Abstract
An 11-mo-old, intact male captive kinkajou (Potos flavus) was submitted for postmortem investigation because of emaciation and hindlimb overgrooming. Histologically, alveolar airspaces were filled with fungal structures that were morphologically and histochemically consistent with Pneumocystis spp. PCR of pulmonary tissue was negative for canine distemper virus and positive for Pneumocystis spp. Molecular testing yielded amplification of the Pneumocystis spp. mitochondrial large-subunit rRNA (mtLSU rRNA, 510 bp) and the small-subunit rRNA (mtSSU rRNA, 565 bp). Phylogenetic analysis suggested a potentially novel Pneumocystis lineage associated with P. flavus. Additional nuclear loci are required to confirm its taxonomic status. Gastric and colonic histologic findings included concurrent candidiasis and colonic nematodosis. An underlying immunosuppressive disease was suspected. Further investigation is required to clarify the role of kinkajous in the ecology of fungal pathogens and the causes of immunosuppression in this species, particularly in the context of human–wildlife interactions. Enhanced surveillance and interdisciplinary collaboration are essential to evaluate potential zoonotic risks and inform conservation and public health strategies.
Kinkajous (Potos flavus)—medium-sized, predominantly frugivorous, quadrupedal mammals in the family Procyonidae—are related to raccoons, coatis, red pandas, ringtails, and olingos.5,16 Singular anatomic features encompass a prehensile tail and a long, narrow tongue adapted to the consumption of honey. Native habitats include the canopies of neotropical primary rainforests throughout Central America and northern South America.5,16 Kinkajous are often found in zoologic collections and as privately owned pets. 18
The scientific literature describing pathologic conditions in kinkajous is scarce and includes: natural and vaccine-induced morbilliviral infection, hypertrophic cardiomyopathy with heart failure, nasal adenocarcinoma, mandibular cystic fibrous osteodystrophy of nutritional origin, and infection by the zoonotic roundworm Baylisascaris procyonis.4,6,9,10,12 Zoonotic cutaneous blastomycosis and hand cellulitis with abscess formation associated with kinkajou bites have been reported in humans.7,8 Additionally, a novel rabies virus variant was reported in a kinkajou from Brazil in 2020, raising concerns about the capacity of kinkajous to serve as a reservoir or spillover host for lyssaviruses. 3 Overall, the documentation of kinkajou lesions remains highly fragmented, often limited to isolated case reports and anecdotal observations. We provide histologic and molecular descriptions of a proposed novel Pneumocystis lineage in a kinkajou with Candida spp. coinfection.
An 11-mo-old, 475-g, intact male kinkajou was submitted to the Veterinary Pathology Centre at the University of Surrey (
Premortem zinc sulphate fecal flotation (Idexx) was negative for parasite oocysts and cysts. Antemortem bloodwork included manual interpretation of a blood smear as well as a biochemistry panel (Idexx;
Grossly, the animal was in moderately poor body condition based on scant subcutaneous and visceral adipose stores as well as decreased axial and hypaxial muscle mass. The lung lobes had poorly delimited, tan-to-pale-yellow, slightly firm, raised areas, prominently distributed within the dorsal areas of the caudal lung lobes. The stomach was markedly distended by gas and had numerous, well-demarcated, dark-red, and slightly depressed ulcers covered by strings of fibrin. Samples of brain, heart, trachea, lungs, liver, kidneys, urinary bladder, testes, spleen, stomach, small intestine, large intestine, pancreas, thyroid and parathyroid glands, skeletal muscle, and sciatic nerve were placed in 10% neutral-buffered formalin. Samples of kidneys, liver, brain, lungs (including charcoal swabs of the affected areas for potential molecular or microbiologic analysis), spleen, blood, and small intestine were stored at −20°C. Selected formalin-fixed tissues (lung, kidney, liver, stomach, small and large intestine, heart, brain [cerebral cortex, pons, cerebellum, thalamus, hippocampus]) were processed routinely, sectioned, and stained with H&E for histologic evaluation.
