Abstract
Suspected Streptomyces spp infections were identified in 4 cats at UC Davis Veterinary Medical Teaching Hospital between 1982 and 2011. Three had ulcerated, dark red mycetomas involving the dermis, subcutis, and fascia with fistulous tracts and/or regional lymphadenopathy. One cat had pyogranulomatous mesenteric lymphadenitis. Granulomatous inflammation in all cats contained colonies of Gram-positive, non-acid-fast organisms. All 4 cats failed to respond to aggressive medical and surgical treatment and were euthanized. Laser capture microdissection (LCM) was used to selectively harvest DNA from the affected formalin-fixed, paraffin-embedded (FFPE) tissues. Cloned amplicons from LCM-derived tissue confirmed the presence of Streptomyces spp in the dermatitis cases. Amplicons from the remaining cat with peritoneal involvement aligned with the 16S ribosomal RNA gene for Actinomycetales. Usually considered a contaminant, Streptomyces spp can be associated with refractory pyogranulomatous dermatitis and cellulitis in cats with outdoor access. LCM is useful in the diagnosis of bacterial diseases where contamination may be an issue.
Streptomyces species are pervasive bacterial saprophytes usually considered environmental contaminants when cultured from human and veterinary patients. 3,7 Only 13% of microbial cultures that were positive for Streptomyces spp were considered to represent true infections by this agent in a retrospective study of human cases. 3 Human Streptomyces spp infections typically present as mycetomas from environmental traumatic inoculation, particularly in exposed feet (Madura foot) of farmers in East Africa. 3,9 In Central America, Streptomyces spp proctitis has been associated with the use of plant material as toilet paper. 9 Disseminated Streptomyces-associated disease, including pneumonia, endocarditis, arthritis, pericarditis, lymphadenitis, and peritonitis, has been documented in immunocompromised human patients. 2,3,9 Risk factors for infection include human immunodeficiency virus, cancer, indwelling catheters, and Crohn disease. 2,3
Streptomyces spp infections pose a diagnostic challenge due to their ubiquitous environmental presence and morphologic, histochemical, genetic, and clinical similarities to other pathogenic bacteria within the order Actinomycetales. 3 Gram-positive bacilli of this order include Corynebacterium, Mycobacterium, Nocardia, Actinomycetes, Dermatophilus congolensis, and Streptobacillus moniliformis. 1 –3 Streptomyces are aerobic, long, filamentous Gram-positive, acid-fast negative bacilli that form dense clusters. 3 Actinomycetes are facultatively anaerobic and non–acid fast; Nocardia are aerobic and variably acid fast. 3 Clinical distinction is relevant because Streptomyces isolates are generally considered resistant to sulfa drugs used to treat Nocardia. 2,3,7
Streptomyces infections are poorly characterized in veterinary species with only sporadic anecdotal reports in cats, dogs, donkeys, dolphins, and ostrich eggs. 6 –8 This retrospective study identifies Streptomyces spp as an intralesional (pathogenic) agent rather than an environmental contaminant in cases of feline pyogranulomatous dermatitis and cellulitis. In this study, we specifically harvested and identified intralesional bacterial DNA by laser capture microdissection (LCM), cloning, and sequencing. Clinical outcomes, gross lesions, histologic examination, and microbial culture results were reviewed from suspected Streptomyces-infected cats to provide a clinical picture of this disease.
Medical records of the University of California, Davis, William R. Pritchard Veterinary Medical Teaching Hospital were searched for the terms feline and Streptomyces from 1980 to 2011. Four suspected clinical cases were identified between 1982 and 2011 (Suppl. Table S1), with 2 male and 2 female cats ranging from 2 to 11 years of age. All 4 cats had outdoor access. Cat Nos. 1, 3, and 4 were moderately anemic. There was no other clinical or postmortem evidence of immunosuppression. Cat Nos. 3 and 4 were feline immunodeficiency virus (FIV) and feline leukemia virus (FeLV) negative. Cat Nos. 1 and 2 had unknown FIV/FeLV status. Three cats were initially diagnosed as Streptomyces suspects based on culture of fresh tissue. Cat No. 2 was tentatively diagnosed based on histologic examination alone. Three cats (cat Nos. 1, 2, and 4) had surgical intervention, some multiple times, and all were treated with a variety of antibiotics (Suppl. Table S1). Disease duration averaged 12.75 months and ranged from 6 to 18 months. All animals failed to respond to surgical and/or antibiotic therapy and were euthanized because of disease progression.
Three of the 4 cats (cat Nos. 1, 3, and 4) presented with severe, regionally extensive, chronic, ulcerative dermatitis, cellulitis, and alopecia (Figs. 1, 2). The lesions consistently affected the limbs, potentially from environmental inoculation. Fistulous tracts communicated with large, deep, multinodular, gray, subcutaneous masses (mycetomas) that contained myriad brown-black granules (Fig. 3) similar to sulfur granules present in Actinomyces bovis–infected tissue. In addition to limb lesions, subcutaneous lesions in the lower abdomen and pelvis, as well as thoracic skin, were present in cat Nos. 1 and 4, respectively. Cat No. 2 had large masses involving mesenteric fat, lymph nodes, and spleen but no skin lesions. Peripheral or abdominal lymphadenomegaly was present in all 4 cats.
