Abstract
Intensely eosinophilic and glassy intracytoplasmic inclusions were present in the neurons of the peripheral autonomic ganglia, Meissner’s and Auerbach’s plexus, and spinal ganglia in 20 aged white-nosed coatis (Nasua narica, 7–19 years old) and in 4 of 7 brown-nosed coatis (Nasua nasua, 2–21 years old) from multiple zoological institutions. Inclusions were single to numerous, sometimes distorting the cell. Pheochromocytomas were present in 5 of 16 white-nosed and 2 of 6 brown-nosed coatis, although no inclusions were present in the adrenal glands. Histochemically, immunohistochemically, and ultrastructurally, these inclusions were consistent with dense neurosecretory granules. Although similar inclusions have been reported sporadically in the adrenal medulla of humans and several other mammalian species as both incidental and pathologic findings, ganglionic inclusions reported herein appear to be unique and related to age in these species.
Keywords
Hyaline globules, also known as adrenomedullary inclusions or adrenal bodies, were first described by Oberndorfer in 1909 in the adrenal medulla of patients who died of infectious disease. 5 They have additionally been noted in pheochromocytomas in humans as well as in the adrenal medulla of patients with multiple sclerosis, amyotrophic lateral sclerosis, Parkinson disease, and other Lewy body disorders. 3,7 Although patients with Parkinson disease may have documented autonomic nervous dysfunction, Lewy bodies but not adrenomedullary inclusions are present in peripheral autonomic neurons. 7,9 Intracytoplasmic hyaline globules have been reported in the adrenal medulla of rats, hamsters, guinea pigs, rabbits, dogs, opossums, and rhesus macaques dying of polychlorinated biphenyl or dioxin toxicosis, severe bacterial infections, or unknown causes. 4 In many of the animals, globules correlated with adrenal necrosis and hemorrhage. These globules were periodic acid-Schiff (PAS) positive, diastase resistant, phosphotungstic acid hematoxylin positive, and autofluorescent. Histologically similar but histochemically and ultrastructurally distinct inclusions have also been rarely noted in hepatocytes and chromaffin cells of otherwise normal liver and adrenal medulla, respectively, as well as Kaposi’s sarcoma, yolk sac carcinoma, and carcinomas of the lung, liver, and breast. 3,6
White-nosed and brown-nosed (or South American) coatis (coatimundi) are procyonidae within the procyoninae subfamily and are related to ringtail cats, raccoons, kinkajous, and olingos. These species are popular in zoological parks. According to the World Association of Zoos and Aquariums, there were 207 white-nosed and 638 brown-nosed coatis in captivity in 2007 (www.waza.org). Glycogen storage disease has been reported in a brown-nosed coati, and Niemann-Pick disease (sphingomyelin lipidosis) has been reported in a raccoon (Procyon lotor). 1,8 We report and characterize herein hyaline globules in the peripheral autonomic neurons and ganglia of predominantly aged captive white-nosed and brown-nosed coatis of both sexes from 13 different zoological institutions.
Tissues were fixed in 10% neutral buffered formalin. Tissues were processed in an automated Tissue-Tek VIP processor and paraffin embedded with a Tissue-Tek TEC embedding station (Sakura Finetek USA, Torrance, CA). Sections were cut at 6 μm for routine hematoxylin and eosin, PAS with and without diastase, Ziehl-Neelsen acid fast, PAS-Luxol fast blue, and Sevier Munger staining.
For electron microscopy, the paradrenal ganglion was deparaffinized, postfixed in glutaraldehyde followed by osmium and potassium ferrocyanide, and embedded in Embed 812 plastic (Electron Microscopy Sciences, Hatfield, PA). Thin (1-μm) sections were cut and stained with toluidine blue to confirm orientation before imaging by transmission electron microscopy. Thin sections (70–90 nm) were mounted on copper grids and stained with 2% aqueous uranyl acetate and lead citrate. The sections were examined with a JEOL JEM 1400 transmission electron microscope (JEOL USA, Peabody, MA). An Orius SC 1000 bottom-mounted CCD camera (Gatan, Inc., Pleasanton, CA) was used to capture the images.
