Abstract
Pax8, napsin A, and CD10 are useful immunohistochemical markers of human renal cell carcinoma (RCC); however, their diagnostic utility in canine RCC is unclear. Forty formalin-fixed paraffin-embedded renal cell carcinomas from dogs (15 papillary, 12 solid, and 13 tubular) and 10 metastases were evaluated for expression of Pax8, napsin A, and CD10. Thirty-nine (98%), 24 (60%), and 19 (50%) tumors expressed Pax8 (nuclear labeling), napsin A (cytoplasmic labeling), and CD10 (cytoplasmic and membranous labeling), respectively. Pax8 was expressed in 92% of solid, 100% of papillary, and 100% of tubular tumors. Napsin A was expressed in 58% of solid, 60% of papillary, and 62% of tubular RCC. CD10 was expressed in 33% of solid, 47% of papillary, and 62% of tubular RCC. Pax8 was expressed in 80% of the metastatic tumors, napsin A in 60%, and CD10 in 50%. Additionally, Pax8 immunoreactivity was stronger overall than that of napsin A or CD10. In summary, Pax8 is a more sensitive marker than napsin A or CD10 for primary and metastatic canine RCC; its nuclear and more intense reactivity also makes it easier to interpret. Tubular and papillary RCCs were more likely than solid RCC to express all 3 markers. These findings highlight the utility of Pax8 as an immunohistochemical marker in diagnosing all major subtypes of canine primary and metastatic renal cell carcinoma.
Renal cell carcinoma (RCC) is the most common human and canine primary renal tumor, comprising up to 1.5% of all canine neoplasms. 2,20,21 Diagnosis is often straightforward in hematoxylin and eosin (HE) stained sections. However, distinguishing primary RCC from neoplasms metastatic to the kidney or confirming metastatic RCC in other organs can be difficult, especially in core biopsy or other non-excisional specimens. Immunohistochemical (IHC) profiles are part of the diagnostic workup for human RCC but are still under development for dogs. 10,11,23,27,35,38 The immunohistochemical profile of canine RCC has been reported, 14,15 but only 2 reports included more than 1 dog. 10,11
Papillary (pRCC), tubular (tRCC), and solid (sRCC) are the most common histologic types of human and canine RCC. 10,38 –43 Each histologic type, especially solid RCC, may be subclassified by cytologic features; clear cell, eosinophilic, and chromophobe variants exist. 20 Clear cell RCC (ccRCC) is the most common human subtype, comprising approximately 70% of human renal tumors and associated with decreased survival times. 1,8 Human ccRCC is thought to originate from proximal tubular epithelium, and most cases are associated with a somatic mutation of VHL. 3,19,33,38,41 Less is known about the pathogenesis of canine ccRCC; 7,10,29 however, ccRCC in both species carries a poorer prognosis than other RCC subtypes, is more likely to metastasize, and is difficult to distinguish from other neoplasms with clear cells (eg, adrenocortical, male and female urogenital, and gastrointestinal carcinomas) based on histology alone. 4,10,20,36,38,39
Different RCC subtypes are hypothesized to arise from different segments of the nephron (eg, proximal or distal convoluted tubules) or collecting ducts and different anatomic regions of the kidney (eg, cortex, medulla, or renal pelvis). 19,38 Whereas ccRCC and pRCC are hypothesized to originate from the proximal tubules in humans, the chromophobe (crRCC) subtype of RCC and oncocytomas originate from intercalated cells of the distal nephron. 8,15,19 The origin of histologically similar canine RCCs has not been thoroughly investigated. 14,15 Additionally, certain RCC-associated markers, such as CD10, napsin A, and RCC antigen (RCCa), label proximal convoluted tubular epithelium and are therefore more specific for human clear cell and papillary RCCs, whereas other markers, such as kidney-specific cadherin (Ksp-Cad) and CD117 (c-Kit), may be useful in diagnosing RCCs of the distal nephron, such as chromophobe RCC or oncocytoma. 1,4,8,18,38,41 These differences in immunoexpression provide a useful means of differentiating RCC from other tumors that share a similar histologic pattern, clear cell morphology, or are otherwise indistinguishable. 8,35,36,38
