Abstract
Canid alphaherpesvirus 1 (CHV) causes morbidity and mortality in susceptible puppies. While the neuropathology of experimentally infected puppies has been detailed, characterization of naturally acquired infections is limited. The aim of this study was to describe the histologic, immunohistochemical, and in situ hybridization features of CHV encephalitis in the dog. Six female and 11 male puppies ranging in age from stillborn to 57 days old were included. Histologically, lesions included multifocal glial nodules (16/17, 94%), meningeal infiltrates (15/17, 88%), and cerebellar cortical necrosis (6/9, 67%); however, robust inflammation was not a significant feature in any of the cases. Immunohistochemistry for CD3, CD20, MAC387, and Iba1 was performed. Although T cells predominated over B cells, the overall number of cells was small in all cases both within the glial nodules and the meninges. In 16 of 16 (100%) cases, glial nodules were diffusely immunoreactive for Iba1; however, limited or no immunoreactivity for MAC387 was present. In situ hybridization directed at the CHV thymidine kinase gene revealed CHV nucleic acid in the granule neurons of the cerebellar folia (8/9; 89%), endothelial cells in the meninges and parenchyma (12/17, 71%), and individual randomly distributed neurons (6/17, 35%). These results clarify the pathology of naturally acquired CHV infection and indicate that developing cerebellar granule neurons are an important site of viral replication.
Keywords
Canid alphaherpesvirus 1 (CHV) is a common viral infection of wild and domestic canids with a worldwide distribution. CHV infection of older puppies and adults generally results in mild recurrent genital, respiratory, or ocular disease while systemic infection in neonatal puppies is almost always fatal and frequently affects the entire litter. 2,3,13 Similar to other α-herpesviruses, CHV remains dormant in ganglia following infection and can recur after periods of stress or immunosuppression. 5,9 Neonatal puppies can acquire the infection in utero, during passage through the birth canal, from direct contact with secretions from infected littermates or the dam, or rarely from fomites or local contamination. 2 –4
Infection of newborn puppies typically between the ages of 1 and 3 weeks results in a systemic, cell-associated viremia with highest viral concentrations localized to the adrenal glands, kidneys, lungs, spleen, and liver. 2 Cellular damage in these tissues manifests as characteristic multifocal areas of hemorrhage and necrosis. 2 Clinical manifestations include persistent crying, dull or depressed attitude, loss of interest in suckling, soft yellow-green stool, and abdominal discomfort on abdominal palpation. 2 The virus has also been implicated as one of a plethora of potential causes for fading puppy syndrome.
In most cases, puppies die before neurological signs develop, although a variety of reports in predominately experimental settings describe the neuropathology of CHV to varying degrees. 10 –12 The bulk of the experimental data on CHV encephalitis was performed in the 1960s in which widespread nonsuppurative meningoencephalomyelitis characterized by destruction of gray and white matter with focal gliosis was described. 10 –12 The most severe changes were located in the cerebellar cortices and the nuclei of the pons, midbrain, and thalamus. 11 Inflammatory infiltrates consisted mostly of lymphocytes within foci of gliosis and mildly expanding the perivascular space. 11 Direct fluorescent-antibody testing of the experimentally inoculated pups revealed that regardless of inoculation route (intraperitoneal or oronasal), the distribution of virus within the central nervous system had the highest frequency in the cerebral cortex followed by the diencephalon, mesencephalon, spinal cord, and olfactory bulb. 11,12 The pattern of distribution varied from diffuse to discrete aggregations of infected cells in the leptomeninges, brain, and spinal cord. 11 Within the cerebellar cortex, immunofluorescence for CHV was concentrated in the granular layer with extension into the molecular layer. 11
To complement the experimental studies, we sought to define the histologic lesions, inflammatory infiltrate, and localization of viral nucleic acid in cases of naturally acquired CHV. Although the experimental data indicate widespread neurologic lesions and viral infection, natural cases of CHV encephalitis are poorly described and lack the diffuse nature described experimentally. Therefore, we hypothesized that the distribution of viral nucleic acid would be more limited and would include endothelial cells and areas of robust neurogenesis in the developing puppy brain—namely, the cerebellar cortex and subventricular zone.
