Abstract
This study describes clinical and histopathological features, treatment, and outcome of cats diagnosed with ocular mycobacteriosis. Cases diagnosed from 2012 to 2017 were reviewed for (a) histopathological evidence of ocular (pyo)granulomatous inflammation containing acid-fast bacilli with mycobacterial morphology, (b) positive mycobacterial culture and/or mycobacterial DNA identified by polymerase chain reaction of ocular tissue, or (c) presumed mycobacteriosis based on ophthalmic examination and positive interferon-gamma release assay. Twenty-five cats (31 eyes) were included; 14 cats (17/31 eyes, 55%) were blind at presentation (unilateral: n = 12 cats; bilateral: n = 2 cats); one unilaterally affected cat later became bilaterally blind. Another 5 cats (7/31 eyes, 23%) became blind after initially being bilaterally visual (unilateral: n = 3 cats; bilateral: n = 2 cats). The commonest ocular finding was uveitis (87%). The main histopathological features were granulomatous to pyogranulomatous chorioretinitis with retinal detachment, anterior uveitis, optic neuritis, episcleritis, scleritis, and/or retrobulbar cellulitis. Nineteen cats (76%) had systemic signs, with disseminated disease being diagnosed in 9, defined by interstitial pulmonary disease, generalized lymphadenopathy, and/or nonocular infection. Nine cats were diagnosed with Mycobacterium bovis, 2 with Mycobacterium microti, 1 with Mycobacterium tuberculosis complex, and 1 with Mycobacterium avium-intracellulare complex. The infecting species was unknown in the remaining cats. Combined surgery (enucleation: n = 5 cats; biopsy: n = 3 cats) and systemic treatment with 2 or 3 appropriate antibiotics for 2 to 7 months resulted in remission in 8 of the 10 cats treated; however, the cat treated with dual therapy relapsed after 8 months. A total of 16 cats (64%) were euthanized; 2 were lost to follow-up.
Keywords
Mycobacteria are aerobic, non-spore-forming acid-fast bacilli (AFB), some of which can cause clinical disease (ie, mycobacteriosis) in many species of animal, including domestic cats. 46 Based on the causative species, disease in cats can be classified as being caused by members of the Mycobacterium tuberculosis complex, causing feline tuberculosis (TB) due to infection with either Mycobacterium bovis or Mycobacterium microti, or as nontuberculous mycobacterial disease (NTM), which includes disease caused by Mycobacterium avium-intracellulare complex, and mycobacteria that cause feline leprosy syndrome (FLS), among others. 37,46 Infections with M. avium-intracellulare complex are strongly associated with profound immunosuppression, unlike those caused by members of the M. tuberculosis complex. 37
The majority of feline mycobacterial infections in the United Kingdom present with cutaneous nodular lesions with or without superficial dermal ulceration. 20,37,46 Associated regional lymphadenopathy is common, and in some cases, persistent lymphadenopathy may be the only clinical sign. 20,37,46 Less common presentations include primary respiratory disease, gastrointestinal manifestations, or disseminated systemic disease. 3,17,20,37,46
Studies documenting ophthalmic mycobacteriosis in cats are limited but include reports of retinal and subretinal hemorrhages; exudative retinal detachment; granulomatous chorioretinitis; scleral, eyelid, conjunctival, and corneal granulomas; and keratopathy. 8,9,13 –15,25,33,34 Mycobacterium tarwinense, endemic to Australia, usually induces nonulcerated nodules particularly on the head, often involving ocular and periocular structures with no clinical evidence of systemic spread. 34
Gunn-Moore et al 20 reported that 18 of 271 cats (6.6%) with mycobacterial infection displayed ocular signs, suggesting that this is an important area for further research.
Ocular TB in cattle and swine has been described as causing similar clinical signs to those listed above, including choroidal infiltrates, anterior uveitis, subretinal exudation, and retinal detachment. 7,44 Ocular mycobacteriosis has occasionally been observed in birds, ferrets, and a horse. 27,29,38 Experimental studies in nonhuman primates, guinea pigs, and zebrafish have revealed that animals infected with mycobacteria display similar histopathological ocular lesions to humans with extrapulmonary TB. 6,39,47 Ocular TB in experimentally challenged rabbits is reported to be severe and progressive, with the first clinical signs seen in the iris. 12
A diagnosis of feline mycobacteriosis can be challenging. 16,21 A high index of suspicion can be held with the identification of histopathological findings of granulomatous to pyogranulomatous inflammation, dominated by the presence of epithelioid macrophages. 10,21,24,33,37,46 Similarly, a high index of suspicion can be obtained from cytological samples demonstrating granulomatous to pyogranulomatous inflammation; 25,37,43,46 mycobacteria may be seen as nonstaining intracytoplasmic bacilli within macrophages when stained with standard Romanowsky stains (eg, Diff Quick). 25,30,32,46 A histopathological or cytological diagnosis of mycobacteriosis is confirmed by the identification of morphologically typical AFB on sections or smears stained with Ziehl-Neelsen (ZN) or similar stains.* As reported by Gunn-Moore et al, 21 histopathology and the presence of AFB are not sufficient to determine which species of mycobacteria is present in any given tissue sample. This diagnosis must be made via special mycobacterial culture, mycobacterial polymerase chain reaction (PCR), and subsequent sequencing or by interferon gamma release assay (IGRA). †
Most mycobacterial infections require treatment with a long course of multiple appropriate antibiotics and, where indicated, surgical excision of affected tissues. 16,21,31,37,46 Tuberculous mycobacterial infections in companion animals are routinely treated with a combination of a macrolide or azalide, a fluoroquinolone, and rifampicin, 16,21,46 which should be given for at least 3 months or longer if indicated by the presence of ongoing systemic disease. 37 Where pulmonary involvement is present, as diagnosed by radiography or computed tomography (CT), treatment is extended to a minimum of 6 months and for 2 months beyond the resolution of radiographic lesions. 37
Given the paucity of comprehensive clinical and histopathological studies of feline ocular mycobacteriosis, the primary aim of this study was to summarize these findings in cases of feline mycobacterial infections where ocular involvement was confirmed and to report treatment and the clinical outcomes following treatment.
Materials and Methods
Case records of cats diagnosed with ocular mycobacteriosis between 2012 and 2017 were included. Cases were diagnosed by primary care (general practitioner) veterinarians and brought to the attention of the authors during the course of case diagnosis and/or management. All cats were resident solely in the United Kingdom, and records were obtained with owner consent from the relevant veterinary pathology laboratories; further details were subsequently requested from the primary care veterinarians, veterinary ophthalmologists (diplomates or certified specialists), and authors, as required. For all cases, clinical, laboratory, and histological records were obtained and stored in accordance to data protection and with the owners’ consent.
