Abstract
Amphibians in the family Ranidae (true frogs) seem highly susceptible to oxalosis, particularly when fed a diet high in oxalic acid during the premetamorphic (tadpole) stage. The authors describe the mortality of 150 captive-raised wood frogs (Rana sylvatica or Lithobates sylvaticus) from oxalate nephrolithiasis and renal tubular necrosis caused by consumption of boiled spinach during tadpole development. Renal lesions were due to intraluminal transparent crystals which were birefringent under polarized light and were identified morphologically and histochemically as composed of calcium oxalate. Evidence of early fibrosis or squamous metaplasia, and a presentation at least 2 weeks after spinach consumption had ended, suggested a subacute course. Tadpole-feeding protocols should avoid plants with high oxalate content (eg, spinach and rhubarb leaves), and any episode of high mortality in captive amphibians along with nephrolithiasis should prompt an evaluation of the feed sources for material with high oxalate content.
Oxalate crystals—originating from the consumption of feedstuff rich in oxalates (eg, ruminants and horses) or the accidental ingestion of ethylene glycol, which is later metabolized into calcium oxalate (eg, dogs and cats)—cause acute and chronic renal failure in domestic animals. 7 Calcium oxalate crystallizes in renal tubules, causing tubular obstruction and renal failure; it may also chelate calcium and magnesium, thus interfering with oxidative phosphorylation of the renal tubular epithelium. 7 Oxalate nephrolithiasis results in acute renal tubuloepithelial necrosis and leads to death or chronic renal failure associated with tubular loss and interstitial fibrosis. Nondomestic species, including amphibians, may be affected by oxalate nephrolithiasis, but published reports and descriptions of histologic lesions are scarce. We describe fatalities associated with renal tubular necrosis in recently metamorphosed wood frogs (Rana sylvatica or Lithobates sylvaticus) raised in captivity from wild-collected egg masses, caused by consumption of boiled spinach during the premetamorphic (tadpole) stage.
Recently metamorphosed wood frogs (froglets) (Fig. 1) housed in the Biology Building at Acadia University, Wolfville, Nova Scotia, Canada, and originating from 2 egg masses collected in a pond near the university began dying abruptly approximately 2 to 3 weeks after metamorphosis. Approximately 150 froglets died, most within a 48-hour period. Although, in general, little to no obvious clinical signs were observed, occasional froglets exhibited hydrocoelom, and a few lost their righting reflex or were unable to get out of a very shallow Petri dish of water. Four froglets were fixed in 70% ethanol immediately after death and referred to the Canadian Cooperative Wildlife Health Centre (Atlantic Veterinary College, Prince Edward Island, Canada) for examination. As tadpoles, they had fed almost exclusively on boiled spinach, with a weekly supplementation of bloodworms (Glycera sp). Immediately after metamorphosis, the froglets had been housed in glass terrariums with a substrate of aquarium moss, water provided in shallow (Petri) dishes changed daily, and fed wingless fruit flies (Drosophila melanogaster) and grasshopper nymphs of various local species ad libitum.
