Abstract
Historically, it was thought that lens protein was sequestered, and injury to the lens capsule causing release of lens material into the eye would always result in ocular inflammation. Currently, it is believed that lens antigens are recognized as self, subject to normal T-cell tolerance. Three different single-dose intravitreal injection/implantation studies of 4 different test materials, ranging from 4 to 6 weeks in length, were performed in New Zealand White rabbits. The test materials included polymer microspheres, polymer rods, a solvent, and a hydrogel. Intravitreal injection/implantation procedures were performed on day 1, and indirect ophthalmoscopy and slit-lamp biomicroscopy examinations were performed by board-certified veterinary ophthalmologists periodically throughout the course of each study. None of the affected animals received corticosteroids or other immunomodulatory agents during the course of the studies. Four rabbits had perforation of the posterior lens capsule during the injection/implantation procedure on day 1, visible on clinical ophthalmic examination as lens capsule alterations described as “lens hits” and/or incipient posterior cataracts. Findings on slit-lamp biomicroscopy examination were limited to vitreous cells in 2 of the animals, although not centered on the area of lens capsule disturbance. Histologically, there was no evidence of inflammation in association with extruded lens protein material in any of the affected eyes. These results indicate that iatrogenic damage to the lens capsule during aseptically performed intravitreal injections/implantations does not appear to induce inflammation in rabbits.
Keywords
Historically, it was believed that lens proteins were sequestered and thus not recognized as self. Any compromise of the lens capsule was presumed to result in significant immunoinflammatory response due to escape of intact lens proteins into the eye. However, much evidence has been presented over the years to show that lens antigens are actually recognized as self, subject to normal T-cell tolerance. 1,5,15,22,28 Lens-induced inflammation is believed to represent a breakdown of normal tolerance to lens proteins. 1,26,28 Evidence that lens proteins are not sequestered includes the detection of lens crystallins in the aqueous humor, other ocular locations of normal eyes, 21,36 and other organs of the body. 7 In addition, anti–lens protein antibodies have been detected in the serum of healthy humans, 18,37 and lymphocytes that can bind lens crystallins have been detected in the spleen of rats. 30 Despite the fact that disruption of the lens capsule is fairly common, inflammation in association with it is rare, 12,22,45 consistent with the recognition of lens proteins as self.
In animals such as dogs, cats, horses, and others, inflammation occurs following trauma to the cornea and anterior lens capsule. 2,9 –11,17,32,42 It has been recognized that bacteria or other organisms are introduced into the lens as part of the trauma, and it is believed that the microorganisms are important in triggering the inflammation rather than, or in addition to, the release of lens proteins. This scenario is now commonly referred to as septic implantation syndrome. 2 Although it had previously been recommended by veterinary ophthalmologists that lens material be removed from the eye as quickly as possible following disruption of the lens capsule because it was presumed to be the primary inciting cause of the inflammation, 9,41,42 it has more recently been recognized that medical therapy is often effective and that lens material removal is usually not necessary and may in fact be detrimental to the eye. 33
Lens capsule rupture with associated inflammation in rabbits has been determined to be related to infection with Encephalitozoon cuniculi. 11,14,16,44 It is uncertain whether the resulting inflammation is purely in response to the microorganisms or if some combination of organisms and escaped lens proteins is required. The lens capsule rupture is usually anterior, and dwarf rabbits are especially susceptible. E. cuniculi organisms have been detected by gram stain, immunohistochemistry, polymerase chain reaction (PCR), and electron microscopy. The mechanism by which E. cuniculi infects the lens of rabbits is unknown but occurs in the absence of trauma and may be present in pups, raising the possibility of vertical transmission during development.
The objectives of the 3 experimental studies discussed here were to evaluate the efficacy and safety of prospective new materials for the long-acting delivery of medications to the interior of the eye. Of 60 total animals in the 3 studies, 4 eyes had iatrogenic rupture of the posterior lens capsule without associated inflammation.
