Abstract
To investigate cases of acute oxalate nephrosis without evidence of ethylene glycol exposure, archived data and tissues from cheetahs (Acinonyx jubatus) from North America (n = 297), southern Africa (n = 257), and France (n = 40) were evaluated. Renal and gastrointestinal tract lesions were characterized in a subset of animals with (n = 100) and without (n = 165) oxalate crystals at death. Crystals were confirmed as calcium oxalate by Raman spectroscopy in 45 of 47 cheetahs tested. Crystals were present in cheetahs from 3.7 months to 15.9 years old. Cheetahs younger than 1.5 years were less likely to have oxalates than older cheetahs (P = .034), but young cheetahs with oxalates had more oxalate crystals than older cheetahs (P < .001). Cheetahs with oxalate crystals were more likely to have renal amyloidosis, interstitial nephritis, or colitis and less likely to have glomerular loop thickening or gastritis than those without oxalates. Crystal number was positively associated with renal tubular necrosis (P ≤ .001), regeneration (P = .015), and casts (P ≤ .001) but inversely associated with glomerulosclerosis, renal amyloidosis, and interstitial nephritis. Crystal number was unrelated to the presence or absence of colitis and was lower in southern African than American and European animals (P = .01). This study found no evidence that coexisting chronic renal disease (amyloidosis, interstitial nephritis, or glomerulosclerosis), veno-occlusive disease, gastritis, or enterocolitis contributed significantly to oxalate nephrosis. Oxalate-related renal disease should be considered as a potential cause of acute renal failure, especially in young captive cheetahs. The role of location, diet, stress, and genetic predisposition in the pathogenesis of oxalate nephrosis in cheetahs warrants further study.
Since the early 1970s, worldwide sporadic cases of oxalate nephrosis have been documented in captive cheetahs and other large felids. 21,37,42,63,72,76,77 Oxalate nephrosis has also been recorded in other nondomestic mammals including mink 67 and koalas. 74 Whereas small numbers of oxalate crystals are relatively common in cheetahs dying of glomerulosclerosis and/or renal amyloidosis, large numbers of oxalate crystals and associated tubular pathology have been described in cheetah kidneys, without other renal disease being present (KAT, KL, EPM, and L. Munson, personal communication). Approximately 10% of captive cheetahs in the United States and South Africa were estimated to have oxalate crystals in renal tubules at necropsy (KAT and EPL, unpublished data). This warranted further investigation of possible risk factors for renal oxalate formation given the vulnerable conservation status of this species. 22
Endogenously produced oxalate is a metabolic by-product of amino acid (hydroxyproline, glycine, and serine) and vitamin C catabolism. 5,11,33 Oxalate nephrosis results from intratubular calcium oxalate deposition when calcium oxalate burdens exceed excretory capacity, 5,11 either due to genetic defects in glyoxylate metabolism that result in increased hepatic endogenous production of oxalate or secondary or acquired disease. 5,10,11,32 Most human forms of primary hyperoxaluria occur due to mutation of the pyridoxine (vitamin B6) dependent enzyme alanine: glyoxylate aminotransferase (AGT) or the enzyme glyoxylate/hydroxypyruvate reductase (GRHPR). 5,44,66 Familial tendencies, a point mutation in the GRHPR gene, and associated reduced enzyme function have also been identified in some domestic cats with oxalate nephrosis. 6,15,27,49
Secondary or acquired oxalate nephrosis can be due to severe liver disease 46 or pyridoxine deficiency, 5,7,24,65 which results in compromised oxalate metabolism, or increased absorption of oxalates due to ingestion of oxalates (or their precursors) including plants, 2,3,11,40,58 ethylene glycol and other glycols, 1,10,11,31,50 xylitol, 13 ascorbic acid, 4,47,48,55 and collagen or feathers. 19,85 An altered intestinal microbiome, 43,69 excessive bile or long-chain fatty acids, 10,56 or low intestinal calcium content, 33 enterocolitis, 5,10 and intestinal surgical resection 9,32 can also increase oxalate absorption and result in oxalate crystal deposition in the kidney. Ethylene glycol toxicity was considered, although never proven, 72,76 in cheetah cases with large numbers of crystals but was thought unlikely in cases from the United Arab Emirates 37 and southern Africa, where ethylene glycol (antifreeze) is less widely utilized. Toxicological evaluation of feed and water supplies in several cases from the United States, southern Africa, and France have failed to find evidence of contaminating ethylene glycol (D.G.A. Meltzer, M. Kinsel, and S. Terrell, personal communications). 42 Similarly, ingestion of oxalate-rich plants or sweeteners, pancreatic insufficiency, ascorbic acid toxicity, high fat or protein diets, and pyridoxine deficiency were considered unlikely primary causes in cheetahs given the widely differing environments and management practices in the United States, southern Africa, and France.
However, captive cheetahs suffer from renal diseases such as glomerulosclerosis and amyloidosis, 8,51,59 which could result in increased oxalate deposition due to reduced glomerular filtration and urinary pH, pyridoxine depletion, and hypercalcemia due to secondary hyperparathyroidism. 5,10,11,53,57 Cheetahs also suffer from gastrointestinal tract and liver disease, 51,52 which could be risk factors for oxalate formation. This retrospective pathologic study was conducted to describe the lesions in cheetahs with and without oxalates in their kidneys and to investigate the relationship between oxalate crystal deposition and concurrent renal, hepatic, or gastrointestinal tract disease.