In this study, 3 IHC markers of human RCC—CD10, Pax8, and napsin A—were evaluated in canine RCC. CD10 (acute lymphocytic leukemia antigen), a cell-surface zinc metallopeptidase, is expressed by normal and neoplastic lymphoid tissue, 1,8,19 renal glomerular epithelium, the brush border of proximal tubular epithelium, and human clear cell and papillary RCC. 1,16,27 However, CD10 immunoreactivity also has been reported in numerous human nonrenal carcinomas with clear cell morphology. 8,19 CD10 expression has been evaluated in a small subset of canine RCC (n = 13), in which it was demonstrated that a fraction of tumors (n = 3) were positive. 11
Pax8 (paired box gene 8) is a transcription factor involved in embryogenesis and maintenance of renal tubular epithelium, upper urinary and Müllerian tracts, nervous system, and thyroid gland; it is also expressed in carcinomas from these tissues in humans. 6,17,18,38 Pax8 has strong nuclear expression in most neoplastic cells in all histologic types of human primary or metastatic RCC. 9,17,26,42,45 Pax8 molecular and immunohistochemical expression also has been observed in normal canine thyroid follicular cells and thyroid carcinomas as well as in some RCC. 28,30
Napsin A, an aspartic proteinase involved in surfactant protein B maturation, is expressed in the cytoplasm of pulmonary epithelial cells and their neoplasms as well as in human renal proximal tubular epithelial cells, ccRCC, and pRCC. 3,22 –25,44 Napsin A immunoreactivity has also been reported in human carcinomas of the breast, pancreas, colon, and thyroid, as well as mesotheliomas. 4,12,24 Napsin A immunoexpression has been reported in canine parafollicular C cells and thyroid tumors, particularly medullary carcinomas; 2 of 5 RCCs were also positive for napsin A. 30
The aim of this study was to evaluate the immunoexpression of CD10, Pax8, and napsin A in the different subtypes of primary and metastatic canine RCC.
Materials and Methods
Sources of Cases
Forty formalin-fixed paraffin-embedded (FFPE) canine primary RCC and 10 metastases were selected from Purdue University and Colorado State University archives (1996–2014). Records for each animal, including age, breed, and sex, as well as clinical information, when available, including metastases or other tumors at the time of diagnosis, are provided in Supplemental Table S1. Nineteen of the 40 RCC specimens were from cases previously investigated by Edmondson et al 10 in a related study. Thirty-six specimens were obtained via nephrectomy and 4 via autopsy. Specimens that were poorly preserved or had insufficient tissue and core biopsies were excluded. In many instances, normal renal tissue was included with the tumor. Diagnosis of primary RCC for each case was based on characteristic morphologic features and lack of evidence of other primary malignant neoplasms via physical examination, radiography, laparotomy, or autopsy. Cases with other masses in organs in which primary tumors may be mistaken for RCC, such as adrenal gland, thyroid gland, male and female genital organs, urinary bladder, and gastrointestinal tract, were excluded.
Histologic Analysis
Tumor classification was based on the World Health Organization histological classification of renal neoplasms. 20,21 HE glass slides were reviewed independently by 3 authors (T.J.P., M.A.M., and J.A.R.); discrepancies among authors were solved by consensus. In tubular RCCs, tubular formations were lined by cuboidal cells, partially filled with mucin or cellular debris, and separated by fibrous stroma. Papillary RCCs had branching fibrovascular projections bordered by cuboidal epithelial cells. Solid tumors consisted of dense nests of clear cells or chromophobic cells in fine fibrovascular stroma. Tumors with mixed patterns were classified by the predominant histologic type.
