Abstract
The wild and captive koala population of the Mt Lofty Ranges in South Australia has a high level of renal dysfunction in which crystals consistent with calcium oxalate have been observed in the kidneys. This study aimed to describe the pathological features of the renal disease in this population, confirm the composition of renal crystals as calcium oxalate, and determine whether any age or sex predispositions exist for this disease. A total of 51 koalas (28 wild rescues, 23 captive) were examined at necropsy, of which 28 (55%) were found to have gross and/or histological evidence of oxalate nephrosis. Histopathological features included intratubular and interstitial inflammation, tubule dilation, glomerular atrophy, tubule loss, and cortical fibrosis. Calcium oxalate crystals were demonstrated using a combination of polarization microscopy, alizarin red S staining, infrared spectroscopy, and energy-dispersive X-ray analysis with scanning electron microscopy. Uric acid and phosphate deposits were also shown to be present but were associated with minimal histopathological changes. No significant differences were found between the numbers of affected captive and wild rescued koalas; also, there were no sex or age predispositions identified, but it was found that oxalate nephrosis may affect koalas <2 years of age. The findings of this study suggest that oxalate nephrosis is a leading disease in this koala population. Possible causes of this disease are currently under investigation.
Renal disease does not appear to be a common cause of morbidity and mortality in populations of koalas (Phascolarctos cinereus) in the eastern states of Australia. Two previous studies performed in New South Wales reported less than 4% of koalas were found to have renal disease as the only necropsy finding.3,4 In addition, the majority of renal disease in these eastern populations has been considered to be secondary to lower urinary tract infection due to chlamydophilosis, 15 with one study showing that 34% of cystitis cases had renal involvement. 6 Hence, the prevalence of primary renal disease in koalas appears to be low and only a minor contributor to poor health in these populations.
In contrast, the Mt Lofty Ranges koala population in South Australia (SA) has been found to have a high occurrence of renal dysfunction, with an estimated 11% of the koala population affected in 2000. 14 In this previous study, the koalas exhibited clinical signs typical of renal insufficiency such as weight loss, polydipsia, and polyuria that were unassociated with signs of infections, including chlamydophilosis. 14 One koala in this previous study was found to have renal crystals that reacted positively to Pizzolato’s stain for calcium oxalate. This finding, taken in conjunction with accounts from wildlife staff and unpublished veterinary reports, was suggestive that oxalate nephrosis occurs in this population of koalas.
Oxalate nephrosis, characterized by renal deposition of calcium oxalate, has been identified in a small number of koalas in several eastern states surveys.2 In New South Wales, 1 necropsy survey of 110 koalas reported less than 3% prevalence of oxalate nephrosis, 9 and similarly, 2 other studies each reported a prevalence of less than 2%.3,4 Isolated cases of oxalate nephrosis in koalas have also been reported in other regions of Australia. 18
A previous report of oxalate nephrosis in 1 koala described renal pathological changes, including grossly reddened cortical areas with pale corticomedullary streaks and, histologically, intratubular crystals with necrosis of renal tubule epithelial cells. 5 Since the pathological features and pathogenesis of oxalate nephrosis in koalas have not been fully characterized, the aim of the current study was to further investigate the features of this disease, including gross renal pathological and histopathological changes, crystal composition, and age or sex predispositions in the captive and rescued wild koala populations of the Mt Lofty Ranges, South Australia.
Methods
Pathology
A total of 51 koalas, euthanized on welfare grounds, were examined between March 2008 and October 2010. These included 23 captive koalas originating from Cleland Wildlife Park, SA, and 28 rescued wild koalas from the surrounding Mount Lofty Ranges area. Of the 51 koalas, 21 were female and 30 were male, including 1 castrated male. Koalas were aged by classification of tooth wear based on studies by Martin 20 and Martin and Handasyde 19 or from captive animal management records. Of 44 koalas for which age was determined, 25 (57%) were of tooth wear class I (<2 years old approximately), 6 (14%) of class II (<3 years old approximately), 5 (11%) of class III (<4 years old approximately), 7 (16%) of class IV (<6 years old approximately), and 1 (2%) of class V (<12 years old approximately).