Materials and Methods
Study Animals
Cases were selected from pathology databases of cheetahs kept as part of the North American Association of Zoos and Aquariums’ Species Survival Plan (n = 297) and from cheetah pathology databases in southern Africa (n = 257) and France (n = 40). Tissues from cheetahs over 1 month old that died between 1990 and 2013 were used as the study population. Histologic sections of kidneys from all cheetahs were screened using polarized light for the presence or absence of crystals. A subset of cheetahs greater than 1 month old that died between 1990 and 2013 (n = 265) was selected for detailed histologic examination based on the quality and availability of relevant histologic material. Cases were defined as cheetahs with renal oxalates at death (n = 100). Controls were defined as cheetahs with no renal oxalates at death (n = 165) and similar demographic characteristics to cases including age, sex, housing facility at death, and year of death; however, matching was approximate and not possible in all instances. Geographic distribution of the final data set of cases and controls was North America (n = 94; 40 cases, 54 controls), southern Africa (n = 167; 56 cases, 111 controls), and France (4 cases, no controls). All of the North American and French cheetahs and all but 2 of the southern African cheetahs were captive at the time of death.
Pathologic Evaluation
Formalin-fixed tissues were processed routinely and stained with hematoxylin and eosin and von Kossa and Masson’s trichrome stains. 21 Crystal number and the presence and severity of renal lesions, veno-occlusive disease (VOD), gastritis, enteritis, and colitis were evaluated by two pathologists (KAT and EPM) using a detailed scoring scheme (Supplemental Table S1) and previously published criteria. 8,51,59 A subset of southern African cases was examined by both KAT and EPM to confirm evaluator concordance.
Raman Spectroscopic Characterization
Archived, formalin-fixed paraffin-embedded kidney sections from cheetahs (n = 47; 28 North American, 15 southern African, 4 French) were used for Raman spectroscopic characterization. Unstained, deparaffinized sections (6-8 µm) on quartz slides (25 × 6 × 1 mm3) (SPI, West Chester, PA, USA) were examined by light microscopy for crystal presence and location. A case of confirmed canine ethylene glycol toxicity was used as a control. Raman spectroscopy measurements on the kidney sections were performed on a Nicolet Almega XR Raman spectrometer equipped with an Olympus BX51 confocal microscope (Thermo Electron, Madison, WI, USA). The camera temperature was set at –50°C through the instrument control software OMNIC 8 for Dispersive Raman. The complimentary Atlµs program was employed for imaging the samples and focusing on the birefringent crystals in the tissue. Spectra were obtained with a 532-nm solid state, diode pumped laser (24 mW) at 100% power, 50-µm pinhole aperture, 2400 lines/mm resolution grating, spectral range of 2000 to 300 cm−1, and a 100× Olympus objective. Individual spectra were acquired with a 20-second interrogation time and three exposures after a 30-second photobleach. All spectra were baseline corrected using the OMNIC software. Library comparison for spectra was performed using the OMNIC software and spectral libraries purchased from Thermo Electron. Specifically, the HR Raman Inorganics Library (containing a calcium oxalate spectrum) as well as additional spectra obtained from a known ethylene glycol poisoning in a dog (07N-0544-Ti, US Food and Drug Administration) were used for confirmation that a spectrum obtained from a crystal was a spectral match with calcium oxalate. Additional spectral processing, including peak identity evaluation, was performed with Spectrus Processor 2012 (ACD/Labs, Toronto, Ontario, Canada).
Statistical Analysis
Because matching of cases (n = 100) and controls (n = 165) was approximate only in this retrospective study, unmatched analysis was performed. Age of animals at death was categorized as < 1.5, 1.5 to < 8, 8 to < 11, and ≥ 11 years old. Univariate analyses were conducted to compare lesion prevalence and severity in cases and controls (Supplemental Table S2) as well as to compare lesion prevalence and severity with crystal number (Supplemental Table S3). Renal edema, hemorrhage, arteritis, fibrinoid necrosis, thrombosis, cortical and medullary intratubular mineralization, the presence of other crystals in tubules, intratubular and intracellular pigment in tubules, tubulitis, and VOD were excluded, as these lesions were uncommon to rare. The cellular characteristics of interstitial inflammation were excluded, as this was invariably lymphoplasmacytic. The association between the presence of oxalate crystals and the presence and severity of each remaining histologic lesion (case/control status) was then assessed using mixed-effects logistic regression (Table 1). First, all potential predictors associated with the outcome with P < .20 in the initial analysis were included and sequentially eliminated until all remaining predictors were significant (P < .05). Every candidate predictor was then retested one-by-one in the model and retained if significant. The scores for renal amyloidosis, interstitial inflammation, and gastritis were collapsed into dichotomous (0/1) scores indicating the presence or absence of each lesion, since the original ordinal scores did not contribute any additional information. Odds ratios were generated describing the strength of association between the presence and absence of crystals and each lesion.
Lesions Significantly Associated With the Presence of Renal Oxalate Crystals in Cheetahs.a
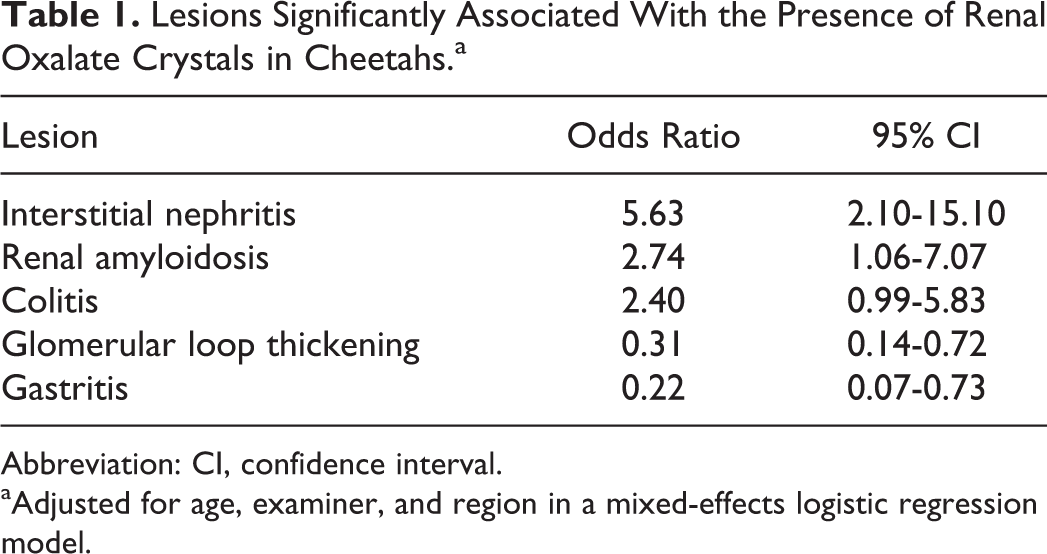
Abbreviation: CI, confidence interval.
aAdjusted for age, examiner, and region in a mixed-effects logistic regression model.