For inclusion in this study, cases had to conform to at least one of the following inclusion criteria: (a) histopathological evidence of granulomatous or pyogranulomatous inflammation containing AFB with mycobacterial morphology within ocular tissue, (b) positive mycobacterial tissue culture and/or positive mycobacterial DNA identified by polymerase chain reaction (PCR) performed on ocular tissue, or (c) presumed ocular mycobacteriosis based on ophthalmic examination performed by diplomates or certified specialists in veterinary ophthalmology and a contemporaneously positive IGRA test.
For cases meeting the inclusion criteria, laboratory and clinical records were reviewed. Results of mycobacterial culture, PCR, IGRA, postmortem examination (where applicable), and details of histopathological findings of ocular tissue, including the number of AFB per slide, were recorded. For the purpose of this article, fewer than 5 AFB per slide was referred to as paucibacillary infection, and the presence of 5 or more AFB was referred to as multibacillary infection. Signalment including age, breed, sex, neuter status, outdoor and/or indoor activity, lesion history (features and localization of the wounds, history of ocular trauma), and ophthalmic and physical examination findings were extracted from clinical records. Any further diagnostics that had been performed to assess systemic involvement were recorded, including thoracic and/or abdominal radiography or ultrasound, CT, cytology of fine-needle aspirates, or histopathological findings of biopsies.
Information regarding infectious disease status, including feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV), was obtained where these had been assessed. Data relevant to treatment and outcome, including times from diagnosis until euthanasia, remission, or until lost to follow-up; whether euthanasia was performed; treatment details (surgery, drugs given, duration of treatment, and the presence of any complications); and eventual outcome were recorded. Disease was reported as disseminated when a cat presented, in addition to ocular mycobacterial pathology, with generalized mycobacterial lymphadenopathy identified clinically, on cytology and/or histology or by CT, with mycobacterial skin lesion(s), with antemortem pulmonary interstitial infiltration on thoracic radiography or CT, or with postmortem evidence of systemic spread of mycobacterial infection into the thoracic and/or abdominal cavity.
Remission criteria required that the cat remained systemically healthy with no mycobacterial (ocular) disease based on clinical examination, and if pulmonary lesions had been present initially, with normal resting respiratory rates, and no pulmonary pathology on repeated thoracic radiography or CT, and no other signs of mycobacterial disease, for 2 months after stopping treatment.
In some cases, the requested information was not available or data were missing; therefore, the number of data points used in each analysis is noted. Where applicable, descriptive statistics were calculated and reported.
For all cases, data were reviewed with the owners’ consent. All cats were pets living in the United Kingdom with no known history of foreign travel. Local ethical approval for the current study was obtained from the Animal Health Trust (AHT; study number AHT 48-2015).
Results
This study reviewed the records of 25 cats (31 eyes) with feline ocular mycobacteriosis. Detailed data are provided in Supplemental Table S1. Definitive mycobacterial species identification was attempted in 13 cases and achieved, at least partially, in all 13 cases.
Diagnosis of Mycobacterial Disease
Histopathology was performed on ocular biopsy samples taken from 21 cats (affecting 24 eyes); the samples included enucleated globes (n = 14) and biopsies of affected ocular tissue (n = 7) (Suppl. Table S1). AFB with mycobacterial morphology were identified in stained sections in 20 of the 21 samples (95%). Four of the 20 cats (cases 1, 2, 3, 4) were infected with M. bovis (2 diagnosed by IGRA, 2 diagnosed by culture), 2 cats (cases 5, 6) were infected with M. microti (both confirmed by both IGRA and culture), and case 7 had M. tuberculosis complex DNA isolated by PCR, but specification to M. bovis or M. microti was not possible. Case 8 was diagnosed with a M. avium-intracellulare complex infection by IGRA. The section of ocular tissue from case 9 was negative for AFB, but the histopathology was typical for mycobacterial infection, and an IGRA test indicated infection with M. bovis.
Histopathological examination was not performed in the remaining 4 cats (7 of the 31 eyes). Mycobacterial culture confirmed M. bovis infection in 2 of these cats (cases 10, 11), one of which was also IGRA positive. For the remaining 2 cats (cases 12, 13), an IGRA test was indicative of M. bovis infections.
Case History
Of the 25 cats included in the study, 19 were males (76%) and 6 were females, with a median age of 5 years (range, 6 months to 10 years). All cats were neutered and comprised the following breeds: Domestic Shorthair (n = 20), Bengal (n = 2), British Shorthair (n = 2), and Burmese (n = 1).
Information about indoor and/or outdoor activity was available for 22 cats, with 13 (59%) known to have mixed outdoor and indoor access and the remaining 9 living outdoors full-time. No cats were reported as living solely indoors.
The geographic location of residence was provided for 23 cats; cases came from the South West of England (n = 8), West Midlands (n = 4), north of London (n = 2), Scotland (n = 2), Wales (n = 2), East Midlands (n = 2), North West of England (n = 1), East Anglia (n = 1), and South East of England (n = 1). Cats diagnosed with M. bovis infection lived in the South West of England (n = 5), the West Midlands (n = 2), and Wales (n = 1); data were unavailable for 1 cat. The cat diagnosed with M. tuberculosis complex was from the north of London. M. microti was identified in 1 cat from Scotland (n = 1) and 1 from the North West of England (n = 1). The cat infected with M. avium-intracellulare complex was from the East Midlands.
Clinical Findings
Clinical signs were restricted to the eye in only 6 of 25 cats (24%; cases 1, 3, 13, 18, 20, 23); 3 of them were diagnosed with M. bovis. An additional 4 of 25 cats (16%; cases 5, 12, 21, 22) that were clinically healthy, other than their ocular disease, later developed systemic signs, including depressed mentation, weight loss, muscle wastage, dyspnea, and/or clinical signs consistent with chronic kidney disease. The remaining 15 of 25 cats (60%) initially presented with a range of clinical signs, including respiratory signs, neurological signs, musculoskeletal disease, pyrexia, skin or subcutaneous lesions, oral mucosal lesions, persistent lymphadenopathy, lethargy and weight loss, depression, and/or gastrointestinal signs, including hyporexia or anorexia.
Lymphadenopathy was noted in 7 of 25 cats (28%; cases 2, 4, 6, 8, 9, 11, 16), with 3 of them (cases 2, 4, 6) having multiple peripheral and internal lymph nodes affected; hepatic or sternal with or without mediastinal and mesenterial lymphadenopathies were recorded (Suppl. Table S1). Cases. 8, 11, and 16 had a mandibular lymphadenopathy while case 9 had an enlarged popliteal lymph node.