The right and left kidneys were cut in half in the transverse and longitudinal planes, respectively. They were scored for the amount of grossly visible crystal deposition on a scale from 0 to 3 (none, mild, moderate, severe). Where large renal crystal deposits were present, these were harvested and dried at 37°C in preparation for infrared spectroscopy analysis (see below). For histopathology, both kidneys were fixed in 10% neutral buffered formalin and processed by routine histological techniques. Kidney halves were then embedded in large paraffin wax blocks and 7-μm sections of the entire surface of each kidney cut on a rotary microtome (Leica, Wetzlar, Germany) and stained with hematoxylin and eosin. Kidney tissue sections were assessed blind for pathological changes and for crystal birefringence using partial or full polarization microscopy with an Olympus BX51 microscope (Olympus, Center Valley, Pennsylvania).
Crystal Composition Analyses
Alizarin red S was used to demonstrate crystals composed of calcium oxalate and differentiate them from those of calcium carbonate and calcium phosphate (see Proia and Brinn 25 ) by staining kidney tissue sections, which showed large crystal aggregates (n = 12, 1 wild, 11 captive) at both pH 7.0 and pH 4.2. Additional tissue sections were treated with acetic acid and hydrochloric acid to assess differential solubilities of crystals to further confirm crystal composition as calcium oxalate or to identify calcium carbonate or calcium phosphate. 25
Infrared spectroscopy was used to determine the presence of calcium oxalate in renal deposits from 11 koalas (7 wild, 4 captive). For this, the deposits were dried, ground, and incorporated into potassium bromide disks for qualitative analysis using infrared spectroscopy 13 on a Varian 800 Scimitar series spectrophotometer (Varian, Palo Alto, California) using Resolutions software. Transmittance spectra were obtained over 4000 to 600 cm–1 and compared with reference spectra for calcium oxalate and other urinary stones.
For scanning electron microscopy, additional unstained kidney tissue sections containing large crystal aggregates (n = 9, 1 wild, 8 captive) were deparaffinized, hydrated, and re-dehydrated through a graded series of alcohols to 100% absolute ethanol. These sections were then air dried, carbon coated, and examined on a Philips XL30 field emission scanning electron microscope (SEM; Philips, Amsterdam, the Netherlands). For elemental analysis of the composition of crystals, energy-dispersive X-ray analysis (EDX) was performed at an accelerating voltage of 10 kV at 5 different crystal locations as well as at a control location for each sample. Data analyses were performed using EDAX Genesis software (EDAX, Mahwah, New Jersey).
Koala Factors
Koalas with oxalate nephrosis were analyzed for differences between age groups, sex, and origin (captive or rescued wild) using χ 2 analysis with statistical significance as P < .05. Also, the current prevalence of renal dysfunction in wild koalas rescued by Cleland Wildlife Park between 2008 and 2010 was estimated from koala management records.
Results
Pathology
Pale yellow crystal precipitation was seen at gross examination of the kidneys in 24 of the 51 koalas (47%) (Fig. 1). Of these, mild crystal deposition, characterized by a very small amount of crystals just visible most commonly at the papilla, was present in 9 of 24 (38%) koalas and moderate crystal deposition, shown by clearly visible deposition in the papillary and medullary regions, in 11 of 24 (46%). Severe cases, showing streaks of heavy crystal precipitation in the papilla and medulla, speckling in the cortex, and gritty yellow precipitate in the renal pelvis, were seen in 4 of 24 (17%).