Second, where oxalate crystals were present (n = 100), the association of each lesion score with crystal number (average number of cortical tubules containing crystals in three 100× fields) was assessed using mixed-effects negative binomial regression, adjusting for age, examiner, and institution (random effect). Factors potentially associated with the severity of oxalate nephrosis were assessed using a mixed-effects negative binomial regression model, with the same candidate variables and modeling approach as above. All statistical analyses were done using Stata 14 (StataCorp, College Station, TX, USA). Significance was assessed at P < .05. The scores for renal amyloidosis were collapsed into dichotomous (0/1) scores indicating the presence or absence of amyloidosis in the final multiple negative binomial regression model of factors associated with the numbers of crystals present (Table 2). Negative binomial regression in this context yields a count ratio (CR), interpreted as the ratio of the crystal number in a category with the lesion to the crystal number in the reference category (without the lesion); therefore, a CR < 1 indicates a negative or inverse association and a CR > 1 indicates a positive association with crystal number.
Age, Region, and Lesions Associated With the Number of Crystals in Cheetah Kidneys.a
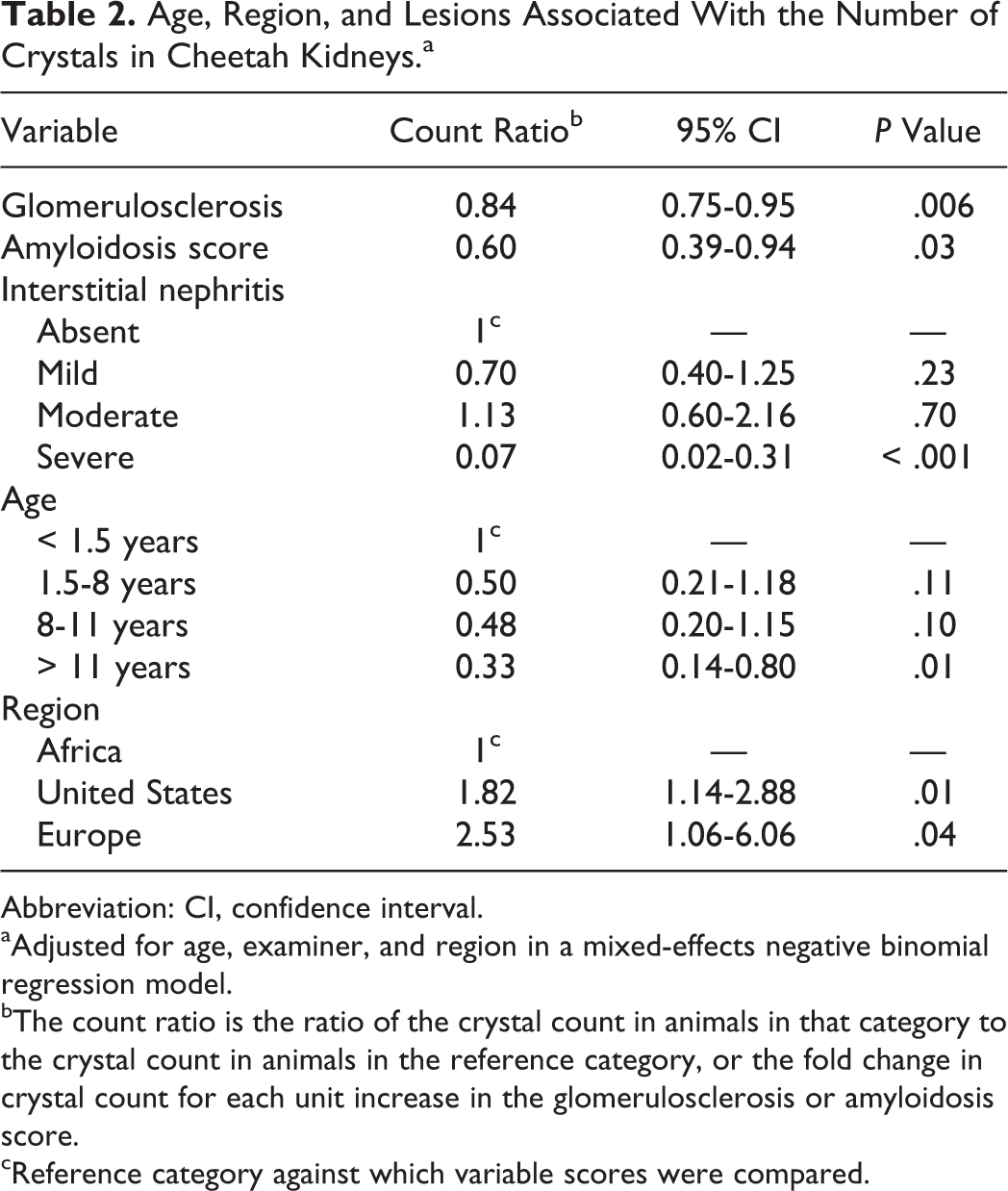
Abbreviation: CI, confidence interval.
aAdjusted for age, examiner, and region in a mixed-effects negative binomial regression model.
bThe count ratio is the ratio of the crystal count in animals in that category to the crystal count in animals in the reference category, or the fold change in crystal count for each unit increase in the glomerulosclerosis or amyloidosis score.
cReference category against which variable scores were compared.