Histologically, the alveolar septa were multifocally expanded by fibrosis and moderate numbers of histiocytes, occasional multinucleate giant cells, lymphocytes, and plasma cells. Lymphoplasmacytic perivascular infiltration was also often observed. The alveolar airspaces were often filled with proteinaceous fluid and fibrin, moderate numbers of foamy macrophages, scant neutrophils, and myriad, round-to-oval, 5–10-µm, extracellular and intracellular fungal organisms with a thin eosinophilic outline and a central slightly basophilic core (
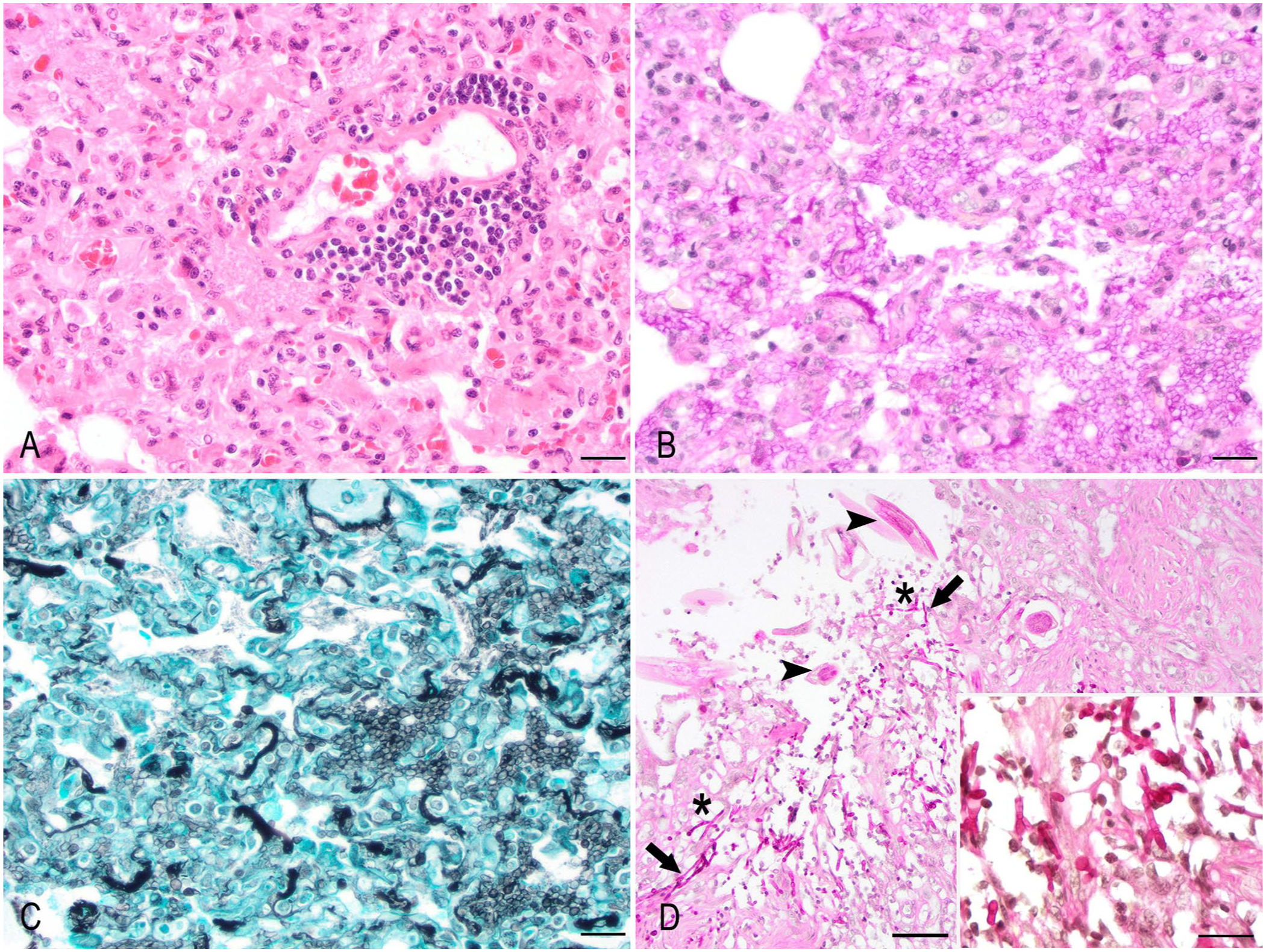
Pneumocystis pneumonia and colonic candidiasis in a kinkajou (Potos flavus).