Immunohistochemistry
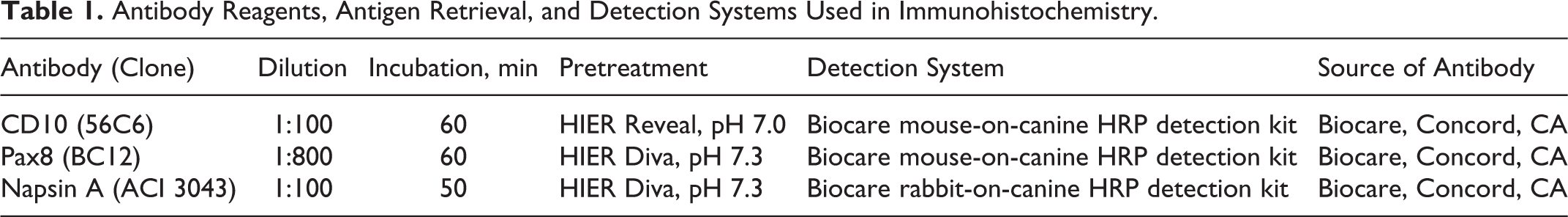
Antigen retrieval was performed on 5-µm FFPE tissue sections in a decloaker chamber (Biocare, Concord, CA) (Table 1). Three primary antibodies were used: mouse monoclonal anti-Pax8, clone BC12; rabbit monoclonal anti-napsin A, clone ACI3043; and mouse monoclonal antibody anti-CD10, clone 56C6. For each of the 3 IHC protocols, negative controls were randomly selected sections of RCC, stained with the Biocare Medical polymer negative control serum (mouse and rabbit) in place of the primary antibody. A mouse-on-canine immunoperoxidase polymer detection kit was used for CD10 and Pax8 immunohistochemistry; a rabbit-on-canine immunoperoxidase polymer detection kit was used for napsin A. The chromogen for all 3 markers was diaminobenzidine (DAB). Normal canine kidney was used to standardize the CD10 IHC method. 10,11,38 The IHC protocol for Pax8 and napsin A has been reported. 30
Antibody Reagents, Antigen Retrieval, and Detection Systems Used in Immunohistochemistry.
Evaluation of Immunoreactivity
The percentage of immunoreactive neoplastic cells in primary and metastatic RCCs was semiquantitatively evaluated for each marker as 0 = no labeled neoplastic cells, 1 = 1% to 15%, 2 = 16% to 50%, and 3 = >50%. The mean percent reactivity (MR) scores for CD10, Pax8, and napsin A for each tumor subtype were calculated from reactivity scores of positive tumors. Mean reactivity for all RCCs was calculated for each marker. The intensity of immunoreactivity in positive tumors was scored as 1+ = weak, 2+ = moderate, and 3+ = strong. Mean intensity (MI) scores were calculated for each marker for each tumor subtype; MI for all RCCs was calculated for each marker. The same reactivity and intensity scoring was performed for all RCC metastases.
Results
In normal dog kidney, CD10 was expressed in glomerular parietal epithelium and in the cytoplasm and brush border of proximal tubular epithelial cells (Supplemental Figure S1). Pax8 was detected in nuclei of cortical and medullary tubular epithelial cells and scattered nuclei of parietal epithelium. Granular napsin A expression was restricted to the cytoplasm of cortical tubular epithelium, particularly that of distal convoluted tubules.
Nuclear pleomorphism, mitotic activity, necrosis, hemorrhage, and invasiveness varied among RCCs. The 40 primary RCCs were classified as 15 papillary (37.5%), 12 solid (30%; 10 chromophobe and 2 clear cell variants), and 13 tubular RCCs (32.5%). Of these, 6 had a mixture of 2 phenotypes (4 tubular-papillary and 2 solid-papillary). Of 10 RCC metastases selected, 5 were papillary (4 bilateral to lung, 1 liver), 2 were solid chromophobe (spleen, lumbar lymph node), and 3 were tubular (1 bilateral to lung, 2 in contralateral kidney) subtypes. Metastases typically had a similar degree of differentiation based on the phenotype and growth pattern as the primary tumors, although there was a tendency for the metastatic tumors to have a single pattern (eg, solid, papillary, tubular) even if the primary tumor had a mixed pattern.