Eight of 25 cats (32%) had apparent bite wounds, skin lacerations, or ocular injury after fights or trauma; these were localized on the outer skin of the eyelids (cases 1, 17), cornea (case 20), eyelid and body (case 8), or limited to the body (case 14). Three cats (cases 4, 6, 9) had a chronic nonhealing wound on the head or body.
Disseminated mycobacterial disease was diagnosed in 9 of 25 cats (36%) with, in order of decreasing frequency, interstitial pulmonary disease (cases 4, 6, 9, 11, 16, 17, 19) and/or generalized lymphadenopathy (cases 2, 4, 6) and/or nonocular tissue involvement (case 7) being the most common signs (Suppl. Table S1).
Ophthalmic Findings
Twenty of 25 cats (80%) presented with unilateral ocular signs while 5 (20%) had bilateral involvement; 1 cat (case 10) that presented initially with unilateral ocular signs later progressed to have bilateral disease. Therefore, a total of 31 eyes were included in this study (Suppl. Table S1).
In 20 cats (27 of the 31 eyes, 87%), ocular examination revealed findings that were compatible with uveitis (panuveitis or anterior uveitis) (Figs. 1–4). Case 6 later developed a corneal granuloma (Fig. 5). A unilateral eyelid granuloma was present at initial presentation in case 11, which was later re-presented with bilateral retinal detachment and retinal granulomas. Case 9 had unilateral conjunctivitis and bilateral panuveitis associated with retinal detachment. Retinal detachment was diagnosed on fundoscopy in 13 of the 20 cats with uveitis (18 of the 27 eyes, 67%) (Fig. 3).
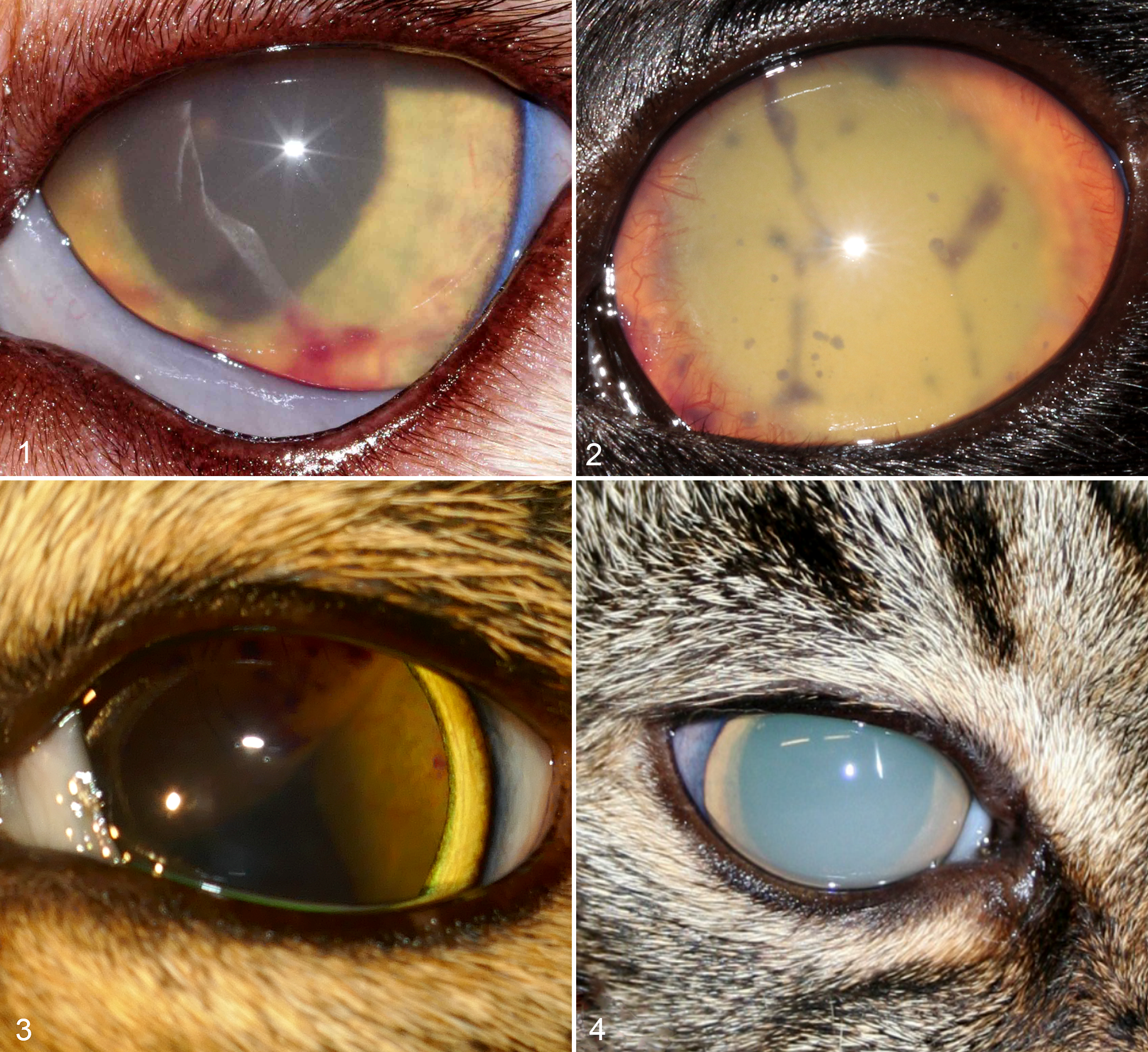
Mycobacterial uveitis, eye, cat.
Mycobacteriosis, eye, cat.
Fourteen cats (17 of the 31 eyes, 55%) were blind at presentation (unilateral: n = 12 cats, bilateral: n = 2 cats); 1 unilaterally affected cat later became bilaterally blind (case 10). Another 5 cats (7 of the 31 eyes, 23%) became blind after initially presenting as bilaterally visual (cases 7, 11, 13, 14, 15; unilateral: n = 3 cats; bilateral: n = 2 cats). One cat (case 23) was diagnosed with a solitary focal raised corneal stromal granuloma (Fig. 6), while 10 of 25 cats (40%) had unilateral (n = 8 eyes) or bilateral keratitis (n = 4 eyes). Varying degrees of palpebral, bulbar, and nictitating membrane conjunctivitis were present in 12 cats (15 of the 31 eyes, 48%). Solitary conjunctivitis initially presented as chemosis and hyperemia in 4 cats (4 of the 31 eyes, 13%; cases 1, 5, 8, 16), which later progressed to a proliferative firm lesion in all cases (Figs. 7, 8). Case 1 was diagnosed with unilateral entropion secondary to rapidly progressive conjunctivitis.
