Abstract
Metallosis is the accumulation of metallic debris in soft tissues resulting from wear following total joint replacement. A dog was evaluated for lameness 4 years after total hip arthroplasty using a titanium alloy and cobalt chromium total hip system. Radiographs revealed severe acetabular component wear, implant-bone interface deterioration, and peri-acetabular osteolysis. During surgical revision, black periarticular tissue surrounded the implants. Histologically, there was fibrosis and granulomatous inflammation with abundant, intra- and extracellular, black, granular material and smaller amounts of clear punctate to acicular material. Laser capture microdissection followed by x-ray fluorescence microscopy indicated the material contained large amounts of titanium with smaller amounts of vanadium, cobalt, and chromium, confirming the diagnosis of metallosis. The clear material was birefringent under cross-polarized light, stained positive with Oil-Red-O, and thus was consistent with polyethylene. Metallosis exhibits characteristic gross and histologic lesions and is a differential diagnosis for aseptic loosening of hip implants.
Keywords
Metallosis is a postoperative complication in humans following arthroplasty with metallic implants. Metallosis is one form of “particle disease” associated with arthroplasty, where metallic debris and/or particles from other arthroplasty components (eg, polyethylene) are released by implant wear and delivered throughout the joint via synovial fluid. Subsequent histiocytic inflammation and activation of osteoclasts results in osteolysis, implant loosening, and failure, pain, and decreased range of motion. 3 –5 The metallic debris has been shown in humans via reverse transcription polymerase chain reaction (RT-PCR) to stimulate expression of proinflammatory cytokines, as well as T-cell and osteoclastic responses. 4
Metallosis in humans is typically recognized upon surgical revision of a prior arthroplasty by the accumulation of black periarticular soft tissues. It can sometimes be identified radiographically as a radiodense line along the joint capsule (“bubble sign”). Arthrocentesis may reveal gray or black synovial fluid. Microscopic evaluation of the abnormal tissue demonstrates granulomatous inflammation with black and birefringent particles. 3
Joint replacement surgery has become routine in surgical specialty centers as treatment for degenerative joint disease in dogs. To date, the veterinary literature contains 2 reports with descriptions of a total of 4 dogs with metallosis following arthroplasty. 8,10 In these cases, black periarticular pigment was seen and assumed to represent metallic wear debris. In this case report, we describe the pathology of post-arthroplasty metallosis in a dog and confirm the metallic origin of the observed pigment using x-ray fluorescence microscopy.
A client-owned, 5-year-old, male, castrated Greater Swiss Mountain Dog was evaluated for a 1-week history of right hind lameness at the Foster Hospital for Small Animals at the Cummings School of Veterinary Medicine at Tufts University. The patient had bilateral, moderate elbow dysplasia treated with bilateral arthroscopic removal of fragmented medial coronoid processes and bilateral hip dysplasia treated with bilateral total hip arthroplasty using cementless total hip systems (BFX; BioMedtrix, Whippany, NJ) 4 years prior. The total hip system had a titanium alloy cup (containing approximately 6% aluminum and 4% vanadium) with sintered chemically pure titanium beads, a polyethylene liner, a cobalt chromium stem with sintered cobalt chromium beads, and a wrought cobalt chromium head. The patient had excellent clinical function following perioperative pain management, exercise restriction, and gradual return to activity. There had been no clinical or radiographic evidence of implant complications.
Radiographs of the implant were performed and reviewed by a board-certified surgeon and board-certified radiologist. They revealed that the femoral head was eccentric within the acetabular component, and there was focal osteolysis of the right ilium cranial to the acetabular component, multifocal osteolysis of the right greater trochanter, a poorly defined soft tissue nodule caudomedial to the acetabular component and ischium with overlying mineral-opaque foci, and right hindlimb muscle atrophy (Fig. 1). These findings were consistent with severe polyethylene wear of the acetabular component, resulting in the eccentric head location, osteolysis of the ilium and greater trochanter, and possibly medial granuloma formation.
