Abstract
Ruffed grouse (Bonasa umbellus) population numbers in Pennsylvania dramatically declined during the early 2000s and have subsequently remained depressed throughout much of the state. While this decline has been temporally associated with the presence of West Nile virus (WNV), lack of information on the WNV susceptibility of this popular game bird species has limited the ability to interpret the potential impacts of WNV. To address this knowledge gap, virologic, immunologic, pathologic, and clinical responses as well as protective effects of vaccination following experimental WNV inoculation in ruffed grouse were assessed. Four of 10 (40%) naive, WNV-inoculated grouse succumbed to infection within 8 days and had moderate mean peak viremia titers (107.0 plaque-forming units [PFU]/ml serum); severe necrotizing myocarditis with widespread, corresponding immunohistochemical labeling; and minimal encephalitis. Grouse that survived to the prescribed end point of 14 days postinoculation (6/10; 60%) had slightly lower mean peak viremia titers (106.8 PFU/ml serum), moderate myocardial lesions, and more widespread brain lesions with rare corresponding immunohistochemical labeling. Vaccinated, WNV-inoculated birds (n = 5) had lower mean peak viremia titers (103.6 PFU/ml serum) and minimal lesions, and sham-inoculated, in-contact control birds (n = 3) had no evidence of infection. All surviving, inoculated birds seroconverted, and WNV-specific antibodies were detectable in serum and Nobuto filter paper strip–eluted blood samples. These data suggest that WNV could serve as an additional population pressure on ruffed grouse in regions where transmission levels are high and WNV competent, ornithophilic vectors exist.
Following its unexpected introduction to North America, West Nile virus (WNV) has become well established throughout much of the continent. During this time, the pathogenesis, reservoir competence, and population impacts have been documented in numerous native North American bird species. 5,11 –14,29 The first evidence of WNV in Pennsylvania was in 2000, and the virus was detected throughout most of the state by the summer of 2002 (http://www.westnile.state.pa.us/surv.htm). A corresponding dramatic decline in population numbers of Pennsylvania ruffed grouse (Bonasa umbellus) occurred in 2002 to 2005, as indexed by hunter diary surveys and brood sighting surveys; ongoing monitoring has revealed ongoing lower harvest and survey numbers of grouse following the arrival of WNV to the state (L. M. Williams, personal communication, 2016). The ruffed grouse has a widespread, year-round geographic range across the United States and Canada, where it is a prized game bird and conservation symbol. 27
Ruffed grouse populations in Pennsylvania and other regions are sensitive to habitat alterations such as loss of early successional forest, 9 with possible contributing factors such as climate change. 15 The decline and lack of recovery in Pennsylvania ruffed grouse populations corresponded temporally to the arrival and establishment of WNV in the state and have led to questions regarding the potential contribution of WNV in this decline. The greater sage-grouse (Centrocercus urophasianus) is within the same subfamily Tetraoninae (family Phasianidae) as the ruffed grouse and experienced reduced survival among radio-marked birds naturally infected with WNV and high mortality rates following experimental WNV infection. 6,17 Population declines of greater sage-grouse prompted conservation concerns and consideration of protected status under the US Federal Endangered Species Act. 10 Similar concerns for the ruffed grouse by regional biologists and hunters highlighted the need to better understand the potential impacts of WNV on ruffed grouse.
The present study was initiated to investigate the susceptibility of ruffed grouse to WNV infection. Specific objectives included (1) to assess the susceptibility of ruffed grouse to WNV under experimental conditions; (2) to characterize the pathobiology of WNV infection in ruffed grouse; (3) to determine WNV-induced viremia, oropharyngeal and cloacal shedding profiles and viral titers in select tissues; (4) to compare anti-WNV antibody titers in serum vs Nobuto filter paper strip–eluted samples in experimentally infected grouse; and (5) to assess the level of vaccine-induced immune protection in WNV-challenged birds.
Materials and Methods
Study Animals
Ruffed grouse eggs were collected by personnel of the Pennsylvania Game Commission from the nests of free-ranging birds in 6 counties in central and northwestern Pennsylvania. Eggs were transported to a commercial ruffed grouse propagator either by ground transportation in an incubator (for eggs incubated by the hen) or via UPS ground transportation (for eggs collected prior to incubation). Once at the propagator, the eggs were incubated, hatched, and hand-reared in mosquito-proof enclosures. In total, 18 ruffed grouse (7 males, 11 females) were used in this study. To evaluate the protective effects of vaccination, 5 of the 18 ruffed grouse were inoculated between 23 and 37 days posthatch with 0.01 ml of an inactivated commercial vaccine (West Nile Innovator WNV equine vaccine; Zoetis, Kalamazoo, MI) administered subcutaneously over the breast muscle. At approximately 4 to 6 weeks of age, grouse were transported to the Animal Disease Laboratory, Colorado State University (Ft Collins, CO). Birds were housed in a biosafety level 3 facility; all were kept free-flight in a 3.7 × 5.5 × 3.4-m room containing multiple standing plywood dividers, tree branches for cover, and ground shavings. The birds were maintained at ambient temperature (21–24°C) and humidity (42%–66%) and a natural light cycle (approximately 14 light-hours/d). Grouse were provided game bird feed (Rangen Quality Game Bird Feed; Connatural Products, Buhl, ID) supplemented with clover and grass every other day and water ad libitum. Animal care and experimentation were approved by the Institutional Animal Care and Use Committee (Colorado State University; protocol 14-5352A) and carried out in accordance with the applicable institutional, local, and national guidelines.
Virus
The virus stock consisted of WNV strain 14511270 isolated from a Culex pipiens/restuans mosquito collected in Suffolk County, New York, in September 2014 and passaged once on C6/36 cells. Virus stock was diluted in BA-1 medium (Hank’s M-199 salts, 1% bovine serum albumin, 350 mg/l sodium bicarbonate, 100 units/ml penicillin, 100 mg/l streptomycin, 1 mg/l Fungizone in 0.05 M Tris, pH 7.6) immediately prior to inoculation.
Experimental Design
Grouse acclimated to the laboratory environment for 1 week prior to inoculation, at which time body mass was measured and blood was collected via the jugular vein for preinoculation serology (0 days postinoculation [DPI]). The 18 grouse were randomly divided into 3 treatment groups: (1) naive (ie, nonvaccinated and seronegative prior to inoculation), WNV-inoculated birds (n = 10); (2) vaccinated, WNV-inoculated (ie, challenged) birds (n = 5); and (3) sham-inoculated, in-contact control birds (n = 3). At 0 DPI, birds in treatment groups 1 and 2 were injected with 0.1 ml BA-1 medium containing 105.1 plaque-forming units (PFU)/ml WNV into subcutaneous tissue over the pectoral muscle. Sham-inoculated, in-contact birds (ie, treatment group 3) were injected with the same volume of BA-1 medium in the same location.
Following inoculation, all grouse were monitored at least twice daily (morning and afternoon) for clinical signs, such as lethargy, ataxia, recumbency, and decreased food or water intake; birds with clinical signs (and those surviving to the predetermined end point of 14 DPI) were euthanized by sodium pentobarbital overdose via intraperitoneal or intravenous injection. A blood sample (0.1 ml) was collected daily from all birds via the jugular vein from 1 to 7 DPI and diluted 1:10 in BA-1 medium. The diluted blood was allowed to coagulate at ambient temperature for 30 to 60 minutes and subsequently centrifuged at 8000 × g for 4 minutes. Oropharyngeal and cloacal swabs were collected on 2 to 5 DPI and placed into 1.0 ml BA-1 medium supplemented with 10% fetal bovine serum. Blood samples and oropharyngeal and cloacal swabs were also collected at the time of euthanasia. Serum and swab samples were stored at –80°C within 1 hour of collection until testing. For birds that survived to 14 DPI, blood (0.1 ml) was also soaked in Nobuto filter paper strips (Advantec; Toyo Roshi Kaisha, Dublin, CA; hereafter, “Nobuto strips”), which were allowed to dry at ambient temperature for 48 hours and processed per the manufacturer’s instructions. Briefly, Nobuto strips were placed in 0.4 ml phosphate-buffered saline for 60 minutes, vortexed for 10 seconds, and centrifuged at 10 000 × g for 2 minutes. Remaining whole blood was processed as described above.
