Abstract
Feline infectious peritonitis (FIP) is a fatal, Coronavirus (CoV)-induced systemic disease in cats, characterized by granulomas in organs and granulomatous vasculitis. This study describes the morphologic features of granulomatous vasculitis in FIP as well as its development in the course of monocyte-associated feline CoV (FCoV) viremia in five naturally infected Domestic Shorthair cats with FIP. Monocyte-associated FCoV viremia was demonstrated by immunohistology, RNA in situ hybridization, and electron micropscopy. Granulomatous phlebitis at different stages of development was observed. Vasculitic processes ranged from attachment and emigration of FCoV-infected monocytes to vascular/perivascular granulomatous infiltrates with destruction of the vascular basal lamina. Monocytes as well as perivascular macrophages were activated because they were strongly positive for CD18 and expressed cytokines (tumor necrosis factor-α and interleukin-1β) and matrix metalloproteinase-9. In addition, general activation of endothelial cells, represented by major histocompatibility complex II upregulation, was observed in all cases. These results confirm FIP as a monocyte-triggered systemic disease and demonstrate the central role of activated monocytes in FIP vasculitis.
Keywords
Feline infectious peritonitis (FIP) is a well-known and widely distributed coronavirus (CoV)-induced systemic disease in cats, characterized by fibrinous to granulomatous serositis with protein-rich effusions in body cavities and granulomatous inflammatory lesions in several organs. 24, 45 Not the most obvious finding, but one of its morphologic hallmarks is a granulomatous to necrotizing phlebitis and periphlebitis. 8, 16, 24, 43 Up till now, the pathogenesis of FIP is largely undetermined.
Feline CoV (FCoV) is transmitted via the fecal-oral route and primarily infects enterocytes. 28 Despite the generally high prevalence of FCoV infection among the cat population, especially in multicat facilities, FIP morbidity is low and rarely surpasses 5%. 1, 7, 28 FIP seems to develop in the individual infected animal when FCoV acquires virulence by deletions in open reading frames 3 and 7, coding for nonstructural proteins of unknown function, which occur as mutations primarily during replication in the infected host. 39 Most infected cats develop an FCoV viremia that can be detected by reverse transcriptase-polymerase chain reaction (RT-PCR) on isolated (and cultivated) monocytes regardless of the development of FIP. 7, 15 FCoV-infected circulating monocytes are considered as responsible for viral dissemination within the host. 10, 11, 28, 44 Furthermore, the demonstration of FCoV antigen within intravascular leukocytes and among cells in the lesions of vasculitis in a previous experimental study suggested that the infected monocyte mediates the development of FIP lesions. 44 The degree of macrophage infection and viral replication within infected cells in vitro, however, is associated with the virulence of FCoV strains. 35 Therefore, it seems likely that the development of FIP is related to the extent of monocyte infection.
On the other hand, the development of vasculitis and granulomas in FIP was previously attributed to a type-III hypersensitivity reaction. 10, 29 This hypothesis was based on immunofluorescence studies that showed cell-free fibrinogen and C3 as well as FCoV antigen, immunoglobulin (Ig)G, and complement within mono-nuclear cells in inflammatory lesions. 29, 43, 45 The demonstration of circulating immune complexes, high γ-globulin and C3 serum levels, and C3 deposition in glomerular mesangia have been interpreted as providing further support. 10 However, neither the morphologic features of vasculitis in FIP nor its development has been thoroughly assessed and compared with unequivocal type-III hypersensitivity vasculitis. 4, 12, 13
This study was performed to further characterize the vasculitis in FIP regarding its pathogenic mechanisms and development. Furthermore, it aimed to assess the role of circulating FCoV-infected monocytes in the disease.
Materials and Methods
Animals and tissues
The study was performed on five cats in which immunohistology for FCoV antigen and routine histology had identified FCoV antigen-positive intravascular monocytes as well as multifocal granulomatous vasculitis. 16 These animals were part of a study on 174 routinely necropsied cats with FIP, which aimed at the demonstration of circulating, FCoV antigen-positive monocytes and FIP-associated histologic lesions. Cat Nos. 1–4 originated from a group of 40 specific pathogen-free (SPF) cats, which were housed with clinically healthy cats from animal shelters. 15 Animals died 48 (cat No. 1), 67 (cat No. 2), 76 (cat No. 3), and 107 (cat No. 4) days after first being housed with FCoV-infected cats at the age of 21 weeks. The remaining cat (cat No. 5) was aged 2 years, lived in an animal shelter, and was housed together with other FCoV-infected cats from different sources, all of which had been tested positive for circulating FCoV-specific immune complexes. 15
All animals were subjected to necropsy. Samples from parenchymal organs (heart, lung, liver, spleen, pancreas, kidneys, brain, eyes) and from tissues with gross granulomatous lesions were fixed in 10% buffered formalin for 16–18 hours and subsequently embedded in paraffin wax. For comparative examinations, lungs, liver, small intestine, pancreas, mesentery, kidney, and pancreas from nine SPF cats aged 38 weeks and five SPF cats aged 72 weeks were treated accordingly. For light microscopy, paraffin-embedded tissue sections (5 μm) were either stained with hematoxylin and eosin (HE) or used for immunohistologic examinations and RNA in situ hybridization (RNA-ISH).
From cat Nos. 1–4, additional tissue specimens were fixed in 2.5% glutaraldehyde in cacodylate buffer, routinely dehydrated in ethanol, and embedded in epoxy resin. Ultrathin sections (5 nm) were stained with uranyl acetate and lead citrate.
Immunohistology
Antibodies and detection methods as well as antibody sources are listed in Table 1. In cats with monocyte-associated FCoV viremia and granulomatous vasculitis, the following antigens were demonstrated: FCoV, CD3 of T cells, CD45R of B cells, myeloid/histiocyte antigen of monocytes/macrophages and neutrophilic leukocytes, CD18, major histocompatibility complex (MHC) II, matrix metalloproteinase (MMP)-9, collagen IV, interleukin (IL)-1β, and tumor necrosis factor (TNF-α). 16–18 In SPF cats, immunohistologic examinations were restricted to the demonstration of MHC II, collagen IV, IL-1β, and TNF-α.
Immunohistological methods and antibodies.
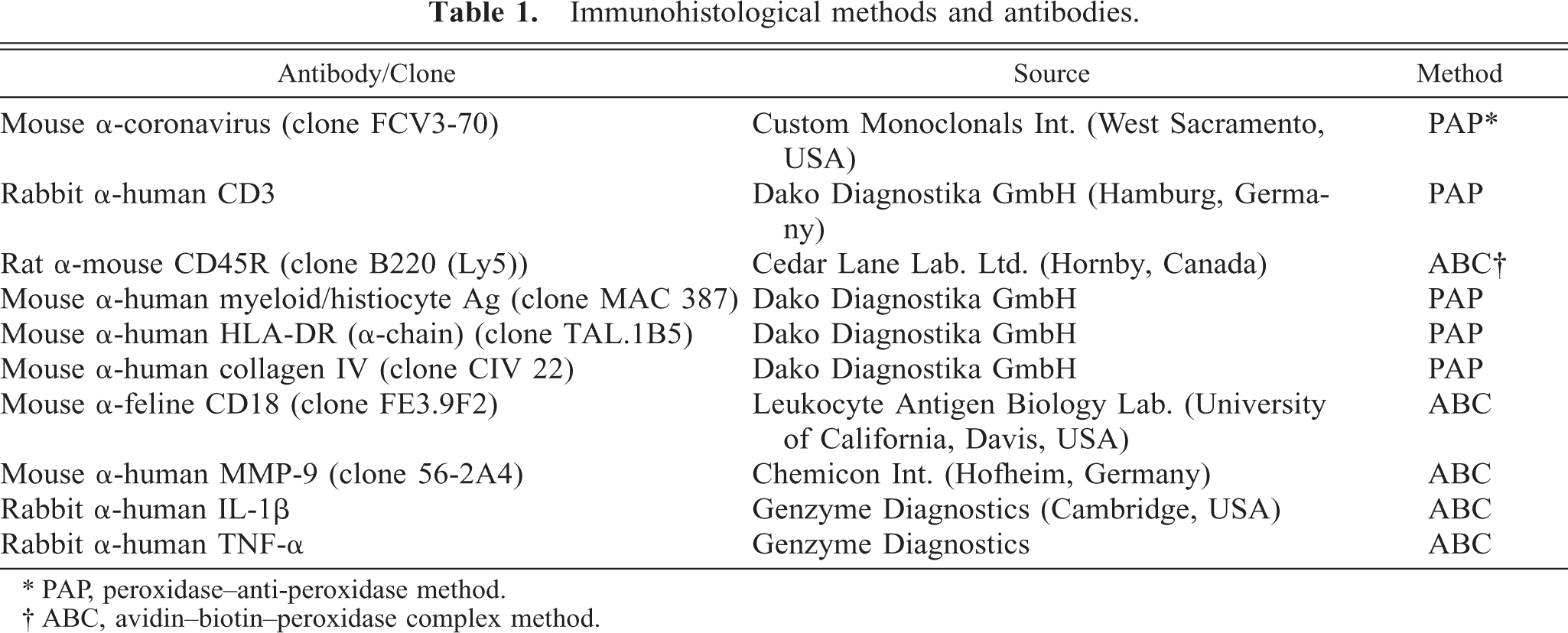
∗ PAP, peroxidase–anti-peroxidase method.