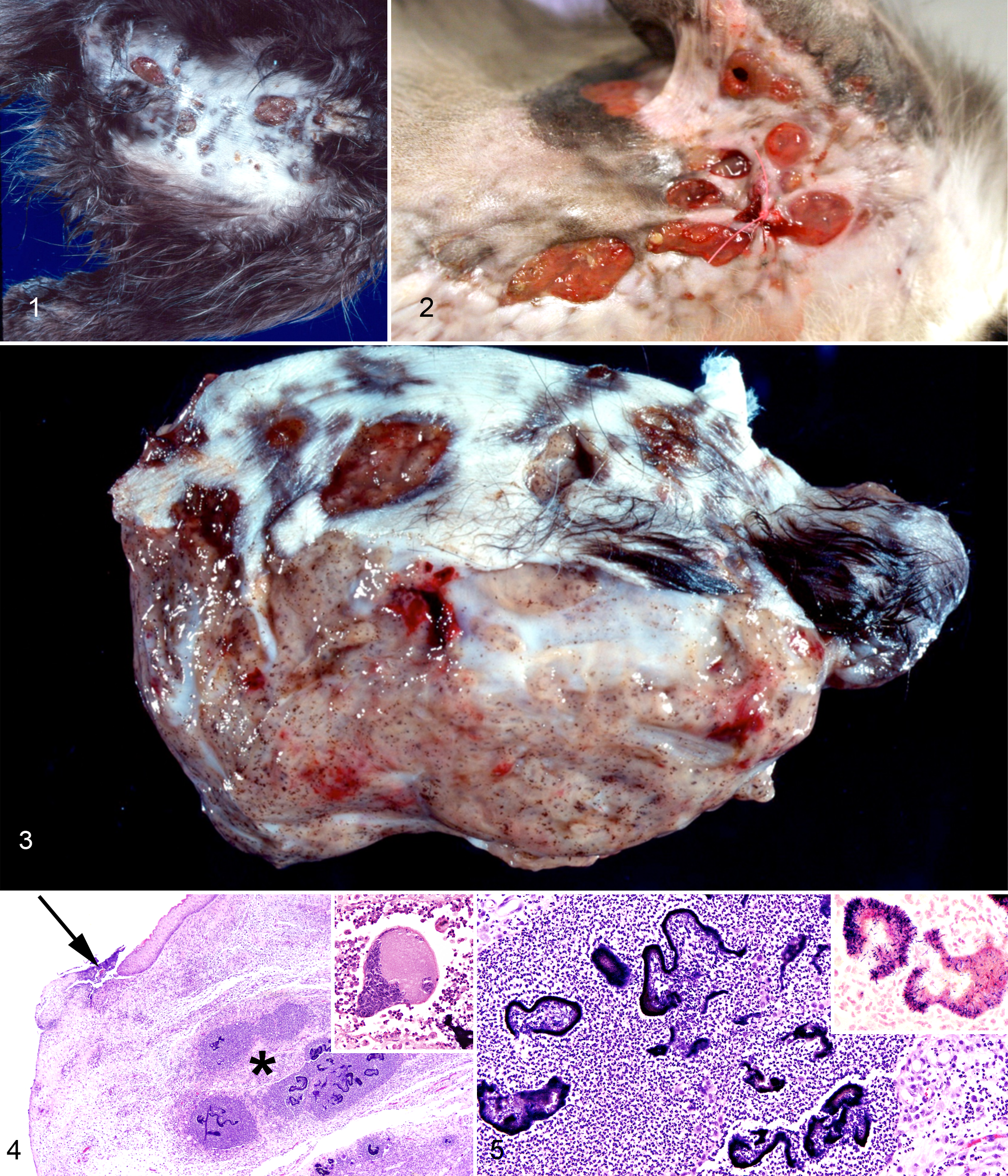
Skin. Dermatitis caused by Streptomyces spp, cats.
Lesions from all 4 cats consisted of severe, nodular, pyogranulomatous inflammation. Dense, deeply basophilic, serpentine bacterial colonies containing abundant, brown to magenta pigment (Figs. 4, 5) were surrounded by large numbers of viable and nonviable neutrophils, further circumscribed by epithelioid macrophages, large multinucleated Langhans cells, and foreign body type giant cells with up to 50 nuclei (Fig. 4, inset). Fibrin, edema, and layers of variably dense fibrous and granulation tissues massively expanded the surrounding regions.