Necropsies were performed within 60 minutes of euthanasia on all grouse. Birds were weighed immediately following euthanasia to calculate percent change in body mass vs preinoculation. During necropsy, gross findings were noted, and 0.5-cm3 samples of kidney, skeletal muscle, heart, and brain and 3 mature breast feathers were collected and stored at –80°C within 1 hour of collection until testing. Tissues were thawed and homogenized in a mixer mill (5 minutes at 25 cycles/s; Retsch GmbH, Haan, Germany) in 1.0 ml BA-1 medium supplemented with 10% fetal bovine serum. Resulting homogenates were clarified by centrifugation (12 000 × g for 3 minutes), and supernatants were stored at –80°C until virus isolation was performed. Representative samples of heart, liver, lung, kidney, spleen, pancreas, duodenum, jejunum, cecum, large colon, adrenal gland, gonad, skeletal muscle (pectoral), cloacal bursa, cerebrum, cerebellum, and brainstem were placed in 10% neutral buffered formalin for 48 hours and then into 70% ethanol.
Virus Isolation
Sera, swabs, and tissue homogenates were stored at –80°C until virus isolation. Virus isolation and serial titrations were performed on a Vero cell plaque assay as previously described. 3 Minimum levels of WNV detection by virus isolation were 101.7 log PFU/ml serum, 100.7 log PFU/swab, and 100.7 log PFU/0.5 cm3 tissue.
Histopathology and Immunohistochemistry
After fixation, tissues were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Duplicate sections of heart, spleen, kidney, adrenal gland, brain, duodenum, and, in some cases, gonad were cut and immunohistochemically labeled using published procedures 16 with modifications as previously described, 18 in addition to the following: (1) the primary antibody was WNV B-956 diluted 1:200 in blocking solution, (2) an additional blocking step was performed with 1% fetal bovine serum and 10% bovine serum albumin in Tris-buffered saline with 10% Tween, (3) a 0.3% H2O2 10-minute incubation step preceded a 30-minute tissue incubation step with 0.15 M glycine in phosphate-buffered saline to minimize nonspecific staining, and (4) Tris-buffered saline with Tween was used in all washes. Sham-inoculated (ie, known virus-negative), in-contact control grouse were used for negative control tissues; positive control tissues were from experimentally WNV-infected American crows (Corvus brachyrhynchos). 24
Histopathology was scored subjectively based on approximated percent involvement of tissue sections as “0” for no lesion observed, “+” for mild lesion(s) and ≤5% of tissue section(s) affected, “++” for moderate lesion(s) with 6% to 25% of tissue section(s) affected, and “+++” for severe lesion(s) with >25% of tissue section(s) affected.
Immunoreactivity for anti-WNV antibodies was scored similarly to histopathology, through assessing the estimated percentage of tissue that contained immunoreactive cells, with “0” for no immunoreactivity observed, “+” for ≤5% of tissue section(s) with immunoreactive cells, “++” for 6% to 25% of tissue section(s) with immunoreactive cells, and “+++” for >25% of tissue section(s) with immunoreactive cells.
Serology
All serum samples and Nobuto strip eluate samples were assessed for WNV-neutralizing antibodies by a plaque reduction neutralization test (PRNT) as previously described. 2 Briefly, sera were diluted 1:10 in BA-1 medium with a challenge dose of approximately 100 PFU of the same WNV strain as used for inoculation, incubated at 37°C for 1 hour, and tested by plaque assay as described above. Preinoculation sera that neutralized ≤70% of WNV PFU at a 1:10 serum dilution were considered negative for anti-WNV antibodies. Postinoculation sera (14 DPI) and blood eluted from Nobuto strips each underwent serial 2-fold dilutions starting at 1:10; antibody titers were expressed as the reciprocal of the highest serum dilution showing greater than 90% reduction (PRNT90) compared with control wells.
Results
Clinical Signs
Four of 10 (40%) naive, WNV-inoculated grouse were euthanized due to acute-onset (<12 hours), severe disease on 7 to 8 DPI (case Nos. 1–4). Two of these birds (case Nos. 1 and 2) were recumbent and reluctant or unable to stand (ie, hindlimb paresis; Fig. 1). The other 2 birds (case Nos. 3 and 4) had fluffed feathers, had dehydration (skin tenting, tacky oral mucosa), and were reluctant to move, and 1 also had intermittent tail and head twitching (case No. 4). All of these birds exhibited loss in body mass from the time of inoculation to euthanasia that ranged from 12% to 22%. Among the 6 subclinically infected, naive, WNV-inoculated grouse (case Nos. 5–10), 3 (case Nos. 7, 8, and 10) increased in body mass from inoculation day to 14 DPI (from 1%–31% of preinoculation body mass), while 3 (case Nos. 5, 6, and 9) decreased 8% to 25% of preinoculation body mass. One of these birds (case No. 9) exhibited decreased activity levels for 3 days prior to euthanasia on 14 DPI, at which time an impacted proventriculus was noted (see gross findings below).