PCR testing for canine distemper virus (CDV) was performed on fresh-frozen lung tissue at Laboklin (Bad Kissingen, Germany), an ISO/IEC 17025–accredited veterinary diagnostic laboratory, using a validated, proprietary assay (Service ID 8003; qualitative real-time PCR), with appropriate positive and negative controls, 14 and was negative. PCR testing for Pneumocystis spp. was performed on formalin-fixed, paraffin-embedded (FFPE) tissues. Nucleic acid extraction was performed on 2 separate 20-mg sections of FFPE lung tissue. Paraffin was manually removed with a sterile scalpel, and the tissue was transferred to 1.5-mL microcentrifuge tubes for xylene-based deparaffinization. After drying, samples were digested with proteinase K, and DNA was extracted (NucleoSpin tissue kit; Macherey-Nagel) following the manufacturer’s instructions. DNA was eluted in 100 µL of ultrapure water, and concentration and purity were assessed (NanoDrop spectrophotometer; ThermoFisher).
Mitochondrial large-subunit rRNA (
Lung tissue samples tested positive for Pneumocystis mtLSU and mtSSU rRNA genes by PCR, yielding identical sequences for each gene based on direct Sanger sequencing, with clean, non-overlapping peaks in chromatograms with no evidence of mixed infection (100% identity;
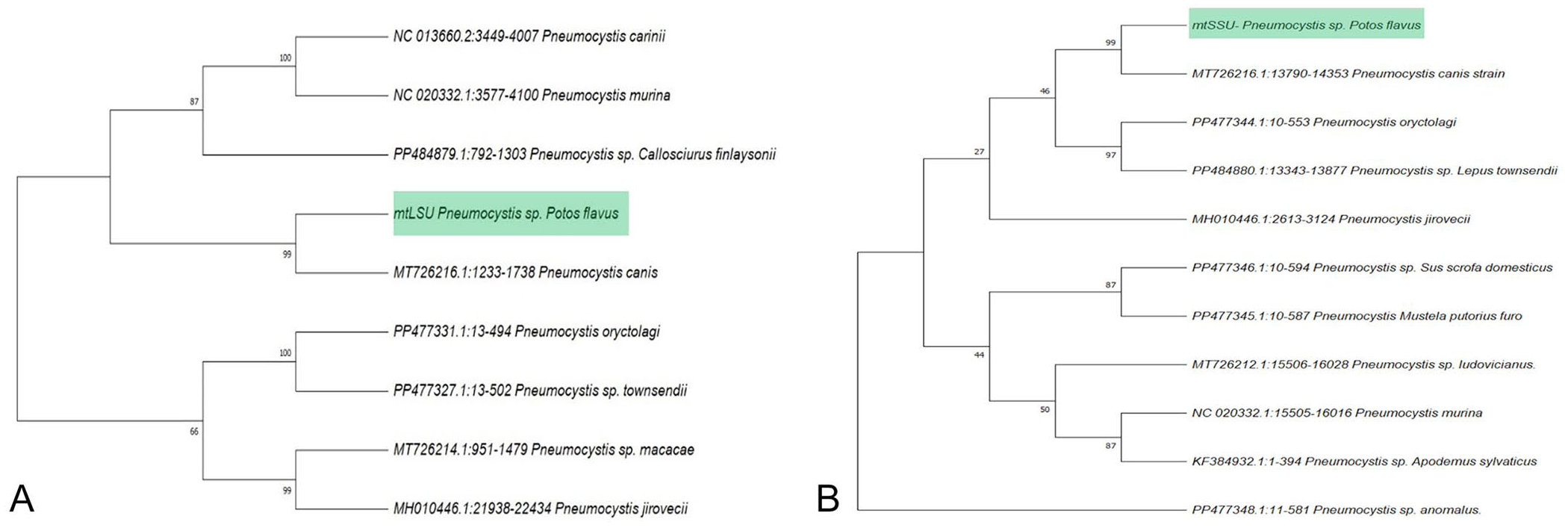
Maximum-likelihood phylogenetic trees of Pneumocystis in a kinkajou.