Results
Histopathology
Histologically, kidneys from cheetah cases contained small to very large numbers of colorless refractive crystals (average 0.3-81 crystals in three 100× fields) in cortical and medullary tubules forming rosettes, globules, and acicular fragments (Fig. 1) that were birefringent with polarized light (Fig. 2). Crystals were not uniformly distributed in the renal cortex, with clusters apparently occurring in single or groups of tubules; crystals were not associated with tracts of inflammation. Affected tubules contained small amounts of sloughed necrotic cellular debris mixed with variable amounts of pale amorphous eosinophilic material and, in many cases, were lined by a discontinuous layer of epithelial cells with variable degrees of necrosis characterized by shrunken cells with hypereosinophilic cytoplasm and dark basophilic pyknotic nuclei (Fig. 1). Adjacent tubules were variably dilated and lined by regenerative low cuboidal epithelial cells with pale basophilic cytoplasm and large crowded oval nuclei (Fig. 3). Variable degrees of mild interstitial fibrosis and small numbers of intratubular cellular casts were present. Crystals and fine mineral deposits variably present on tubular basement membranes in the cortex and medulla stained variably positive with von Kossa, which was interpreted as the presence of calcium phosphate and/or carbonate salts, since melanin pigment was not visible in crystals or on tubular basement membranes on hematoxylin and eosin stains (Fig. 4). Renal lesions were present histologically in 46 of 165 (28%) cheetahs without oxalate crystals and in 87 of 100 (87%) cheetahs with oxalate crystals.
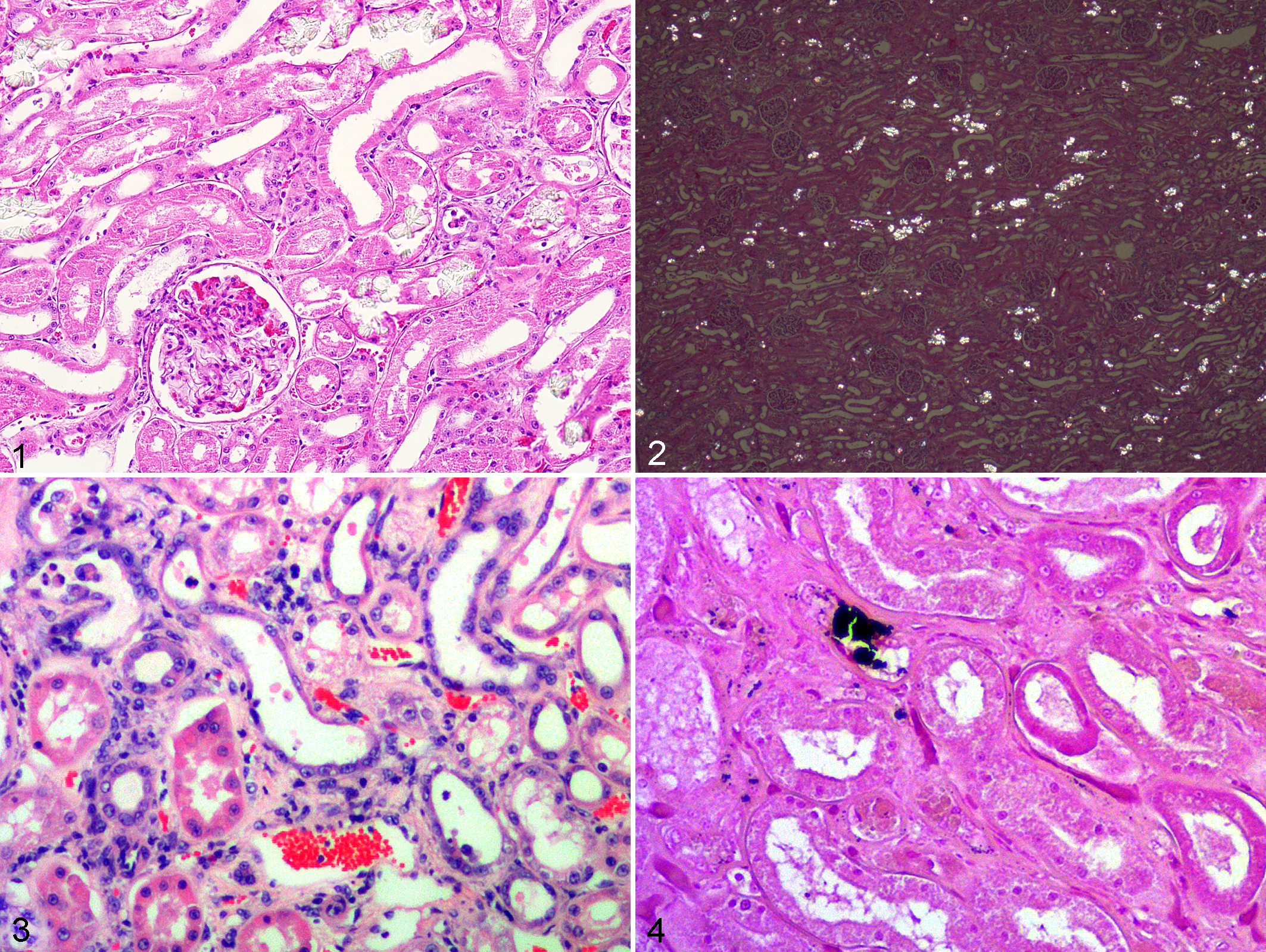
Additional renal lesions included small numbers of deeply basophilic mineralized crystals in the lumina of both cortical and medullary tubules, rare pale eosinophilic or yellow pigment in renal tubular epithelial cells or in tubular lumina, and variable microvesiculation consistent with lipidosis or hydropic degeneration in renal tubular epithelial cell cytoplasm. Tubulitis, renal edema or hemorrhage, arteritis, and vascular fibrinoid necrosis were rarely noted.
