells in the cats. This result is in agreement with previous observations made in other organs with FIP lesions. 7,25 T cells may have been in the minority in fatal cases of FIP because they are important in protection against the virus, and cats with a strong cell-mediated immune response might not have been included in this study. In cases of FIP, T cells are depleted in lymphoid organs due to apoptosis. 20
In summary, this study has shown that in cases of FCoV-induced retinitis, expression of GFAP in Müller cells is increased, and in cases of retinal detachment, these cells also proliferate. Both reactions are evidence of gliosis. Unlike in other organs with FIP lesions, macrophages are usually not the most numerous cells in inflammatory infiltrates in the eye. However, macrophages increase in number in cases of severe and massive inflammation, when damage to blood vessel walls occurs. Only some of these macrophages, mostly those recently derived from blood, express FCoV antigens. The predominance of B cells and plasma cells over other inflammatory cells in the ocular lesions of many examined cats suggests that the humoral immune response plays a key role in ocular inflammation in cats with fatal FIP.
Footnotes
Acknowledgements
We would like to thank Dr Monika Leśnik-Strzeżek for providing us with the clinical material, Prof Aneta Andronowska for providing us with antibodies against calprotectin, Mark Leonard for help with graphics and for editorial support, and Jacek Sztorc for his technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by Research Grant No. 15.660.106 from KNOW (Leading National Research Centre) and the decision of Ministry of Science and Higher Education No. 05-1/KNOW2/2015.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