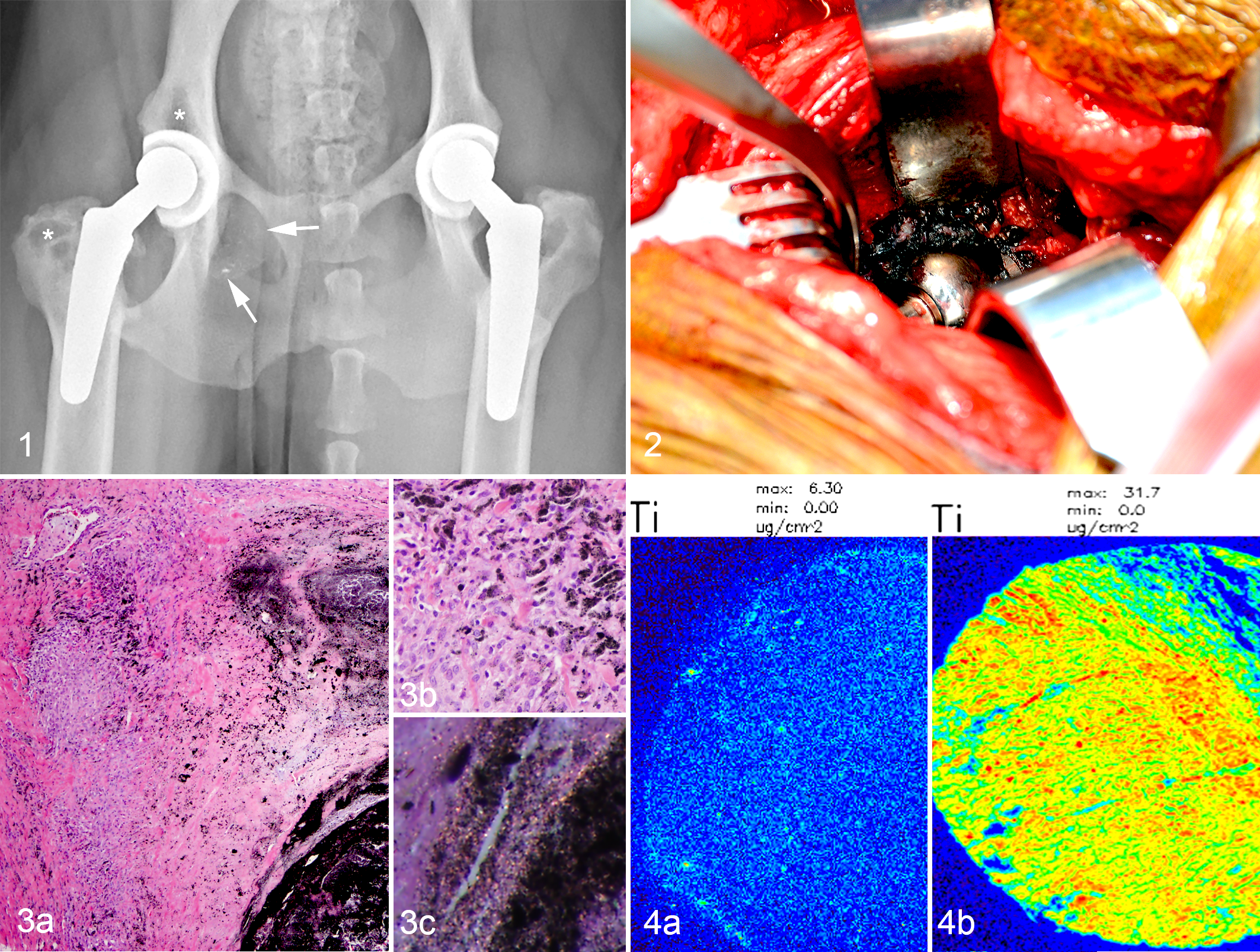
At surgery, the implant was surrounded by black, irregular tissue (Fig. 2) that was sampled for histopathology. The acetabular component and femoral head were replaced, and the patient was discharged with strict exercise restriction and a nonsteroidal anti-inflammatory.