Raman Spectroscopy
On Raman spectroscopic examination, crystals were easily observed as bright spheres in the dark tissue background. Crystals in the control case of canine ethylene glycol poisoning and in 27 (96%) North American, 14 (93%) southern African, and 4 (100%) French cases were confirmed as calcium oxalate with no laser photodegradation of the crystals (Fig. 5). Laser photodamage prevented crystal identification in 2 cheetahs (1 North American and 1 southern African). In addition to calcium oxalate crystals, calcium carbonate, calcium phosphate, or calcium sulphate crystals were present in the kidney tubules of 3 North American cheetahs, and 1 additional North American cheetah sample had a Raman multicomponent spectrum that contained calcium oxalate bands. All of these cheetahs were > 10 years of age and had other underlying renal disease including glomerulosclerosis and amyloidosis.
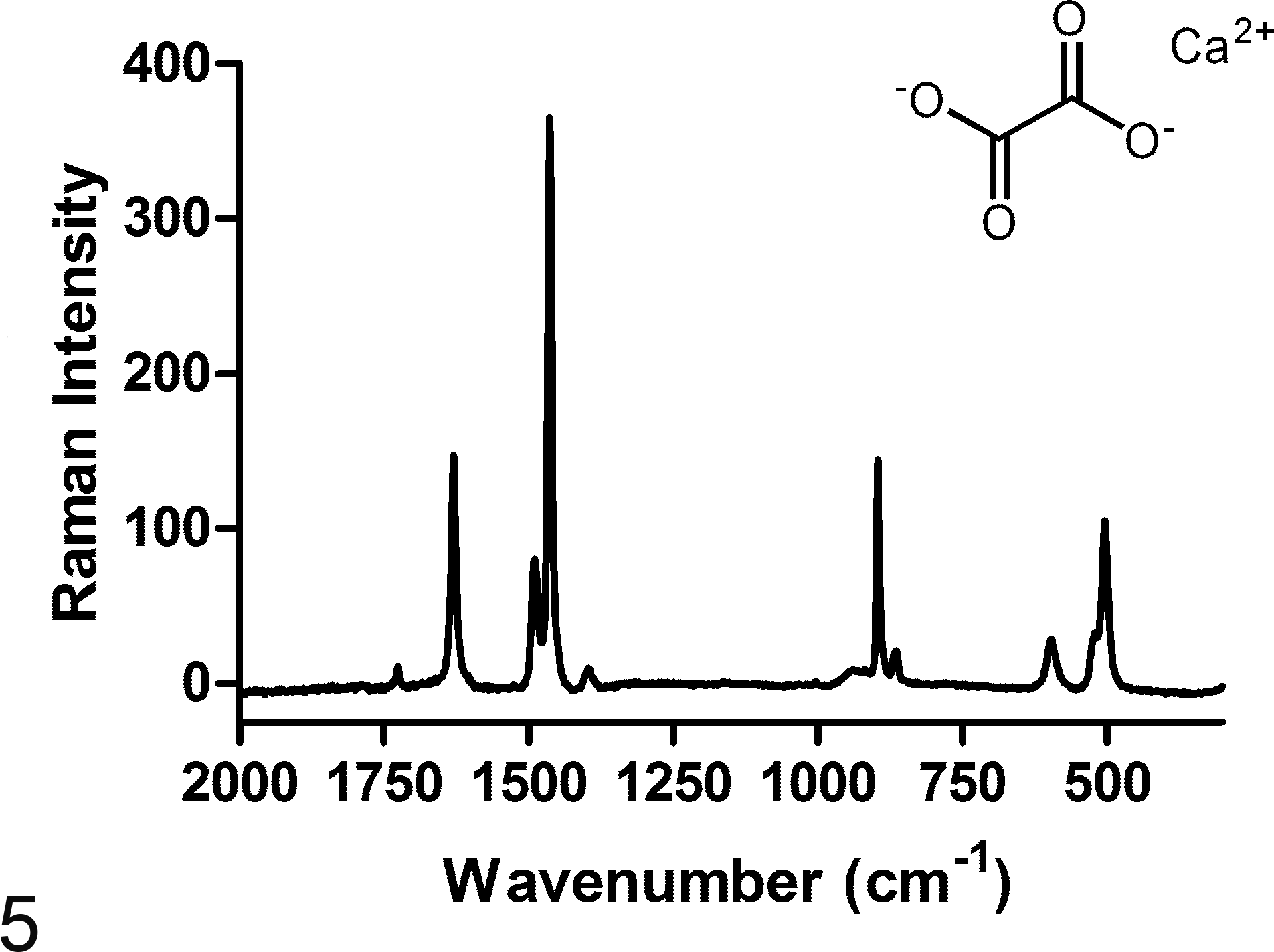
Representative Raman spectrum of a crystal from an 8-year-old captive female cheetah from South Africa. Characteristic bands for calcium oxalate monohydrate are assigned to the C-O asymmetric stretch (1628 cm–1), C-O symmetric stretch (1490 and 1463 cm–1), C-C stretch (896 cm–1), and O-C-O in-plane bending (502 cm–1).
Statistical Analysis
The presence and numbers of crystals were not significantly different between male and female cheetahs (P = .454). The average age of cheetahs with renal oxalate crystals at death was similar in the North American, French, and southern African populations (3.7 months-15.9 years old; median [interquartile range] = 8.7 [6.4-11.3] years). Age distribution was similar in cheetahs without oxalates (median [interquartile range] = 8.6 [3.6-11.3] years). Cheetahs older than 1.5 years were much more likely to have oxalates than younger cheetahs (P = .034, Supplemental Table S2), but cheetahs < 1.5 years old had significantly more crystals than older cheetahs (P < .001, Supplemental Table S3). Although 3 young cheetahs from 1 North American institution had very high numbers of crystals (average of 69-81 per three 100× fields), no grouping of crystal number based on age was present (Fig. 6). On the multivariate mixed-effects negative binomial regression analysis, cheetahs > 11 years old had significantly fewer crystals than those < 1.5 years old (P = .01, Table 2).
