Abstract
This case report describes an unusual case of late-onset larynx angioedema after ranibizumab intravitreal injection. A 72-year-old female patient presented to our clinic for decreased vision; right eye (RE) fundoscopy and optical coherence tomography (OCT) revealed mild chorioretinal atrophy and choroidal neovascularization with subretinal fluid. A ranibizumab injection was planned in the RE, with standard pretreatment with daily oral administration of betamethasone, cetirizine, and ranitidine because her medical history revealed two adverse drug reactions (ADRs) to contrast media (CM). Despite the premedication, 2 h after injection, the patient referred throat closing sensation and dyspnea that resolved within few hours by betamethasone 4 mg intramuscular injection, without further reoccurrence. In occasion of the second intravitreal injection, video rhinofibrolaryngoscopy revealed subglottic edema that resolved within few hours by betamethasone 4 mg intramuscular injection. This report suggests that, even in cases of intravitreal injection, patients with history of allergy, despite the anti-allergic treatment, should be hospitalized to detect late onset of such a life-threatening complication.
Introduction
Exudative maculopathies are one of the most sight-threatening complications worldwide. Current trends of treatment suggest the use of intravitreal anti-vascular endothelial growth factor (VEGF) as the first-line therapy and, due to the increasing number of patients affected and the need of repeated treatments, in clinical routine they are often treated as outpatients. 1 We report a case of late-onset larynx angioedema in a patient with history of allergy that underwent ranibizumab intravitreal injection, which to the best of our knowledge has not been previously described.
Case report
A 72-year-old woman was referred to our clinic for decreased vision in the right eye (RE). The subject provided a written informed consent for information and images, in accordance with the Declaration of Helsinki. Her medical history revealed two adverse drug reactions (ADRs) with contrast media (CM) injection, which occurred with submandibular and neck swelling, loss of voice, dyspnea, and fainting episode; no other symptoms and signs were present. The second ADR occurred despite patient’s premedication with oral corticosteroids ad histamine blockers, but in both cases, adverse events were solved treating her with intramuscular corticosteroids.
The patient regularly assumed an angiotensin-converting enzyme (ACE) inhibitor/diuretic combination (perindopril 5 mg/indapamide 1.25 mg) plus a beta blocker (bisoprolol 1.25 mg) as hypertension therapy over the past several years, without ADRs occurrence. The ocular anamnesis revealed the presence of severe myopia, retinal detachment in the left eye (LE) treated with surgery, and cataract surgery in both eyes, with no ADRs reported. Her best-corrected visual acuity was 20/70 RE and 20/20 LE; RE fundoscopy and optical coherence tomography (OCT) revealed mild chorioretinal atrophy and choroidal neovascularization (CNV) with subretinal fluid (Figure 1). The diagnosis of myopic CNV was entertained and ranibizumab injection was planned.
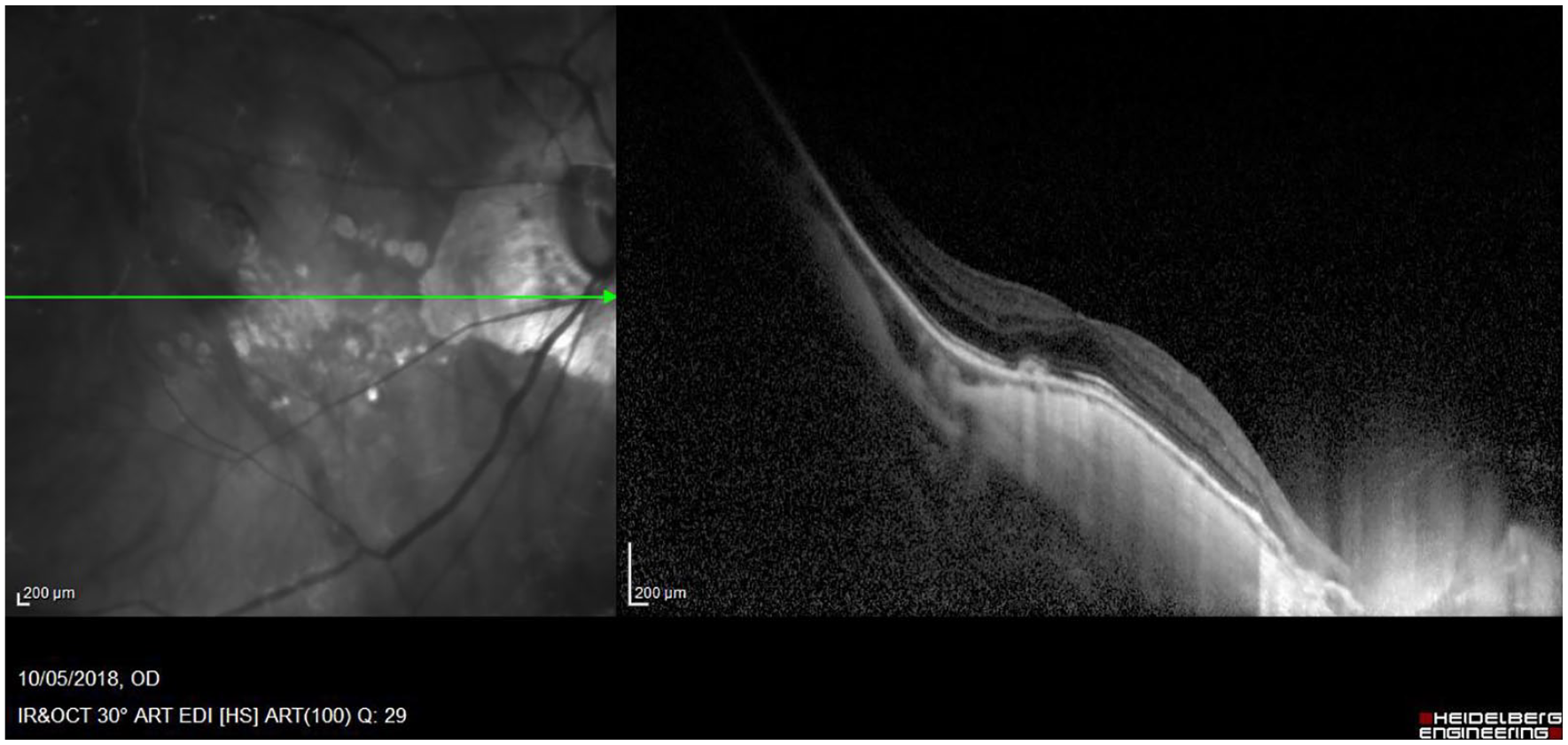
Fundus photography and OCT image of the right eye.
Premedication regimen with oral corticosteroid and antihistamines (betamethasone, cetirizine, ranitidine) was performed. The patient received intravitreal injection of ranibizumab in pre-filled syringe (containing a single dose of 0.05 mL solution); 2 h after injection, the patient referred throat closing sensation and dyspnea, treated and resolved with an intramuscular injection of betamethasone (one dose of 4 mg), without further reoccurrence.