Secondary glaucoma was clinically diagnosed in 4 cats (6 of the 31 eyes, 19%; cases 2, 3, 4, 7) and cataract formation was identified in 4 cats (5 of the 31 eyes, 16%; cases 10, 20, 22, 24).
Supplementary Diagnostic Tests
Thoracic radiography was performed in 17 of 25 cats (68%), revealing an abnormal pulmonary interstitial pattern consistent with mycobacteriosis in 7 cats (cases 4, 6, 9, 11, 16, 17, 19); only 2 cats presented with clinical respiratory signs (cases 16, 17) (Suppl. Table S1). Abdominal ultrasound, when performed (n = 2), showed no findings consistent with mycobacteriosis. CT (n = 1) of the head and thoracic cavity revealed internal lymphadenopathy in case 2. Cytological examination, including ZN staining, of ocular and nonocular tissue was performed in 4 cats; AFB were found in the fine-needle aspirates of the facial lesion and direct lung smear in one of them (case 6). Postmortem examination performed in 3 cats confirmed disseminated mycobacteriosis in cases 4 and 6. FeLV antigen and FIV antibody status was negative in all tested cases (n = 14).
Gross and Histopathology
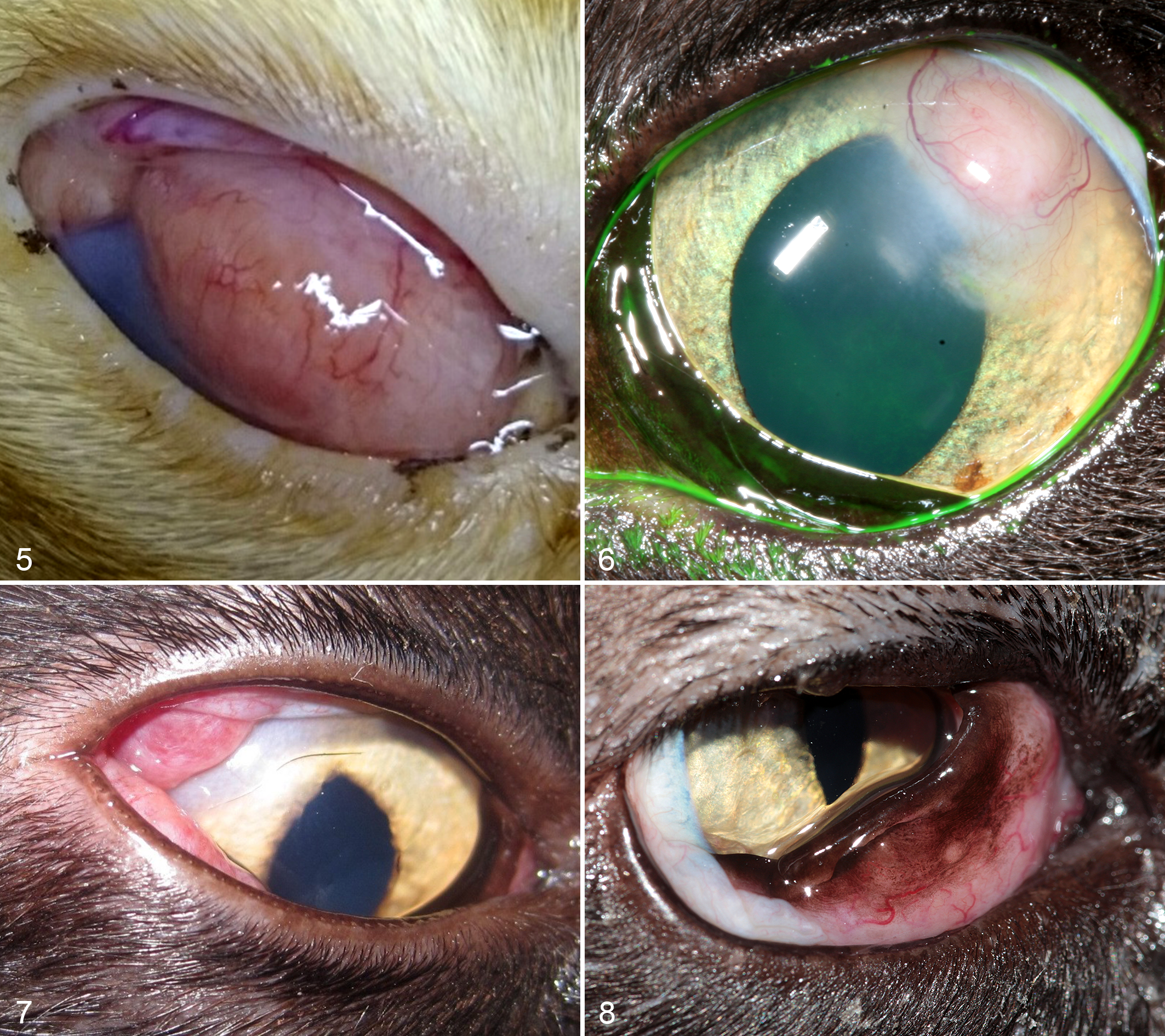