Immunoreactivity of Renal Cell Carcinomas for Pax8, Napsin A, and CD10
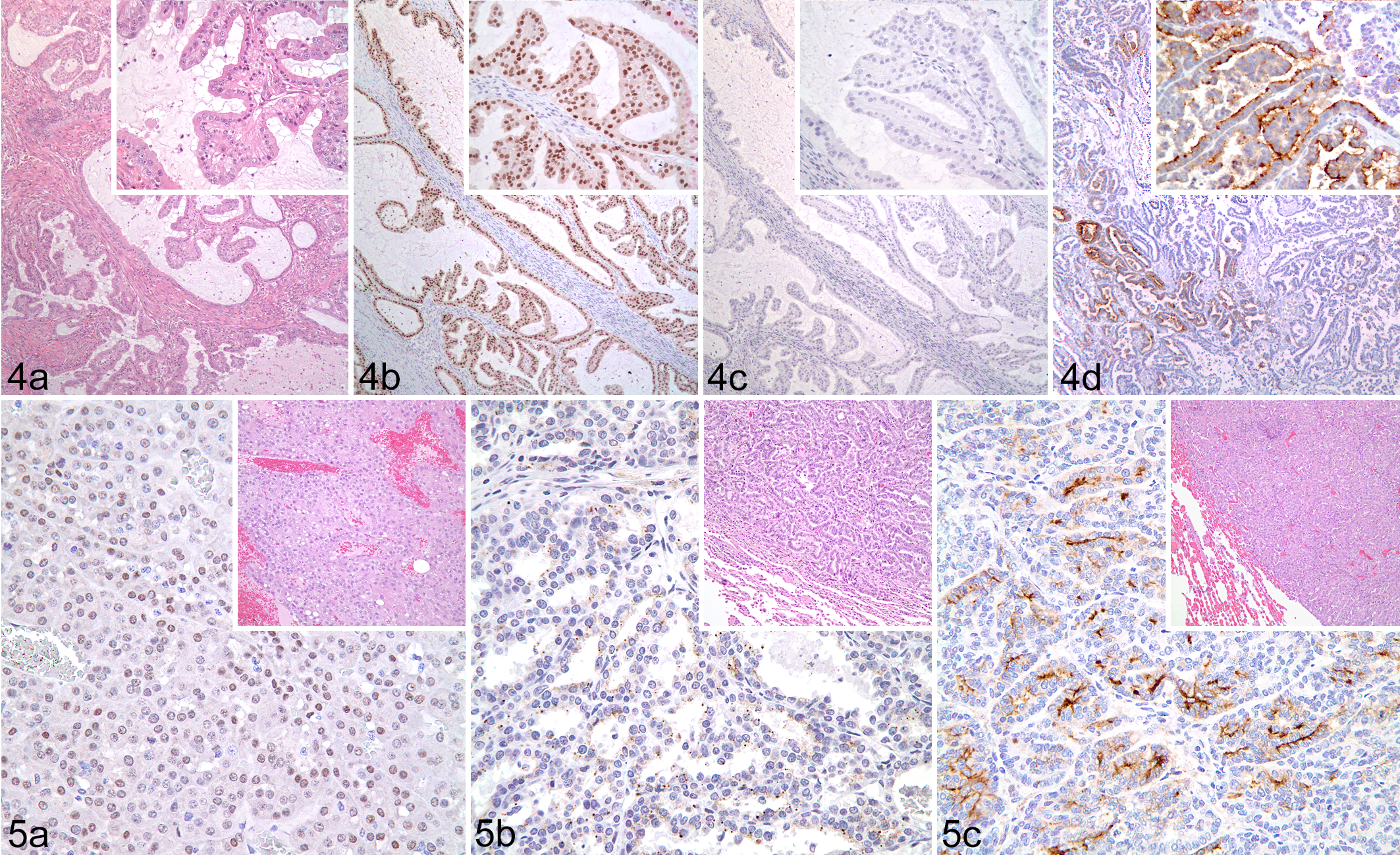
Pax8 was expressed in 11 of 12 solid, 15 of 15 papillary, and 13 of 13 tubular RCCs (Table 2, Figs. 1–4). Napsin A was expressed in 7 of 12 solid, 9 of 15 papillary, and 8 of 13 tubular RCC. CD10 was expressed in 4 of 12 solid, 7 of 15 papillary, and 8 of 13 tubular RCC. The weighted MR and MI scores for all primary RCCs positive for Pax8 were 2.83 and 2.64, respectively; 2.33 and 2.43 for napsin A; and 2.10 and 2.50 for CD10. In solid RCCs, Pax8 was expressed in nuclei of most neoplastic cells throughout the tumor, whereas napsin A expression (cytoplasmic) varied from patchy to more diffuse, and CD10 expression (membranous and cytoplasmic) was restricted to patchy areas.