Materials and Methods
Three different single-dose intravitreal injection studies of 4 different test materials were performed in New Zealand White rabbits. The test materials included polymer microspheres, a polymer rod, a solvent, and a hydrogel. The studies were either 4 or 6 weeks in length. A total of 60 animals were included in the 3 studies. Although exact ages are not known for animals used in experimental studies, the animals in the studies, including rabbit Nos. 1 and 2, had initial body weights ranging from 2.2 to 2.8 kg, with male rabbits in this weight range usually being approximately 12 to 16 weeks of age. The animals in the study including rabbit Nos. 3 and 4 had initial body weights ranging from 3 to 3.4 kg, with female rabbits in this weight range usually being approximately 16 to 20 weeks of age. Intravitreal injection/implantation (in the case of the rods) procedures were performed the same way in each study. Prior to the dosing procedures, rabbits were anesthetized with ketamine and xylazine. A mydriatic (1% tropicamide) was administered to each eye. The eyes were cleaned with a 5% Betadine solution and rinsed with sterile saline. A topical anesthetic (proparacaine or equivalent) was then applied to each eye. A wire speculum was used to retract the eyelids. The eyes were cleaned with a dilute, approximately 1% povidone iodine solution, and rinsed with sterile saline prior to the first dose. The periorbital region was cleaned with a dilute, approximately 1% povidone iodine solution. Doses were administered by intravitreous injection in the inferior temporal quadrant of each eye (in approximately the 7- or 8-o’clock position in the right eye and approximately the 4- or 5-o’clock position in the left eye). This resulted in the deposition of the test material in the inferior, anterior, and temporal aspect of the posterior compartment, just posterior and inferior to the lens.
For the polymer rod implantation, anesthesia and eye preparation procedures were the same as described for the intravitreal injection procedures. The rod was implanted surgically, in the superior temporal quadrant. The conjunctiva was incised and a scleral incision made using a surgical blade. The rod implant was gently grasped with a smooth-tipped forceps and introduced into the vitreous cavity through the scleral incision. The sclera was sutured closed with 1 or more sutures.
Ophthalmic examinations were performed by board-certified veterinary ophthalmologists periodically throughout the course of each study (as noted below for each animal) and included indirect ophthalmoscopy and slit-lamp examination. None of the affected animals received corticosteroids or other immunomodulatory agents during the course of the studies. All procedures and protocols for all studies were approved by the appropriate IACUCs (Institutional Animal Care and Use Committees: Genentech and Charles River Laboratories, Reno, NV) and were carried out in accordance with all applicable institutional, local, and national guidelines. Standard housing and husbandry practices were used for this species.
Following fixation of the eyes in Davidson’s solution at necropsy, eyes were trimmed and sectioned the same way for each study: eyes were sectioned in a vertical plane, with 3 cuts made to show the point of entry of the optic nerve (central), the nasal region of the eye, and the temporal region of the eye (ideally containing the injection or surgical site). Three-step sections were taken of each of these cuts, at 200-micron intervals, for a total of 9 sections examined per eye. The sections were 3 to 5 microns thick and were stained with hematoxylin and eosin (HE).
Rabbit No. 1 received a single dose of solvent in both eyes on day 1. Ophthalmic examinations were performed predose and on days 1 (postdose), 8, 15, 21, and 29. Necropsy was performed on day 29.
Rabbit No. 2 received a single dose of hydrogel in both eyes on day 1. Ophthalmic examinations were performed predose and on days 1 (postdose), 8, 15, 22, 30, and 43. Necropsy was performed on day 43.
Rabbit No. 3 received a single dose of polymer microspheres in the right eye on day 1. Ophthalmic examinations were performed predose and on days 1 (post dose), 5, 10, 13, 18, 24, 29, and 41. Necropsy was performed on day 42.
Rabbit No. 4 was implanted with a polymer rod in the right eye on day 1. Ophthalmic examinations were performed predose and on days 1 (post dose), 5, 10, 13, 18, 24, 29, and 41. Necropsy was performed on day 42.