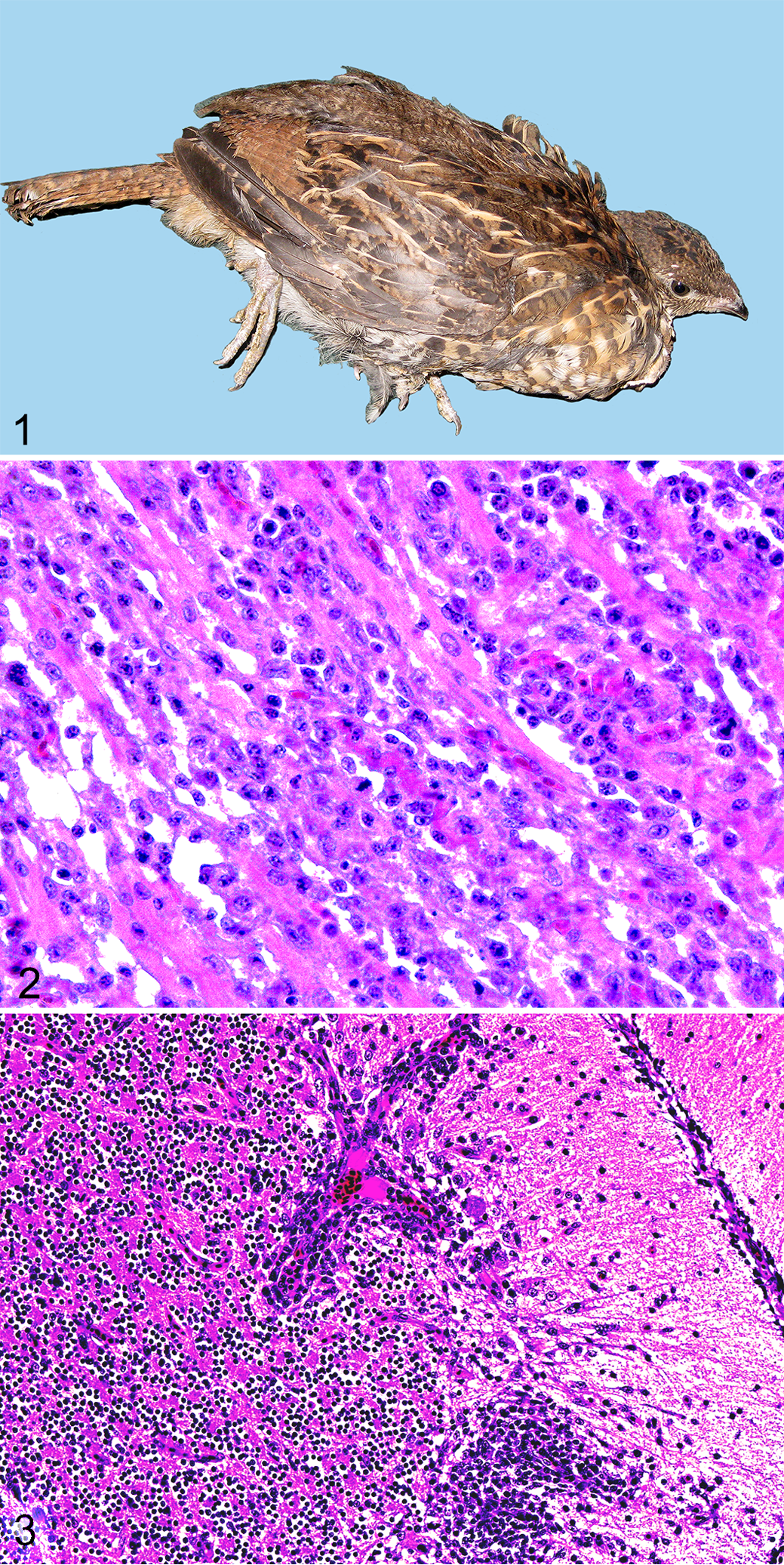
None of the vaccinated, WNV-inoculated (case Nos. 11–15) or sham-inoculated, in-contact control birds (case Nos. 16–18) exhibited clinical disease prior to or during the study. The 5 WNV-inoculated, vaccinated birds gained 19% to 37% of body mass, and sham-inoculated, in-contact control birds gained between 30% and 49% of body mass throughout the 14 days of the study.
Viremia, Viral Shedding, and Virus Isolation From Tissues
All 10 naive (ie, nonvaccinated), WNV-inoculated grouse had detectable viremia titers between 1 and 4 DPI (Table 1). Among these, the 4 grouse with clinical disease on 7 to 8 DPI (case Nos. 1–4) had a mean peak viremia titer approximately 1.4-fold higher than those with no detectable clinical signs. Mean peak viremia titers occurred consistently on 2 to 3 DPI. Viremia lasted an average of nearly 2 days longer in clinically affected grouse (case Nos. 1–4) vs their subclinically infected counterparts (case Nos. 5–10). Among the 6 grouse in the latter group, 1 inoculated bird (case No. 7) had an unusually low, transiently detected viremia titer (102.7 PFU/ml serum on 6 DPI); this bird exhibited oropharyngeal shedding on 5 DPI and seroconverted and is therefore considered to have been infected and is included in the analysis.
Summary of Viremia Profiles (Mean Peak Titer, Timing, and Duration) in Ruffed Grouse Experimentally Inoculated With West Nile Virus.a

DPI, day(s) postinoculation; PFU, plaque-forming units.
aViremia was assessed from 1 to 7 DPI and, when possible, at the time of euthanasia if due to morbidity (ie, 7–8 DPI).
bViremic vaccinated birds included case Nos. 12 to 14.
Among the 5 vaccinated, WNV-inoculated birds (case Nos. 11–15), 3 (60%) had detectable viremia titers (case Nos. 12–14) of limited duration, including 1 day (3 DPI) in 2 birds (case Nos. 13 and 14) and 3 days (1–3 DPI) in the third (case No. 12). Mean peak viremia titers were approximately 2000-fold lower among vaccinated vs nonvaccinated inoculates.
Oropharyngeal shedding of infectious virus occurred in all naive, WNV-inoculated grouse (Table 2). Mean peak titers were approximately 2-fold higher in subclinically infected grouse (vs clinically affected), with the highest titers usually detected on 4 to 5 DPI. Cloacal shedding of infectious WNV was detected in all but 1 (9/10; case No. 7) naive, WNV-inoculated birds of variable duration; shedding was also detected on 7 to 8 DPI in the 4 grouse euthanized due to clinical disease. Mean peak viral titers in cloacal swabs were over 2-fold higher for birds with clinical disease vs those that were subclinically infected. Oropharyngeal shedding was detected in all 5 vaccinated birds (case Nos. 11–15) and cloacal shedding in 2 of 5 vaccinates (case Nos. 12 and 14). Vaccinated birds had approximately 1800- and 850-fold lower mean peak oropharyngeal and cloacal titers, respectively, vs nonvaccinated inoculates.
Summary of Virus Shedding Profiles in Ruffed Grouse Experimentally Inoculated With West Nile Virus.a
DPI, day(s) postinoculation; PFU, plaque-forming units.
aOral and cloacal viral shedding was assessed from 2-5 DPI and at the time of euthanasia if due to clinical disease (ie, 7–8 DPI). Titers are expressed as log PFU per swab.
bNaive, inoculated, subclinically infected birds that shed cloacally included: case Nos. 5, 6, 8, 9 and 10; vaccinated birds that shed included case Nos. 12 and 15.
Infectious WNV (determined by virus isolation) was detected in at least 2 of 5 tissues tested from all 4 of the clinically affected birds (case Nos. 1–4). Tissue tropism was most consistent in heart (4/4; range, 103.0–6.9 PFU/cm3) and kidney (4/4; range, 103.3–4.7 PFU/cm3). Skeletal muscle and feathers were also positive in 3 of 4 birds (range, 102.0–3.6 PFU/cm3 and 102.5–3.6 PFU/3 feathers, respectively; case Nos. 1–3) and brain in 2 of 4 (range, 103.4–4.7 PFU/cm3; case Nos. 2 and 3). Virus detected in tissues was limited to the kidney of 2 of 6 of the subclinically infected inoculates on 14 DPI (102.0 PFU/cm3; case Nos. 6 and 8). None of the 5 vaccinated, challenged birds (case Nos. 11–15) had virus detected in muscle, heart, kidney, or feathers; 1 of 5 (case No. 12) had low viral titers in brain (102.0 PFU/cm3).
None of the 3 noninoculated, in-contact control birds (case Nos. 16–18) had detectable viremia, oropharyngeal or cloacal shedding, or virus in tissues.
Gross Pathology
The 4 naive, WNV-inoculated grouse euthanized on 7 to 8 DPI (case Nos. 1–4) were dehydrated and in poor nutritional condition with serous atrophy of pericardial fat and minimal gastrointestinal tract contents. Three of these birds had grossly small spleens (case No. 1: 0.4 × 0.5 × 0.5 cm; case Nos. 3 and 4: 0.5 cm3) relative to sham-inoculated, control birds (case No. 16: 0.5 × 0.6 × 0.9 cm; case Nos. 17 and 18: 0.4 × 0.5 × 0.7 cm). Three of these birds (case Nos. 2–4) also had pale foci on the epicardium extending into the superficial myocardium. One of these grouse (case No. 2) had a diffusely pale myocardium; another (case No. 4) had multifocal, pale, tan streaks or mottled foci that extended to the myocardium; and a third (case No. 3) had both diffusely pale myocardium and variable off-white streaks that were most prominent at the apex (Suppl. Fig. S1).