Materials and Methods
Case Selection and Histology
The pathology database at the New York State Animal Health Diagnostic Laboratory, Cornell University College of Veterinary Medicine (Ithaca, NY) was searched for cases of CHV-associated disease in the dog from 2007 to 2016. Only cases in which brain was collected and trimmed for processing were included regardless of the presence or absence of histologic lesions in the brain. Locations of the brain sections examined in each case are recorded in Supplemental Table S1 based on the 5 subdivisions of the brain. Necropsies were performed within 24 to 72 hours of death. Tissues were collected, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained via hematoxylin and eosin (HE). All cases had gross and histologic evidence of disseminated canine herpesvirus, including adrenal, hepatic, and renal necrosis and hemorrhage.
Immunohistochemistry
In total, 5-μm paraffin sections of formalin-fixed tissues were used for immunohistochemistry. Sections were deparaffinized in xylene and hydrated in graded ethanol. Endogenous peroxidase activity was blocked with a 3% peroxide solution for all antibodies. Staining protocols are presented in Supplemental Table S2: for all antibodies, sections were treated with an avidin/biotin blocking kit (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions and serum-free protein block (Dako, Carpinteria, CA) for 10 minutes at room temperature prior to the application of the primary antibodies. Secondary antibodies were either biotinylated horse anti-mouse (CD3, MAC387; Vector Laboratories, Burlingame, CA) or biotinylated goat anti-rabbit (CD20, Iba1, GFAP; Vector Laboratories), diluted at 1:200 for 30 minutes at room temperature. Tertiary antibody was Vectastain ABC Elite (Vector Laboratories) incubated for 30 minutes at room temperature. 3,3′-Diaminobenzidine (DAB) chromogen (Dako) was used to develop the slides, and Mayer’s hematoxylin (Dako) was used as a counterstain. Positive control for the antibodies consisted of canine lymph node (CD3, CD20, MAC387, Iba1) and brain (GFAP), and isotype-matched antibodies were used as negative controls in adjacent sections of tissue.
Scoring based on the composition of CD3, CD20, and MAC387 immunoreactivity within glial nodules was designated as absent (0% of the cell population), rare (<5% of cells), mild (<20% of cells), moderate (20%–50% of cells), and marked (>50% of cells). There were no glial nodules with greater than 50% of cells with immunoreactivity for CD3, CD20, or MAC387. All glial nodules were greater than 90% positive for Iba1, so this parameter was not scored.
In Situ Hybridization
Based on previously published data used in designing in situ hybridization probes for canine herpesvirus, we used sequences of the CHV thymidine kinase gene as the basis for the in situ hybridization. 8,14 Probes were designed in collaboration with Advanced Cell Diagnostics (ACD, Newark, CA). They used the sequence of the CHV thymidine kinase gene (Genebank X75765.1) to design 18 different probes that bound to various regions of this gene. The sequence of these probes is proprietary, and additional information can be found at https://acdbio.com/search/site/%252A457121%252A/cms/probes. The 5-μm sections were cut and stored at –80°C prior to staining. The sections were deparaffinized in xylene, washed with ethanol, and dried. The staining was performed according to the manufacturer’s protocol for colorimetric in situ hybridization (ACD) for brain tissue. Slides were treated with H2O2 (ACD) to block endogenous peroxides for 10 minutes. Slides were antigen retrieved by boiling for 15 minutes in antigen retrieval solution (ACD) and then treated with proteinase K (ACD) for 30 minutes. Slides were incubated for 2 hours with CHV probes (ACD), and then 6 amplifications steps were performed with ACD reagents. The bacterial gene DapB probe (ACD) was used as a negative control. Slides were developed with DAB chromogen (ACD) for 10 minutes and counterstained with Mayer’s hematoxylin (Dako). Positive control tissue was adrenal gland from a puppy infected with CHV.
Molecular Characterization
DNA was purified from two 15-μm-thick scrolls using QIAampe DSP DNA FFPE Kit (cat. 60404; Qiagen, Hilden, Germany) according to the manufacturer’s instructions. We designed primers for the thymidine kinase gene (Genebank X75765.1) using NCBI primer blast. The primer pairs generated a 100-bp product; the forward primer sequence was 5′-TTCCGGAACCTATGGCATTCT, and the reverse primer sequence was 5′-AGGCTTAATTCACCACGCAG. All polymerase chain reactions (PCRs) were carried out in a 50-μL volume using DreamTaq Green PCR Master Mix (cat. K1081; Thermo Fisher Scientific, Waltham, MA). Cycling conditions were as follows: denaturing step at 95°C for 3 minutes; 35 amplifications consisting of 95°C for 30 seconds, 55°C for 30 seconds, and 72°C for 1 minute; and the final extension at 72°C for 5 minutes. Positive control was naturally occurring cases of CHV in which virus isolation confirmed infection. Negative control was uninfected canine tissue. QIAquick Gel Extraction Kit (Qiagen) was used to purify DNA from the gel. DNA was then sequenced and aligned to known CHV sequence via nucleotide BLAST.