FFPE lung tissue and histologic slides from our case are archived at the VPC-US. Given that only mitochondrial loci were obtained, we conservatively refer to this organism as Pneumocystis sp. (Potos flavus lineage). Further support for its designation as a distinct species would require sequencing and phylogenetic analysis of nuclear loci, which were not available for this sample and represent a major limitation for definitive species identification. 11
Pneumocystis is a genus of highly host-specific, ubiquitous fungi that infect mammals opportunistically. The 7 formally named species include: P. jirovecii (human), P. murina (mice), P. carinii (swine and rats), P. oryctolagi (rabbits), P. canis (dogs), P. wakefieldiae (rats), and P. ‘macacae’ (macaques). 2 Presumably, the life cycle alternates between 2 major stages including the cyst or ascus (5–8 µm), which are thick-walled cysts containing intracystic bodies, and the metabolically active trophic forms (2–8 µm), which attach to type I pneumocytes. 2 Although pathologic effects of Pneumocystis spp. are most prevalent in young and immunosuppressed individuals, subclinical infection in presumed immunocompetent individuals has been reported. 17 Animals, including humans, may acquire Pneumocystis spp. through several routes: horizontal airborne transmission from infected or subclinical hosts, vertical transmission through the placenta, which has been postulated in rabbits and humans, or perinatally through contact with colonized or infected parents during the neonatal period. 1
Hematologically, our case had left-shifted neutrophils with toxic changes, suggestive of rapid neutropoiesis in response to the pneumonia and mycotic gastrocolitis. Hyperglycemia was likely part of an acute stress response, which has been documented in both companion and wildlife animals. 15
Lymphoplasmacytic-to-granulomatous interstitial pneumonia with fibrosis is typically described in pneumocystosis in animals. 17 The granulomatous and lymphoplasmacytic interstitial pneumonia in our case was prominent within caudodorsal areas of the lungs, with abundant intra-alveolar pneumocystis organisms, aligning with published reports. 17
The multifocal perivascular accumulation of lymphocytes and plasma cells within the lung tissue in our case raised additional concern for an underlying primary infectious condition that could have predisposed this animal to opportunistic infections. Reported infectious comorbidities in animals infected with Pneumocystis spp. include CDV, canine parvovirus, feline panleukopenia virus, feline leukemia virus, porcine circovirus 2, and porcine reproductive and respiratory virus, among others. 17 Vaccine-induced and naturally occurring CDV have been reported in kinkajous. 9 Interestingly, our case was housed in a collection with ferrets that died following a feline panleukopenia outbreak. Testing for potential parvoviral infection was not performed in our case as neither gross nor histologic findings (i.e., enteritis and/or Peyer patch necrosis) were consistent with such infection.
Additionally, ulcerative gastritis and colitis with intralesional fungal elements morphologically compatible with Candida spp. were observed in our case, with concomitant nematodosis in the colonic lesions. Coinfection of Pneumocystis spp. and Candida albicans has been described in a wide range of animal species with compromised immune status. 16 Kinkajous have been documented to harbor gastrointestinal nematodes (e.g., Ancylostoma spp., Strongylus spp.), resulting in damage to the intestinal mucosa and chronic enteritis, potentially creating idoneous conditions for opportunistic organisms.12,20 In addition, recent acquisition and dietary change were reported, potentially resulting in increased stress. Gastric ulcers related to husbandry stress and abrupt dietary change have been described in other captive exotic species. 19 We suspect opportunistic colonization of the damaged gastric mucosa by Candida spp. in our case.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387251413289 – Supplemental material for Pulmonary pneumocystosis in a captive kinkajou with molecular evidence of a novel Pneumocystis lineage
Supplemental material, sj-pdf-1-vdi-10.1177_10406387251413289 for Pulmonary pneumocystosis in a captive kinkajou with molecular evidence of a novel Pneumocystis lineage by Pablo Díaz-Santana, Alejandro Suárez-Bonnet, Javier Déniz-Marrero, Francisco J. Salguero, Bernat Martí-Garcia, Vincente Friaza, Enrique J. Calderón and Sai Fingerhood in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the histology and postmortem technicians at the Veterinary Pathology Centre, University of Surrey, for their contributions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