For immunohistochemistry, sections were cut and mounted on plus slides. Sections were stained for phenylethanolamine N-methyltransferase (PNMT; Abcam ab119784, 1:50) and catechol o-methyltransferase (COMT; Abcam ab126618, 1:50) with citrate antigen retrieval using the Vectastain Elite ABC kit and DAB chromagen (Vector Laboratories, Burlingame, CA) with Mayer’s hematoxylin counterstaining. Negative controls omitted the primary antibody. Sections were also stained with rabbit polyclonal antibodies to ubiquitin (Dako Z0458) and alpha-synuclein (Millipore AB5038). Negative controls used universal negative control serum containing mouse and rabbit antibodies (Cell Marque, Rocklin, CA).
All light microscopic images were obtained with an Olympus BX51 microscope and DP71 digital camera using cellSens Standard 1.6 imaging software (Olympus America, Center Valley, PA).
The index case (WN8) was euthanatized for humane and diagnostic purposes following a 3-month clinical history of weight loss and inappetence. She had been spayed 9 months prior for pyometra. Gross necropsy findings included poor body condition and moderate dental tartar and gingivitis; striking neuronal inclusions were the only notable histopathologic finding. Inclusions were present in the paradrenal (splanchnic) ganglion (Fig. 1), periductal ganglia of the pancreas and submandibular salivary gland, and the Auerbach’s and Meissner’s plexus of the esophagus, stomach, and small and large intestine. The inclusions were variably sized, 2–25 μm, often with a large clear halo (shrinkage artifact). Inclusions were glassy and brightly eosinophilic, ranging in number from single to more than 20. The inclusions would sometimes compress and distort the nucleus. Inclusions were most notable in the paradrenal (splanchnic) ganglion.
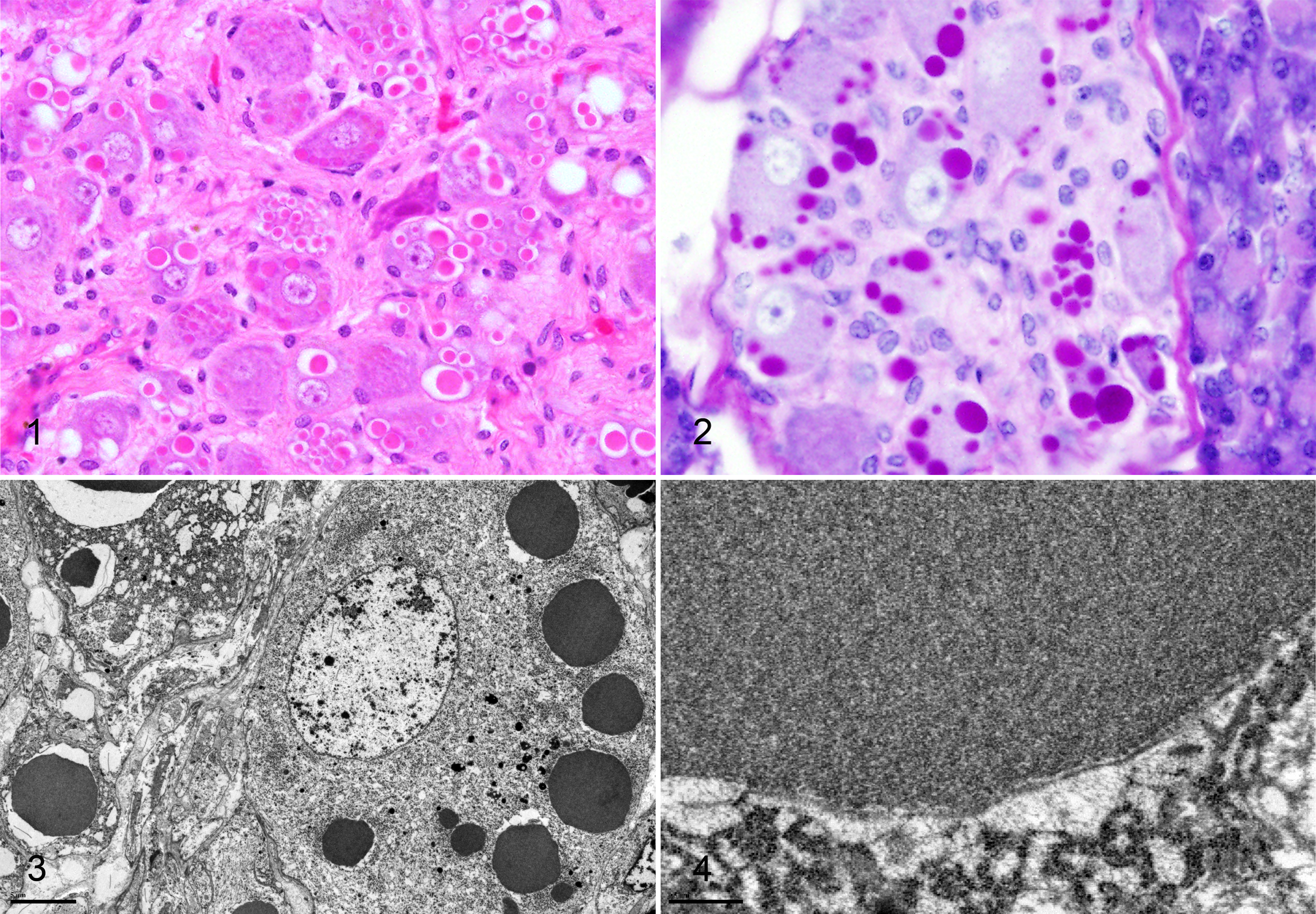