† ABC, avidin–biotin–peroxidase complex method.
Consecutive sections were used as negative controls where a nonreacting mouse monoclonal antibody against chicken lymphocytes and normal rabbit or rat serum replaced the primary antibodies. 16 Feline lymphatic tissues served as positive controls for leukocyte markers, MHC class II and CD18; a case of cutaneous mycobacteriosis was used as positive control for IL-1β and TNF-α; and FIP lesions tested positive for FCoV antigen served as positive controls for the demonstration of FCoV antigen. 16, 18
RNA-ISH for FCoV
RNA-ISH was performed, using an RT-PCR system for the detection of a 234-bp fragment of the FCoV N protein gene (NCBI accession No. A22378). 19 Primers were as follows: FIPVNa (upper), 5′-AAC GCG TCA ACT GGG GAGAT-3′; FIPVNb (lower), 5′-TAC GCT GGC CTT TTA CAATAC GA-3′. After ligation into a plasmid vector (TOPO TA Cloning® Kit pCR® II-TOPO®; Invitrogen BV, Groningen, NL), in vitro transcription of a digoxigenin-labeled riboprobe was performed, using an SP6/T7 polymerase system (DIG RNA Labeling Kit; Roche Diagnostics GmbH, Mannheim, Germany) according to the manufacturer's instructions.
The protocol follows a previously published report for RNA-ISH. 26 Slides were deparaffinized and digested in proteinase K (1 μg/ml) at 37° C for 15 minutes, postfixed, acetylated, and prehybridized. Hybridization was performed at 52° C for 15–18 hours with 3 μl RNA/100 μl hybridization mix. After posthybridization washing, staining was performed with alkaline phosphatase-coupled anti-digoxigenin antibody (anti-digoxigenin-AP FAb fragments; Roche Diagnostics GmbH) and nitroblue tetrazoliumchloride (NBT)/5-bromo-4-chloro-3-indolylphosphate (BCIP) (Sigma-Al-drich Chemie GmbH, Steinheim, Germany). Slides were counterstained for 10 seconds with Papanicolaou's hematoxylin (1 in 20 in distilled water; Merck Eurolab GmbH, Darmstadt, Germany).
Results
Macroscopical findings
At necropsy, cat No. 1 exhibited a fibrinonecrotizing esophagitis and epiglottitis, cat No. 2 showed a suppurative pneumonia, and cat Nos. 3, 4, and 5 exhibited granulomatous lesions in several organs, consistent with FIP (Table 2).
Distribution of vasculitis and granulomatous lesions in organs of cats examined in this study.
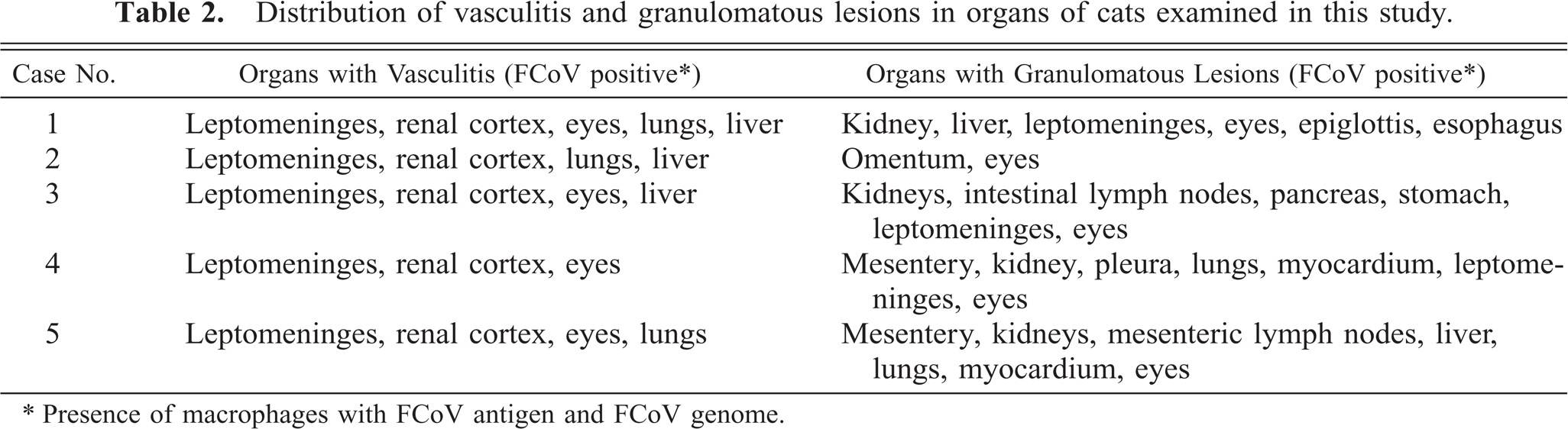
∗ Presence of macrophages with FCoV antigen and FCoV genome.
Demonstration of monocyte-associated FCoV viremia
FCoV infection of circulating monocytes was confirmed by immunohistology for FCoV antigen, RNA-ISH for FCoV genome, and ultrastructural demonstration of CoV particles within cytoplasmic vesicular structures (the so-called budding compartment) in monocytes (Figs. 1–3). 19 FCoV-positive monocytes were predominantly seen within the lumen of leptomeningeal, renal cortical, and pulmonary vessels (cat Nos. 1–3 and 5). In cat No. 4, FCoV-positive monocytes were only demonstrated within vessels in the brain parenchyma.