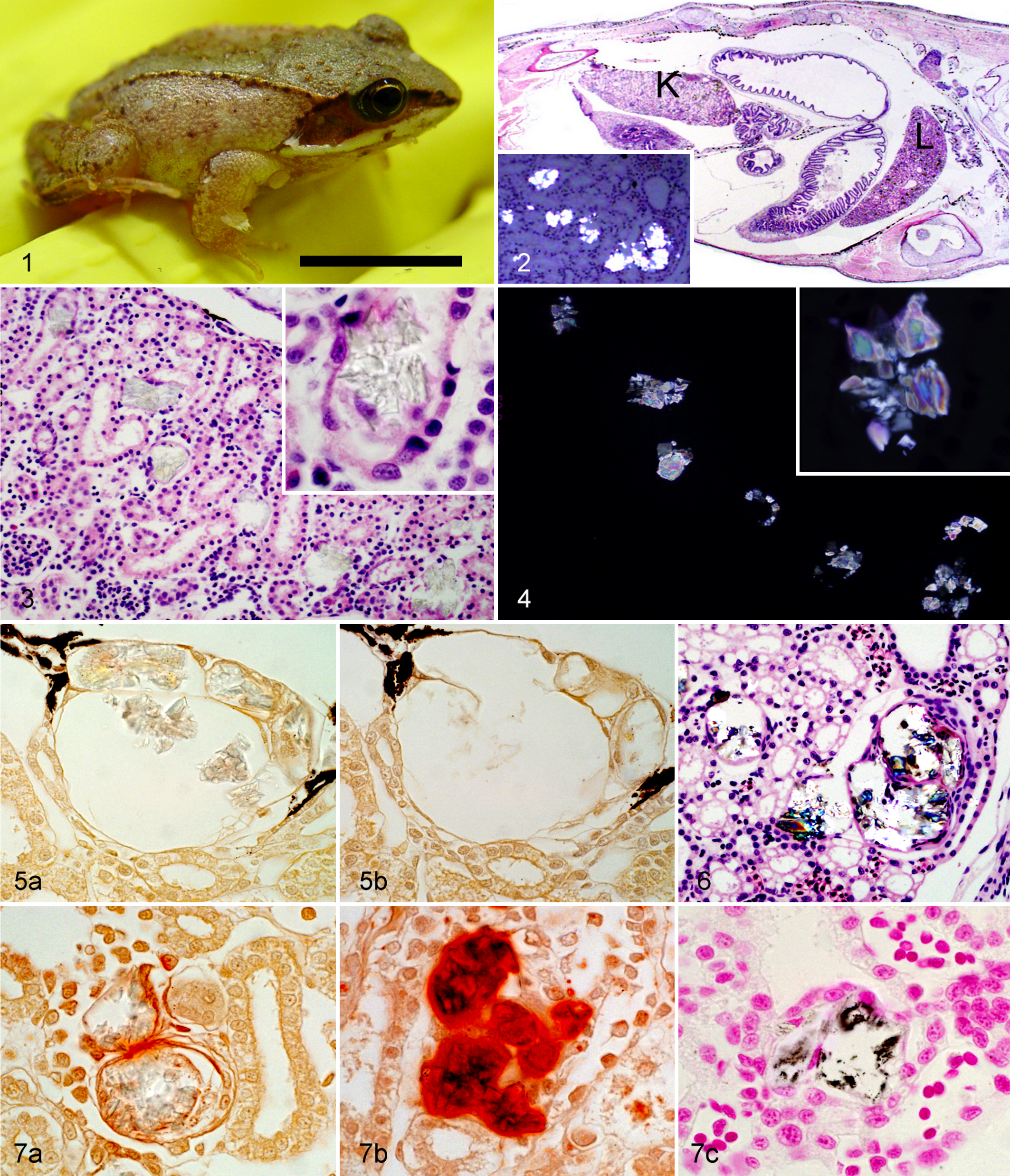
Grossly, there were no obvious abnormalities. Histologic examination of longitudinal full-body sections followed routine tissue processing (Fig. 2). The froglets lacked any histologic evidence of coelomic adipose tissue, and their digestive tracts were either empty or contained small amounts of food, suggesting a poor body condition. Numerous crystals were present in the lumen of the renal tubules, with an average of 15/mm2 of parenchyma (range, 8–26/mm2). These crystals—ranging from transparent to green-tinged when stained with hematoxylin and eosin—were occasionally arranged in a starburst or rosette pattern, but more commonly formed irregular overlapping plates (Fig. 3). Under polarized light, they were positively birefringent, with either an aquamarine or indigo core surrounded by violet and brown-yellow rims and a white outer edge (Fig. 4). The tubules containing crystals had segmental or circumferential attenuation or loss of epithelium (Fig. 3, insert), and some were segmentally dilated (Fig. 5). Around a few crystals, there were concentric layers of flattened cells suggestive of squamous metaplasia or early fibrosis (Fig. 6). No significant lesions were observed in any other organ, and death was concluded to be the result of acute to subacute renal failure due to calcium oxalate nephrolithiasis. Although the poor body condition of the affected animals could have been partly due to anorexia resulting from renal disease, insufficient feed or excessive competition may have been contributing factors, since these were among the first cohorts of froglets of this species to have been raised by the researchers. The high mortalities occurring in such a short period, however, are unlikely to have been the result of primary malnutrition.
