Abstract
Finding new strategies to counteract periprosthetic infection and implant failure is a main target in orthopedics.
Introduction
The ability of
The internalization within host cells acts protecting bacteria from the host immune response as well as from the medical treatments, as eukaryotic cells are often impermeable to conventional extracellular antibiotics. 6 Furthermore, it was demonstrated that cultured osteoblasts infected with bacteria secrete immune modulators of the inflammatory response, namely, chemokine and cytokine molecules, which trigger and feed bone inflammation and destruction.7,8
In this study, we investigate new approaches that might be used to counteract internalization of osteoblasts by
Methods
Bacterial strains and culture conditions
Bacterial strains used in this work were the following:
Eukaryotic cells
The human osteoblast cell line MG-63 12 was cultured in minimal essential medium with Earle’s balanced salt solution (MEM/EBSS), supplemented with 10% fetal calf serum (FCS), 1% glutamine and 1% penicillin–streptomycin in an atmosphere of 95% air, and 5% CO2 at 37°C. All media were from Euroclone, Italy. Monolayers were used 48 h after seeding.
Chemicals
SPEP (2540 U mg−1), obtained from Takeda Italia Farmaceutici (Italy), was dissolved in phosphate-buffered saline (PBS; pH 7.2) at a stock concentration of 20,000 U mL−1 and stored at −20°C.
Cellular infections in vitro
Staphylococcal strains from 18 h cultures in BHI broth, were grown in the absence of SPEP. Subsequently, bacteria were diluted at 1:100 in BHI and sub-cultured up to OD600 = 1.0 at 37°C with or without 200 U mL−1 SPEP (SPEP-pretreated and SPEP-untreated bacteria, respectively). Before infection, human osteoblast cells were maintained in basal medium containing 10% FCS without antibiotic (penicillin–streptomycin) for 24 h at 37°C and 5% CO2. Subsequently, human cells cultured in 24-well plates (BD Falcon, USA) to obtain semi-confluent monolayers (1.75 × 105 cells/well) were separately infected with 0.05 mL of SPEP-pretreated and SPEP-untreated bacterial suspension at a multiplicity of infection (MOI) of about 30 bacteria per cell (MOI 30:1) for 2 h at 37°C and 5% CO2.
After incubation, osteoblast cell monolayers were washed with PBS, and 0.5 mL of fresh medium containing 50 µg/mL of gentamicin was added to each well and incubated for 1 h at 37°C and 5% CO2 to kill extracellular bacteria. First, the sensitivity of bacteria to gentamicin was verified, as well as the non-toxicity toward MG-63 cells. Cells were then lysed by the addition of 0.025% Triton X-100 and the collected supernatants were plated on tryptic soy agar (TSA; Oxoid, UK) followed by an overnight incubation at 37°C to count viable intracellular bacteria. The internalization efficiency is expressed as the percentage of the inoculated bacteria. Data represent the mean of three independent experiments.
Cell proliferation and cell viability
For cell proliferation experiments, osteoblasts were incubated with staphylococcal strains. The infection was performed as previously described. After incubation, the number of living cells was determined by colorimetric 3-(4,5-dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay (Sigma-Aldrich, Italy). After removing the supernatant, the resulting intracellular purple formazan was solubilized with dimethyl sulfoxide (DMSO) and spectrophotometrically quantified at 560 nm. Uninfected osteoblasts were used as a control. All experiments were performed with or without SPEP. The results were expressed as the mean percentage in comparison with no infected cells. The cell viability was also assessed by Trypan Blue exclusion dye. 13
Measurement of cytokine concentration
The release of pro-inflammatory chemokine MCP-1 was evaluated after the exposure of the osteoblast cells to staphylococcal strains. Following the incubation, the concentration of the chemokine was determined in the culture supernatants by an enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s recommended procedure (Thermo Fisher, Life Technologies Italia Fil, Italy). As positive control, cells stimulated with lipopolysaccharide (LPS) from
Statistical analysis
The significance of the differences in the results of each test and the relative control values was determined with Student’s t-test. Values of
Results
SPEP action on staphylococcal internalization of osteoblasts
Preliminary experiments were addressed to assess the cytotoxicity of SPEP on MG-63 cells using the same SPEP concentration adopted in the internalization assays (200 U mL−1). Cell morphology, viability, and proliferation remained unaffected by SPEP treatments.
At first, the ability of

Effect of SPEP on cell internalization. MG-63 cells were infected with
In the subsequent experiments, the internalization efficiency of SPEP-pretreated bacteria was compared with that of SPEP-untreated bacteria. Conversely, when bacteria were pretreated with SPEP, they showed a different behavior. In particular, as shown in Figure 1, the internalization efficiency was neatly reduced for all SPEP-pretreated
SPEP did not interfere with the internalization process of
Results obtained seemed to stress the hypothesis the effect of SPEP is on bacterial invasive features rather than on eukaryotic cells permeability to the infection.