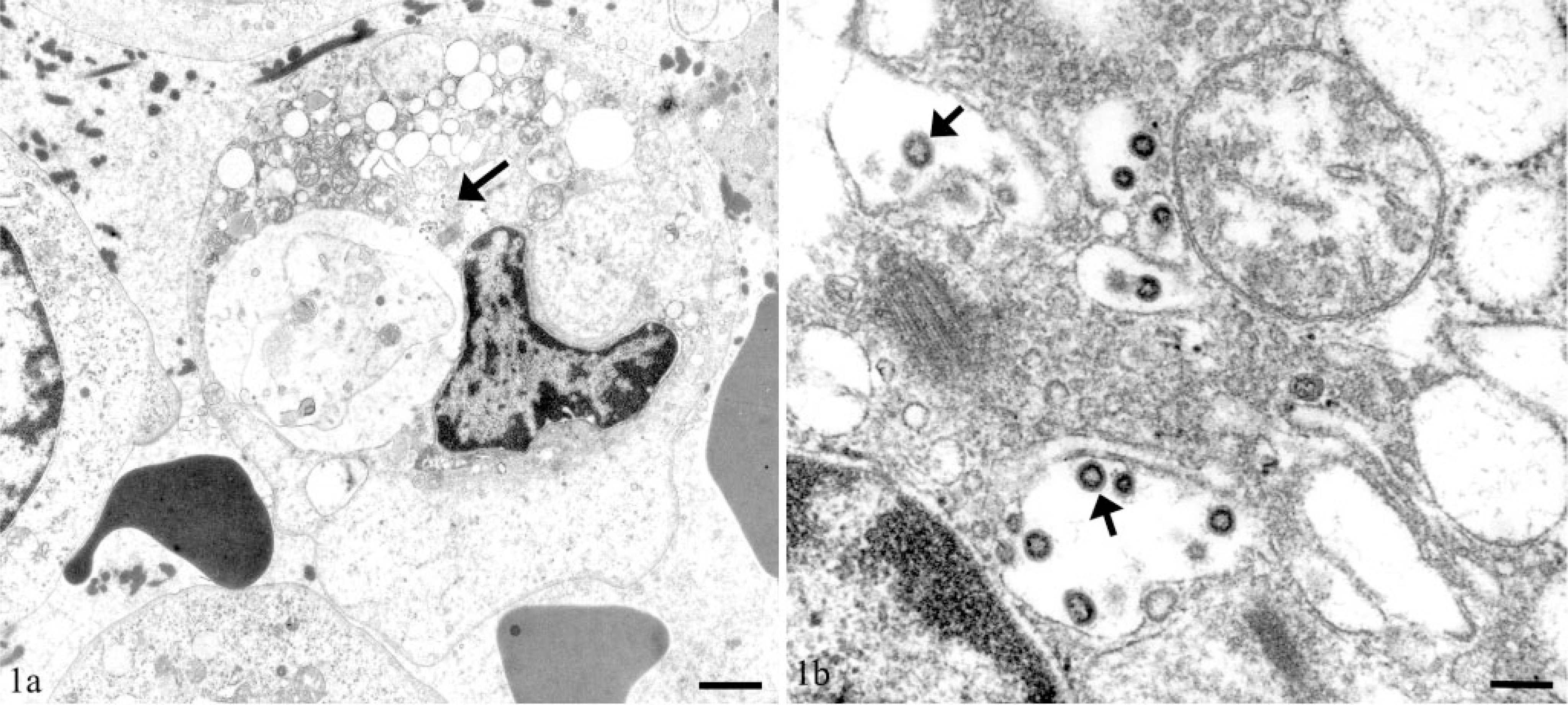
Vein; cat No. 2. Demonstration of viral particles within cytoplasmic vesicles (arrows) of monocytes. Transmission electron micrographs.
Vein, leptomeninx; cat No. 2. Monocyte-associated viremia. Demonstration of FCoV antigen in monocytes attached to endothelial cells (arrows). Peroxidase-anti-peroxidase method, Papanicolaou's hematoxylin counterstain. Bar = 30 μm.
Numerous small pulmonary veins in cat No. 1 and a few small veins in the brain parenchyma in cat No. 4 were completely obliterated by an agglomerate of myeloid/histiocyte antigen-positive monocytes among which numerous cells were FCoV positive (Fig. 4).
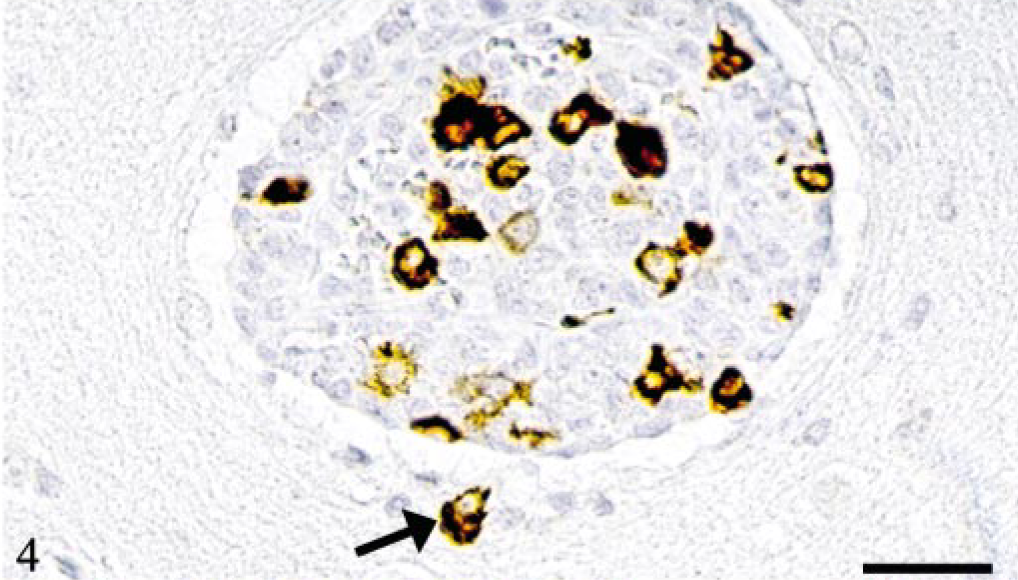
Brain parenchyma; cat No. 1. Occlusion of vein by monocytes, several of which are positive for FCoV antigen. Demonstration of an emigrated, FCoV-infected monocyte (arrow). Peroxidase-anti-peroxidase method, Papanicolaou's hematoxylin counterstain. Bar = 30 μm.
In addition, scattered FCoV-infected macrophages were seen in splenic red pulp of cat Nos. 1–4 and in the lymph node sinuses of cat No. 1. Electron microscopy in cat No. 2 identified CoV particles in a few sinus macrophages in mesenteric lymph nodes.
Characterization of inflammatory cells in vasculitis
Inflammation was restricted to small and medium-sized veins, and veins in leptomeninges and renal cortices were affected in all the cats. In addition, ocular (four of five cats), pulmonary (three of five cats), and hepatic (three of five cats) veins were affected (Table 2). In the kidney, only veins in the outer cortex (Venae capsulares) exhibited infiltrates. In the eyes, venules in iris, chorioidea, or retina (or all) (each three of five cats), around the optic nerve (two of five cats), and in the sclera (one of five cats) were involved.
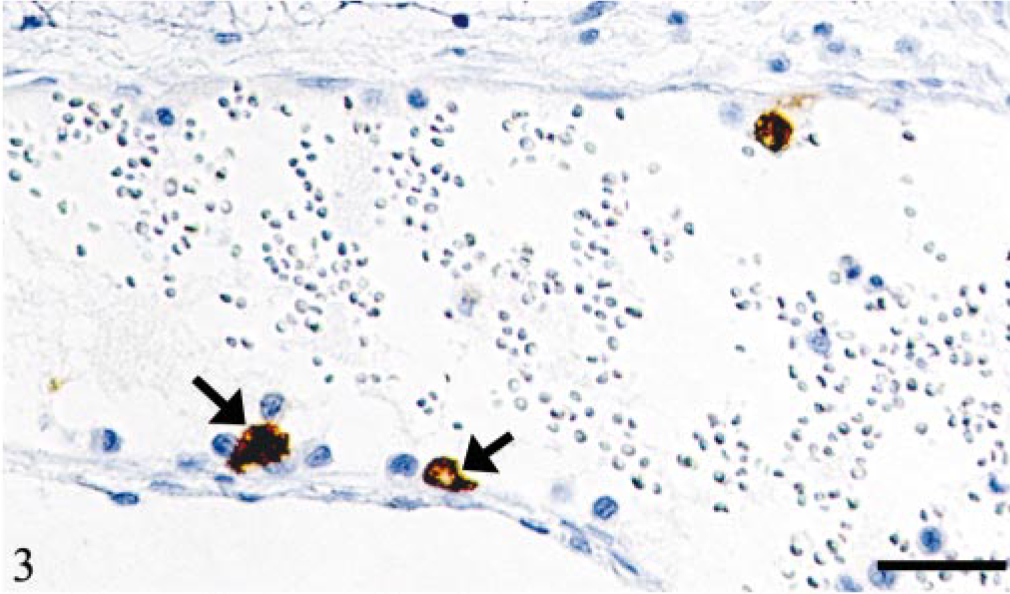
Inflammatory lesions were dominated by monocytes/macrophages intermingled with a few neutrophils and lymphocytes. Lesions were represented by monocytes, attached to endothelial cells (EC) or emigrating from the vessel, by a circular or-in larger veins-focal infiltration of the vascular wall or by perivenous cellular agglomerates (or a combination of all). Occasionally, inflammatory cells completely replaced the walls of affected veins. Among infiltrating cells, a substantial proportion of monocytes/macrophages was FCoV positive (Figs. 2–6). Especially in cat No. 1, FCoV-infected monocytes formed intravascular agglomerates, which were focally attached to the endothelium (Fig. 5); cross sections of larger veins also depicted several agglomerates in the same vessel.