Results
Rabbit No. 1
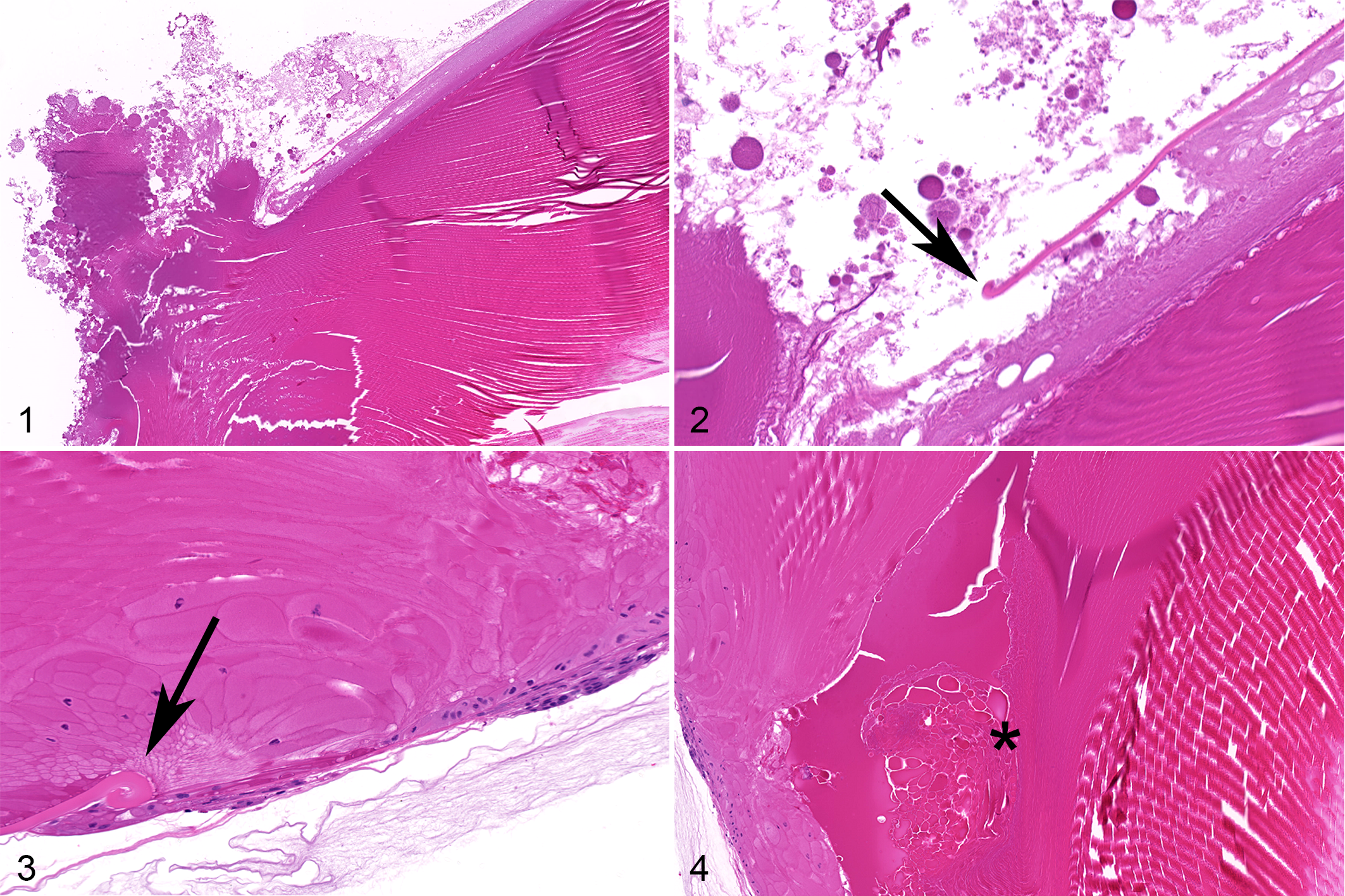
Lens alterations (denoted as “lens hits”) and subsequently incipient posterior cataracts were identified by ophthalmic examination in both eyes starting on day 8 and continuing throughout the remainder of the study. (Ophthalmologists performing in-life ophthalmic examinations in preclinical ocular toxicology studies use the term lens hits to describe alterations of the posterior surface of the lens/capsule that they have learned are associated with iatrogenic in-life trauma to the lens capsule during the intravitreal injection or implantation process [ie, “hitting” the lens with the needle or implanted structure.]) On slit-lamp examination in-life, the left eye had vitreous cells, ranging from 4+ at day 15, decreasing to 1+ by day 29; these cells were diffusely distributed and not centered on the area of lens capsule disturbance. Histologically, this animal had rupture of the posterior lens capsule in the left eye, with extrusion of lens material into the posterior compartment. This extrusion of lens material as well as curling of the edge of the torn lens capsule confirmed that it was compromised in-life, prior to necropsy and fixation of the eye. No inflammation was apparent despite the presence of lens material in the posterior compartment (Figs. 1, 2). There were no cellular infiltrates correlating with the 1+ vitreous cells seen on ophthalmic exam on day 29. In the right eye, there was mild degeneration of the posterior lens fibers, without any apparent compromise of the lens capsule.