Because of the small size of the froglets and their kidneys, the crystals could not be dissected and removed for physicochemical analysis. Crystals were identified as calcium oxalate based on their location in the lumina of renal tubules, birefringence under polarized light with second- and third-order colors (Michel-Levy scale) in the thicker core of the plates but first-order white along the thinner edges 6,8 (Fig. 4), and staining and solubility with various dyes and solvents. Specifically, the crystals stained inconsistently with Von Kossa, positively with Alizarin red S at pH 7.0, but negatively with Alizarin red S at pH 4.2, and they were soluble with 1N hydrochloric acid but insoluble with 2M acetic acid (Figs. 5 and 7, Table 1). 3,10 Histochemical staining characteristics and, specifically, insolubility in acetic acid distinguish calcium oxalate crystals from other calcium salts and renal calculi (ie, cystine) and are the bases of their histologic identification. 3
aStaining and solubility assays were slightly modified from Proria and Brinn. 10 Alizarin red S stain was applied for 2, not 3, minutes to the tissue; HCl concentration was 1N instead of 0.1N, as the latter failed to dissolve any crystals.
bSolubility: soluble (+), insoluble (–).
Although a few oxalate crystals may be found in the renal tubules of healthy amphibians, 5 renal tubular necrosis associated with calcium oxalate crystals causing 92% to 95% mortality has been described in recent metamorphs of Northern leopard frogs (Rana [Lithobates] pipiens) that were fed spinach as tadpoles. 1,2 Spinach is high in oxalic acid, a substance that, when consumed in large amounts, results in oxalosis: the precipitation of the highly insoluble calcium oxalate salt in tissues. 3 Interestingly, both in previous reports 1,2 and in the present case, mortality does not occur during the tadpole stage when the spinach is actually being consumed but, instead, 2 to 3 weeks postmetamorphosis, after the froglets have been on a fully insectivorous diet for at least 14 days. Berns et al 1 observed that most mortalities from oxalosis in Northern leopard froglets clustered around days 15 to 16 postmetamorphosis, resulting in high mortalities in a short period, similar to what occurred with our wood frogs. Tadpoles fed spinach seem able to maintain homeostasis and even accelerate their development as oxalate crystals deposit in their renal tubules, 2 whereas froglets die from subacute oxalate nephrolithiasis. The delay between consumption of spinach and development of clinical nephrolithiasis might be related to the differences in the renal morphology and physiology of pre- and postmetamorphic frogs. The anterior portion of the tadpole kidney (pronephros) is lost during metamorphosis, so postmetamorphic kidneys consist only of the middle and posterior nephrostomes (mesonephric kidneys). 4 Tadpoles, living in an aquatic environment, have little need to preserve water and thus excrete nitrogen waste as ammonia (ammonotelism); the move to a terrestrial environment after metamorphosis is accompanied by a switch to urea excretion (ureotelism) and an increase in plasma osmolarity, both aimed at water conservation. 4 Requiring further investigation is, first, whether the postmetamorphic structural changes and increased demands on the kidneys are the underlying causes of renal failure and death in the presence of oxalosis and, second, why this should present so regularly a fortnight after metamorphosis.
In Northern leopard frogs, the crystals seem to be more numerous on the ventral half of the kidneys. 2 In the wood frogs in this report, the deposition of the crystals seemed to follow a linear pattern, close to the dorsal surface of the kidney and dorsal to the majority of the glomeruli, which are most numerous along the ventral third of the organ. The reason behind this pattern is uncertain. Perhaps an anatomic characteristic of the tubules in the affected area, such as a sharp bend, makes this site particularly susceptible to crystal deposition.
The present case highlights the high susceptibility of ranid amphibians to death from dietary calcium oxalate nephrolithiasis. Tadpole-feeding protocols should avoid plants with high oxalate content, such as spinach and rhubarb leaves; similarly, insects to be fed to froglets and adults must not be allowed to feed on oxalate-rich vegetation. 9 An episode of high mortality in captive amphibians, be it pets or research animals, along with nephrolithiasis should prompt an evaluation of the feed sources for material with high oxalate content.
Footnotes
Acknowledgements
We thank Mrs. Ramona Taylor and Sarah Bernard, whose histologic and chemical skills were indispensable to the preparation of all tissue slides and special stains.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Acadia University Research Fund and the Canadian Cooperative Wildlife Health Centre.