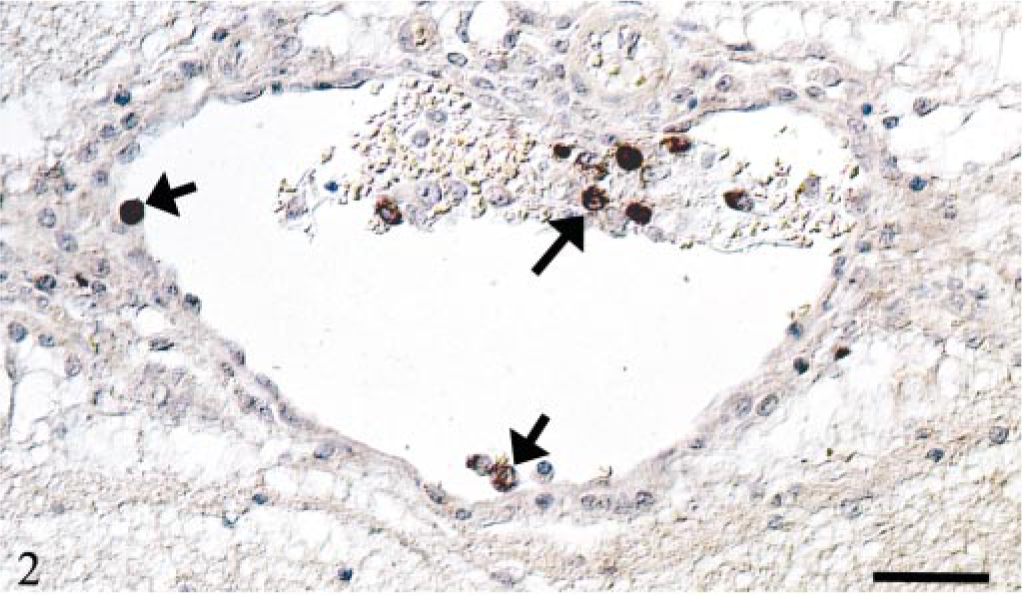
Vein, brain parenchyma; cat No. 1. Monocyte-associated viremia. Demonstration of FCoV genome in monocytes in the vascular lumen and attached to endothelial cells (arrows). RNA-ISH, digoxigenin-labeled probe, NBT/BCIP, Papanicolaou's hematoxylin counterstain. Bar = 40 μm.
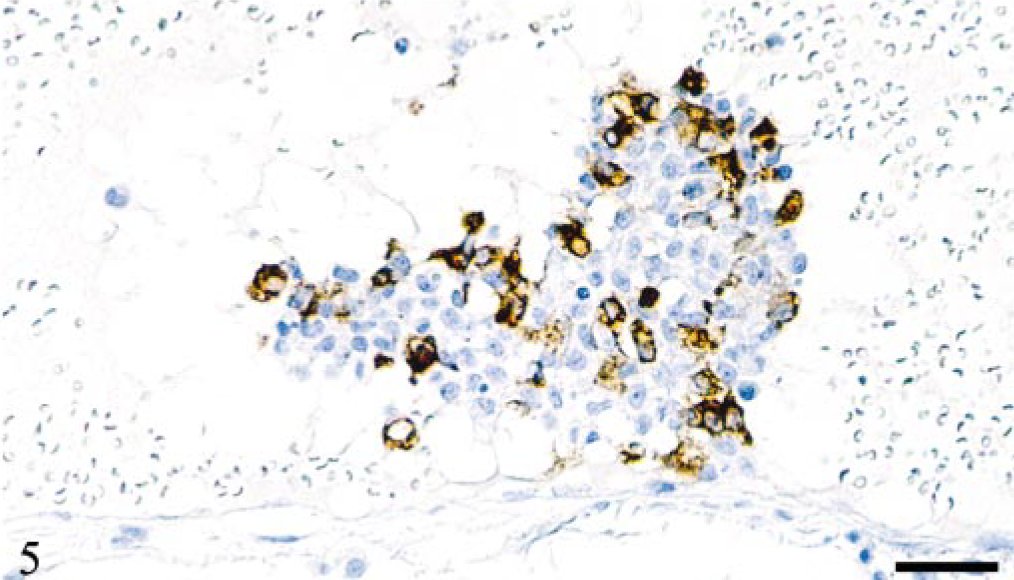
Leptomeninx; cat No. 1. Agglomerate of partly FCoV-infected monocytes, which is focally attached to the endothelium and protrudes into the vascular lumen. Peroxidase-anti-peroxidase method, Papanicolaou's hematoxylin counterstain. Bar = 30 μm.
Kidney; cat No. 3. Renal cortical vein with focal perivenous granulomatous infiltration. Numerous monocytes/macrophages express FCoV antigen. Peroxidase-anti-peroxidase method, Papanicolaou's hematoxylin counterstain. Bar = 40 μm.
Both intravascular monocytes as well as perivascular macrophages were strongly positive for the myeloid/histiocyte antigen (Fig. 7). The renal veins of cat No. 5 and a few leptomeningeal veins of cat Nos. 1 and 3, which all exhibited relatively intense circular perivascular infiltrates, were occasionally surrounded by a peripheral rim of B cells, whereas T cells were disseminated among the other inflammatory cells in low numbers. In cat No. 4, this feature was most obvious because some vessels in the brain parenchyma not only exhibited a broad perivascular rim of B cells (Fig. 8a) but were also completely occluded by myeloid/histiocyte antigen-positive monocytes among which numerous cells were FCoV positive (Fig. 8b).
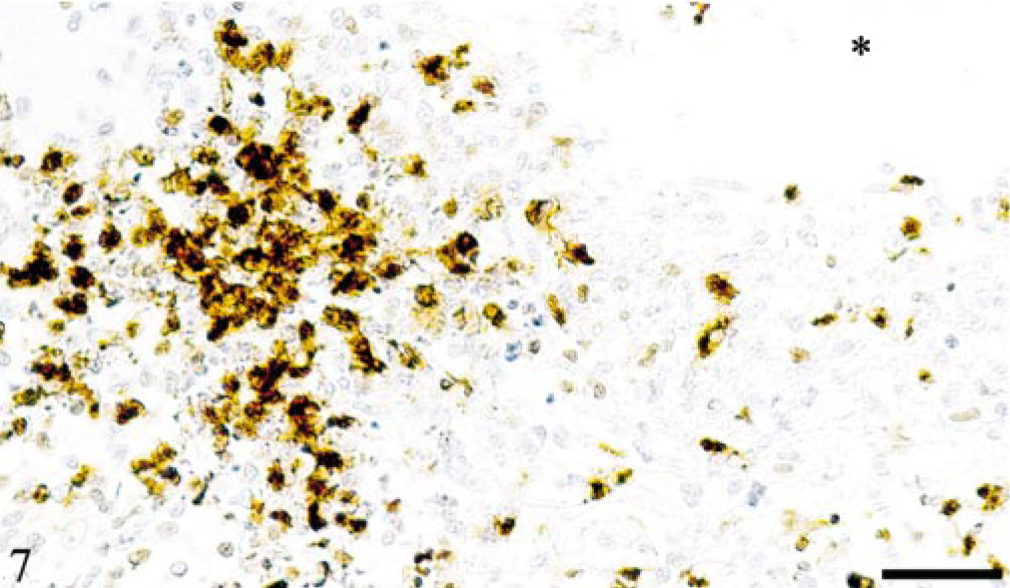
Kidney; cat No. 3. Renal cortical vein (∗) with focal perivenous granulomatous infiltration. The majority of cells are myeloid/histiocyte antigen positive, typical of recently blood-derived macrophages. Peroxidase-anti-peroxidase method, Papanicolaou's hematoxylin counterstain. Bar = 80 μm.