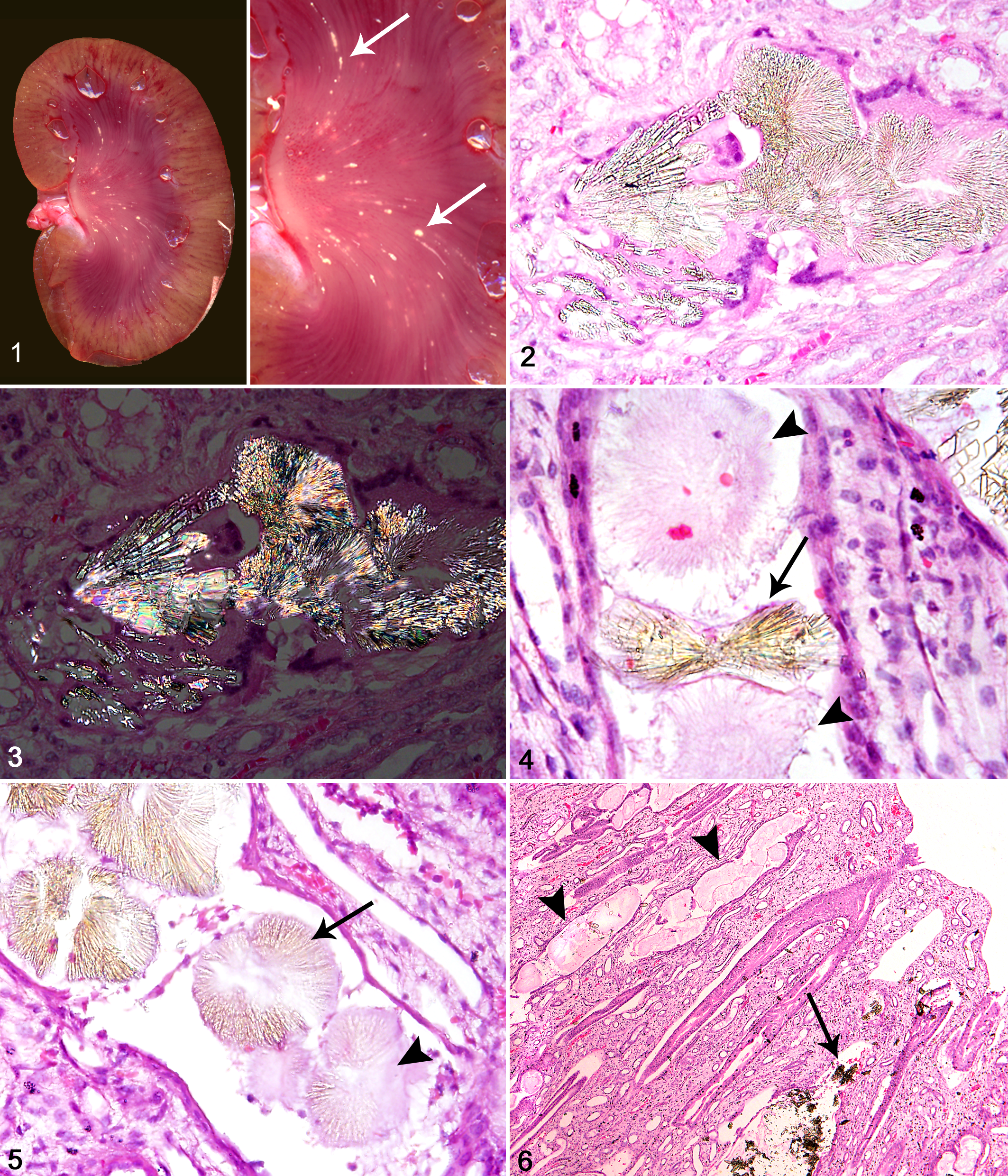
Kidney; koala No. 10. Kidney with crystal deposition. Inset: arrows
indicate the deposits in the medulla.
The majority of the kidneys from these 24 koalas appeared otherwise macroscopically normal, but several were pale and firm, whereas some others showed a segmental pattern of pale areas extending from the corticomedullary boundary to the outer cortex. One koala (case No. 10) had an abnormally small left kidney with severe crystal deposition, whereas the right kidney showed a moderate amount of crystals.
Histological examination showed evidence of renal crystals in 28 of the 51 koalas (55%), identifying 4 additional affected koalas (case Nos. 9, 16, 18, and 26) and indicating that only 86% of the 28 affected koalas showed grossly visible crystal deposition at necropsy. Histological examination showed that in all 28 koalas, the crystals were transparent yellow and usually arranged in rosettes or fan shapes with radiating bands (Fig. 2). They were birefringent under polarized light (Fig. 3), suggestive of calcium oxalate. Variations in crystal morphology included wheat-sheaf or bow-tie formations (Fig. 4) or rectangular shards. Histological evidence of oxalate-like crystals included direct visualization of crystals as well as observation of crystal “ghosts,” which were characterized by pale eosinophilic outlines in the shape of crystals (Fig. 4), some of which contained crystal remnants (Fig. 5). Crystal ghosts were observed in 93% of the 28 cases altogether, with 13 cases (46%) showing crystal ghosts only.