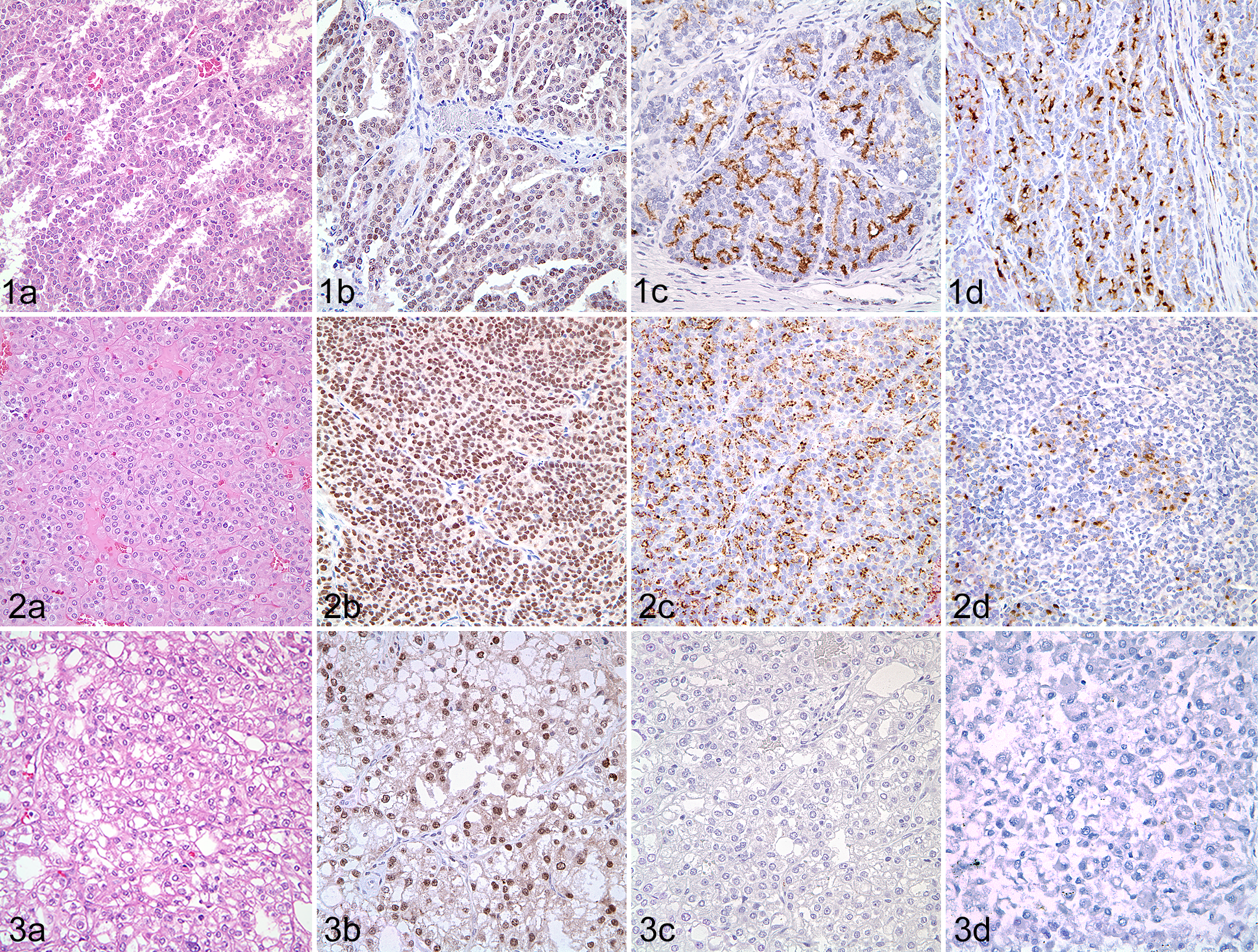
Renal cell carcinoma (RCC), kidney; dog.