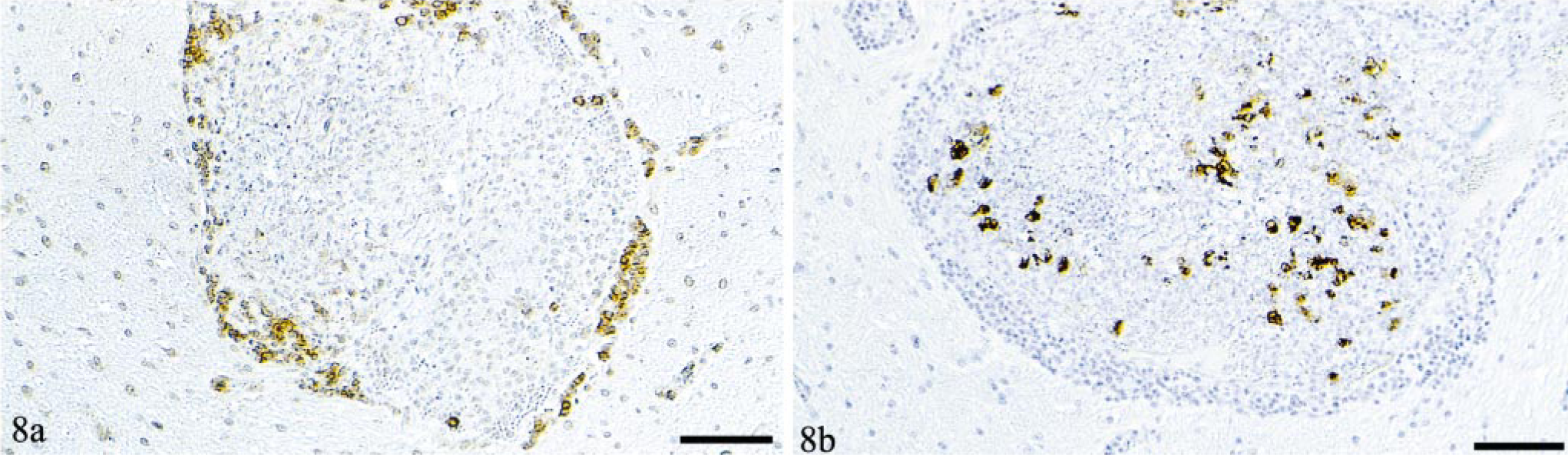
Brain parenchyma; cat No. 4. Completely occluded vein.
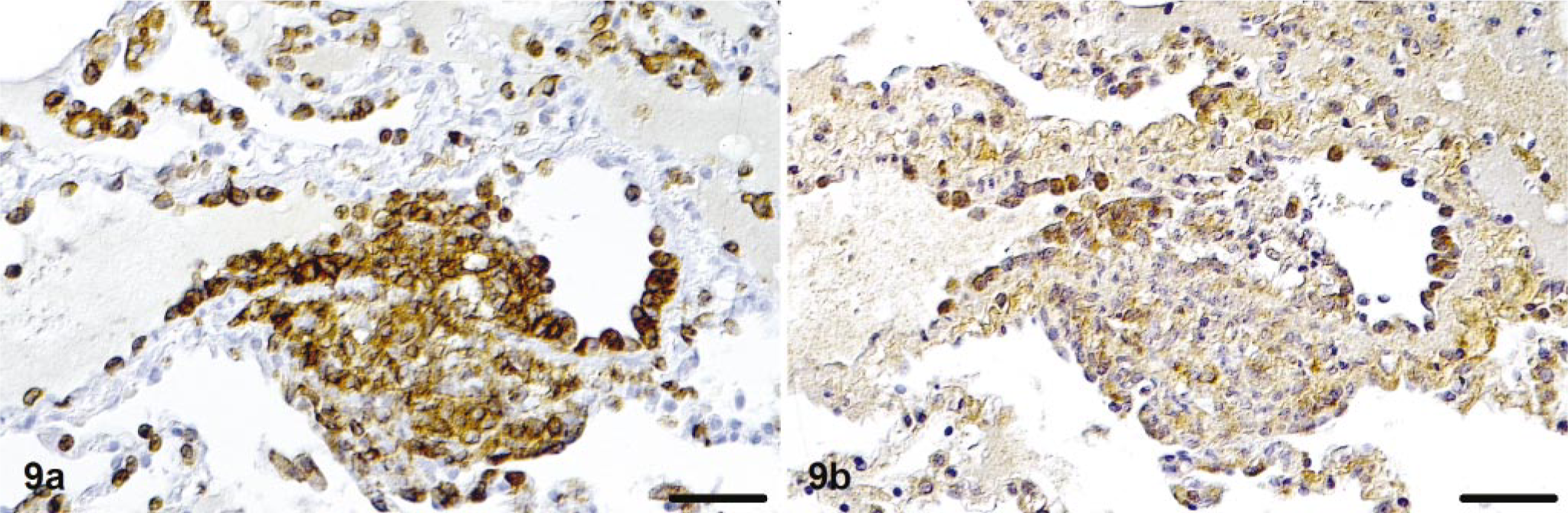
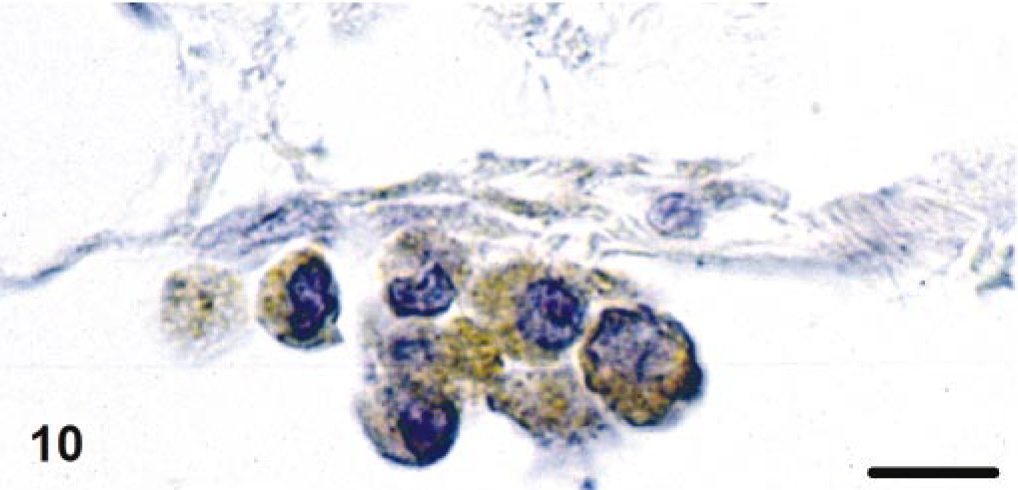
Intravascular monocytes and macrophages in perivascular granulomatous infiltrates were strongly positive for CD18 (Fig. 9a), with pronounced staining on the cell surface, and were often positive for IL-1β and TNF-α (Figs. 9b, 10). In cat No. 4, lymphocytes in the broad perivascular B cell rims showed a mild to moderately positive staining for IL-1β. A few intravascular monocytes and a few macrophages in perivascular infiltrates were also positive for MMP-9 (Fig. 11). Both monocytes/macrophages and lymphocytes expressed MHC class II antigens.
Lung; cat No. 1. Granulomatous phlebitis and periphlebitis.
Leptomeninx; cat No. 3. Leptomeningeal veins. Agglomerate of TNF-α-positive monocytes attached to venous endothelium. Avidin-biotin-peroxidase complex method. Papanicolaou's hematoxylin counterstain. Bar = 10 μm.