SPEP action on osteoblast proliferation
At first, an assay was performed to evaluate the effect of SPEP on the proliferation of MG-63 cells in the absence of bacteria in order to exclude a conditioning of osteoblast proliferation due to the protease (data not shown). The proliferation remained unaffected following SPEP treatment. Then, the effect of bacterial strains pretreated or untreated with SPEP on proliferation of MG-63 was assessed. Figure 2 shows MG-63 proliferation in the presence of
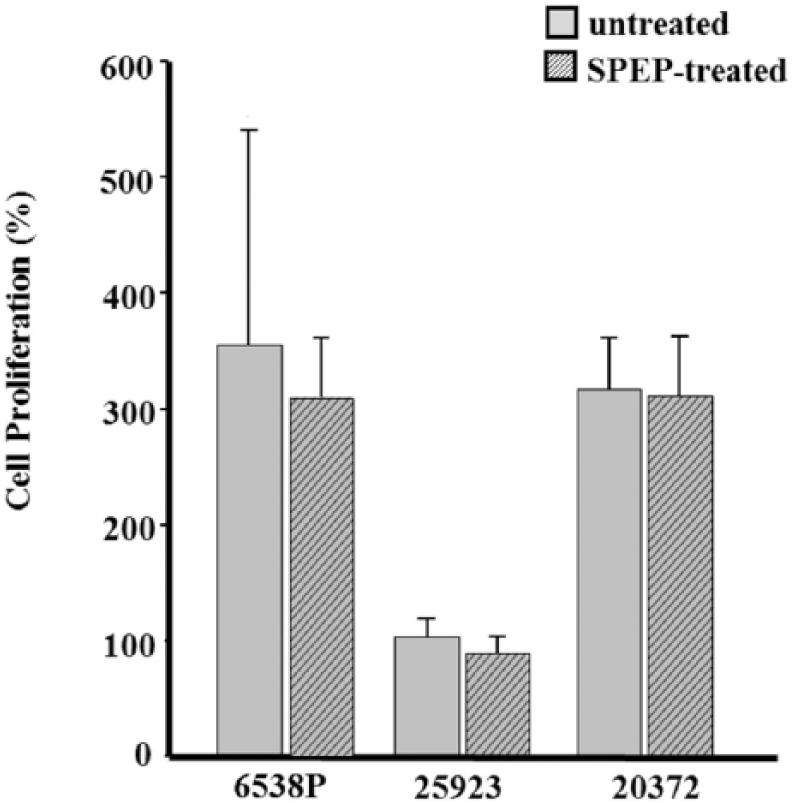
Effect of SPEP on cell proliferation. Osteoblasts were incubated with
When bacteria were pretreated with SPEP, a slight, statistically non-significant, decrease of proliferation was observed for all
Effect of SPEP on chemokine release
In order to evaluate the levels of expression for chemokine MCP-1 after staphylococcal infection, a specific ELISA test was performed. Figure 3 shows MCP-1 levels in supernatants from MG-63 cells infected with

Effect of SPEP on MCP-1 release. MCP-1 secretion in MG-63 cells stimulated with
Afterwards, the content of chemokine MCP-1 in osteoblasts infected with SPEP-pretreated and SPEP-untreated
Discussion
Several studies have also demonstrated that
In this article, the capacity of SPEP to affect the staphylococcal internalization of bone cells and to regulate the proliferation and the production of a pro-inflammatory molecule by infected osteoblasts was investigated. Although the main roles of osteoblasts are the synthesis of bone components and the regulation of osteoclasts activity, it was demonstrated that osteoblasts infected with bacteria secrete inflammatory cytokines and chemokines that contribute to feed inflammation and promote bone destruction. 7 For this reason, we have chosen to monitor MCP-1 release because this chemokine is involved in the recruitment of osteoclasts and monocytes/macrophages (but also of neutrophils and lymphocytes), when eukaryotic cells are exposed to bacteria.
Our data show that when
The use of SPEP in synergy with traditional antibiotics may be suggested as a good strategy to reduce the progression of infection in bone infection and control the progression of inflammatory diseases. Further in vitro and in vivo studies should support this view.
In conclusion, our research demonstrates that SPEP affects the ability of
Footnotes
Acknowledgements
Carla Renata Arciola would like to acknowledge financial support from “5 per mille” contribution for Health Research to the Rizzoli Orthopaedic Institute of Bologna.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