Results
Case Features and Histologic Findings
Seventeen cases were included in the current study based on the availability of brain sections with the complete necropsy (Supplemental Table S1). Six female and 11 male puppies ranging in age from stillborn to 57 days old were included. Breeds represented were Labrador Retriever (7/17), Bernese Mountain Dog (3/17), Boxer (2/17), French Bulldog (2/17), and 1 each of Doberman Pinscher, Siberian Husky, and Queensland Heeler (Table 1). PCR testing of paraffin-embedded brain tissue was positive for CHV in all cases (17/17) (Supplemental Fig. S1).
Clinicopathologic Features of Canine Herpesvirus Encephalitis.
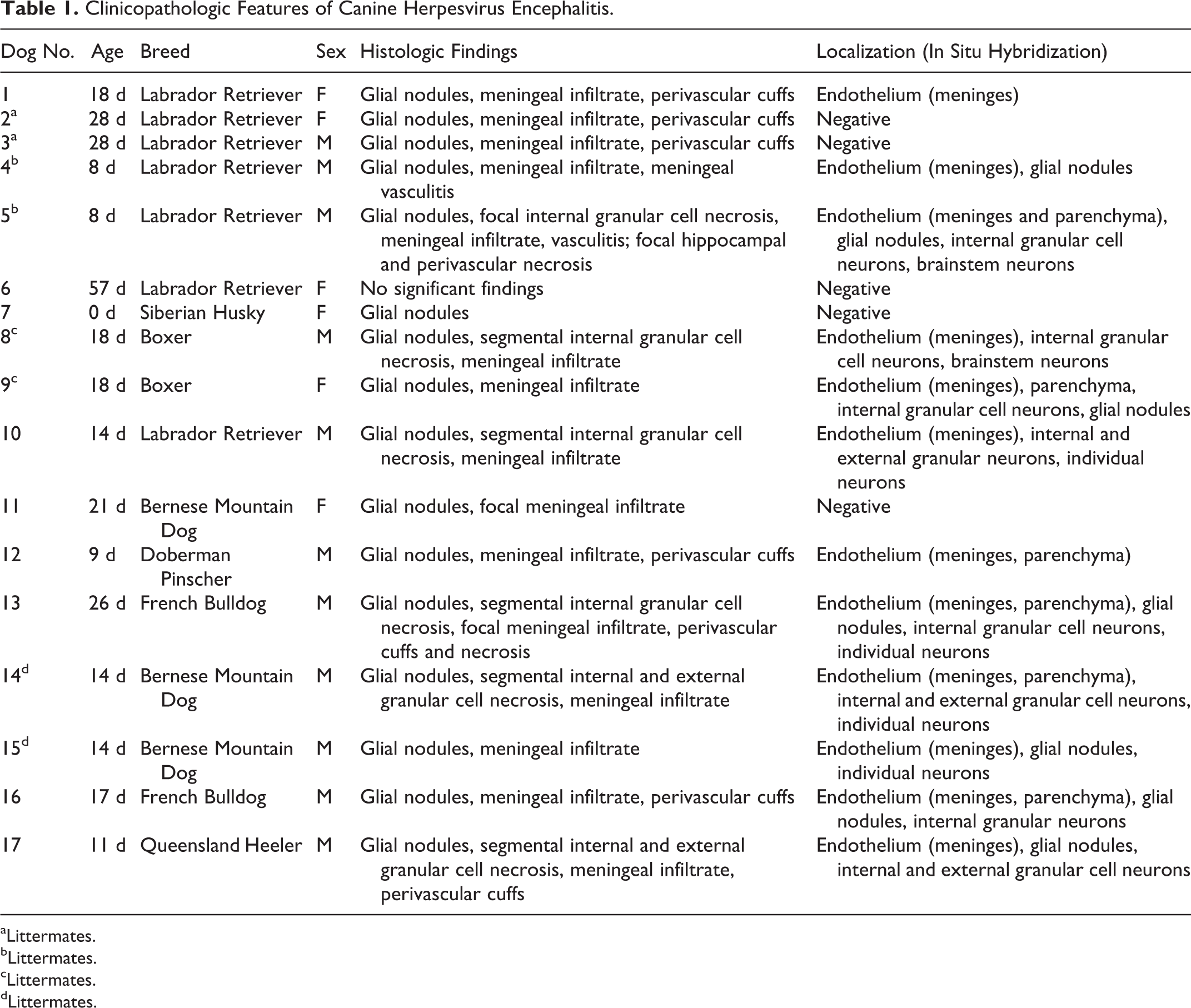
aLittermates.
bLittermates.
cLittermates.
dLittermates.