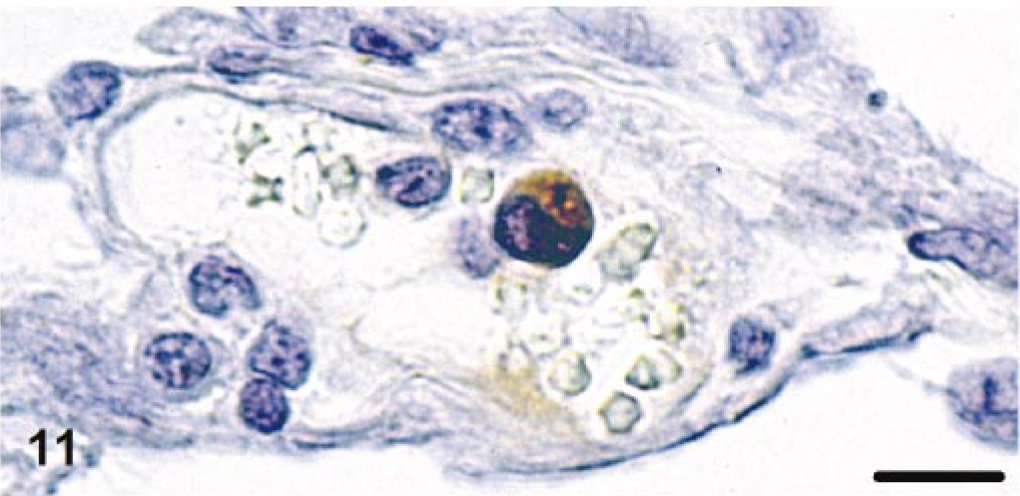
Leptomeninx; cat No. 5. Monocyte expressing MMP-9 within leptomeningeal vein. Avidin-biotin-peroxidase complex method. Bar = 15 μm.
Characterization of blood vessels with immunohistology
In uninfected SPF cats, vessels generally had an intact basal lamina, identified by distinct circular sub-endothelial staining for collagen IV. EC were negative for MHC II antigen, except for occasional venous and arterial EC in brain parenchyma and leptomeninges, which stained very faintly positive for MHC II antigen. Staining for cytokines was restricted to a faint to moderate reaction for TNF-α in arterial/arteriolar smooth muscle layers.
In cats with monocyte-associated FCoV viremia and granulomatous vasculitis, staining of vessels with granulomatous inflammation for collagen IV identified inflammatory cells between layers of disassembled basal lamina (Fig. 12) and complete destruction of the basal lamina in areas of intense vascular/perivascular infiltration (Fig. 13). FCoV antigen and RNA were not detected in cells of vessel walls (EC, pericytes, smooth muscle cells).
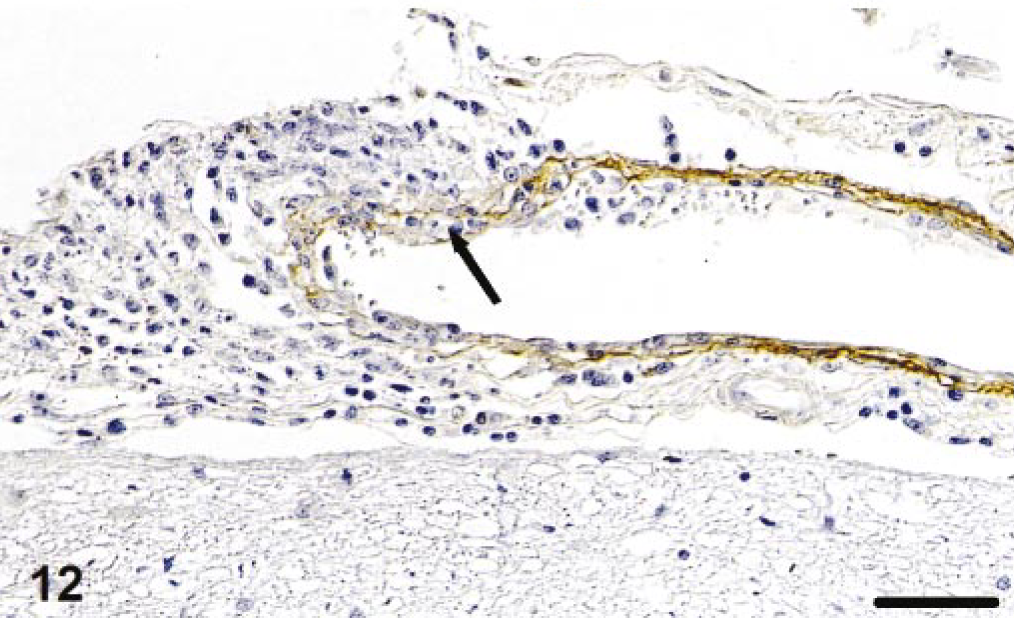
Leptomeninx; cat No. 5. Demonstration of collagen IV in affected veins shows disassembly of the basal lamina and inflammatory cell infiltration between layers of basal lamina (arrow). Peroxidase-anti-peroxidase method. Papanicolaou's hematoxylin counterstain. Bar = 40 μm.
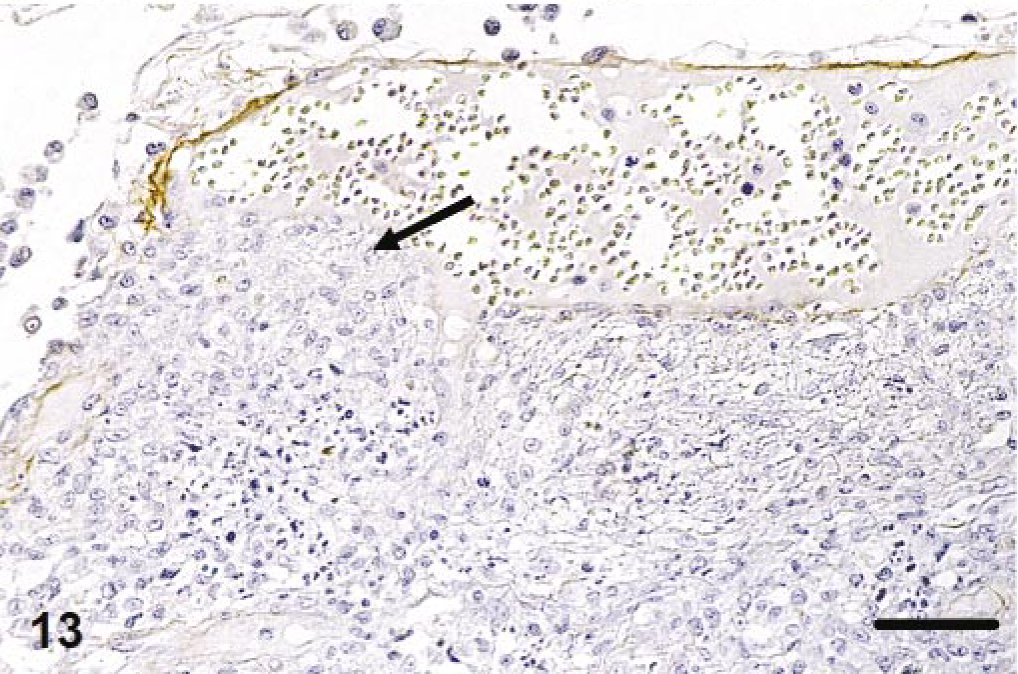
Leptomeninx; cat No. 3. Staining for collagen IV. With intense (peri)vascular infiltration, the basal lamina is completely destroyed (arrow). Peroxidase-anti-peroxidase method. Papanicolaou's hematoxylin counterstain. Bar = 40 μm.
Regardless of the presence of inflammatory infiltrates, EC in veins and arteries of different calibers in all tissues showed a mild to moderate expression of MHC II antigen (Fig. 14) in affected cats. In some vessels, EC also exhibited features of activation because they were bulging with a tombstonelike morphology (Fig. 14). In the kidney, glomerular EC were generally negative for MHC II antigen, whereas EC in veins and arteries in the medulla and to a lesser extent in the cortex stained positive. In cat Nos. 3 and 4, perivenous leptomeningeal inflammatory infiltrates often stretched along arteries. There, the whole arterial wall was moderately to strongly MHC II antigen positive.