Among the 6 naive, WNV-inoculated, subclinically infected birds (case Nos. 5–10), all but 1 (case No. 9) were in fair to good nutritional condition with abundant food contents throughout the gastrointestinal tract. The bird with loss of body condition (case No. 9) had a proventricular impaction with 2 wood shavings. Two of the 6 birds (case Nos. 5 and 8) had pale streaks in the epi- and myocardium involving the apex and/or atria; 1 of these (case No. 8) had a grossly enlarged spleen (1.2 × 1.0 × 0.8 cm) relative to sham-inoculated, control birds. The remainder of the naive, WNV-inoculated, subclinically infected birds had spleen sizes similar to those of sham-inoculated control birds (case No. 5: 0.6 × 0.6 × 0.9 cm; case Nos. 6 and 10: 0.5 × 0.6 × 0.9 cm; case No. 7: 0.5 × 0.6 × 0.7 cm; case No. 9: 0.4 × 0.5 × 0.6 cm).
The 5 vaccinated, WNV-inoculated (case Nos. 11–15) and 3 sham-inoculated birds (case Nos. 16–18) euthanized on 14 DPI were in good nutritional condition with no gross lesions.
Histopathology
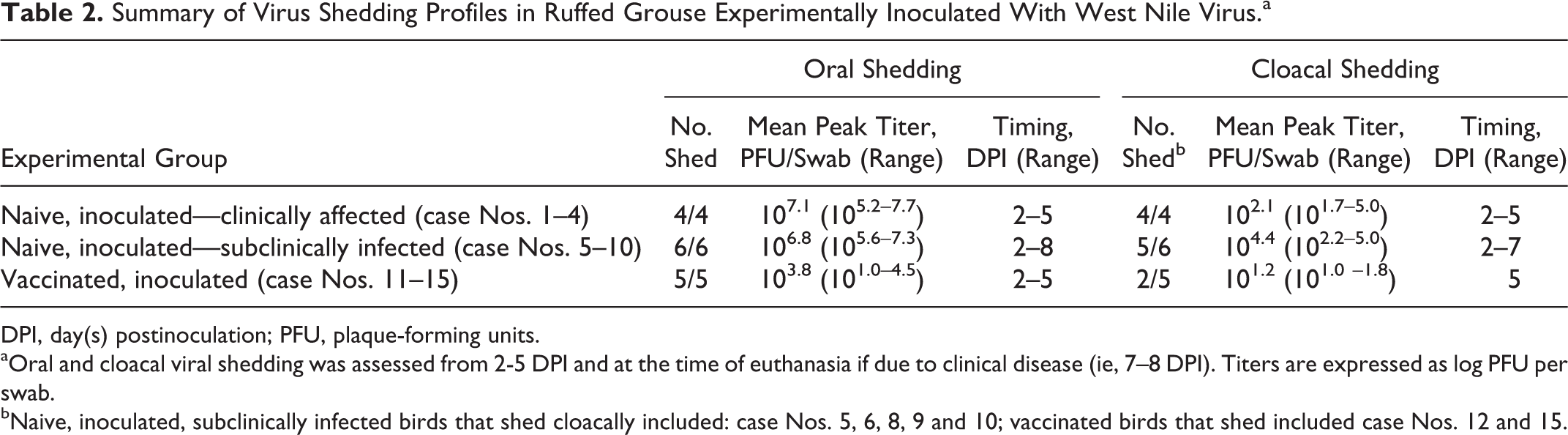
Microscopically, hearts from the 4 naive, WNV-inoculated grouse that exhibited severe disease (case Nos. 1–4) had severe, multifocal to coalescing (subjectively affecting up to 80% of the myocardium within the sections examined), lymphoplasmacytic myocarditis including perivascular distribution, with myofiber degeneration (Fig. 2), necrosis, and mineralization (Table 3). Hearts of 5 of 6 of the naive, WNV-inoculated, subclinically infected birds (case Nos. 5, 6, 8, 9, and 10) euthanized on 14 DPI had 10% to 30% of the myocardium (including left and right free walls and septum) infiltrated by lymphocytes and plasma cells, which were often associated with myocyte degeneration and necrosis and were occasionally located around vessels. The remaining grouse among these (case No. 7) had less severe lesions that included mild, multifocal perivascular cuffing with lymphocytes and plasma cells and moderate, multifocal myofiber degeneration. Two of 5 vaccinated, inoculated grouse (case Nos. 12 and 13) had rare (1–2 foci) accumulations of small numbers of lymphocytes and plasma cells in the perimysium of the myocardium.
Summary of Microscopic Lesions and Immunohistochemical Labeling in Tissues From Ruffed Grouse Experimentally Inoculated With West Nile Virus.a
DPI, day(s) postinoculation; IHC, immunohistochemistry; INF, inflammation; NEC, necrosis; NT, not tested.
aImmunohistochemistry assessment: 0 = no immunoreactivity; + = ≤5% of tissue section(s) with immunoreactivity; ++ = 6% to 25% of tissue section(s) with immunoreactivity; +++ = >25% of tissue section(s) with immunoreactivity.
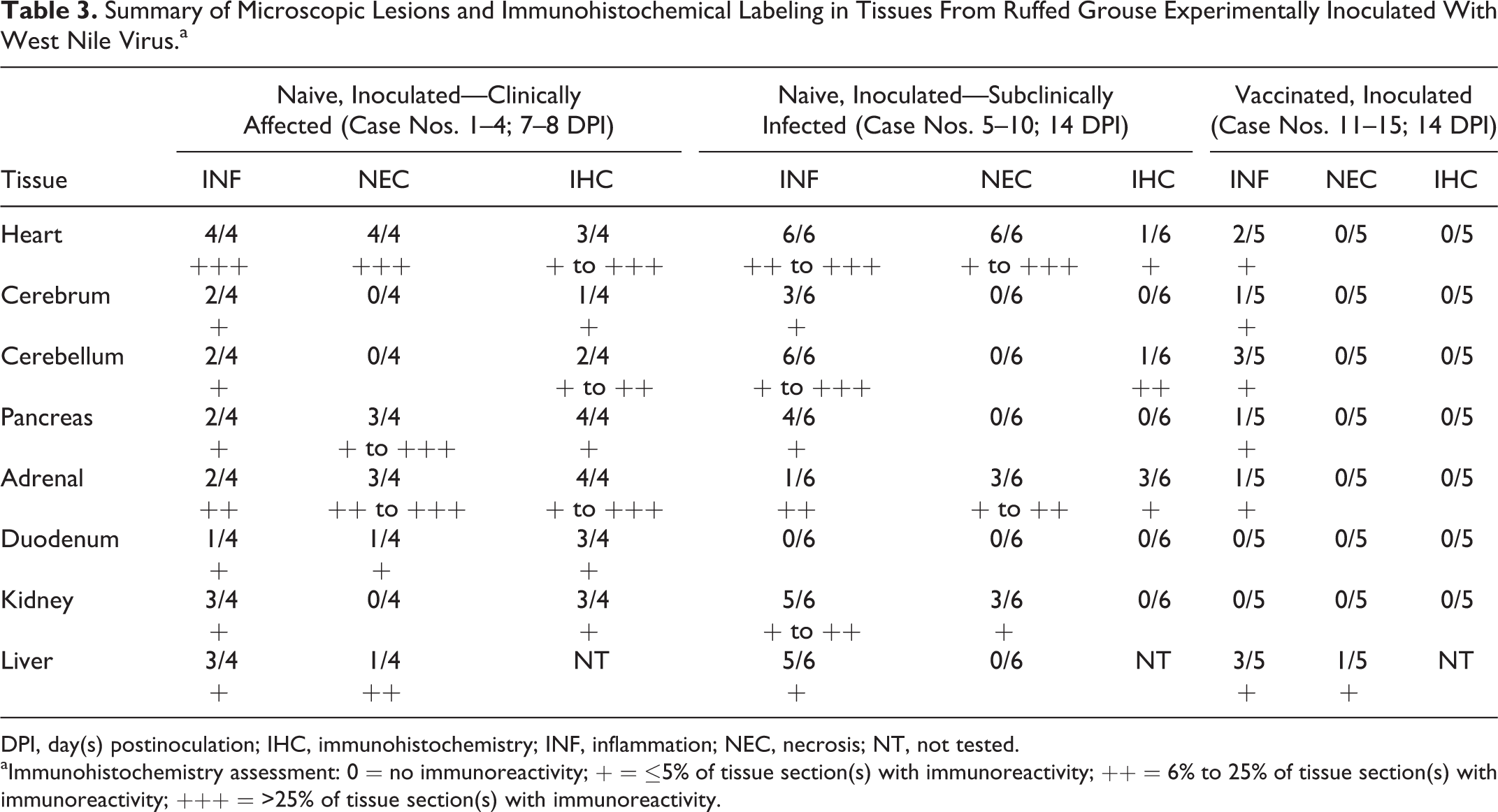