Histologically, cases had lesions that were characterized as multifocal glial nodules (16/17, 94%; Fig. 1), meningeal infiltrates of lymphocytes (15/17, 88%; Fig. 2), and cerebellar cortical necrosis (6/9, 67%; Fig. 3). In all cases except 1 (dog No. 6), glial nodules were randomly distributed throughout the white and gray matter of the parenchyma and adjacent to blood vessels of the cerebrum, brainstem, and cerebellum. The nodules were predominantly composed of mixed mononuclear cells with variable numbers of degenerate cells. In general, the degree of meningeal changes was equivalent to the severity of necrosis or formation of glial nodules in the subjacent parenchyma. The meninges in a majority of cases (14/17) were mildly to moderately, diffusely or segmentally thickened by lymphocytes and macrophages, with the most significant infiltrates overlying regions of necrosis in the cerebellum. Less common features included individual necrotic cells in perivascular infiltrates (2/17) and fibrinoid necrosis of meningeal vessels (1/17; Fig. 2). Inclusion bodies were rare and identified in presumptive endothelial cells only in 1 case (dog No. 10, Fig. 4). Satellitosis and neuronophagia were not observed.
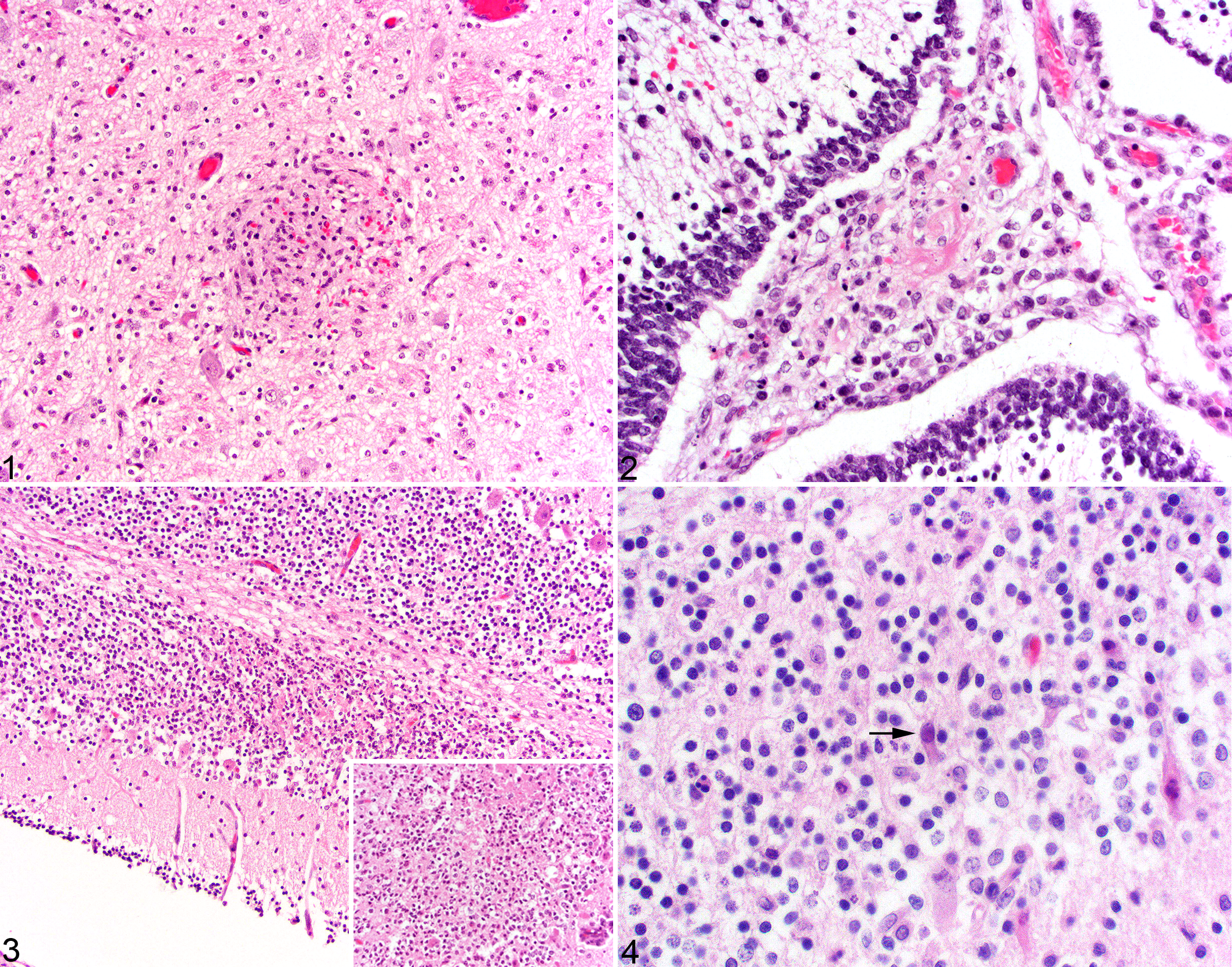
The cerebellar lesions consisted of discrete segments of the cerebellar folia that had mild to severe necrosis of the granular layer with abundant karyorrhexis, nuclear debris, loss of cellular definition, and small amounts of fibrin (6/9). In 4 of 6 (67%) of these cases, only the internal granular layer was involved, while the remaining cases (dog Nos. 14 and 17) had narrow segments of necrosis in the overlying external granular layer. Adjacent to these regions of necrosis were small numbers of degenerate Purkinje cells and a few, scattered lymphocytes.