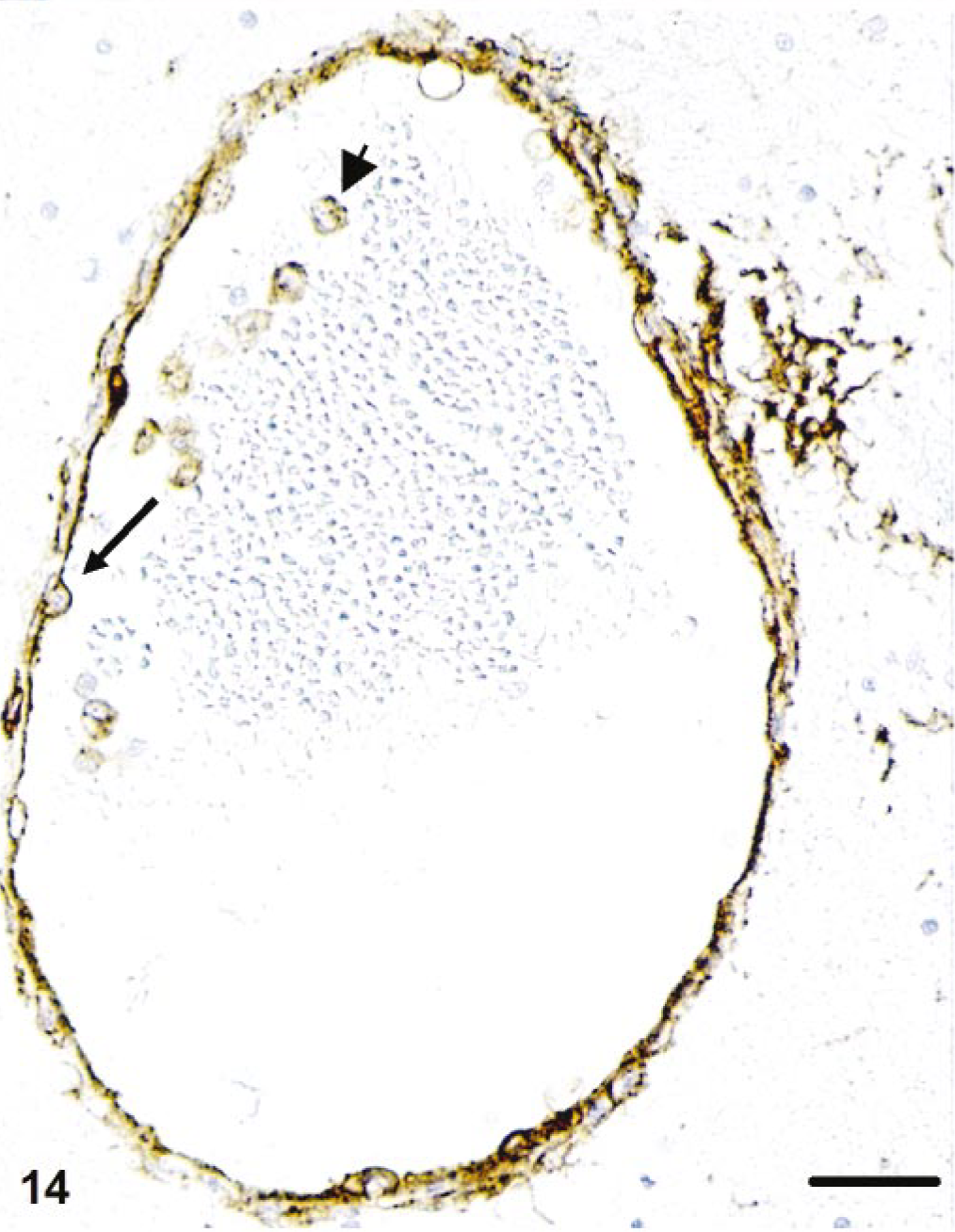
Liver; cat No. 2. Endothelial cells are strongly MHC II antigen positive and occasionally exhibit the tombstone-like morphology of activated cells (arrow). Monocytes are also MHC II positive (arrowhead). Peroxidase-anti-peroxidase method. Papanicolaou's hematoxylin counterstain. Bar = 30 μm.
Arterial walls often showed a mild to moderate staining for IL-1β and occasionally also for TNF-α (layers outside the internal elastic membrane). EC staining for IL-1β was restricted to a faint reaction predominantly in leptomeningeal vessels; TNF-α was occasionally faintly expressed by venous EC in intestinal wall and liver.
Additional findings
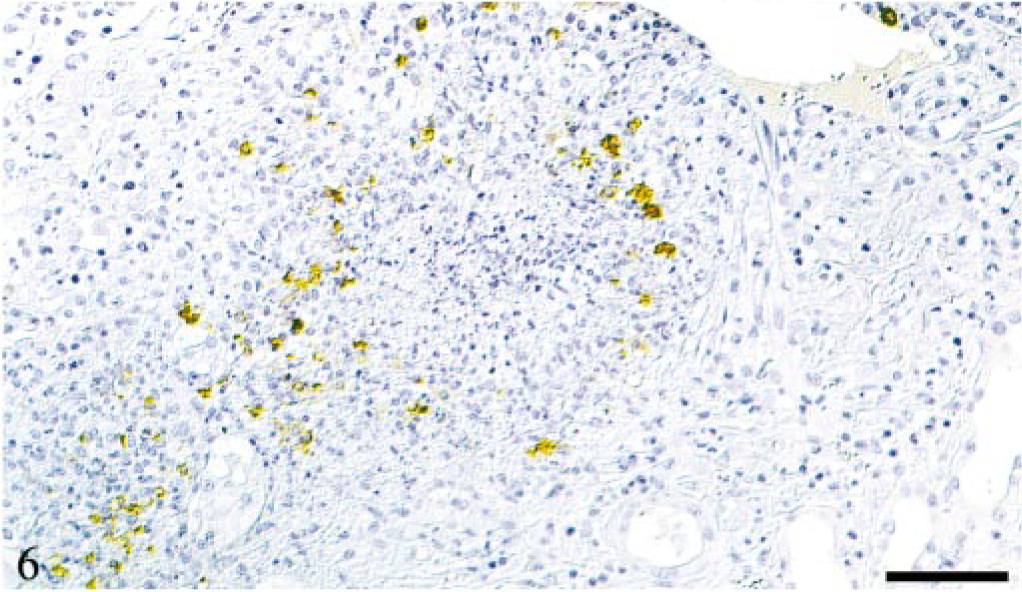
In addition to granulomatous vasculitis, all five animals exhibited granulomatous or granulomatous-necrotizing inflammation (or both) with FCoV-positive macrophages in the eyes and various organs (Table 2). In these lesions, venous structures were not identified.
Cat No. 2 additionally had a focal purulent bronchopneumonia and cat No. 3 a focal aspiration pneumonia.