Immunoreactivity of CD10, Pax8, and Napsin A in Primary and Metastatic Canine Renal Cell Carcinomas.
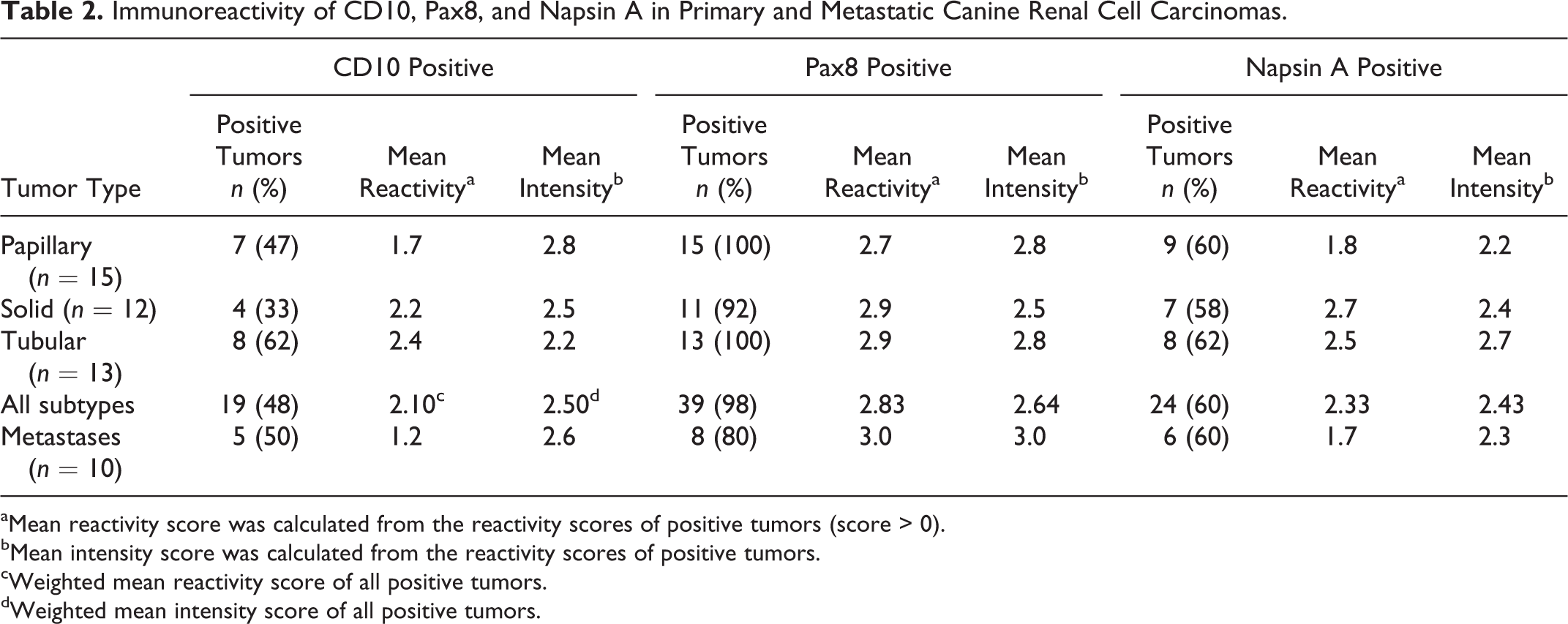
aMean reactivity score was calculated from the reactivity scores of positive tumors (score > 0).
bMean intensity score was calculated from the reactivity scores of positive tumors.
cWeighted mean reactivity score of all positive tumors.
dWeighted mean intensity score of all positive tumors.