In the brain, 4 of 4 naive, WNV-inoculated, clinically affected birds (case Nos. 1–4) had rare, mild perivascular cuffs consisting of lymphocytes and plasma cells and rare accumulations of small numbers of microglial cells in the gray matter of the cerebrum, cerebellum, and brainstem. All 6 naive, WNV-inoculated, subclinically infected birds (case Nos. 5–10) had encephalitis of varying distribution and severity, with cerebellar involvement in all cases. Encephalitis was mild in 4 of 6 (case Nos. 5, 8, 9, and 10) and moderate to severe in 2 of 6 grouse (case Nos. 6 and 7). Aggregates of lymphocytes and plasma cells were scattered within the molecular layer affecting numerous folia (case Nos. 5–10) and less commonly the Purkinje and granular cell layers (case No. 6; Fig. 3). Small numbers of similar inflammatory foci were within the white matter of the cerebellum and brainstem (case Nos. 5, 6, and 7) and gray matter of the brainstem (case Nos. 5 and 10; Fig. 4). The cerebral gray matter had 4 to 8 small foci of perivascular cuffs consisting of lymphocytes and plasma cells in 3 of 6 grouse (case Nos. 5–7). Vaccinated birds had rare, minimal accumulations of lymphocytes, plasma cells, and microglial cells in the cerebrum (2 small foci; 1/5; case No. 12), cerebellum (1–6 small foci; 3/5 birds; case Nos. 12–14), and brainstem (6–7 small foci; 2/5 birds; case Nos. 12 and 13).
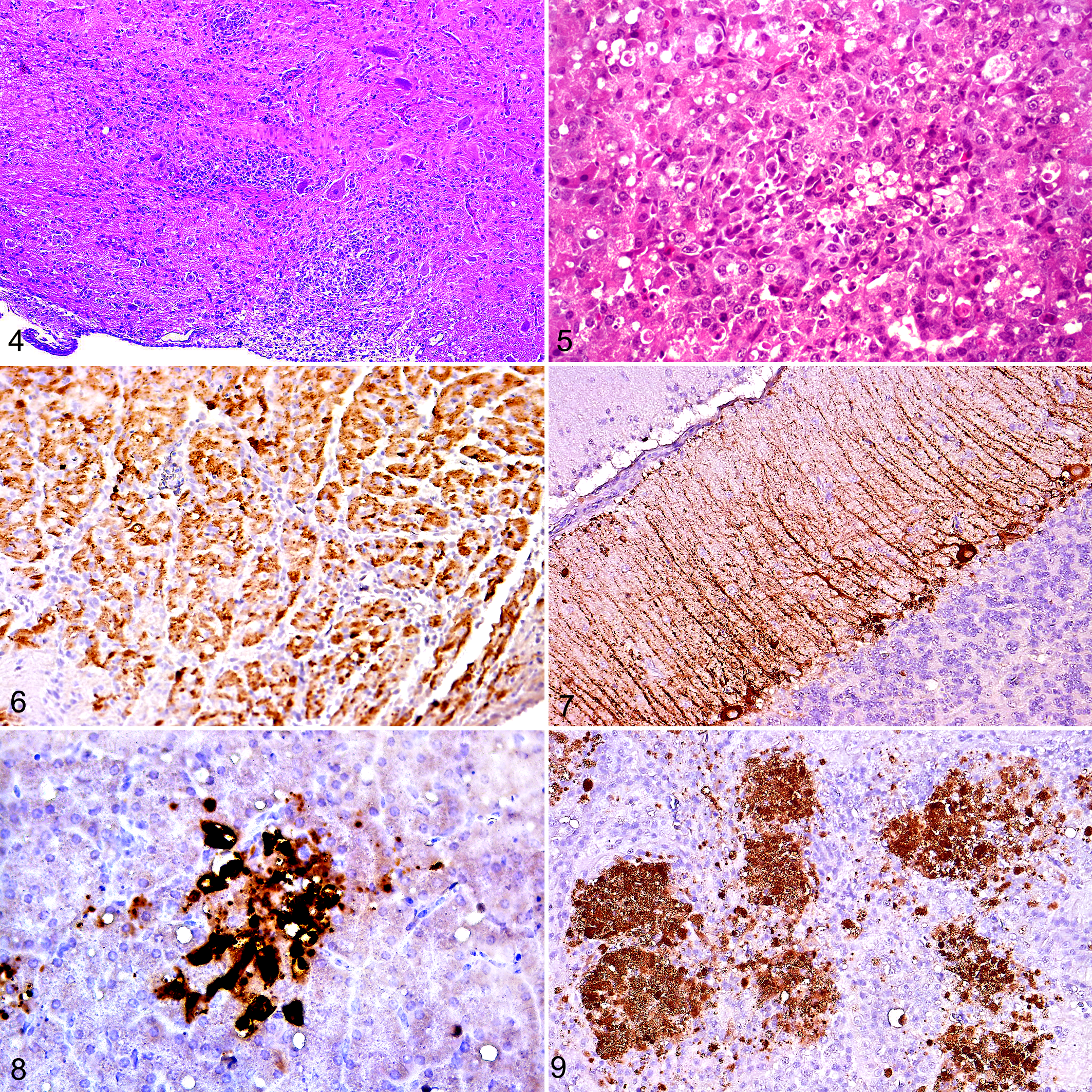
All 4 naive, WNV-inoculated, clinically affected birds had pancreatic lesions, 2 with mild lymphoplasmacytic pancreatitis (case Nos. 1 and 4) and 3 with varying degrees of necrotizing pancreatitis (from focal in 1 bird to 50% of parenchymal involvement in 1 bird; case Nos. 2–4; Fig. 5). Three of these 4 birds had moderate to severe adrenal necrosis affecting 10% to 15% of the parenchyma in 2 birds (case Nos. 1 and 3) and 50% in the third (case No. 2); 2 of these (case Nos. 1 and 2) had accompanying lymphoplasmacytic inflammation. One in 4 of these (case No. 3) had moderate necrotizing mucosal duodenitis. Four of 6 naive, WNV-inoculated, subclinically infected birds (case Nos. 6–9) had mild (1–3 foci; perivascular in 1; case No. 6) lymphoplasmacytic pancreatitis; 3 of 6 had adrenal cortical (interrenal) cell degeneration and necrosis, which was mild in 2 birds (case Nos. 6 and 8) and moderate in the third (case No. 10; affecting 20% of the section) accompanied by infiltrates of lymphocytes, plasma cells, and fewer heterophils. Three of 6 (case Nos. 5–7) grouse had periganglionic lymphocytes and plasma cells in the myenteric plexus. Vaccinated birds had rare accumulations of lymphocytes and plasma cells and fewer heterophils in the pancreas (case No. 13; 1/5 birds; 1 perivascular focus) and adrenal gland (case No. 12; 1/5 birds; 3 small foci of lymphocytes and plasma cells among chromaffin cells).