Rabbit No. 2
Incipient posterior cataract was identified by ophthalmic examination in the left eye starting on day 15 and continuing throughout the remainder of the study. On slit-lamp examination in-life, the left eye had 2+ vitreous cells at day 43; these cells were diffusely distributed and not centered on the area of lens capsule disturbance. Histologically, there was evidence of perforation of the lens capsule, as well as degeneration of lens fibers. Curling of the edge of the torn lens capsule confirmed that it was compromised in-life, prior to necropsy and fixation of the eye. No inflammation was apparent despite the compromise of the lens capsule (Figs. 3, 4). There were no cellular infiltrates correlating with the 2+ vitreous cells seen on ophthalmic exam on day 29.
Rabbit No. 3
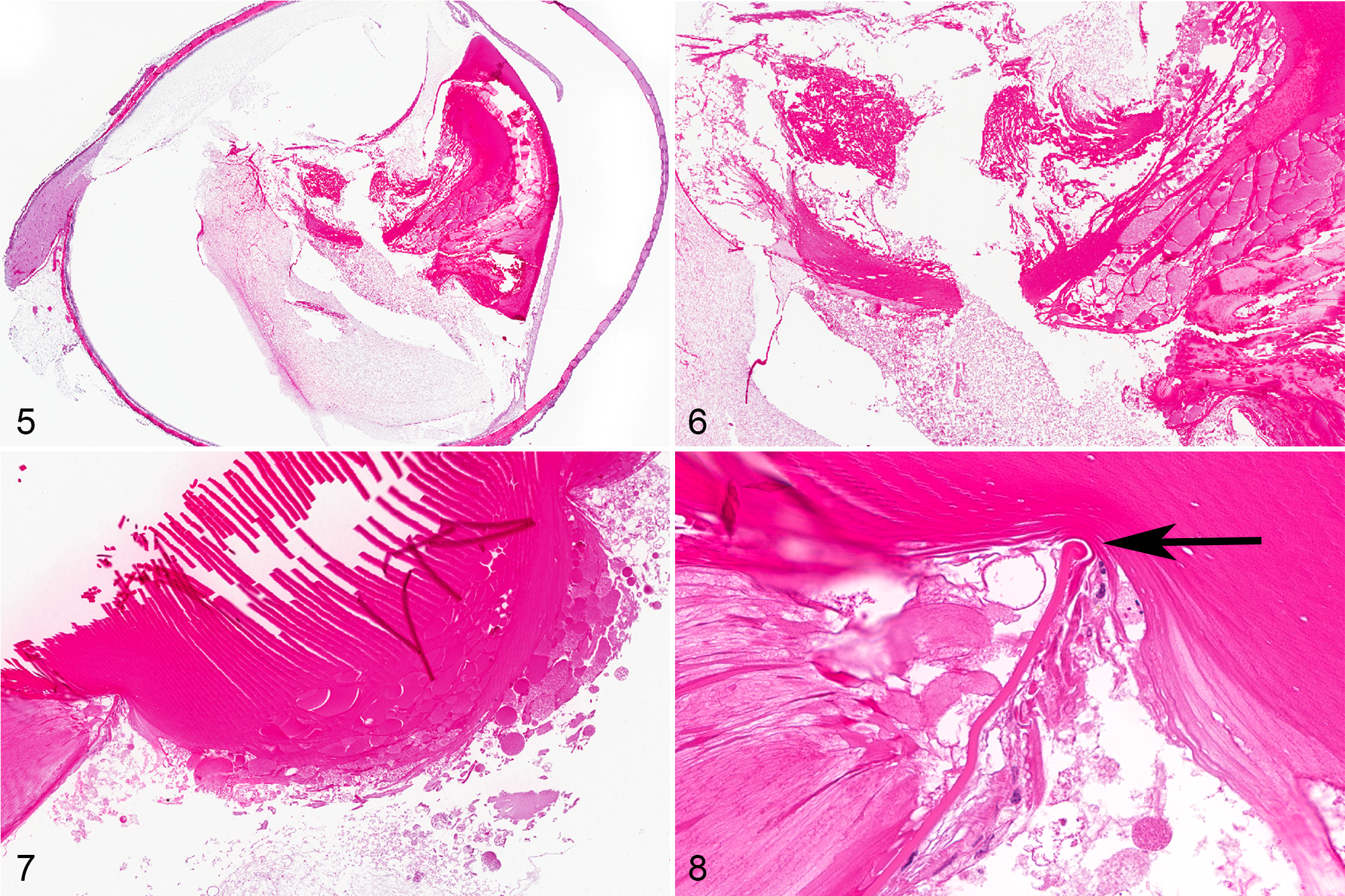
Posterior cataract was noted by ophthalmic examination in the right eye beginning on day 18 and continuing throughout the remainder of the study. No other abnormalities were noted on ophthalmic examination. Histologically, this animal had rupture of the posterior lens capsule in the right eye, with extrusion of lens material into the posterior compartment as well as degeneration of lens material still contained within the lens proper. No inflammation was apparent in association with the presence of lens material in the posterior compartment (Figs. 5, 6). There were 1+ mononuclear cells in the periphery of the vitreous, lining the retina. These cells were typical of what is seen in control and dosed animals on intravitreal injection studies and were not associated with the extruded lens material.