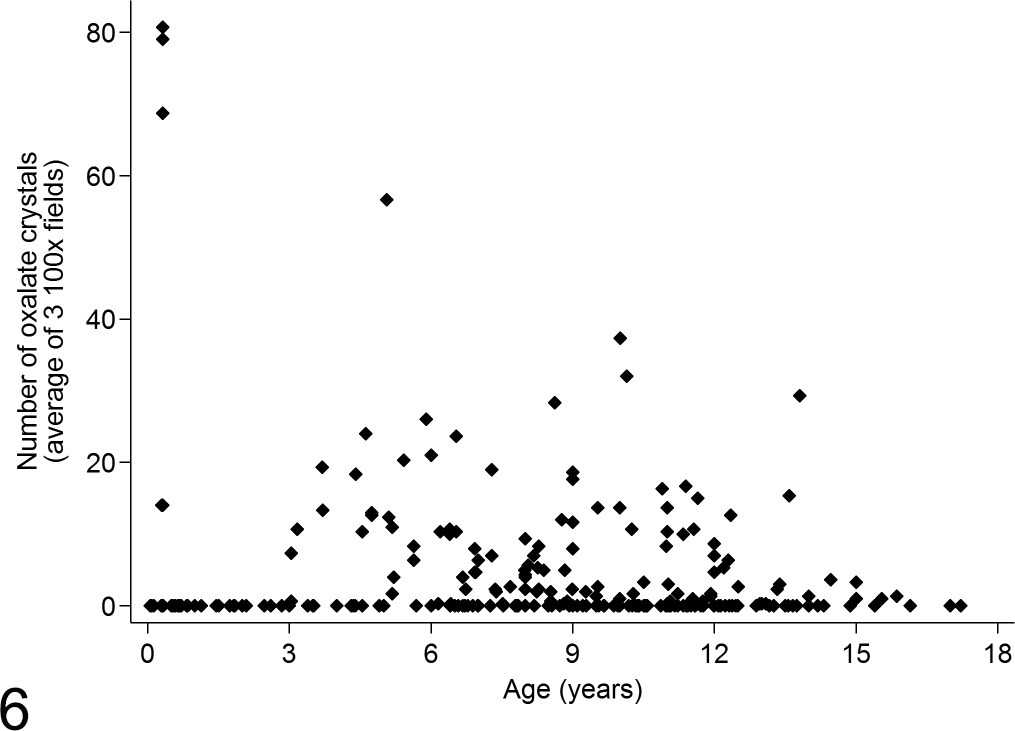
Association between cheetah age and average number of oxalate crystals in renal tubular lumina.
When comparing cheetahs with renal oxalate crystals (cases) with those without such crystals (controls), tubular changes were significantly associated with the presence of crystals (Supplemental Table S2). Cases had significantly more renal tubular necrosis, regeneration, dilatation, and protein and cellular casts than controls (P ≤ .001 for each parameter). Compared with controls, cases also had significantly more lymphoplasmacytic interstitial nephritis (P ≤ .001), amyloidosis (P ≤ .001), chronic infarcts (P ≤ .001), efferent arteriole arteriolosclerosis (P ≤ .001), arcuate artery arteriosclerosis (P = .011), cortical (P = .02) and medullary (P = .005) tubular basement membrane mineralization, cortical fibrosis (P = .049), hyperplastic collecting tubule epithelium (P = .063), enteritis (P = .001), and colitis (P = .017). In contrast, gastritis was significantly more common in controls than cases (P = .012). Sex (P = .899), glomerulosclerosis (P = .952), glomerular loop thickening (P = .273), glomerular hypercellularity (P = .743), and medullary fibrosis (P = .898) had no effect on the likelihood of having renal oxalate crystals. In the 250 cheetahs for which liver was available, hepatic VOD was present in 22 of 88 (25%) cases and 32 of 162 (20%) controls. In addition, 6% of cheetahs without oxalates but only 3% of cheetahs with oxalates had severe VOD. In the multivariable mixed-effects logistic regression analysis comparing cases and controls, the only factors associated with an increased likelihood of having renal oxalates were interstitial nephritis and, to a lesser extent, renal medullary amyloidosis and colitis (Table 1). Case cheetahs had reduced odds of having glomerular loop thickening and gastritis compared with controls.
When crystal number was evaluated in cheetahs with oxalate crystals in the kidney (Supplemental Table S3), the number of crystals present was positively associated with tubular necrosis (P ≤ .001), regeneration (P = .015), cellular casts (P ≤ .001), glomerular hypercellularity (P = .007), and arteriosclerosis of the efferent arteriole (P ≤ .001). Crystal number was inversely associated with protein casts (P = .043), medullary tubular basement membrane mineralization (P = .002), collecting duct epithelial hyperplasia (P = .023), glomerulosclerosis (P = .009), glomerular loop thickening (P ≤ .001), amyloidosis (P ≤ .001), chronic infarcts (P ≤ .001), renal cortical (P ≤ .001) and medullary (P = .002) fibrosis, enteritis (P = .001), and colitis (P = .019). In 31 cheetahs with large numbers of oxalates (average of 10-81 per three 100× fields), there was no evidence of any underlying renal disease. Crystal number was unrelated to sex (P = .455), tubular dilatation (P = .409), cortical tubular basement membrane mineralization (P = .382), arteriosclerosis of the arcuate artery (P = .118), or gastritis (P = .826). The relationship between crystal number and interstitial nephritis was significant but nonlinear (P ≤ .001), since cheetahs with grade 2 nephritis had more crystals than those with nephritis grades 0, 1, and 3. In the multivariable mixed-effects negative binomial regression analysis, geographic region was the only factor positively associated with crystal number (Table 2). On average, cheetahs from the United States and France had significantly more oxalate crystals than those from Africa (P = .01). Nine of the 10 cheetahs with the highest average number of cortical tubules containing oxalate crystals (26-81) were from North America (5 different institutions), and the remaining 1 was from a South African institution (24). Among the 10 southern African cheetahs with the highest number of crystals (14-24), cheetahs from 1 institution were overrepresented (50%). The 4 cheetahs from 3 French institutions had an average number of cortical tubules containing crystals of 15 (10-20). Glomerulosclerosis and amyloidosis were negatively associated with crystal number in the multivariate analysis (Table 2).