Bacteria in lesions from cat Nos. 1, 3, and 4 were filamentous rods that formed dense tangled clumps. The bacteria from cat No. 2 were coccobacilli. The bacteria from all 4 cats were Gram positive (Fig. 5, inset) and Fite’s and Ziehl-Neelsen acid fast negative (Suppl. Table S1).
Intralesional bacteria were harvested from all 4 cats using LCM. Since the primary benefit of LCM is the selective molecular analysis of target tissue, it is important to ensure that the surrounding tissue has not contaminated the sample. Controls and procedures employed in the current study included cutting each section with a fresh microtome blade and using separate sterile DNA/DNAase-free reagents during all steps of processing. Three unstained 10-μm-thick sections of FFPE tissue were mounted on uncharged glass slides and deparaffinized. Deparaffinized slides were microdissected with a P.A.L.M. Microbeam IP 115 V Z laser (P.A.L.M. Microlaser Technologies AG, Bernried, Germany) and Zeiss Axiovert microscope (Carl Zeiss, Jena, Germany). To assess capture clarity after LCM, target tissue was visually confirmed to be selectively captured (Suppl. Fig. S1). Positive and negative controls were used during initial polymerase chain reaction (PCR) amplification (Suppl. Fig. S2). Genomic DNA from LCM samples was extracted and purified using the QIA amp DNA extraction kit (FFPE tissue kit; Qiagen, Venlo, Netherlands).
For amplification of target sequences, a single primer set was designed to distinguish a 208-bp region of Streptomyces 16S ribosomal RNA (rRNA) (GenBank: JN566155.1) from morphologically similar Nocardia 16S rRNA (GenBank: AB632394.1) and Actinomyces 16S rRNA (GenBank: HQ616392.1). Primers designed for longer 400-bp, 800-bp, 1205-bp, and 1327-bp amplicons failed to amplify DNA likely due to DNA shearing.
Based on sequence comparison and size restrictions, the forward primer 5′-TAGTGGCGAACGGGTGAGTAAC-3′ and reverse primer 5′-GCCCTCTCAGGCCGG-3′ were used (Integrated DNA Technologies, Coralville, IA). PCR-mediated target sequence amplification was performed using recombinant Taq DNA polymerase (Invitrogen, Carlsbad, CA). PCR products were purified and cloned into the pCR2.1 vector using the TOPO TA Cloning Kit (Invitrogen). See supplemental materials for a detailed description of methodology.
Amplicons from lesions isolated from cat Nos. 1, 3, and 4 had 99% to 100% homology with the Streptomyces 16S rRNA gene (GenBank: JN566155.1) (Suppl. Fig. S3). The 16S rRNA gene sequence from cat No. 2 did not have sequence homology to the 16S rRNA gene of the Streptomyces spp and was consistent with the divergent morphology of the bacteria and site involvement in cat No. 2. These findings suggest that the bacteria present in cat No. 2 were other members of Actinomycetales and not Streptomyces. PCR-amplified sequences from all cases had only 85% to 86% homology with the Mycobacteria 16S rRNA gene (GenBank: GU358075.1), further ruling out this agent as causative.
LCM offers a unique opportunity to molecularly confirm a histopathologic diagnosis of bacterial infection when the etiologic agent is unculturable, mixed with other bacteria, or an environmental contaminate. 4 Isolating high-quality bacterial DNA from FFPE tissue poses a number of challenges. 4 Formalin fixation causes depurination and fragmentation of DNA by hydroxyl ions. 5 DNA is also trapped by cross-linked proteins. 5 Authors have described a size limit of 400 bp for DNA extraction from FFPE. 5 Large quantities of nontarget DNA (including other bacteria) found in the whole section can have a dilutional effect if the bacteria of interest are not selectively targeted. 4 Also, target prokaryotes yield a lower amount of DNA compared with eukaryotic host tissue. 4 If the bacteria implicated in a tissue section are completely unknown, universal 16S rRNA gene primers can be used for amplification and sequencing. 4
In conclusion, Streptomyces spp can be associated with feline pyogranulomatous dermatitis and cellulitis and should not be summarily dismissed as environmental contaminants. Based on the Veterinary Medical Teaching Hospital clinical records during this 29-year period, the prevalence of Streptomyces-associated disease was approximately 0.05% of all cats seen and 0.36% of all feline dermatitis cases. Although feline Streptomyces spp infections appear rare based on the population evaluated in this study, the clinical outcomes suggest the prognosis is poor despite aggressive therapy. Outdoor access is a significant risk factor for disease.
Footnotes
Acknowledgements
We thank Diego Castillo, Chadwick Hillman, Barbara Elliott, Alicja Omanska-Klusek, Dr Emir Hodzic, the late Dr Peter Ihrke, and the National Institutes of Health Comparative Biomedical Scientist Training Program.
Author Contribution
Conception or design: RPT, RV, LJL, BM. Data acquisition, analysis, or interpretation: RPT, CMR, RV, EMS, CTS, CO, VKA, LJL, SDW, BM. Drafting the manuscript: RPT. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