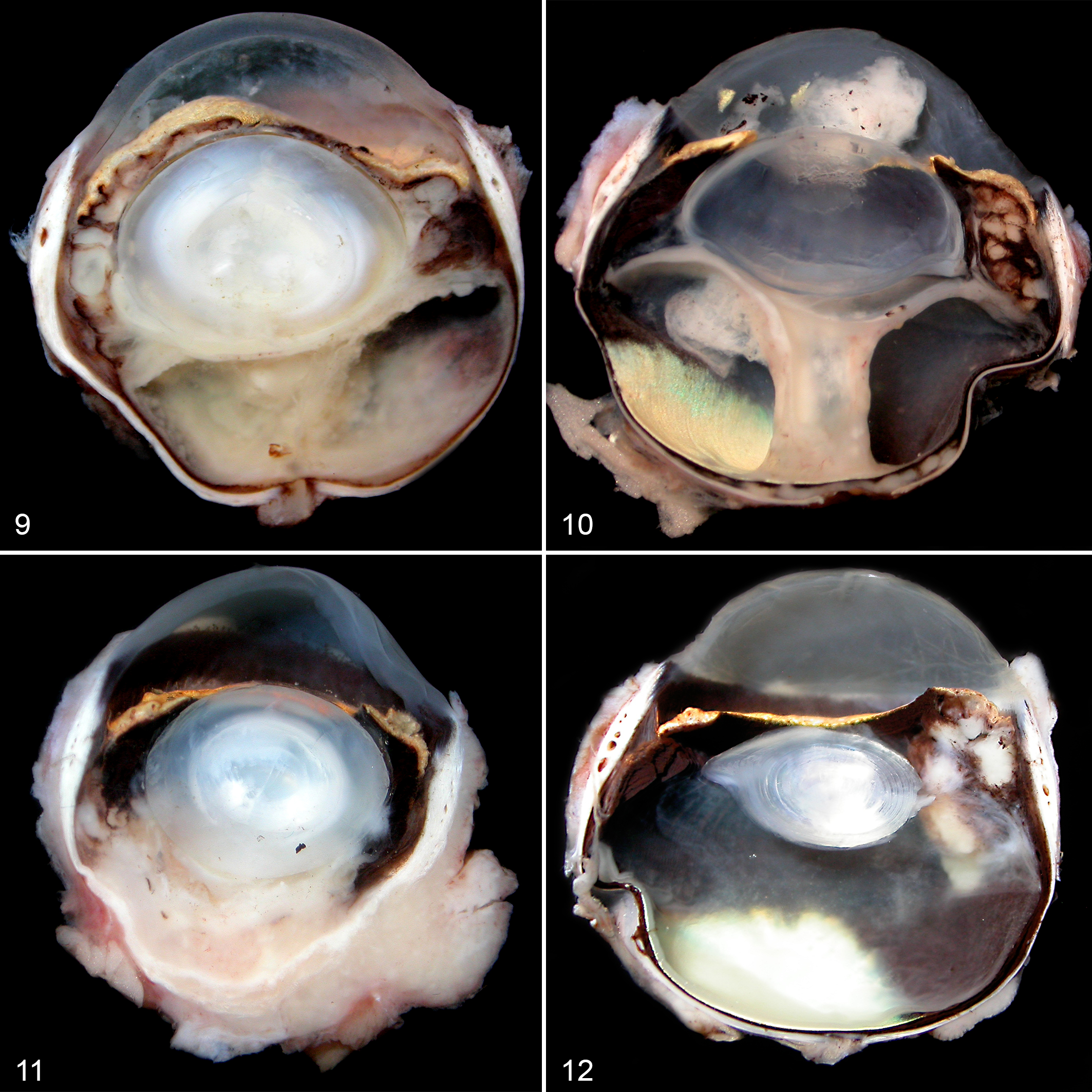
On gross examination of 14 enucleated globes, the posterior segment was most commonly involved, characterized by expansion of the optic nerve, sclera, choroid, and/or retina with an infiltrate that was cream-yellow on cut surface and was commonly associated with retinal detachment (Figs. 9–11). Anterior uveal involvement with no or minimal involvement of the posterior segment was less common (cases 7, 18; Fig. 12).
Mycobacteriosis, eye, cat. The globes have been sectioned parasagittally adjacent to the optic nerve.
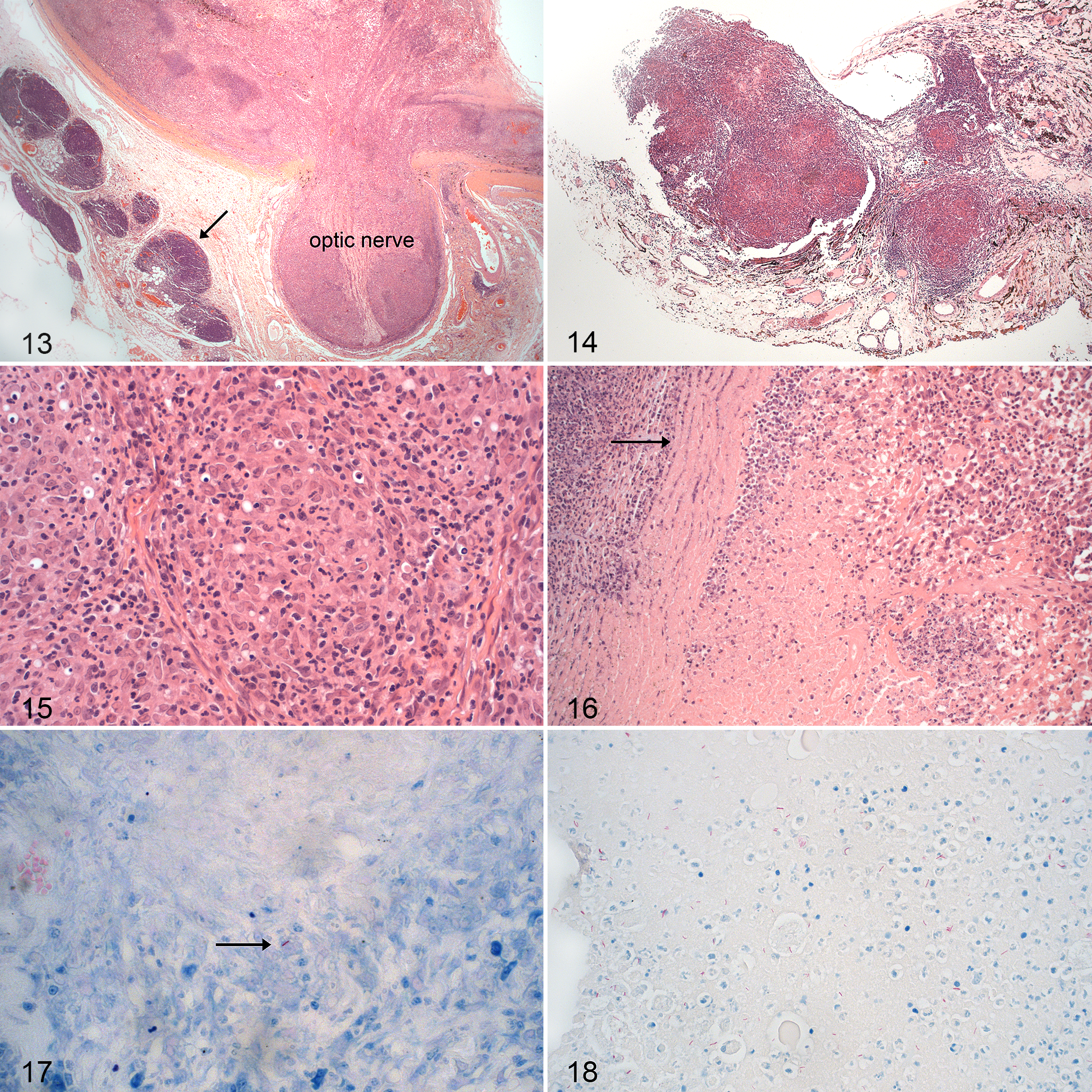
Ocular histopathology was assessed in 21 of the 25 cats (Suppl. Table S1). On examination of hematoxylin and eosin (HE)–stained sections, granulomatous to pyogranulomatous inflammation associated with necrosis and occasional microgranuloma formation were diagnosed in all lesions examined (Figs. 13–16). The inflammatory infiltrate was characterized by large numbers of epithelioid macrophages admixed with fewer neutrophils and lymphocytes, with areas of central necrosis (Figs. 15, 16). There was no evidence of mineralization or multinucleated giant cells in any of the sections.