The renal interstitium in 3 of 4 naive, WNV-inoculated, clinically affected birds (case Nos. 1, 2, and 4) had rare (1–4), minimal foci of lymphocytes and plasma cells, and 1 of these birds (case No. 4) also had moderate mucus and urate accumulation in tubular lumens of the medullary cones and ureter. Five of 6 naive, WNV-inoculated, subclinically infected birds (case Nos. 5–8 and 10) had mild to moderate lymphoplasmacytic interstitial nephritis, which was occasionally perivascular.
Three of 4 naive, WNV-inoculated, clinically affected birds (case Nos. 1–3) had mild, multifocal, periportal, lymphoplasmacytic hepatitis, which was accompanied by moderate necrosis (affecting 10% of liver) in 1 of these (case No. 3). Five of 6 naive, WNV-inoculated, subclinically infected birds (case Nos. 5–9) had mild (1–8 foci), random to periportal, small lymphoplasmacytic accumulations in the liver. Three of 5 vaccinated birds (case Nos. 12, 14, and 15) had minimal (1–4 foci), random, lymphoplasmacytic hepatitis.
Compared with sham-inoculated, control birds, all inoculated birds (despite grossly small spleens) had splenic lymphoid hyperplasia to varying degrees: moderate in 4 of 4 naive, clinically affected birds (case Nos. 1–4); mild in 3 of 6 (case Nos. 5, 6, and 10) and moderate in 2 of 6 naive, subclinically infected birds (case Nos. 7 and 8); and mild in 4 of 5 vaccinated birds (case Nos. 11–13 and 15). Similarly, bursal lymphoid depletion varied among groups: moderate in 3 of 4 naive, clinically affected birds (case Nos. 1–3); mild in 4 of 6 naive, subclinically infected birds (case Nos. 5–8); and mild in 3 of 5 vaccinated birds (case Nos. 12, 13, and 15). GALT lymphoid depletion was also noted in the ceca of 1 of 4 naive, clinically affected birds (case No. 3) and 2 of 6 naive, subclinically infected birds (case Nos. 7 and 8) and none of the vaccinated birds.
Less common lesions among WNV-inoculated birds included moderate interstitial lymphoplasmacytic oophoritis in 1 of 4 of the naive, inoculated, clinically affected birds (case No. 2). One of 6 naive, inoculated, subclinically infected birds (case No. 8) and 2 of 5 vaccinated birds (case Nos. 11 and 15) had mild lymphoplasmacytic perivascular accumulations in the lung.
Microscopic lesions in sham-inoculated, control birds consisted of 1 focus of lymphocytes, plasma cells, and fewer heterophils in the myocardium of 2 of 3 birds (case Nos. 16 and 17); 1 of these (case No. 16) also had focal mineralized concretions surrounded by fibrous connective tissue in the endocardium of the left free wall. One of 3 (case No. 17) had 5 small foci of random, lymphoplasmacytic hepatitis.
No significant lesions were observed in skeletal muscle, lung, jejunum, and large colon of any of the grouse. In addition, vaccinated and naive, WNV-inoculated, subclinically infected birds lacked lesions in gonads and duodenum; vaccinated birds had no significant renal lesions.
Immunohistochemistry
Strong positive immunoreactivity (ie, to anti-WNV antibody) was evident in the cytoplasm of cardiomyocytes in 3 of 4 (case Nos. 2–4) WNV-inoculated, naive, clinically affected birds (affecting from 20%–60% of tissue sections; Fig. 6); was most prominent in the left ventricular free wall and septum; and corresponded to foci of degenerated and necrotic myofibers. One of 6 (case No. 6) naive, inoculated, subclinically infected birds had immunoreactivity for WNV antigen in the heart, which was limited to a single myocyte in the tunica media of an arteriole surrounded by a lymphoplasmacytic cuff in the left free wall.
The cerebrum of 1 of 4 (case No. 2) naive, inoculated, clinically affected birds had positive WNV immunohistochemical labeling of rare, single neurons; this bird was also 1 of 2 (case Nos. 2 and 4) that had labeling of the cerebellum. Cerebellar labeling in case No. 2 involved approximately 20% of the Purkinje cell neurons and their axonal processes, extending through the molecular layer (Fig. 7). The second of these birds (case No. 4) had 3, single, randomly scattered, immunoreactive neurons in the cerebellum and brainstem. The cerebrum of naive, inoculated, subclinically infected birds lacked immunohistochemical labeling, while 1 of 6 (case No. 6) had labeling in the cerebellum that affected approximately 8% of the molecular layer (9 folia) and consisted of sparsely labeled neurons and neuronal processes corresponding to areas of inflammation.
All 4 (case Nos. 1–4) naive, inoculated, clinically affected grouse had WNV-positive immunolabeling in the cytoplasm of pancreatic acinar cells, which corresponded to foci of acinar cell degeneration and necrosis and affected 1% to 5% of sections (Fig. 8). All 4 of these birds also had immunoreactivity within the cytoplasm of cortical cells in the adrenal gland that involved 2% to 25% of tissue sections and corresponded to foci of cell degeneration and necrosis (Fig. 9). No naive, inoculated, subclinically infected birds had immunoreactivity in the pancreas, and 2 of 6 (case Nos. 8 and 10) had minimal immunoreactivity in the adrenal gland that corresponded to foci of cortical cell degeneration.
The duodenum was immunoreactive for WNV antigen in 3 of 4 (case Nos. 2–4) naive, inoculated, clinically affected birds, with labeling limited to a small number of myocytes within the muscularis mucosa. No immunoreactivity was observed in the duodenum of naive, inoculated, subclinically infected birds. Minimal labeling was observed in the kidney of 3 of 4 (case Nos. 1–3) naive, inoculated, clinically affected birds and involved ≤1% of tissue. Immunoreactivity included single or 2 to 3 cell-long segments of tubular epithelium and was more often in distal tubules and medullary cones, as well as in the glomeruli, of 2 birds (case Nos. 1 and 3). No labeling was observed in the kidneys of naive, subclinically infected birds. In the spleen, rare, randomly scattered macrophages (cytoplasmic) and arteriolar walls were immunoreactive in 3 of 4 (case Nos. 1–3) naive, inoculated, clinically affected and 2 of 6 (case Nos. 8 and 9) subclinically infected birds, affecting between 1% and 5% of tissue sections. One (case No. 2) naive, inoculated, clinically affected bird had immunoreactivity in the ovary, involving single or small clusters of stromal cells.
No WNV-inoculated, vaccinated grouse or sham-inoculated, in-contact control grouse had immunohistochemical labeling in the tissues examined.
Serology
All 10 naive, WNV-inoculated grouse had ≤55% neutralization (ie, interpreted as seronegative) at 0 DPI. Among the 6 birds that survived to 14 DPI, serum PRNT90 titers at this time were from 320 to 1280. There was 100% agreement between serologic results for serum and Nobuto strip–eluted samples (ie, positive or negative); however, PRNT90 titers of Nobuto strip–eluted samples were lower than corresponding serum samples in 5 of 6 of these birds (case Nos. 6–10; Table 4).