Following identification of the index case, 18 archived white-nosed coati cases from 2 different pathology services, representing 11 different zoological institutions, were reviewed, and 1 additional animal was necropsied prospectively. Cases submitted to one service (MMG) were necropsied onsite by zoo staff at different zoological institutions, and tissue sampling was variable. Inclusions were present in all 20 white-nosed coatis examined, including both archived and prospective cases. Both sexes were affected, and ages ranged from 7 to 19 years (Supplemental Table S1). Inclusions were noted in a number of peripheral autonomic ganglia in or near the adrenal, salivary glands, pancreas, and gallbladder, as well as the myenteric (Auerbach’s) and submucosal (Meissner’s) plexus of the gastrointestinal tract. In 1 case (WN2), inclusions were present in a spinal ganglion (presumed dorsal root). Inclusions were not documented in the brain (including the hippocampus, cerebellum, thalamus, and cerebral cortex) or in the adrenal medulla. Spinal cord and dorsal root ganglia were not examined in all cases, and the trigeminal ganglion was examined in only a single case (WN14, positive). Five of 16 white-nosed coatis in which the adrenals were examined had pheochromocytomas (8 to 11 years old), although inclusions were not present in normal or neoplastic adrenal medullary cells.
Similar inclusions were present in archived slides from 4 of 7 captive brown-nosed coatis, ages 2 to 21 years old, from 3 different zoological institutions (Supplemental Table S2). Two young animals, 2 and 2.25 years old, were included. In the youngest animal, inclusions were frequent in the Auerbach’s and Meissner’s plexus of the stomach and small intestine; no extraenteric ganglia were available to evaluate. In the older of the two, inclusions were rare and limited to Meissner’s plexus of the small intestine; periductal ganglia in the pancreas were negative. The 2 oldest brown-nosed coatis (13 and 21 years old) had pheochromocytomas, but no inclusions were present in normal or neoplastic medullary cells.