Immunohistochemical Findings
CD3+ T lymphocytes were detected in glial nodules of 13 cases (13/17; Fig. 5) with immunoreactivity ranging from absent to 50% of the cells in the nodule. In the majority of glial nodules, less than 20% of the cell population was composed of CD3+ cells. Across all 17 cases, 41 of 149 (28%) nodules were devoid of CD3 immunoreactivity, and 58 of 149 (39%) had rare, 33 of 149 (22%) had mild, 17 of 149 (11%) had moderate, and 0 of 149 had marked infiltrates of CD3+ cells.
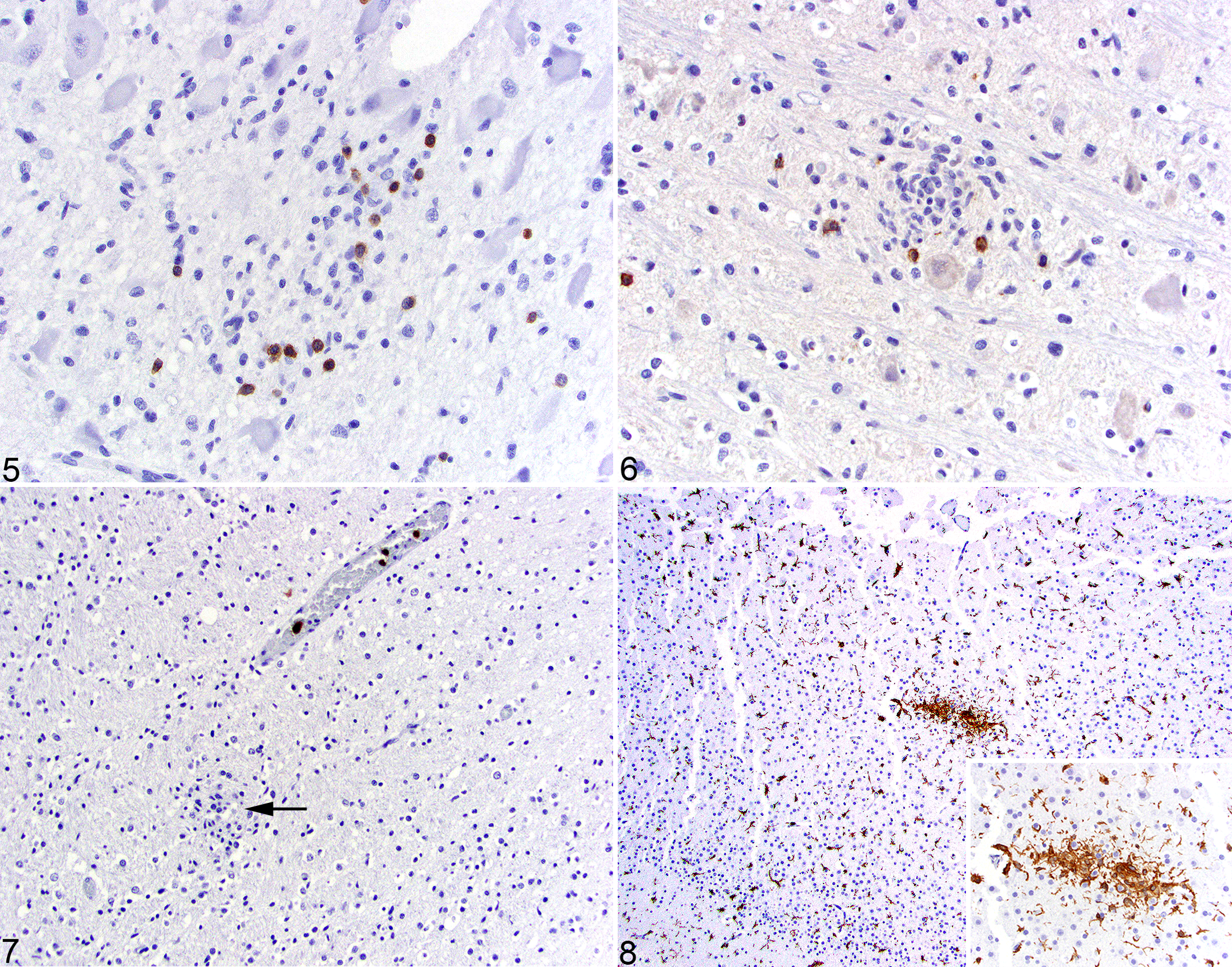
CD20+ B lymphocytes were present in glial nodules in a minority of cases (7/16, 44%; Fig. 6). Within these nodules, B lymphocyte infiltration was designated as absent (115/132, 87%), rare (14/132, 11%), or mild (3/132, 2%). The meninges in a majority of cases (13/16, 81%) contained small numbers of B lymphocytes. Areas of necrosis in the cerebellar internal granular layer had small numbers of individual, scattered B lymphocytes.
MAC387+ macrophages with strong cytoplasmic immunoreactivity were absent in the majority of glial nodules (84/112, 75%; Fig. 7) but comprised 5% or less of the cellular population in a minority of the nodules (28/112, 25%) represented in 10 of 17 cases (59%). MAC387+ cells were absent in the cerebellar folia except in regions of necrosis, where small numbers of immunoreactive macrophages were present. While there were small numbers of immunoreactive cells in the meninges, there were focal increases in areas overlying cerebellar necrosis.