Anti–West Nile Virus (WNV) Antibody PRNT90 Neutralization Titers Determined From Sera and Nobuto Strip–Eluted Samples Following WNV-Experimental Inoculation of Ruffed Grouse.a
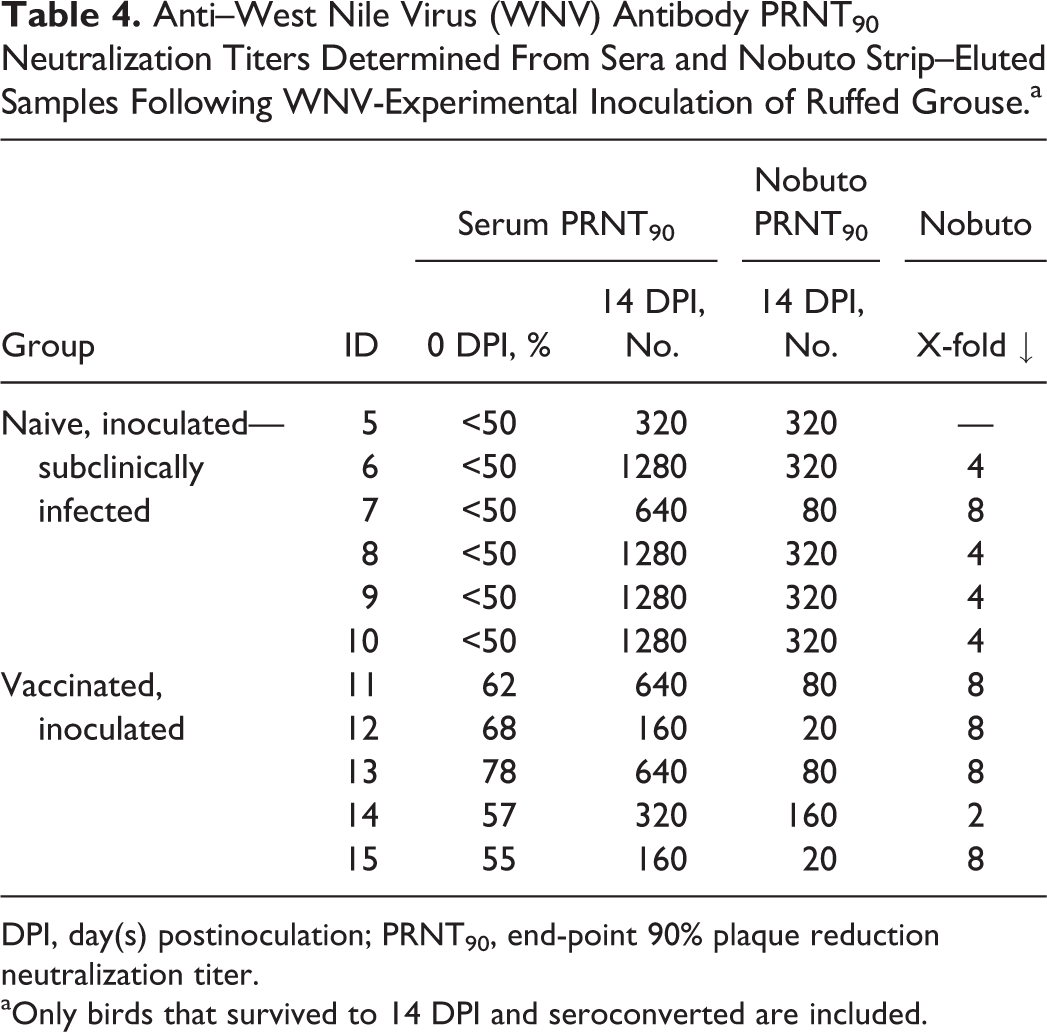
DPI, day(s) postinoculation; PRNT90, end-point 90% plaque reduction neutralization titer.
aOnly birds that survived to 14 DPI and seroconverted are included.
Vaccinated birds had from 55% to 78% serum neutralization of WNV at 0 DPI (ie, pre-WNV challenge); 1 of these birds was considered to have detectable anti-WNV antibodies at this time (case No. 13; 78% neutralization). All of these vaccinated birds seroconverted by 14 DPI, with serum PRNT90 titers from 160 to 640. As with the WNV-inoculated, naive birds, there was 100% agreement between serum and Nobuto strip–eluted samples; however, Nobuto strip–eluted samples yielded lower PRNT90 titers in all birds (by 2- to 8-fold; Table 4).
All sham-inoculated, in-contact control birds were seronegative at 0 and 14 DPI.
Discussion
The ruffed grouse is highly valued as a game bird species and is the state bird of Pennsylvania. Although their continued existence is highly dependent on the availability of suitable habitat, WNV has had a suspected role in the precipitous declines in ruffed grouse populations that corresponded temporally with the arrival and continued seasonal circulation of WNV in Pennsylvania. Grouse populations in most of the state have since failed to recover (L. M. Williams, personal communication, 2016), and ongoing surveillance efforts (primarily mosquito pool testing) have revealed positive pools across much of the state from 2002 to 2016 (http://www.westnile.state.pa.us/surv.htm). The present laboratory study provides information toward a better understanding of the potential effects of WNV on free-ranging ruffed grouse in Pennsylvania. Such studies are needed to merge with field-collected data and observations to provide a more holistic and realistic view of the health risks of infectious diseases to ruffed grouse and other free-ranging wildlife. For example, ongoing field studies revealed marked losses of radio-marked greater sage-grouse and American crows corresponding with the arrival of WNV to their respective study sites, while experimental studies in these species have revealed high mortality rates. 5,6,13,17 However, studies focused on forest-dwelling birds, such as the ruffed grouse, present unique challenges, and thus the impacts of WNV infection in such species are understudied compared with more visible, peri-domestic passerine birds and raptors, some of which have reportedly undergone WNV-associated population declines. 11,14,21
Results from the present study support the suspicion that WNV poses an additional ruffed grouse population pressure to the well-recognized and longstanding conservation challenge of habitat loss. 8 Based on clinical and pathological responses to experimental infection observed in the present study, estimated WNV-associated morbidity and mortality of ruffed grouse may be as high as 90%. Forty percent of naive, inoculated birds succumbed to severe clinical disease in the more acute phase of infection, most likely due to a culmination of systemic infection leading to a poor plane of nutrition and hydration and severe cardiac lesions consistent with heart failure. These cardiac lesions were widespread, severe, and associated with abundant immunohistochemical labeling in most of these birds. In addition to these more acute deaths, and based on poor weight gain and severity of microscopic lesions in the heart and brain, an additional 50% of naive, inoculated grouse may have had longer-term (ie, >2 weeks postinfection) health effects associated with chronic WNV-associated disease. Underlying chronic disease may be exacerbated by seasonal challenges such as juvenile fall dispersal and postdispersal location of food and cover, as well as location of winter food and cover resources by juveniles and adults. WNV should be considered a potential additional pressure on ruffed grouse in ongoing and future population management strategies.