Crystals were distributed throughout the renal cortical, medullary, and papillary regions with particularly large aggregates of crystals and crystal ghosts within collecting ducts in the papilla observed in several cases (Fig. 6). Widespread dilation of collecting ducts and cortical tubules was observed in 82% of the 28 cases, suggestive of an obstructive process. Occasionally, large aggregates of crystals were seen in the medullary interstitium where they had presumably ruptured from tubules. These were associated with a granulomatous inflammatory reaction consisting of lymphocytes, neutrophils, macrophages, and occasional multinucleated giant cells.
Crystals located within tubules were associated with a mixed inflammatory reaction in 89% of the 28 cases (Fig. 7), with epithelial necrosis adjacent to intratubular crystals seen in 86% of cases. Interstitial fibrosis and tubule loss, as well as multifocal areas of mononuclear inflammation in the cortex, were common features (Fig. 7) and present in 86% and 82% of cases, respectively. In 57% of cases, radiating and segmental cortical interstitial fibrosis interspersed with less affected renal parenchyma was observed. Fibrosis was also prominent in the subcapsular region in 18% of cases, usually associated with widespread cortical fibrosis. Glomerular changes included fibrous thickening of Bowman’s capsule (29%) and dilation of Bowman’s space (39%) (Fig. 8), whereas 46% of cases showed glomerular atrophy or loss. Histopathological examination of the small abnormal kidney from koala No. 10 indicated renal dysplasia.
Kidney; koala No. 28. Focal interstitial mononuclear inflammatory cell
aggregate next to intratubular crystal ghost (arrow)
associated with predominantly neutrophils. Hematoxylin-eosin
(HE).
In 16 of the 28 (57%) cases with oxalate-like crystals, small basophilic deposits were also observed (Fig. 9). These deposits were intratubular; not associated with tubule dilation, fibrosis, or inflammation; and not birefringent when examined with polarization microscopy, suggestive of calcium carbonate or calcium phosphate. Occasionally, basophilic deposits were co-located with the oxalate-like crystals. In 3 koalas (case Nos. 6, 11, and 15), particularly heavy deposition of the basophilic deposits were observed in collecting ducts in the papilla, causing tubule dilation and epithelial necrosis, and were probably indicative of dystrophic calcification.