Discussion
This study found that the presence of intratubular renal oxalate crystals was associated with renal tubular necrosis, regeneration, and the presence of tubular casts in cheetahs in North America, southern Africa, and France and that increased numbers of crystals were associated with increased severity of these tubular changes. This effect of oxalate crystals on tubular epithelium is well documented in humans and animals. 11,16,29,44,49,66,78,83
Raman spectroscopy confirmed that the majority of crystals were calcium oxalate. Melamine and other renal crystals were ruled out because the Raman spectral fingerprints did not match these compounds. 38,61,68 The 2 cases in which oxalate crystals were not found were cheetahs with only a few crystals in the hematoxylin and eosin stained sections, and it is possible that sections prepared for spectroscopy contained no crystals or that they were damaged by the laser. Calcium carbonate, calcium phosphate, or calcium sulphate crystals, present in the kidney tubules of 3 North American cheetahs, were not distinguishable from calcium oxalate on routinely stained sections but were readily identifiable by their Raman spectra. Therefore, whereas calcium oxalate monohydrate was the predominant crystal noted, other crystal types can occur in cheetahs. Compound oxalate crystals have also been described in domestic dogs. 64
Oxalate crystals and associated tubular damage occurred in 31 cheetahs without other renal diseases, all of which were reported to have clinical evidence of renal failure (inappetance, weakness, vomiting, azotemia, dehydration, polyuria, and depression) and showed varying degrees of parathyroid gland hyperplasia and metastatic mineralization but no other renal disease. Similar findings have been described previously in cheetahs 37,42,76 and other animals with oxalate nephrosis. 14,21,23,28,29,34,35,41,67,73 –75 Oxalate nephrosis, therefore, occurs independently of other renal diseases in some captive cheetahs and should be considered in the differential diagnosis of renal disease. Although rare scattered oxalate crystals may occur secondary to many types of chronic renal disease, 11 11 cheetahs with chronic renal disease also had large numbers of crystals, suggesting that oxalate nephrosis may coexist with other renal diseases at least in some cheetahs. Tubular lesions, glomerular hypercellularity, and efferent arteriolosclerosis could contribute to oxalate crystal formation in chronic renal disease, as these were the only renal lesions positively associated with crystal number in these cheetahs. Whereas a few oxalate crystals may occur secondary to concurrent chronic renal disease in some cases, factors other than concurrent renal disease likely affect renal oxalate deposition in cheetahs.
No clear evidence was found in our study that oxalate nephrosis occurs secondary to gastrointestinal or hepatic disease in cheetahs. Gastritis was more common in controls than cases and was unrelated to crystal number. On the univariate analysis, more cheetahs with renal oxalate crystals had enteritis and colitis than controls, and on the multivariate analysis, colitis was associated with an increased likelihood of having renal oxalates. However, both lesions were inversely associated with crystal number and therefore likely do not play a key role in the development of oxalate nephrosis. In humans, fat malabsorption due to enterocolitis or pancreatic insufficiency may result in reduced intestinal calcium, which increases intestinal oxalate absorption because calcium binds intestinal oxalates. 5,10,46,56,81 However, no relationship between enterocolitis and renal oxalate crystal number was found and pancreatic insufficiency is rare in the parent population of cheetahs. 51,52 Veno-occlusive disease was uncommon in animals with and without renal oxalate crystals. Increased intestinal absorption of oxalates may also result from loss of intestinal lactic acid bacteria, including Oxalobacter formigenes, that degrade oxalate into carbon dioxide and formate. 43,46,70,82 Research is needed to determine whether or not cheetahs that receive broad-spectrum antibiotic therapy for Helicobacter-associated gastritis or other infections have depleted O. formigenes populations, which may predispose them to oxalate nephrosis. 36
Significant geographic and institutional clustering of cheetahs with abundant renal oxalate crystals suggests that genetic, dietary, or management factors may influence the prevalence of oxalate nephrosis in captive cheetahs. So far, no proof of ethylene glycol toxicity has been found in cheetahs, but exposure to ethylene glycol, xylitol, or other oxalate precursors should be thoroughly investigated in cases of oxalate nephrosis. Identification of dietary factors was beyond the scope of this retrospective necropsy study. However, evaluation of the fluid intake and protein, fat, collagen, feathers, calcium, magnesium, beet pulp, pyridoxine, ascorbic acid, and arachidonic acid levels in captive cheetah diets is needed as these factors can influence oxalate excretion. 17 –20,22,32,33,46,60,71,84 Dietary calcium to oxalate ratios affect O. formigenes colonization of the intestine in rats, so this ratio may be important in cheetah diets. 43 Obesity and stress may contribute to oxalate nephrosis in humans, 54 and their roles in oxalate nephrosis may warrant further study since obesity may be present in underactive captive cheetahs and stress has been documented in captive animals. 79