Discussion
This study describes the morphologic features and development of multifocal granulomatous vasculitis in FIP, a fatal, CoV-induced, systemic disease in cats. 8, 16, 24
Vascular inflammatory processes were restricted to small and medium-sized veins predominantly in the leptomeninges, renal cortex, and eyes but also frequently in the lungs and liver. Morphologically, vasculitis presented as venous and perivenous, macrophage-dominated circular infiltrates in small veins and focal infiltrates in larger veins. Neutrophils and T cells represented minorities among inflammatory cells, and B cells mainly occurred as peripheral rims around circular granulomatous infiltrates. The latter most likely reflected the longevity of the inflammatory process because B cells have been shown to surround and progressively replace macrophages in FIP granulomas. 16 Monocyte emigration and perivenous macrophage accumulation were associated with disassembly and destruction of the basal lamina, represented by a loss of staining for collagen IV, the major extracellular matrix component of the vascular basal lamina. 20 Because collagen IV degradation by monocytic cells is mediated by the secretion of matrix metalloproteinase B (MMP-9), destruction of the basal lamina of affected vessels in FIP vasculitis is likely a byproduct of monocyte emigration. 40
FIP has previously been regarded as an immune complex-mediated, type-III hypersensitivity disease: cell-free fibrinogen and C3, as well as viral antigen and IgG within mononuclear cells, have been demonstrated in inflammatory vascular lesions. 8, 10, 29, 43, 45 The demonstration of FCoV-specific immune complexes in the peripheral blood and in glomerular vessels, together with high serum levels of γ-globulin and C3, supported this hypothesis. 10 However, because Igs/immune complexes can also be trapped nonspecifically, especially at sites of necrosis in vessel walls, definitive proof of the responsibility of immune complexes for vasculitis requires the demonstration of cell-free deposition of the specific antigen within vascular lesions. 13, 30, 32, 37
As demonstrated in this study, the morphology, cellular composition, and distribution of granulomatous vasculitis in FIP differs significantly from any immune-mediated vasculitis described in humans and animals. 4, 12, 13, 21, 30, 32, 37 The lack of arterial involvement and the involvement of small and medium-sized veins rather than postcapillary venules excludes the classification of FIP vasculitis as small or medium-sized vessel vasculitis. 13 Instead of being the dominant cell population, neutrophils only represented a minority among infiltrating cells. 4, 12, 13, 30 Furthermore, older vasculitic processes in FIP were represented by a rim of B cells around the granulomatous infiltrate rather than the nonspecific lymphocytic perivascular “cuffing,” which replaces the initial leukocytoclastic inflammation in immune complex vasculitis. 4 The lack of evidence for a direct attack on the vessel wall and the paucity of T cells among the infiltrate, on the other hand, differentiate FIP vasculitis from a cell-mediated vasculitis, as is seen for example in acute vascular allograft rejection. 12 In addition, neither in this nor in an experimental study was there evidence for direct infection of EC by FCoV. 16, 29, 44
FCoV antibodies may still play an important role in the development of FIP vasculitis and granulomas. The increasing number of B cells, which delineate granulomas and vasculitis, and the plasma cells bearing FCoV-specific antibodies, which are present within the infiltrate, might provide newly formed antibodies that could then bind to virus and form immune complexes. 16 Complement and Fc receptor activation can then increase the phagocytic activity of macrophages and thereby promote the infection of further macrophages. 10, 28 FCoV infection may increase the synthesis of complement factors in macrophages, and antibody-mediated lysis of infected macrophages in or around vessels may enhance the inflammatory reaction. 10, 11, 43, 45 However, although all these features may well participate in the disease, they seem not to induce the vasculitis in FIP.
Instead, this study indicates that in FIP, phlebitis is initiated by activated and FCoV-infected, circulating monocytes. Monocyte activation was reflected by their increased adhesion to both EC and other monocytes. The latter was evident because endothelium-bound monocyte aggregates were found protruding into and-in smaller veins-occasionally occluding the vascular lumen. 22 The strong expression of the myeloid/histiocyte antigen, a marker for (recently blood derived) macrophages, identified the macrophages in the venous/perivenous infiltrates as emigrated monocytes. 33 Monocytes/macrophages in vascular lesions also exhibited strong expression of CD18, which was frequently more pronounced on the cell surface. The latter is further evidence for activation of the cells because it has been shown that CD18 is translocated from cytoplasmic pools to the cell surface with activation of leukocytes. 5 Furthermore, the β2 integrins CD11/CD18 are known to be the predominant mediators of interaction between monocytes and EC in adhesion and emigration of monocytes. 9, 41 In cats with FIP vasculitis, both intravascular and emigrated monocytes/macrophages also stained positive for IL-1β and TNF-α. Both cytokines not only increase adhesion of monocytes to EC but are also upregulated in monocytes in response to β2 integrin-mediated adhesion to EC. 9, 25, 27, 42 Accordingly, FIP vasculitis may well develop and persist because of a paracrine and autocrine reaction pattern. Monocyte/macrophage MMP-9 secretion, for example, is not only upregulated by exogenous IL-1β and TNF-α but also by chemokine (e.g., CCL 2, 3, 5)-induced endogenous TNF-α. 31, 34 These chemokines, however, can again be secreted by emigrating macrophages, whereas secreted TNF-α can bind to fibronectin and laminin, thereby limiting the effect of TNF-α to target areas of inflammation. 36, 38
EC also appear to contribute to the development of FIP vasculitis. The intense MHC II upregulation and activated morphology observed in venous and, to a lesser extent, arterial EC independent of inflammatory lesions in the respective vessel indicates generalized activation of EC during the course of monocyte-associated viremia and granulomatous vasculitis. 3 Endothelial MHC II upregulation can be induced by TNF-α, and the wide distribution of endothelial MHC II expression observed indicates a systemic cytokine effect. 3 On the other hand, endothelial TNF-α expression, which was only seen close to FIP lesions, is probably a paracrine effect.
Interestingly, in FIP, inflammatory processes are restricted to veins, and especially to veins in selected organs such as the brain. It is known that venous EC generally induce more intense adhesion of leukocytes than arterial EC. 6 Furthermore, a systemic cytokine effect might be involved. In some organs, such as the brain, EC generally have a significantly low tendency to bind lymphocytes but show a very strong increase in adhesion in response to TNF-α- and IFN-γ. 23 Also, TNF-α selectively upregulates VCAM-1 in venous EC. Its receptor, VLA-4, a β1 integrin, however, is not expressed by neutrophils. 2, 14 This would explain both the restriction of inflammatory lesions to veins and the low amount of neutrophils in the inflammatory infiltrate in FIP vasculitis.
Low-level, monocyte-associated FCoV viremia, detected by RT-PCR on cultured monocytes, can be demonstrated in similar proportions of both clinically healthy cats from households with endemic FIP and cats with FIP. 7, 15 Accordingly, monocyte-associated viremia can be regarded as a general feature of FCoV infection in cats. 7, 15 In contrast, our results show that during the development of FIP lesions, cats undergo at least intermittent high-level, monocyte-associated viremia, where FCoV protein, genome, or viral particles (or all) can be demonstrated in monocytes in situ. This indicates that quantitative differences regarding the degree of monocyte infection occur at some stage in the transition from FCoV infection without disease to FIP. Because vasculitis appears to be associated with high-level, monocyte-associated viremia and because affected animals have lesions at various stages of development, it is likely that cats undergo recurrent bouts of monocyte-associated viremia with development of new lesions in each viremic phase. 16 The demonstration of cerebral veins with both a broad perivascular rim of B cells and complete obstruction of the vascular lumen by monocytes, some of which were FCoV-infected, indicates that an acute phase of monocyte-associated viremia with granulomatous vasculitis can superimpose the residua of an older FCoV-induced phlebitis. 16
It is likely that not only the extent of monocyte infection, but also the viral effect on monocytes/macrophages is important for the development of FIP. In cats with immunohistologically detectable, monocyte-associated FCoV viremia and granulomatous vasculitis, monocytes/macrophages are activated, most likely as a sequel of viral infection. Activation of monocytes may therefore represent a feature of the virulent bio-type of FCoV, which is known to bear deletions in genome sequences coding for nonstructural proteins of unknown function. 39
Summarizing, this study proves that FIP is a monocyte-triggered disease and identifies the central role of activated monocytes in the development of FIP vasculitis.
Footnotes
Acknowledgements
We are grateful to Dr. Margaret Hosie and Prof. Os Jarrett, Department of Veterinary Pathology, University of Glasgow, Veterinary School, Glasgow, UK, for kindly providing the SPF cat tissues and wish to thank Dr. Haimo Enbergs for helpful discussions. Thanks are also due to Mrs. M. Wipplinger, Mrs. P. Herzog, and Mrs. I. Langhof for excellent technical assistance. We wish to thank Mr. Rob Pearson for photographic assistance.
1Department of Veterinary Pathology, Faculty of Veterinary Science, University of Liverpool, Liverpool, UK