Mycobacteriosis, eye, cat.
Granulomatous to pyogranulomatous chorioretinitis with retinal detachment with or without granulomatous anterior uveitis, optic neuritis, scleritis or episcleritis, and/or retrobulbar cellulitis was identified in 12 complete globes. Histopathology of 2 globes (2 of 21 cats, 9.5%; cases 7, 18) with clinical signs of anterior uveitis revealed nodular pyogranulomatous anterior uveitis; the anterior uveal tract was focally infiltrated and replaced by multiple granulomas to pyogranulomas with no or minimal involvement of the posterior segment in both cases.
Granulomatous to pyogranulomatous conjunctivitis was diagnosed in 5 ocular tissue samples (5 of 21 cats, 24%; cases 1, 5, 8, 9, 16), including case 9, which had bilateral panuveitis associated with retinal detachment, from which only conjunctival tissue was submitted for histopathological examination. Granulomatous to pyogranulomatous keratitis was diagnosed in 2 tissue samples (2 of 21 cats, 9.5%; cases 6, 23); case 6 had panuveitis, but only corneal tissue was submitted postmortem for histopathological examination.
Other histopathological findings included corneal vascularization and ulceration, lens subluxation, various degrees of cataract formation, anterior and posterior synechiae, hyphema, preiridal fibrovascular membrane, and secondary glaucoma. In addition, case 3, which had chorioretinitis associated with retinal detachment, anterior uveitis, and orbital involvement, also had a posterior lens capsule rupture associated with a hypermature cataract and abundant intralenticular AFB.
AFB with mycobacterial morphology were found in 20 of the 21 ocular tissue samples examined (95%), located in the areas of necrosis and/or granulomatous inflammation. Eighteen of 20 samples (90%) had paucibacillary infections (Fig. 17), while multibacillary infections were identified in only 2 ocular samples (10%; cases 3, 21) (Fig. 18).
Treatment and Clinical Outcome
Four of the 25 cats (16%; cases 6, 10, 11, 13) were euthanized after diagnosis of mycobacterial infection without any surgical or medical intervention.
Twenty-one of the 25 cats underwent ocular surgery as part of their diagnosis and/or treatment (84%). Fifteen of 25 cats (60%) underwent unilateral enucleation, resulting in 15 of 31 affected eyes being removed (48%; the enucleated eye from 1 cat was not assessed histopathologically, and hence only 14 globes were examined). Five of 25 cats (20%; cases 1, 5, 8, 9, 16) had incisional biopsies of unilateral conjunctival lesions. Case 23 underwent a superficial keratectomy for a corneal granuloma.
Following a histopathological diagnosis subsequent to enucleation (n = 7) or incisional biopsy (n = 3), 10 of the 21 cats detailed above (48%) were then treated medically with oral antibiotics. Untreated cats were either euthanized at this time point (n = 10) or lost to follow-up (n = 1). Hence, a total of 14 cats (14/25; 56%) were euthanized at the time of diagnosis.
Triple antibiotic therapy was administered to 8 cats (cases 2, 3, 5, 8, 12, 15, 16, 19). Seven of those cats were treated with rifampicin (Rifadin; Aventis Pharma Limited, Guildford, UK) at 10 to 15 mg/kg orally (PO) every (q) 24 hours, pradofloxacin (Veraflox; Bayer, Leverkusen, Germany) at 3 to 5 mg/kg PO q24 hours, and either azithromycin (Zithromax; Pfizer Limited, New York, US) at 5 to 10 mg/kg PO q24 hours or clarithromycin (Klaricid; Mylan, Potters Bar, UK) at 5 to 10 mg/kg PO q12 hours. Case 16 was treated with rifampicin 9 mg/kg PO q24 hours, clarithromycin 9 mg/kg PO q12 hours, and marbofloxacin (Marbocyl; Vetoquinol UK Limited, Towcester, UK) at 2.5 mg/kg PO q24 hours.
The duration of the triple antibiotic therapy ranged from 2 to 7 months. Two of the 8 cats (cases 2, 12) were treated for only 2 months. While case 2 achieved remission, case 12 developed lethargy, ataxia, and (mild) hepatopathy, followed by hyporexia and cachexia associated with azotemia and bilateral renomegaly, and was euthanized. Four of the 8 cats (cases 3, 8, 15, 19) were treated with triple antibiotic therapy for 5 to 7 months, and all achieved remission. In one of the remaining cats (case 5), rifampicin was withdrawn after 3 months due to a suspected adverse drug reaction (neutropenia, thrombocytopenia, pruritus, and self-mutilation); the remaining 2 drugs were continued for a further 3 months and the cat achieved remission. The final cat (case 16), which initially received marbofloxacin as part of its triple therapy, had this withdrawn after 2 months for an unspecified reason and was continued on the remaining 2 drugs for 6 months; this cat also achieved remission.
Case 20 was given dual therapy with enrofloxacin (Baytril; Bayer, Leverkusen, Germany) at 5 mg/kg PO q24 hours and azithromycin (Zithromax; Pfizer Limited, New York, US) at 5 mg/kg PO q24 hours for 2 months; the cat achieved remission but developed an enucleation site abscess 8 months after antibiotic treatment was discontinued and was euthanized without further investigation.
Case 21 was given monotherapy with pradofloxacin suspension (Veraflox; Bayer, Leverkusen, Germany) at 5 mg/kg P’O q24 hours for 1 month. The cat developed further clinical signs, including dyspnea and coughing, and was subsequently lost to follow-up.
Remission was therefore achieved in 8 of the 10 cats (80%) where surgical intervention (enucleation n = 5 cats [cases 2, 3, 15, 19, 20] or incisional biopsy n = 3 cats [cases 5, 8, 16]) was combined with the systemic administration of 2 or 3 drugs that were active against mycobacteria being given for 2 to 7 months. Notably, the only cat to receive a short course of dual therapy later relapsed.