Iba1 had strong, diffuse, membranous, and cytoplasmic immunoreactivity in all of the glial nodules (Fig. 8). The density of cell processes within these nodules was more pronounced compared to those of the normal microglia in the adjacent parenchyma. Similarly, Iba1+ cells were found in discrete perivascular clusters in the meninges. Discrete segments of necrosis within cerebellar folia had small numbers of scattered Iba1+ cells; however, there were frequently small numbers of glial nodules throughout the folia not associated with necrosis. Seven of 17 cases (41%) had mild perivascular expansion of the Virchow-Robin space by Iba1+ cells.
In all cases, GFAP immunoreactivity exhibited no significant changes in the distribution and density of astrocytes, including in areas of glial nodules and cerebellar necrosis.
Distribution of Viral Nucleic Acid
CHV nucleic acid was consistently present in the granule neurons of the cerebellum with a predilection for the internal granular layer compared to the external granular layer (8/9, 89%; Fig. 9). Cells with strongly positive nuclear hybridization formed multifocal, well-demarcated regions within the cerebellar folia. These regions of necrosis ranged from small foci to larger regions (up to 15%) of the cerebellum in some sections. The pockets of positively hybridized granule cells within the folia were densely packed and comprised the majority of the cells within these areas of necrosis. Less commonly, CHV hybridization was present in single granule neurons throughout the cerebellum.