The biopsies were formalin fixed, paraffin embedded, sectioned at 5 μm, and stained with hematoxylin and eosin. Histopathology of the periarticular tissue showed coalescing nodules of dense fibrous tissue and granulomatous inflammation with fine, black intrahistiocytic pigment and large, extracellular lakes of similar material (Fig. 3). Smaller amounts of fine, clear, punctate to acicular material that exhibited faint orange to white birefringence with cross-polarized light were admixed. Foci of metaplastic cartilage and segments of eroded to hyperplastic synovium were also present.
A previously described, 48-hour Oil-Red-O staining protocol stained the clear material orange-red, supporting that this material was polyethylene. 9 The black pigment was not attenuated by exposure to bleach and did not stain with Prussian blue, making melanin and iron unlikely, respectively.
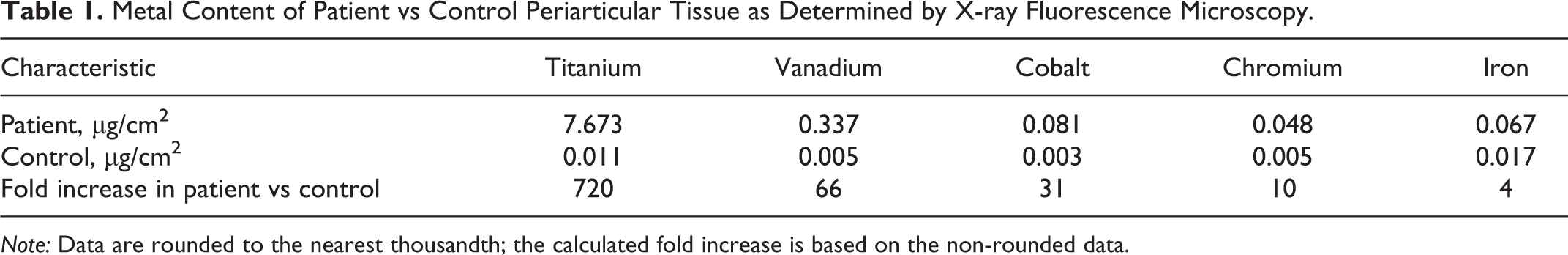
X-ray fluorescence microscopy was performed on the black material. Formalin-fixed, paraffin-embedded blocks of tissue were sectioned at 9 μm, placed on polyethylene naphthalate slides, and deparaffinized. Laser capture microscopy was performed (Arcturus Veritas platform; Arcturus Bioscience, Inc., Mountain View, CA) using a UV laser to excise areas of tissue containing black pigment. The excised areas were mounted on Si3N4 membrane grids (2.0 × 2.0 mm). 6 Samples were excited with incident synchrotron x-ray of 10 keV for elemental Kα characteristic emission lines. Elemental profiles were obtained using synchrotron scanning x-ray fluorescence microscopy (μ-XRF) at the Advanced Photon Source of the Argonne National Laboratory. The analysis showed markedly elevated levels of titanium (Fig. 4) with smaller elevations in vanadium, cobalt, and chromium and mild elevations in iron in the patient sample relative to the control (Table 1). The proportion of titanium and vanadium was roughly consistent with the alloy used in the cup, which was composed of approximately 90% titanium, 4% vanadium, 6% aluminum, and less than 1% of various other elements by weight. This suggested cup wear as the major contributor to the periarticular material with smaller contributions from the femoral components shown by the more modest cobalt and chromium elevations. The mild iron elevation could be from either the titanium alloy used in the cup (up to 0.3% by weight) or the femoral components (up to 0.75% by weight).
Metal Content of Patient vs Control Periarticular Tissue as Determined by X-ray Fluorescence Microscopy.