After this event, the patient underwent lidocaine 4% eye drops instillation and skin and fornix disinfection with povidone–iodine 5% solution, without ranibizumab injection, to determine whether the symptoms were related to preoperative preparation or ranibizumab injection; no reaction was detected in the following 3 h and in the days after discharge. The second injection of ranibizumab, preceded by the same premedication regimen, was followed by an extended period of observation in the hospital. The patient experienced the same symptoms; she underwent video rhinofibrolaryngoscopy (RFL), which revealed a subglottic edema. The patient was treated with betamethasone 4 mg intramuscular injection and the subglottic edema resolved in few hours, without further reoccurrence (Figure 2(a)–(d)).

(a) Rhinofibrolaryngoscopy (RFL) before treatment showing a very light edema of the subglottic region. (b) RFL 10 min after the ophthalmological procedure showed an unmodified picture of the subglottic region. (c) RFL 1 h after the injection showing a clearly visible edema under the right true vocal cord, whereas the contralateral was unchanged. (d) 150 min after the injection, RFL showed a marked edema with a significant increase under the right true vocal cord.
Discussion
Only two cases of hypersensitivity reactions (HRs) to intravitreal injection of bevacizumab were described.2,3 In the first case, angioedema and conjunctival chemosis developed 50 min after the injection, which resolved within 48 h with intravenous methylprednisolone and diphenhydramine. 2 In the second case, the patient developed dyspnea 5 min after the first and even after the second injection, despite pretreatment with cetirizine and diphenhydramine. After another injection with ranibizumab, the patient experienced throat tightness and coughing. Therefore, he was shifted to verteporfin and photodynamic therapy. 3
In our case report, angioedema and its timing of onset (at the end of the 2-h time frame considered immunoglobulin E (IgE) mediated by World Allergy Organization (WAO) definition) are still potentially compatible with a type 1 HR, taking into account the implemented premedication regimen. However, the same clinical presentation previously occurred twice, with longer time lag (~6 h), following CM administration, which frequently triggers non-allergic hypersensitivity reaction (NAHR). 4 Indeed, the patient carried several risk factors potentially relevant in the HR to ranibizumab: female sex, previous HR to CM, cardiovascular conditions, use of beta blocker and ACE inhibitors, which can trigger severe facial and laryngeal angioedema. 5
Differentiation between allergic and NAHR is difficult due to superimposable timing and clinical manifestations; specificity and sensitivity of skin prick test for ranibizumab are yet to be established. 6 However, desensitization protocols can successfully deliver monoclonals in patients experiencing HRs. Premedication with corticosteroids and H1-antihistamines is useful mainly for NAHRs 5 although may not prevent IgE-dependent anaphylaxis.7–9 In these patients, alternative anti-VEGF monoclonals could be cau-tiously administered. 2
In conclusion, this report documents endoscopically the localization of laryngeal swelling, and suggests that even in cases of intravitreal injection, patients with history of allergy, despite the anti-allergic treatment, should be hospitalized to detect late onset of such a life-threatening complication.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Cometico Campania Sud, Italy (prot. no. 16544).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the FARB, University of Salerno.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