In addition, a third deposit type was found to be co-located with clear oxalate-like crystals (Fig. 10) in several unstained tissue sections (koala Nos. 4 and 10). This deposit was yellow-orange, amorphous, and water soluble and hence was not seen in histological examination of stained tissue sections. No evidence of inflammation or other pathological changes were associated with this deposit type.
Crystal Composition Analyses
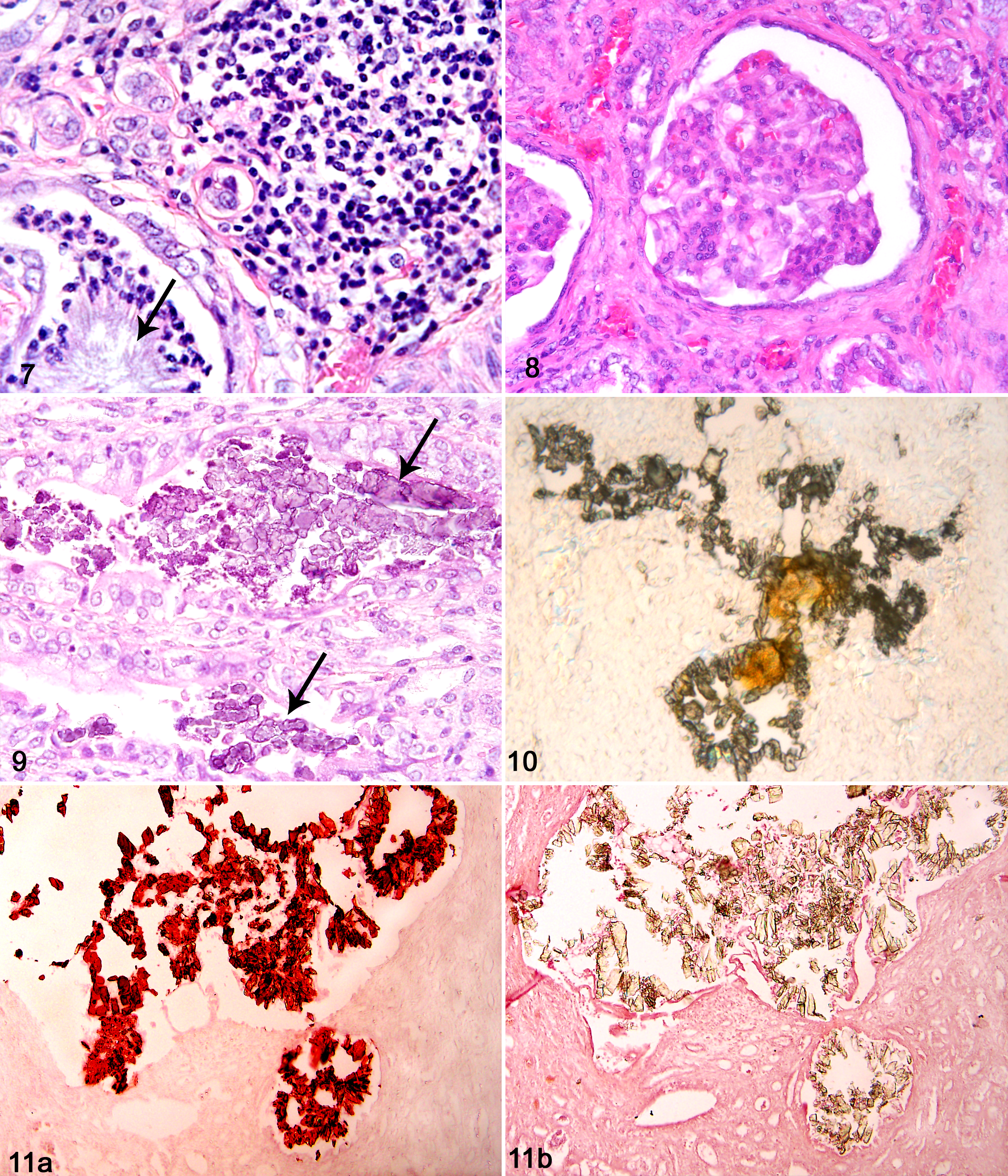
Results for analyses of the 3 types of renal deposits observed in koala kidney tissue sections are summarized in Table 1. Alizarin red S staining of renal tissue sections with calcium oxalate–like crystals showed positive staining of these crystals at pH 7.0 but not at pH 4.2 in all 12 koalas (Fig. 11), suggestive of calcium oxalate. A composition of calcium oxalate was further supported by treatment of additional kidney tissue sections containing large crystal aggregates with hydrochloric acid, which resulted in significant crystal dissolution, and also with acetic acid, which minimally affected crystals. In 4 cases in which basophilic deposits were also present, these stained positively with alizarin red S at both pH 7.0 and pH 4.2, consistent with either calcium carbonate or calcium phosphate. Further testing showed that their likely composition was calcium phosphate since carbon dioxide bubbles were not produced with addition of acetic acid.
Crystal Composition Analyses Demonstrating Presence of Calcium Oxalate, Phosphate, and Uric Acid in Koala Kidneys.

N, result of analysis was not supportive of the deposit type; NA, analysis was not performed; SEM/EDX, scanning electron microscope/energy-dispersive X-ray analysis; Y, result of analysis was supportive of the deposit type.
Calcium oxalate and phosphate were also shown to be present by analysis of renal deposits with infrared spectroscopy. In all 11 samples analyzed, the spectra demonstrated peaks corresponding to those of calcium oxalate and phosphate and, in addition, showed peaks consistent with those of uric acid (Fig. 12). Furthermore, the peaks of the infrared spectra were almost identical between all samples analyzed, which strongly suggested a similar if not equivalent composition and, hence, common pathophysiological process.