Based on these findings, a diagnosis of metallosis was made, with associated osteolysis, fibrosis, and inflammation. The much smaller amounts of polyethylene debris seen histologically potentially contributed to a lesser degree. The gross and histologic appearance of the periarticular tissue in this case was similar to descriptions of metallosis in humans and the few canine reports. 4,8,10
Nine months later, the patient presented again for reduced range of motion of the right coxofemoral joint. Computed tomography revealed progressive right hindlimb muscle atrophy, subluxation of the femoral implant, progressive lysis surrounding the acetabular component and of the femoral trochanter, and multiple punctate mineral to metal attenuating foci within the soft tissues dorsal to the greater trochanter. Caudal to the right acetabular component and extending slightly into the pelvic canal was an ovoid, 3-cm in diameter soft tissue attenuating, non-contrast-enhancing structure containing mineral- and metal-attenuating foci. The right medial iliac lymph node was slightly enlarged and contained mineral foci. Biopsies of the greater trochanter, acetabulum, and periarticular tissues indicated progressive fibrosis, black pigment-laden granulomas, and bone necrosis. The persistence and progression of these changes are thought to have been associated with residual metal and polyethylene particles from the original implant components despite surgical revision. Following luxation of the right coxofemoral joint 3 months later, another revision was performed. The dog was bearing weight well on both legs with minimal lameness 4 months after this last procedure (the last recorded follow-up visit).
The patient had potential risk factors for metallosis. First, as the dog had bilateral elbow dysplasia in addition to bilateral hip dysplasia, the bearing surfaces of the hip implants were likely loaded to a greater degree than those in patients with only hip disease. This may have resulted in more rapid and greater wear. Second, the implant used in this dog had textured surfaces that were manufactured by sintering metallic beads onto the surface. In humans, this technique has been associated with shedding of metallic debris into the joint space as beads loosen secondary to micromotion or poor bead fixation. When bead or bead fragments interpose between bearing surfaces (eg, femoral head and polyethylene liner of acetabular cup), wear is accelerated due to the resulting abrasive action of the debris during implant motion (so-called third body wear). Sintered bead fixation may have contributed to the occurrence of metallosis in this dog, but the majority of the wear debris likely resulted from wear of the femoral head on the metallic surface of the acetabular component once the polyethylene liner was worn through. Debris generated between broken metallic beads and the acetabular cup on the back or bone ingrowth surface (so-called backside wear) is another potential source of wear debris, but motion at this interface is in the micrometer range (vs millimeter range between femoral head and acetabular component), so the production of wear debris is expected to be less. The potential for development of complications due to shedding of sintered beads is addressed in newer implants using electron beam melting to texture the surface of the cup and stem. The effect of different texturization processes of biologic fixation implants on the potential for metallosis in veterinary patients requires further study.
In humans, the most prevalent particle type contributing to aseptic implant loosening is thought to be polyethylene; however, it is considered a less potent initiator than metal when considering similarly sized particles. 1 Evidence of polyethylene was seen in this case, but as it was present in much smaller amounts histologically than the metallic debris, it is assumed that the lesions were primarily due to released metallic particles.
Accumulation of metal particles within tissues in humans with metallosis has led to concern about the potential for systemic effects, including toxicity, carcinogenesis, or teratogenesis. 2,7 In this case, the computed tomography scan performed 9 months after the initial diagnosis of metallosis showed mineral to metallic dense material in a local lymph node. Although no systemic concerns exist for this dog at this time, this may indicate spread of implant-associated particles beyond the joint itself. Evaluation of metal levels in circulation or tissues in veterinary patients with metallic implants could warrant future study.
Metallosis in this case was diagnosed as a cause of lameness as a long-term complication of a total hip arthroplasty. Radiographically, there was implant loosening and osteolysis. Black periarticular tissue was identified grossly and was histologically composed of granulomatous inflammation and fibrosis associated with black material. X-ray fluorescence microscopy indicated the material around the joint contained titanium, vanadium, cobalt, and chromium and was thus secondary to implant wear. The potential for development of metallosis in dogs emphasizes the importance of regular radiographic and clinical monitoring of joint replacement patients, especially if the procedure is performed in a young animal or one with other orthopedic disease resulting in increased loading of the bearing surface.
Footnotes
Acknowledgements
We extend our appreciation for the technical support provided by the histopathology laboratory at the Cummings School of Veterinary Medicine at Tufts University and the Advanced Tissue Resource Center at Massachusetts General Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: Support was provided by Harvard NeuroDiscovery Center. This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under contract DE-AC02-06CH11357.