Four of the 8 cats that achieved remission had their mycobacterial species identified; 3 were infected with members of the M. tuberculosis complex (cases 2, 3, 5), and 1 was infected with M. avium-intracellulare complex (case 8). In the remaining 4 cats, the infecting species was not investigated.
Two of 8 cats (cases 2, 8) that reached remission were tested with IGRA after completing the antibiotic course and were both found to be negative.
Long-Term Follow-Up
Sixteen of the 25 cats (64%) were euthanized a median of 12 days after diagnosis (range, 0–529 days, with case 20 being euthanized on apparent relapse following a remission of 8 months). No antimycobacterial therapy was attempted in 14 (14/16; 88%) of these cases. The main reasons for euthanasia were stated as compromised animal welfare, patient or owner noncompliance with therapy, and/or public health concerns regarding zoonotic risk.
Of the remaining 9 cats, 7 (cases 2, 3, 5, 8, 15, 16, 19) were alive and in remission at the end of the study, a median of 439 days after diagnosis (range, 128–1258 days). The final 2 cats were alive when they were lost to follow-up after 30 days (case 21) after diagnosis and 36 days after diagnosis (case 18, which received no antimycobacterial therapy) (Suppl. Table S1).
Discussion
This study is the first to investigate a case series of feline ocular mycobacteriosis. Similar to the findings of Gunn-Moore et al 20 and other studies, 14,34 most cats in this cohort were young neutered adult male cats with outdoor access and predominantly nonpedigree. Feline mycobacteriosis has a geographical distribution in the United Kingdom; 20 in keeping with these previously reported trends, cats in this study with confirmed M. bovis, M. microti, and M. avium infections all came from regions of the United Kingdom where those pathogens are endemic.
The presenting ophthalmic and systemic signs of mycobacterial disease were highly varied; less than one-third of the cases presented with ocular signs only. Similar to human TB, 45 ocular signs in our study population were most commonly unilateral, at least initially. Other authors have similarly described ocular mycobacterial infections as generally unilateral, 9,14,34 although some have reported ocular findings in affected cats to be occasionally bilateral and almost symmetrical. 13 Progression of disease (ie, from unilateral to bilateral, from visual to blind, and increasing severity of ocular disease) has not been reported previously in feline veterinary ophthalmology for either TB or NTM disease. However, similar patterns of severity and progression have been described in rabbits, 12 and progression from unilateral to bilateral disease has been reported in humans. 1 Progression of ocular signs (unilateral to bilateral, visual to blind, and increasing severity of ocular disease) was recorded in 11 of the current cases, demonstrating the need for primary care clinicians to make early decisions to intervene in these cases so that mildly affected and visual eyes can be retained, whenever possible.
In the current study, ocular signs were similar to those previously reported in sporadic cases of feline ocular mycobacteriosis, 8,9,13 –15,25,33,34 with affected cats most frequently having uveitis and being blind, with corneal, eyelid, and conjunctival lesions being less common. The most characterized of these infections is the fastidious organism Candidatus ‘Mycobacterium tarwinense’, endemic to Australia, which resulted in ocular lesions in 13 of 40 cats with a similar gross appearance to those described in the current series. 34 –36 By contrast, the 2 other frequently diagnosed feline leprosy agents in Australia, Candidatus ‘Mycobacterium lepraefelis’ and Mycobacterium lepraemurium, have not been reported specifically as causing ocular disease. 34 –36
Cataracts and lens subluxation have been previously reported in cats with tuberculous mycobacteriosis. 13 Four cats (5 eyes) in the current series presented with cataracts, which typically developed secondary to uveitis, as has been reported previously in human ocular mycobacteriosis. 11,22,45 Case 7 also had lens subluxation secondary to mycobacterial uveitis. Similarly, the glaucoma observed in this study in 4 cats (6 eyes) was a likely sequel to mycobacterial uveitis. 11,22,45
Histopathological examination of the ocular tissue from these cases consistently found changes typical of the expected cellular infiltrates associated with feline mycobacteriosis. 8,10,21,24,33,37 These infiltrates were dominated by macrophages, often displaying an epithelioid phenotype. Neutrophilic inflammation was also a common feature, with occasional lymphocytic accumulation at the periphery of lesions. Histopathological examination of material from these cases was essential to confirm that lesions were mycobacterial and not due to other conditions, such as penetrating globe injuries, hypertensive retinopathy, feline infectious peritonitis, fungal infection, or neoplasia.
The most common histopathological diagnosis in the current study was of granulomatous to pyogranulomatous chorioretinitis associated with retinal detachment with or without anterior uveitis, optic neuritis, scleritis, and/or retrobulbar cellulitis. Focal granulomatous to pyogranulomatous anterior uveitis associated with no or minimal posterior segment inflammation was less common. Although previously reported in humans, 5,22,40,45 granulomatous to pyogranulomatous vitritis, optic neuritis, scleritis, episcleritis, and retrobulbar cellulitis (extraocular myositis, fasciitis) were all findings observed in this study, which have not previously been described in cases of feline ocular mycobacteriosis. 8,9,13 –15,25,33,34 Granulomatous to pyogranulomatous scleritis, episcleritis, and extraocular myositis have been described in experimental rabbits with TB. 12
Focal anterior uveal or ciliary body pyogranulomas with no or minimal posterior segment involvement has been described in humans as a form of ocular TB; 2,22 however, they have not been reported previously in cats with ocular mycobacteriosis. This feature may be an example of an early diagnosed stage of mycobacterial disease similar to iris lesions described in rabbits with an early stage of experimental TB. 12 Focal anterior uveal or ciliary body pyogranulomas can clinically resemble neoplasia and so reinforces the clinical importance of histopathological examination of lesions.
Gunn-Moore et al 21 reported rare findings of mineralization or multinucleated giant cells in nonocular mycobacteriosis, and Lamagna et al 25 also reported multinucleated giant cells with intracytoplasmic AFB on the cytological examination of a corneo-conjunctival granuloma. The current study did not identify any of those histopathological features. However, such a lack of multinucleated giant cells in nonocular lesions from cats with mycobacterial infections has previously been reported. 17,19,43 Overall, it appears that feline macrophages may infrequently form giant cells in response to mycobacteria. 8,21,26,46
AFB with mycobacterial morphology were present in 95% of the 21 histopathological samples from the current cases; this is much higher than has been reported in a previous study of feline mycobacterial lesions. 21 The reasons for this are not clear. It may be an artifact of the relatively small sample size in the present study. However, the examination of tissues by an experienced pathologist and a high index of suspicion for the disease may also play an important role in identifying mycobacteria in tissues, particularly those from paucibacillary infections. Gunn-Moore et al 21 reported that AFB were found by an experienced pathologist in 47% of 45 cases compared to under a third where the experience of the examining pathologists was unknown. 18
In the current cases, the IGRA was able to identify the mycobacterial species in all 9 cats tested. Although contested in human TB studies, 4 Rhodes et al 42 suggested that the IGRA could potentially be used to monitor treatment in cats. Interestingly, repeated IGRA tests in 2 affected cats in this study became negative after treatment.