Pax8 was diffusely expressed in both clear cell RCCs, whereas napsin A was expressed in only 1, and CD10 immunoexpression was negative in both ccRCC. Papillary RCCs displayed similar distribution of immunoreactivity as solid tumors for each of the 3 markers. Pax8 had diffuse positivity in all pRCC cells, whereas napsin A and CD10 reactivity was weak and scattered. Likewise, most tubular RCC were diffusely positive for Pax8 but contained only scattered apical cytoplasmic napsin A expression. Weak CD10 positivity was patchy and restricted to the cytoplasm and luminal plasma membrane of neoplastic cells in tubular RCCs.
In the mixed tubular-papillary tumors, the tubular component had stronger intensity for napsin A. Pax8 was strongly positive in most tubular-papillary RCC. CD10 had weak cytoplasmic expression in scattered areas of both tubular and papillary components. In the 2 solid-papillary tumors, Pax8 immunoreactivity was strong throughout both components, whereas napsin A and CD10 were weakly positive in scattered foci.
Among the 10 metastatic tumors, Pax8 labeled 4 of 5 papillary, 1 of 2 solid, and 3 of 3 tubular RCC, with an MR and MI of 3.0 and 3.0, respectively, in positive neoplasms (Table 2, Fig. 5). Pax8 expression was observed in the majority of metastatic neoplastic cells. Scattered patchy napsin A expression was noted in 60% of RCC metastases (3/5 papillary, 1/2 solid, and 2/3 tubular), with an MR and MI of 1.7 and 2.3, respectively. Weak cytoplasmic and membranous CD10 expression was observed in only 50% of RCC metastases (3/5 papillary, 1/2 solid, and 1/3 tubular), with an MR and MI of 1.2 and 2.6, respectively.
Discussion
Renal cell carcinomas are relatively uncommon tumors in dogs that can present as a diagnostic challenge, particularly when received as needle biopsy or possible metastases of unclear origin; in such cases, markers specific and sensitive for renal differentiation are desirable. In this study, Pax8 was more sensitive than napsin A or CD10 for the 3 major histologic types of primary and metastatic canine RCC. The nuclear and intense reactivity of Pax8 labeling facilitated interpretation. Pax8 was also the only antibody to label both clear cell carcinomas. The sensitivity of Pax8 for all RCC phenotypes recapitulates observations in human RCC. 8,17,19,25,26 A mouse monoclonal antibody (clone BC12) was used in this study, whereas reported human studies used both monoclonal and polyclonal antibodies, raising the question of the value in comparing Pax8 antibodies. 8,17,19 The high MR of Pax8 in positive tumors and within different histologic patterns of the same tumor, regardless of phenotype, also facilitates diagnosis of RCC, especially in tumors that might have patchy and weak expression of the other markers.
Furthermore, although the percentage of Pax8 immunoreactive cells in metastatic RCC (80%) was less than that in primary RCC (98%), the overall MR and labeling intensity for Pax8 were greater in the metastases. This finding is important as pulmonary metastases have been reported in as many as 38% of dogs with metastatic RCC in an early study, 2 although a more recent study found pulmonary metastases in only 16% of dogs with RCC. 5 Further, the expression of many other tumor markers, especially membranous markers, is reduced in metastases. 8,10,17,20,25 , 30 With only 10 metastases evaluated in this study, however, it is difficult to make inferences about the immunoexpression of Pax8 in the different subtypes.