Rabbit No. 4
Posterior cataract was noted by ophthalmic examination in the right eye beginning on day 18 and continuing throughout the remainder of the study. The only other abnormality noted on ophthalmic examination was mild swelling of the conjunctiva of the right eye on day 5. Histologically, this animal had rupture of the posterior lens capsule in the right eye, with extrusion of lens material into the posterior compartment. This extrusion of lens material as well as curling of the edge of the torn lens capsule confirmed that it was compromised in-life, prior to necropsy and fixation of the eye. No inflammation was apparent in association with the presence of lens material in the posterior compartment (Figs. 7, 8). There were 1+ mononuclear cells in the periphery of the vitreous, lining the retina. These cells were typical of what is seen in control and dosed animals on intravitreal injection studies and were not associated with the extruded lens material.
Discussion
Although many veterinarians have been taught that any compromise to the lens capsule results in substantial inflammation, this idea may have been influenced by the common association of external trauma and introduction of microorganisms into the eye in cases of lens capsule disruption in domestic animal species, the so-called septic implantation syndrome. 2 The true incidence of inflammation associated with lens capsule disruption in domestic animals under sterile conditions is less clear.
In general, it has been determined that lens proteins are immunologically tolerated, 31,34,35 and even though disruption of the lens capsule is fairly common, inflammation associated with it is rare. 12,30,45 Tolerance to lens protein is thought to be centrally mediated in the thymus, via T lymphocytes. While homologous lens crystallins can induce the formation of antibodies from B cells, certain types of T cells have been shown to be nonreactive to the same crystallins. 5,15,20,22,26 In addition, when a depot of antigen was removed from the eye, it did little to change the tolerance to the antigen. 20 With any of these scenarios, 2 or more antigen exposures may be needed to stimulate a response, suggesting a need for sensitization. 16
Regardless of the normal state of tolerance to lens material, factors do occur that result in inflammation following rupture or other compromise of the lens capsule. The terminology to describe such inflammation in humans has been somewhat controversial over the years, but the term phacoantigenic endophthalmitis (PE) (previously called phacoanaphylactic endophthalmitis) is most often used to describe the acute granulomatous uveitis that occurs after surgical or traumatic rupture of the lens capsule, 12,13,40,45 and phacolytic (referred by some authors as phacotoxic) uveitis is used to describe the more chronic inflammation related to the release of lens proteins from a cataractous lens. In the veterinary literature, the term phacoclastic uveitis has been used as the counterpart to human PE, since the inflammation seen in animal cases does not represent exactly that seen in humans. 41
There are examples of spontaneous rupture of the posterior lens capsule in animals, including rats 38 and mice. 19,20,39 In the rats, there was no evidence of trauma or other compromise of the wall of the eye, yet mild inflammation, including macrophages and multinucleated giant cells, was present. In contrast, in the mice, the lens capsule rupture was determined to be secondary to a developmental abnormality of lens fiber arrangement, and there was no associated inflammation. Thus, despite the lack of any evidence of compromise of the blood-ocular barrier, there was inflammation associated with the lens capsule rupture in one species but not the other, for undetermined reasons. In addition to the rats and mice described above, posterior lens capsule rupture has been seen in association with persistent hyperplastic primary vitreous/persistent tunica vasculosa lentis in humans 4 and TgN3261Rpw transgenic mice, 8 with inflammation (compatible with PE) described in the former but not in the latter.