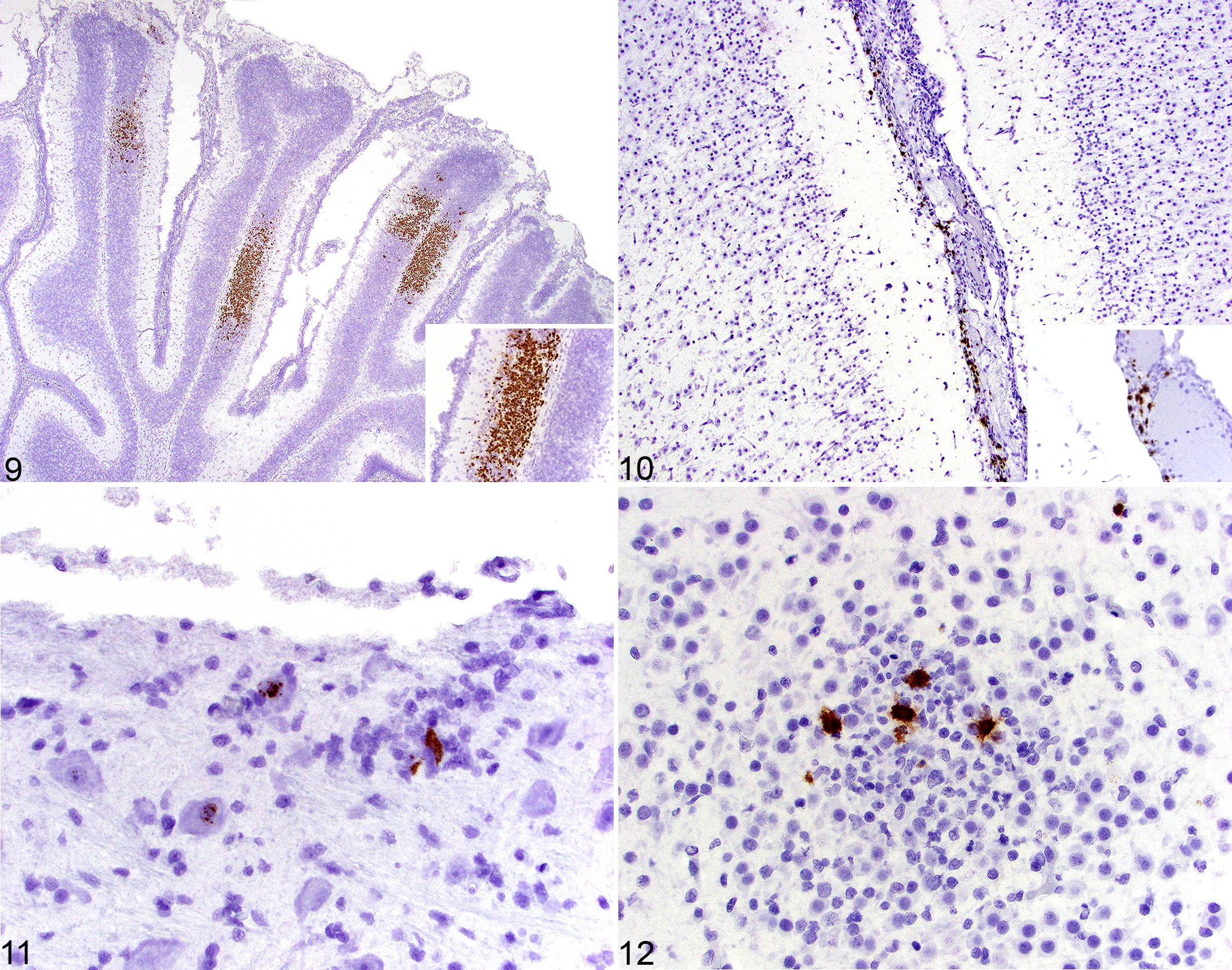
CHV hybridization was less common in other cell populations, including isolated endothelial cells in the meninges (12/17, 71%; Fig. 10) and parenchyma (6/17, 35%), as well as individual neurons (6/17, 35%), including those in brainstem nuclei (4/6, 67%; Fig. 11), hippocampus (2/6, 33%), cerebellar nuclei (2/6, 33%), and thalamus (2/6, 33%). These neurons were frequently located away from areas of necrosis or glial nodules, with no evidence of karyorrhexis. Hybridization of cells in glial nodules was less common (7/17, 41%; Fig. 12) and could not be conclusively identified on morphology alone.
Discussion
In our present study of naturally occurring CHV meningoencephalitis, there were several similarities and differences with respect to the experimental studies. Most similar were the extensive regions of necrosis in the developing cerebellum. These lesions were restricted to the granular layer. In the developing canine brain, the subventricular zone is another region of the canine brain that is rapidly dividing; however, in none of the cases were histologic changes evident in this location, nor was there any positive hybridization signal. The receptor for canine herpesvirus remains poorly elucidated and likely includes heparan sulfate as well as unidentified receptors. 7 Based on the location of the lesions in the developing cerebellar cortex compared to the subventricular zone, the presence of a specific receptor in the granule neurons is possible. Although cerebral cortical lesions are described in experimental infections, our study failed to recognize any animals with segmental necrosis of the cerebral cortex.