Pax8 immunoexpression is detected in only a few normal tissues, renal neoplasms, and neoplasms of the upper Müllerian tract and thyroid gland in dogs. 30 Pax8 is therefore considered a specific marker for differentiating canine RCC from other tumors with similar morphology (eg, pulmonary carcinomas) and/or clear cell features (eg, adrenocortical, urogenital, and gastrointestinal carcinomas). However, since both RCC and thyroid carcinoma consistently express Pax8, further immunohistochemistry is necessary when metastatic disease is suspected. 30 Thyroid transcription Factor-1 (TTF-1) is a sensitive marker for thyroid follicular tumors, with cross-reactivity noted only in canine normal lung and pulmonary carcinomas. 31,32 Together, Pax8 and TTF-1 is expected to differentiate metastatic thyroid carcinoma (TTF-1 positive) from RCC (TTF-1 negative).
Unlike human RCC, which is usually ccRCC and labels consistently with anti-CD10 antibody, 1,19,27 only 2 of the canine solid RCC in this study were of the clear cell type, neither expressed CD10, and fewer than half of the remaining (chromophobe) solid RCC were CD10-positive. Additionally, the percentage of CD10-positive RCC metastases was similar to that of primary tumors, but the overall MR was much lower than that of the weighted MR in primary tumors. Furthermore, CD10 immunoreactivity was patchy. Together, these data suggest that the utility of CD10 as a diagnostic marker for most types of primary and metastatic RCC in dogs is questionable. The relative rarity of ccRCC in this canine series and weak CD10 reactivity in primary or metastatic RCC indicate a potentially different pathogenesis or cellular origin in most canine RCC compared to the human counterpart (VHL mutation, clear cell variant, and arising from proximal tubules), as suggested by previous investigators. 7,9,10,29 None of the evaluated metastases were ccRCC, which contradicts studies that indicated higher metastatic potential for this RCC phenotype. 10,11,13 However, the lack of metastatic ccRCC in our series may be a consequence of the small sample size.
In humans, napsin A is expressed mainly in papillary RCC and less commonly in ccRCC. 24 Likewise, in dogs, napsin A was expressed in a slight majority (60%) of the tested pRCC, but the percentage of reactive neoplastic cells and labeling intensity was less than that of Pax8. Furthermore, although napsin A was expressed in the same percentage (60%) of metastatic RCC as primary tumors, the weighted MR and MI were lower in metastatic RCC. Weak and patchy napsin A expression in primary RCCs and reduced immunoreactivity in metastatic tumors may confound diagnosis in difficult cases. These findings, in light of the strong expression of Pax8 in most primary and metastatic RCC, suggest Pax8 is a better marker than napsin A for the common types of canine RCC.
Napsin A is proposed as a marker of human pulmonary carcinomas and has been detected in some canine pulmonary carcinomas. 30 Therefore, cross-reactivity of napsin A in normal and neoplastic lung tissue may complicate the diagnosis of a metastatic RCC to the lung or, inversely, pulmonary carcinoma to the kidney. 4,37 Because therapy is increasingly tailored to the type of cancer and the lung is a common site of metastasis of RCC and many other neoplasms, further evaluation of napsin A expression in canine pulmonary carcinoma is warranted. 34,40 As mentioned, TTF-1 is a moderately sensitive and specific marker for canine pulmonary carcinoma, as well as thyroid carcinoma, and may aid in differentiating among metastatic pulmonary, thyroid, and renal cell carcinomas as part of an IHC panel with Pax8, napsin A, or other markers. 30 –32
In summary, Pax8 is more sensitive than napsin A or CD10 as an immunohistochemical marker of canine RCC. Furthermore, its nuclear labeling and more intense reactivity facilitate interpretation, especially in small biopsy specimens or distant metastases. Also, based on a recent publication, 30 it appears that Pax8 is highly specific for canine RCC, so with the exception of thyroid follicular carcinomas and sporadic labeling of some Sertoli, pancreatic, and ovarian tumors, it can be used to identify metastatic RCC.
Footnotes
Acknowledgements
We thank the histology technicians at Purdue University Animal Disease Diagnostic Laboratory.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