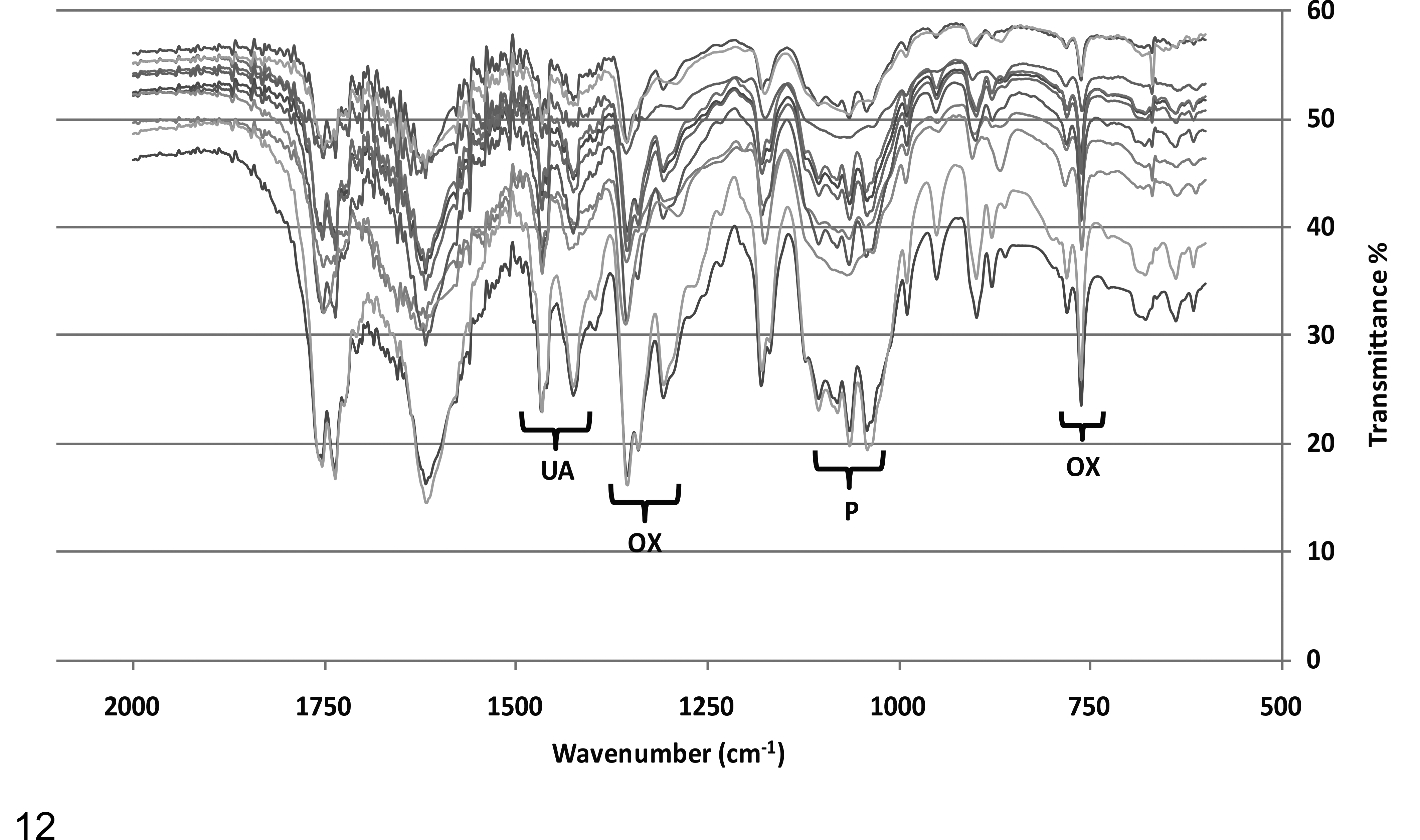
Infrared spectroscopy spectra for 11 koala renal crystal precipitates (koala Nos. 1–5, 10, 13, 14, 21, 25, 28) showing peaks of calcium oxalate (OX), uric acid (UA), and phosphate (P).
Using SEM, EDX analysis was performed on tissue sections with large aggregates of crystals that had oxalate-like morphology and had shown birefringence with polarization microscopy. All 6 koala samples analyzed showed co-location of carbon, oxygen, and calcium within the crystals (Fig. 13), reflecting the composition of calcium oxalate. In 4 samples, silicon was detected at low levels but was also present in the control readings and therefore likely to be a component of the glass slides. Phosphorus was detected in 1 reading and, although supportive of the result of phosphate found with alizarin red S staining and infrared spectroscopy, also was detected in control readings. Nitrogen was not detected in any sample despite the infrared spectroscopy results indicating uric acid.