Other viruses with cerebellar neurotropism in domestic species include feline panleukopenia virus and bovine viral diarrhea virus (BVDV). As a parvovirus, feline panleukopenia has a predilection for rapidly dividing cells, resulting in selective destruction of the external granule cell neurons following perinatal infections. 6 This differs from CHV infection in that alphaherpesviruses do not require a rapidly dividing cell population and further supports the hypothesis that a CHV-specific receptor may be present on the internal granule cells. Retention of axons was noted within the lesions, in cases with enough tissue to perform a Bielschowsky silver stain (data not shown). This further supports that virus distribution is dependent on patterns of cellular receptor expression, because if the viral infection caused only vascular damage, all structures (neurons, axons, and parenchyma) should be affected equally. Like feline panleukopenia, BVDV also has a tropism for the external granule cell neurons, which results in cerebellar hypoplasia following in utero infection with the cytopathic form of the virus. Unlike CHV or feline panleukopenia, BVDV results in significant cerebellar atrophy secondary to massive destruction of the Purkinje neurons and white matter laminae as a result of virus-induced necrosis and subsequent inflammation.
In view of the recognized endothelial tropism of CHV, endothelial cells were studied for viral hybridization, but histologic evidence of vasculitis or vascular degeneration was absent in all but one case. In addition, infarctive changes that would be expected in cases of vascular disease were not present in any of the cases. The role of vascular CHV infection on the pathogenesis of CHV meningoencephalitis is unclear. However, the presence of CHV-positive endothelial cells may reflect the passage of virus into the brain, with resultant changes as described.
The demonstration of glial nodules in the brainstem and cerebrum of affected animals was a significant finding. Glial nodules are a typical feature of viral and protozoal infections in domestic animals. The glial nodules in the CHV cases typically contained large numbers of Iba1+ cells that were morphologically consistent with ramified microglia. Iba1+ cells were absent within the regions of cerebellar necrosis, likely due to the acute nature of the necrosis. Importantly, MAC387+ macrophages were rare in all cases and were generally absent from the glial nodules. MAC387 is expressed by macrophages that have recently emigrated from the bone marrow and differentiated in tissues from monocytes. 15 The lack of significant MAC387+ macrophages indicates that the glial nodule is predominately formed by microglia rather than infiltrating macrophages. Although T lymphocytes were often found in association with these glial nodules as well as in the meninges of affected animals, their numbers were typically small, suggesting that T cell–mediated immune responses is not a significant feature of natural CHV encephalitis in the dog. The low number of B lymphocytes suggests that these cells were not a significant feature of the disease.
CHV hybridization signal was often absent in the glial nodules, supporting that these are healed lesions from previous virus-induced necrosis. CHV-positive cells were far more abundant in endothelial cells and cells of the cerebellar cortex. Although CHV is often a fatal disease, survival following CHV infection has been described and includes remnant glial nodules in affected puppies. 10 The punctate hybridization of viral nucleic acids in the nuclei of isolated neurons, in contrast to the more diffuse hybridization in other affected cell populations, may represent the establishment of latency in certain neurons. These findings raise the possibility that CHV may be an underdiagnosed cause of central nervous system disease in older dogs. 5 Similarly, CHV causes extensive necrosis in the developing retina, and recovery from viral infection could also lead to developmental abnormalities in the eye. 1 In 2 cases where ocular tissue was included (dog Nos. 4 and 5), widespread CHV nucleic acid hybridization was detected in all layers of the retina (data not shown).
A single previous study using in situ hybridization (ISH) to identify viral nucleic acid failed to find significant evidence of CHV within the central nervous system (CNS); however, in our study, all cases except 1 had significant histologic lesions and ISH signal located within the CNS. 14 Although this series was collected over a period of 8 years, no evidence of degradation of signal occurred during this time. This study demonstrates the histologic lesions, inflammatory cell infiltrates, and ISH localization of CHV nucleic acid in a collection of naturally occurring CHV cases. CHV has widespread tropism to CNS tissue, especially developing granule neurons in the cerebellum.
Footnotes
Acknowledgements
We thank Jen Patterson for assistance with the images. We also thank Advanced Cell Diagnostics for their assistance in probe development and optimization.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