Ruffed grouse that succumbed to WNV infection in the present study had systemic infection with corresponding clinical signs (eg, dehydration, poor nutritional condition, and lethargy). Based on the histopathologic observation of severe, diffuse necrotizing myocarditis in these birds, cardiac failure was likely a major contributing cause of severe morbidity, culminating in euthanasia. This pattern was also observed with natural WNV infections in juvenile birds, including domestic geese (Anser anser domesticus), Rouen ducks, and mallards (Anas platyrhynchos). 1,7,32 Experimentally infected American crows also had multisystemic WNV disease, including acute myocardial degeneration with minimal lesions in the central nervous system. 24 Brain lesions were more severe and widely distributed in naive, inoculated, subclinically infected ruffed grouse on 14 DPI vs those euthanized at earlier time points (7–8 DPI) due to morbidity, suggesting that encephalitis may be a more chronic manifestation of WNV infection in ruffed grouse.
Despite the high experimentally induced, WNV-associated morbidity and mortality rates in ruffed grouse, viremia titers were moderate relative to those in other susceptible avian species, such as the American crow, blue jay (Cyanocitta cristata), house sparrow (Passer domesticus), common grackle (Quiscalus quiscula), American robin (Turdus migratorius), red-winged blackbird (Agelaius phoeniceus), and killdeer (Charadrius vociferous). 13 The duration of detectable viremia in ruffed grouse is consistent with other experimentally infected avian species, often lasting from approximately 1 to 5 DPI; 13 however, viremia lasted an average of approximately 2 days longer in clinically affected vs subclinically infected grouse in the present study. In addition, mean peak viremia titers in ruffed grouse with clinical disease were about 1.4-fold higher than those with subclinical infections. Viremia profiles of longer duration and higher titers were also observed in American vs fish crows (C. ossifragus), which was in part attributed to delayed and weakened humoral immune responses and higher mortality rates in the former. 24 Differing clinical outcomes may be based in part on the ability to control and clear infection, which, in turn, may reflect both evolutionary (eg, genetics, immune response) and environmental factors (eg, nutritional condition, stress, underlying or prior infections). 24 The reason(s) for the minimal, late-onset viremia in 1 grouse (case No. 7) is unknown; this bird may have had enhanced immunity or failure of initial inoculation followed by later infection via shared water contaminated with infectious oral secretions or feces from co-housed grouse. The latter has been documented in experimental studies of corvid species and a ring-billed gull (Larus delawarensis); however, in most of these cases, viremia profiles (eg, peak titer and duration) were similar among needle- and mosquito-inoculated and cage mate–infected birds, unlike the present case. 13
The timing of oropharyngeal and cloacal WNV shedding in birds following experimental inoculation tends to parallel those of viremia, with titers higher for viremia than shedding, and oropharyngeal shedding titers higher than cloacal. 13 This pattern held true for experimentally infected ruffed grouse in the present study. The purpose of assessing viral shedding in the present study was to document infectious WNV in oropharyngeal and cloacal secretions in the early stages postinfection that generally become undetectable after approximately 1 week postinoculation, consistent with many other experimentally infected bird species. 13 The 2-fold higher mean peak oropharyngeal shedding titer observed in naive, subclinically infected vs clinically affected grouse was unexpected and may be attributed to small sample size and individual variation. Kidney and heart were selected for virus isolation in ruffed grouse because they have proven to be tissues with consistently high WNV titers in past avian WNV experimental studies and can be rapidly collected from carcasses and, therefore, have been commonly used in WNV surveillance. 13,19,31 In addition, feathers may be useful for WNV testing of field-collected carcasses in poor condition (eg, long interval between death and postmortem exam) as they may be stored at room temperature for extended periods. 25 Results here support that these tissues would be useful for WNV surveillance and diagnosis in field-collected grouse. Skeletal muscle was included due to its application to hunter-handling of carcasses. Although skeletal muscle of grouse (and other birds) that died relatively acutely due to WNV infection contained infectious virus, the risk of infection due to handling and consumption of meat is minimal when appropriate hygiene and cooking temperatures are achieved. 28
Vaccination has been considered in free-ranging corvids for controlling WNV outbreaks; 4 however, the logistical challenges of vaccinating free-ranging birds, including ruffed grouse, are difficult to overcome. A vaccination cohort was included in the present study because this management practice is used in some captive (breeding) grouse flocks and, in dire circumstances, may provide an option to attempt to mitigate the effects of WNV on wild bird populations. Results from the present study suggest that vaccination with a licensed (killed) equine WNV vaccine would provide protection against WNV-associated morbidity and mortality, while excluding vaccinated birds from transmission cycles. The seemingly high level of protection of a single, small dose of equine-licensed vaccine is noteworthy because the same vaccine product failed to protect greater sage-grouse as well as American crows following experimental WNV challenge. 4,6 Previous studies of WNV vaccination in birds have assessed both equine and novel vaccine candidates, with highly variable rates of seroconversion and protection. 22
The present study also included an assessment of the potential utility of Nobuto strips for testing for anti-WNV antibodies via PRNT (ie, the gold standard) in grouse. Similar to previous work, 10 results from the present study suggest that although the PRNT90 antibody titer was often decreased by at least 4-fold in samples eluted from Nobuto strips vs sera, antibodies to WNV were consistently detected in paired samples of both types. Thus, Nobuto strips are currently used in ongoing field research in Pennsylvania to estimate WNV seroprevalence among hunter-harvested, free-ranging ruffed grouse. We believe that the logistical advantages of the use of Nobuto strips for field-collected blood samples (ie, long-term storage at ambient temperatures) outweigh this apparent loss of sensitivity.
Numerous factors appear to affect WNV infection outcome in birds, including taxonomy and age. 13,20 For example, juvenile birds (as used in the present study) may be more susceptible to clinical disease and higher viremia titers associated with WNV infection. 1,7,23 Although fully mature ruffed grouse may have lower susceptibility to WNV-associated morbidity and mortality, the juvenile life stage corresponds to the peak months of WNV transmission in much of the eastern United States. In addition, population studies in the Appalachian region identify productivity (ie, chick survival and fecundity) as the most influential demographic factor on population growth; 9 thus, WNV and other potential challenges to juvenile survival and productivity should be recognized and considered in population management strategies. We recognize that WNV experimental trials may underestimate natural rates of disease, which may be partly related to inoculation methods. However, needle inoculation, as used in the present study, has been shown to lead to similar infection outcomes in birds as mosquito inoculations with WNV and other arthropod-borne viruses. 26,30
Conclusions
The present study is the first to assess the outcomes of WNV experimental infection in ruffed grouse and was prompted by observed population declines with failure to recover in free-ranging grouse in Pennsylvania following the introduction and establishment of WNV. Laboratory-derived data reported herein suggest that WNV may serve as an additional population pressure on ruffed grouse in Pennsylvania. Observed morbidity, as well as pathology and immunohistochemistry following experimental infection, revealed acute mortality as well as potential chronic WNV-associated health effects in grouse. Additional research is needed to expand upon these laboratory data and better characterize WNV infection in ruffed grouse in the field, including how WNV may affect populations along with other factors such as habitat loss.
Footnotes
Supplementary material for this article is available online.
Acknowledgements
We thank our Forestry and Food and Cover colleagues at the Pennsylvania Game Commission for assistance in locating grouse nests. Paul Gordy and Airn Hartwig at Colorado State University provided logistical support and performed immunohistochemistry, respectively. Dan Snyder, John Redmon, and Tim and Debbie Flanigan performed cross-country transport of grouse eggs; Dan Snyder also provided invaluable expertise in grouse transport logistics and husbandry. Dr Laura Kramer (New York State Department of Health) provided the WNV inoculum strain.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Funding partners included Ruffed Grouse Society, Woodcock Limited, and Snyder’s Grouse and Waterfowl Park (Council, Idaho). Additional funding support was provided by Animal Modules Core 21-21500 (Colorado State University) and the Natural Sciences and Engineering Research Council of Canada.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