That 14 of the 25 cats in the current study were euthanized without treatment (56%) suggests that veterinarians may perceive ocular mycobacteriosis to have a poor prognosis, although they also cited compromised animal welfare, patient and/or owner noncompliance, and/or public health concerns regarding zoonotic risk as reasons in their decision making. Certainly, a poor response to treatment was not seen in this study, where 8 of the 10 cases treated with surgery and antibacterial intervention achieved remission (80%). The cat that came out of remission after 8 months had only been treated with a short (2-month) course of dual therapy (which omitted rifampicin), supporting the recommendation for a longer course of appropriate triple therapy. All cats that underwent a surgical intervention alone were either euthanized (n = 10) or lost to follow-up (n = 1), and the time to euthanasia was notably short, with only 1 cat (case 22) surviving beyond 294 days.
These data are similar to those reported by Gunn-Moore et al, 21 where 87% of cats with mycobacteriosis underwent either surgery and/or medical therapy, although in that study, only 42% gained remission. However, few cats in that study 21 were treated appropriately; most received suboptimal drug regimens, such as short (1-month) courses of fluoroquinolone monotherapy. Where appropriate treatment has been undertaken, success rates approaching 90% have been achieved with some cutaneous NTM infections, 34 and ∼70% remission has been recorded in cases of cutaneous TB with or without secondary pulmonary involvement, 37 findings that are in line with the data shown in this cohort. In the current study, most of the cats reaching and sustaining remission underwent enucleation or incisional biopsy, followed by 5 months or more of treatment with appropriate triple antibiotic therapy (although this was then reduced to double therapy in 2 cases). These findings suggest that ocular mycobacterial disease in cats should be considered a treatable manifestation of mycobacterial disease where intensive treatment (ie, combined surgical and prolonged medical treatment) is considered viable.
While the majority of cases presented with some systemic signs (19/25; 76%), disseminated mycobacterial infection was demonstrated in only 9 cats (9/25; 36%). However, due to the retrospective nature of this study, investigations were not conducted in a standardized manner, so the true number of cats with disseminated mycobacteriosis could have been higher. While Gunn-Moore et al 20 reported that at presentation, systemic signs such as weight loss, lymphadenopathy, and/or respiratory disease were seen in comparatively fewer cases (in only 10%–16%), that case series included a predominance of cutaneous disease, which is perhaps likely to be discovered earlier in the disease process than ocular disease, and so have less time to progress before diagnosis is made. The posterior segment of the globe was most commonly affected in the current cases, and this is consistent with hematogenous spread to the eye, even though evidence of clinical disease elsewhere may not have been apparent at the time of diagnosis. In contrast, the cases of focal granulomatous to pyogranulomatous keratitis and/or conjunctivitis were considered most likely secondary to trauma, although it is difficult to exclude hematogenous spread to these sites completely. In agreement with previous work, 19 only 2 of the 7 cats in the current cohort with respiratory mycobacteriosis showed signs of respiratory disease. This finding reinforces the existing recommendation that thoracic radiography (or CT) be performed in all cases with suspected or confirmed mycobacteriosis. 16
The limitations of the current study included variability in the availability of clinical data and incomplete diagnostic evaluations of each patient. Despite this, this is the largest study of feline ocular mycobacteriosis to date, and it adds significantly to our understanding of the condition.
In conclusion, feline ocular mycobacteriosis can involve any structure of the eye and can occur with or without concurrent systemic disease and in the absence of classically recognized immune suppression. This case series of 25 cats demonstrates the importance of considering mycobacterial disease as a differential diagnosis for feline ocular disease, especially in countries with endemic mycobacteriosis. As such, ocular examination should be a part of the diagnostic approach to all cases of systemic feline mycobacteriosis. The main histopathological features identified were granulomatous to pyogranulomatous chorioretinitis with retinal detachment with or without anterior uveitis, optic neuritis, scleritis, episcleritis, and/or retrobulbar cellulitis. The presence of indicative AFB with mycobacterial morphology was useful to confirm the initial diagnosis, while IGRA testing, in combination with the history and clinical signs, was a useful confirmatory test. Combining these tests can prevent delays in making diagnoses, unnecessary suffering, and the administration of inappropriate treatments. Eight cases responded favorably to systemic medication following surgery and achieved remission, with 7 of them maintaining remission. Clinicians should be aware that good clinical outcomes can be achieved in these cases where intensive therapy is possible.
Supplemental Material
Supplemental Material, DS1_VET_10.11770300985819844819 - Feline Ocular Mycobacteriosis: Clinical Presentation, Histopathological Features, and Outcome
Supplemental Material, DS1_VET_10.11770300985819844819 for Feline Ocular Mycobacteriosis: Clinical Presentation, Histopathological Features, and Outcome by Renata Stavinohova, Conor O’Halloran, Jonathan Richard Newton, James Andrew Clive Oliver, Emma Scurrell and Danièlle Audry Gunn-Moore in Veterinary Pathology
Footnotes
Acknowledgements
We thank all of the owners who gave a consent to include a case into this study; all of the veterinary clinics, surgeons, nurses, and reception staff who helped in this study; and Debenham Vets, Donaldson’s Vets Ltd, Eye Veterinary Clinic, Marches Veterinary Group, Rowe Referrals Eye Clinic, South Devon Referrals, and Drs. C. Chiwitt, S. Farr, N. Fisher, M. Karczewska, T. Knott, A. Muguet-Chanoit, R. Non Linn-Pearl, D. Nutbrown-Hughes, M. Rhodes, L. Webb, and P. Wilson. Figures are courtesy of Dr. P. Wilson (Fig. 1), Dr. M. Ames (Fig. 2), Dr. A. Muguet-Chanoit (Fig. 3), Davies Veterinary Specialists (Fig. 4), Dr. C. Chiwitt (Fig. 6), and Animal Health Trust (![]() ).
).
Author Contribution
Emma Scurrell and Danièlle Audry Gunn-Moore contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Conor O’Halloran is supported by a Biotechnology and Biological Sciences Research Council (BBSRC) studentship BB/M014894. The remaining authors received no financial support for the research, authorship, and/or publication of this article. Animal Health Trust (Ophthalmology Department) provided financial assistance toward publication.
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