Inclusions were PAS positive and diastase resistant (Fig. 2) but negative by Ziehl-Neelsen acid fast, Luxol fast blue, and Sevier Munger staining methods. Ultrastructurally, the inclusions in the paradrenal ganglion of the index case were finely granular and osmiophilic (Fig. 3), and a single limiting bilayer membrane was often present (Fig. 4). Although there was tissue-handling artifact related to sample preparation, there was no ultrastructural evidence of neurodegeneration or apoptosis/necrosis in those neurons.
Immunohistochemical staining for rabies virus and canine distemper virus (MSU DCPAH, East Lansing, MI) on the index case (WN8) and case WN2 were negative. Immunohistochemical staining for PMNT was variably positive within the inclusions (Supplemental Fig. S1) and was positive in the surrounding neuronal cytoplasm. Neurons, but not inclusions, were strongly positive for COMT (Supplemental Fig. S2) and weakly positive for ubiquitin (Supplemental Fig. S3) and α-synuclein (Supplemental Fig. S4).
The term hyaline globules was first applied to glassy eosinophilic cytoplasmic inclusions in the adrenal medulla of humans dying of infectious disease. 5 Additional reports identified their presence in the adrenal medulla of animals dying of infectious disease or toxicosis and in humans with a variety of neurodegenerative disorders. 4,7,9 Similar inclusions were noted to be a diagnostic feature of tumors arising from the adrenal medulla 3 ; however, histologically similar but histochemically and immunohistochemically distinct inclusions of a variety of normal and neoplastic tissues have also been identified under the “hyaline globule” rubric. 6 There have been no published reports to date of hyaline globules in the peripheral ganglionic neurons of any species.
The inclusions reported herein share some histologic, histochemical, immunohistochemical, and ultrastructural features with the hyaline globules of the adrenal medulla in laboratory animals. 4 Ultrastructurally, the inclusions in coatis resembled very large dense-core neurosecretory granules and stained positively for PNMT, the enzyme that converts norepinephrine to epinephrine. The inclusions were negative for markers typical of neurodegenerative processes such as ubiquitin and α-synuclein. Features of apoptosis or degeneration (thanatosomes) as suggested by Papadimitriou et al 6 are not present in the coatis. Thanatosomes have recently been reported in the gastrointestinal epithelium (but not ganglionic cells) of humans with various disease conditions. 2 Thanatosomes in the latter study stained positively for carcinoembryonic antigen, cytokeratins AE1/AE3, and the lysosomal associated membrane proteins LAMP1 and LAMP2. Although only 2 coatis younger than 7 years were evaluated, both brown nosed, the limited presence in those individuals compared with the widespread distribution in the older animals suggests that inclusion development may be an age-related process; however, additional cases need to be evaluated. The absence of expression of α-synuclein and ubiquitin suggests that the inclusions are not indicative of neurodegeneration per se. There were no consistent major disease processes in these animals, and causes of death/euthanasia included malignancies, infections, or were undetermined. Although these animals appear to have a relatively high prevalence of pheochromocytoma, inclusions were not present in normal or neoplastic adrenal medullary cells and were noted in animals without pheochromocytoma. There was no clinical evidence for functional neurologic defects in these animals related to the inclusions. Two of these animals were subsequently tested for rabies and distemper virus due to the similar histologic appearance of inclusions in these infectious processes. This similarity underscores the importance of recognizing these inclusions as an age-related background change in these species but should not preclude testing for these viruses in suspect cases. It is unclear if these inclusions are present in wild coatis or other members of the procyonid taxonomic group, and this may be an area of further exploration; however, the presence of the inclusions in animals from 13 geographically diverse zoological institutions suggests that local environmental conditions are not a factor.
Footnotes
Acknowledgements
Dr James W. Griffith performed some of the original necropsies. We thank Weifang Lin and Ellen Mullady (PSHMC) and Roy Brown (Histology Consulting Service) for histologic specimen preparation. We are grateful for assistance from Roland Myers with electron microscopy specimen preparation and photography. Qing Zhong performed immunohistochemistry for COMT and PNMT. The authors also thank Cathy Minogue (Northwest ZooPath) for data and specimen retrieval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