Primary hyperoxaluria (PH) in humans is an autosomal recessive condition associated with mutations in 3 genes involved in oxalate metabolism: AGT gene in PH type 1, GRHPR gene in PH type 2, and 4-hydroxy-2-oxoglutarate aldolase (HOGA1) in PH type 344. Similarly, both PH type 1 and PH type 2 have been described in domestic cats. Clear evidence that oxalate nephrosis in cheetahs is a primary genetic disease was not found in our study; however, we did not test genetic relatedness. In humans and cats with inherited oxalate nephrosis, the disease is seen in juveniles. 6,15,16,27,49,78 In this study, although cheetahs < 1.5 years old had significantly more crystals than older cheetahs, which might indicate an inherited predisposition, relatively large numbers of crystals were seen in cheetahs of all ages. Lesions that are characteristic of primary disease in humans and cats, including granulomatous nephritis associated with interstitial oxalates, widespread tissue oxalate deposition, 10,12,45,66,80 and neurological disease, 30,52 were not seen in the cheetahs in this study. Manifestation of genetic disease is complex with environmental influences, multigenetic inheritance, epigenetic effects, incomplete penetrance, and variable expression resulting in differing disease severity and phenotypes. 30 Therefore, whether variations in crystal number are due to differing manifestations of the same disease or differences in pathogenesis among cheetahs in this study is uncertain. Since US, southern African, and French cheetah populations share common founders, a detailed genetic analysis, including sequencing of key genes involved in oxalate metabolism and resultant enzyme activity, is needed to determine whether or not cheetahs suffer from a genetically limited capacity for glyoxylate metabolism. 48
Early diagnosis of oxalate nephrosis is important since this disease is potentially treated by fluid therapy to reduce oxaluria, 44,85 pyridoxine to stabilize and enhance the activity of AGT, 5,7,24,65 urine alkalinization, 66,85 and n-3 fatty acid supplementation to decrease urinary oxalate excretion and free-radical injury. 5,25,39,46,57,62,66,70 Urinalysis data were limited to a few cases in this retrospective study, but urinalysis could be a valuable diagnostic tool since small numbers of oxalate crystals may be present in the urine of affected cheetahs. 31 However, confirming the presence of oxalate nephrosis and differentiating it from concurrent glomerulosclerosis or renal medullary amyloidosis in azotemic cheetahs are not simple if urinary oxalates are absent. Renal ultrasonography findings are nonspecific in domestic dogs and cats with oxalate nephrosis. 1 Computed tomography scans are used to identify renal crystals in humans 5 but are not a practical diagnostic modality in cheetahs. At one Southern African institution, urinary oxalate was elevated in an acutely azotemic cheetah later diagnosed with oxalate nephrosis without other renal lesions (8000 mg oxalate/g creatinine, compared with 128 mg/g in a healthy enclosure mate; F. Reyers, Golden VetPath, unpublished data). Since the crystal number was higher in young cheetahs and chronic renal disease in cheetahs is age related, 26,51 oxalate nephrosis should be a primary consideration in younger cheetahs with renal failure.
In conclusion, this study found that oxalate nephrosis unrelated to ethylene glycol toxicity occurs in cheetahs. We found no convincing evidence that oxalate nephrosis in captive cheetahs was secondary to renal, hepatic, or gastrointestinal tract disease, although it may occur concurrently with such diseases. Oxalate nephrosis should be considered as a differential diagnosis in captive cheetahs with renal failure, especially in young animals that are less likely to be suffering from renal amyloidosis or glomerulosclerosis. Oxalate nephrosis is of uncertain etiology in captive cheetahs but a multifactorial pathogenesis, including a primary genetic predisposition, diet, altered gut microbiome, and/or stress, is suspected.
Footnotes
Acknowledgements
This study was made possible by the veterinarians and cheetah holding and breeding institutions that submitted clinical data and carcasses from captive and free-ranging cheetahs for examination from the United States: The Association of Zoos and Aquariums’ Cheetah Species Survival Plan (including 5 anonymous facilities), Albuquerque Biopark Zoo, Binder Park Zoo, Brevard Zoo, Caldwell Zoo, Chehaw Wild Animal Park, Cincinnati Zoo and Botanical Garden, Cleveland Metroparks Zoo, Columbus Zoo and Aquarium, Dickerson Park Zoo, Fort Wayne Children’s Zoo, Fort Worth Zoo, Fossil Rim Wildlife Center, Honolulu Zoo, Jackson Zoo, Jacksonville Zoo and Gardens, Kansas City Zoo, The Living Desert, Maryland Zoo in Baltimore, Mesker Park Zoo and Botanic Gardens, Montgomery Zoo, Nashville Zoo at Grassmere, Oklahoma City Zoo and Botanical Garden, Phoenix Zoo, San Antonio Zoo and Aquarium, San Diego Zoo Global, Tulsa Zoo and Living Museum, Utah’s Hogle Zoo, White Oak, Wildlife Safari, Zoo Knoxville, and Zoo New England; from southern Africa: AfriCat, Cango Wildlife Ranch, Cheetah Conservation Botswana, Dr. A. Tordiffe, Dr. D. Zimmerman, Dr. K. Good, Dr. P. Buss, Dr. P. Caldwell, Dr. P. Swartz, Farm Inn, Hoedspruit Endangered Species Centre, Johannesburg Zoo, Letsatsi La Africa, Lory Park Zoo, Matobo Veterinary Centre, National Zoological Gardens of South Africa, Rhino and Lion Park, Seaview Predator Park, The Ann van Dyk Cheetah Centre, and Tshwane Nature Conservation; from France: Dr. C. Vitaud and Dr. D. Sarran of the Safari de Peaugres and Mr. G. Breton and Dr. F. Ollivet-Courtois of Parc des Félins. Data and cases provided by Drs. Linda Munson and Nadia Robert are also acknowledged. Pathology laboratory staff at the National Zoological Gardens of South Africa and Faculty of Veterinary Science, University of Pretoria; at Anipath, France; and at the University of Illinois Zoological Pathology Program and Veterinary Diagnostic Laboratory provided excellent technical assistance. We thank Dr. Chuck Mohr for his guidance in developing the renal scoring system. Professors John Lawrence and Leon Prozesky, Faculty of Veterinary Science, University of Pretoria, provided valuable editorial input. The National Research Foundation, through a core grant to the National Zoological Gardens of South Africa, provided the funding to conduct pathologic examinations in southern African cheetahs. The views expressed in this publication are those of the authors and do not necessarily reflect the official policy of the US Department of Health and Human Services, the US Food and Drug Administration, or the US government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