Kidney; koala No. 27. Energy-dispersive X-ray analysis spectrum with peaks of carbon (C), oxygen (O), and calcium (Ca) from analysis of intratubular crystal aggregate (arrow), shown in the scanning electron micrograph (inset).
The 3 samples with heavy basophilic deposits, for which alizarin red S staining had suggested a composition of calcium phosphate, were also analyzed using EDX. The deposits showed carbon, oxygen, and calcium, as well as a distinct peak for phosphorous, consistent with calcium phosphate. Other elements detected in both the deposits and control readings included sodium, magnesium, silicon, and aluminum.
Koala Factors
This study found that 55% of the 51 koalas examined from the captive and rescued wild population of the Mt Lofty Ranges, SA, were affected by oxalate nephrosis. A similar proportion of renal dysfunction in rescued wild koalas was determined from examination of records at Cleland Wildlife Park for 2008 to 2010, which showed an annual occurrence of 62% ± 11% (mean ± SD), based on an average total of 27 koalas per year.
Results for sex and numbers of wild rescued and captive koalas found in this study to be affected by oxalate nephrosis are presented in Table 2. No significant differences were found in disease prevalence based on sex, and no statistical association was found for origin of koalas (captive or rescued wild) and likelihood of disease. Of the 26 koalas with oxalate nephrosis for which age was able to be determined, 16 (62%) were of tooth wear class I, 2 (8%) of class II, 4 (15%) of class III, 3 (12%) of class IV, and 1 of class V (4%). Despite the high proportion of koalas from tooth wear class I (less than approximately 2 years old), no statistically significant association between age and disease was identified since overall, there were a high number of young koalas in the total cohort.
Rescued Wild or Captive Origin and Sex of Koalas Found to Be Affected With Oxalate Nephrosis.

Values are presented as No. or No. (%).
aCaptive koalas kept at Cleland Wildlife Park, Mt Lofty Ranges, South Australia.
Discussion
This study found 28 of 51 (55%) captive and rescued wild koalas from the Mt Lofty Ranges population in South Australia with gross and/or histological evidence of renal oxalate crystal deposition. Renal histopathological findings were similar among affected koalas and included tubular loss, cortical fibrosis, tubule dilation, glomerular atrophy, and crystal-associated inflammation. These are the hallmark renal lesions associated with oxalate nephrosis in humans,7,27 and the findings in the current study build on those of a previous case study of 1 koala with oxalate nephrosis. 5 An additional observation in the current study was a segmental cortical pattern of fibrosis similar to that described in humans with advanced nephrocalcinosis, thought to be the result of obstruction of tubules with crystals, leading to areas of cortical atrophy. 27
The crystals in the kidneys of affected koalas showed typical calcium oxalate morphology and were birefringent with polarization microscopy, a characteristic of calcium oxalate crystals. 7 Renal crystals were analyzed using a combined approach of differential staining with alizarin red S, infrared spectroscopy, and EDX analysis with scanning electron microscopy, and all results supported a composition of calcium oxalate. Crystal ghosts were also a prominent histological feature and have been reported in other mammalian species with oxalate nephrosis, representing organic residues that retain the shape of the crystals lost during histological processing.17,23 Hence, evidence obtained in this study clearly demonstrated that oxalate nephrosis exists in this South Australian koala population.