Spontaneous lens capsule rupture has also been described in dogs with diabetes mellitus–associated cataracts, most often equatorial and rarely posterior. 43 Inflammation is present but primarily represents a lymphoplasmacytic iridocyclitis and retinitis, consistent with phacolytic uveitis (a response usually seen when degenerated lens protein leaks from a cataractous lens). We have reported here 4 instances of iatrogenic rupture of the posterior lens capsule in New Zealand White rabbits from single-dose intravitreal injection/implantation studies. The eyes of these rabbits were devoid of any inflammatory response to the escaped lens material, either in the vitreous adjacent to the lens material or in the uveal tract or elsewhere in the eye. This was despite the fact that foreign materials had been introduced into the eyes as part of the study procedures, including polymer microspheres, polymer rods, a solvent, and a hydrogel. None of the affected animals received corticosteroids or other immunomodulatory agents during the course of the studies. Aseptic procedure was always followed in our intravitreal injection/implantation studies, and therefore microorganisms should not have been introduced. Since inflammation did not occur, despite the compromise of both the wall of the eye and the posterior lens capsule as well as the introduction of foreign material into the eye, it would follow that the presence of some other type of stimulus is needed to stimulate an inflammatory/immune response. This has been suggested by some investigators, although the additional inflammatory stimulus was not specifically identified. 16,22 Other investigators have determined that it was difficult to experimentally induce phacoclastic endophthalmitis in rabbits simply by disrupting the lens capsule; it was necessary to also introduce mycobacterial components to successfully induce inflammation in this species. 3,24,25,31,35 This supports our finding of a lack of inflammation to released lens protein under aseptic conditions. Phacoantigenic endophthalmitis has been induced experimentally in other laboratory species, including rats, guinea pigs, and mice, but presensitization with lens proteins followed by disruption of the lens capsule was usually required. 23,24,27,29 In rats, it was possible to induce PE using serum. 24
Aside from the possibility of an additional antigen being necessary, it may be that inadequate time had passed between lens rupture and necropsy to allow the formation of an immune response in these rabbits. In fact, in humans, delays of as long as 19 years have been documented between the time of lens trauma and the onset of inflammation. 6 However, we have seen substantial inflammatory/immune responses develop as soon as 5 days following the injection of humanized proteins into rabbit eyes, without prior sensitization (unpublished data), so the 29 and 42/43 day lengths of the studies of this report could be reasonably assumed to be long enough for an immune response to have occurred. It is also possible, in these studies, that a second procedure may have been necessary to trigger an inflammatory response.
It has not been determined whether the location of lens capsule rupture, anterior vs posterior, affects the presence or absence of inflammation, particularly since most instances of inflammation in relation to anterior capsular rupture are associated with trauma and introduction of microorganisms to the eye. It is unclear, however, whether release of lens proteins into the anterior chamber may be more likely to result in inflammation since antigen may theoretically escape into the bloodstream via the iridocorneal drainage apparatus (and thus potentially contact the systemic immune system) more easily and/or quickly than lens protein released into the posterior compartment as a result of a posterior rupture. In addition, since it appears that some cases of inflammation associated with rupture of the lens capsule in animals and humans may not be associated with the presence of microorganisms, it is uncertain whether, if a large amount of lens protein is released all at once, the proteins themselves might be enough to stimulate an immune response. As described previously, there could be yet some other antigen associated with lens capsule rupture that can trigger an immune response. 15 There is certainly more to be understood regarding the mechanisms behind the presence or absence of inflammation following trauma to the lens.
The 4 cases we have reported here, of iatrogenic rupture of the posterior lens capsule in New Zealand White rabbits without associated inflammation, support the findings of previous investigators that lens capsule rupture and lens protein release within the eye is not in itself enough to cause inflammation in this species. Our findings also support the contemporary belief that lens protein is no longer considered a sequestered antigen, the release of which will lead to an immune attack against it. Rather, as proposed by other investigators, some combination of factors must combine to abrogate normal tolerance of the immune system toward lens proteins in order for inflammation to occur.
Footnotes
Acknowledgements
We thank Charles River Laboratories, Reno, Nevada, for the conduct of one of the intravitreal injection studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