In addition to calcium oxalate, composition analyses of the crystals also suggested the presence of phosphate and uric acid. The basophilic deposits observed in koala kidney tissue sections were morphologically consistent with calcium carbonate or phosphate 1 and were shown to be calcium phosphate using alizarin red S staining and EDX analysis. In humans, calcium phosphate is often found in conjunction with calcium oxalate in the kidney17,22 and may be due to the elevated blood and urine phosphate levels that occur in renal failure. 12 However, in the current study, these crystals were also found in the kidneys of healthy koalas, where they were not associated with inflammation and hence may not be directly associated with oxalate nephrosis. In humans, basophilic calcium concretions are not an uncommon finding in otherwise normal kidneys and are regarded as functionally unimportant in most cases. 27
The uric acid results of the infrared spectroscopy analyses were more difficult to confirm using other methods due to its solubility in water. 1 Although nitrogen was not detected by EDX in renal deposit samples, the solubility of the orange deposit examined on the unstained slide was consistent with uric acid. In addition, the absence of this material in the stained tissue sections, which undergo hydration during preparation, is also supportive. A previous study on urinary tract disease in koalas in New South Wales identified both calcium oxalate and uric acid in a bladder stone of a koala. 4 Uric acid deposits may occur similarly as in humans, whereby decreased renal function can lead to hyperuricemia and hyperuricosuria, 24 and dehydration may cause precipitation. 27
EDX analyses showed other elements in some samples, such as sodium, magnesium, and silicon, but these were invariably detected in control locations also and are thus likely to be components of the glass slide. This is also likely to be the case for aluminum, which was detected only in the readings from the small phosphate deposits and controls but has been implicated in koala renal disease in SA in a previous study. 14
The kidneys of most koalas showed gross evidence of crystal deposition and demonstrated similar renal histopathological changes, and infrared spectroscopy showed an identical composition of all kidney precipitate samples, suggesting a common disease process. Although sex does not appear to be a factor and similar numbers of captive and rescued wild koalas were found to have the disease, it was evident that oxalate nephrosis occurred in many young koalas, showing that this disease is not limited to older animals.
This study found that 55% of the total number of koalas examined had renal histopathological changes consistent with oxalate nephrosis, which is a similar prevalence to that of renal dysfunction in rescued koalas at Cleland Wildlife Park per annum (62%). These figures are markedly increased from the previous estimate of 11% for koalas affected by renal dysfunction in this population in 2000, 14 which is consistent with reports from wildlife staff that renal disease is being seen more frequently in this population. To confirm the presence of renal dysfunction in koalas with oxalate nephrosis, blood and urine biochemistry investigations will be undertaken.
The potential causes of oxalate nephrosis in koalas may be similar to those in other mammalian species. Dietary intake of plants containing high levels of oxalate is a cause in other herbivores 21 and has been previously suspected in koala cases. 5 However, the captive koalas in the current study had no access to known oxalate-containing plants, and little is known of the oxalate content of eucalypts. 18 Calcium oxalate crystals may also be found within scarred renal tubules as a secondary process, 21 but the high proportion of cases found in this population would suggest a common inciting cause of primary renal disease.
Although studies in New South Wales koalas have shown that urogenital chlamydophilosis infection may cause inflammation and fibrosis of the kidney,6,15 these previous studies reported either an absence 6 or a low proportion3,4 of koalas with renal calcium oxalate crystals. This indicates that the prevalence of oxalate nephrosis remains low in areas with endemic chlamydophilosis. Another study using immunohistochemical techniques to demonstrate the presence of Chlamydiaceae in koala kidneys could not identify chlamydial antigen in the single koala with intraluminal crystals. 15 In addition, clinical urogenital chlamydophilosis in the Mt Lofty population has not been reported by wildlife veterinarians, and lesions consistent with chlamydophilosis were not observed in the current study. Despite this, positive results from serological and polymerase chain reaction (PCR) testing of SA koalas were found in a previous study, 16 and there is a need for further research to determine the current status.
Another cause of oxalate nephrosis well recognized in humans is primary hyperoxaluria, an autosomal recessive inherited disorder of the hepatic enzymatic pathways involved in glyoxylate metabolism, in which excessive endogenous oxalate production occurs. 8 Forms of this disease have been reported in both purebred dogs 11 and cats 10 and should be considered in koalas due to the low genetic diversity in this population.16,26 On the basis of these causes of oxalate nephrosis in other species and previous investigations of disease in koalas, further studies to determine a potential cause of oxalate nephrosis in the koala population of the Mount Lofty Ranges, SA, are in progress.
Footnotes
Acknowledgements
Many thanks to Amanda Sulley (Cleland Wildlife Park); Dr Lynley Johnson (Zoos SA); Michael Haywood (Institute of Medical and Veterinary Science); Chris Leigh, Tavik Morgenstern, and Nancy Briggs (University of Adelaide); and Angus Netting (Adelaide Microscopy).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly funded by the Holsworth Wildlife Research Endowment - ANZ Trustees Foundation and Zoos SA.
